Abstract
Home and work situations can expose diabetes medical devices to a number of environmental factors that may influence their function and safety. In accordance with regulatory requirements, manufacturing companies take great care in the construction and design of their products so that environmental factors encountered on a daily basis have as little influence as possible. However, more intense environmental conditions, such as undergoing magnetic resonance imaging (MRI), require patients to remove personal electronic medical devices beforehand. During product development, manufacturers thoroughly investigate how various environmental factors may impact a new medical device. Corresponding operational documents and manufacturer guarantees accompany each device. Similarly, manufacturers investigate any adverse interactions that may occur during communications between medical devices, such as those required with another product, smartphone, or another personal medical device, such as a pacemaker. Questions that arise from patients or medical professionals about a medical device’s safety or quality, particularly because of environmental factors, are made to the manufacturer. Manufacturers then often refer to the operating instructions, even though these contain information, such as electromagnetic compatibility, that are difficult to understand for people lacking special technical or physical knowledge. This review highlights the effects of various physical and technical influences on medical devices used in diabetes therapy.
Introduction
Today’s medical device systems are increasingly playing a role in the diagnosis and treatment of chronic diseases, like diabetes mellitus. Many electronic medical products exchange information with their environment. In doing so, they generate their own electromagnetic fields (EMF) on the one hand, while EMF and other environmental factors, like temperature, air pressure and humidity, may influence their function, on the other. Well-known examples in this context are pacemakers and defibrillators.
In diabetes technology (DT), such EMF influences can affect both therapeutic products like insulin pens and pumps and diagnostic medical devices, like blood glucose self-measurement (SMBG) systems or continuous glucose monitoring (CGM) systems. When combining diagnostic and therapeutic products for automated insulin delivery (AID) systems, not only are different medical devices used, but people also use other technical systems like smartphones and personal digital assistants in the process with intensive data exchanges between the various system components.
In their everyday lives, patients with diabetes (PwD) use DT systems under markedly different environmental conditions. They encounter diverse physical and technical influences whose strength and interactions are not always easy to estimate. In the private environment, induction stoves are used in the kitchen at home, strong electromagnetic fields occur when driving streetcars or trains, electric cars are connected to fast charging stations with high charging power and high current flows, at passing through airport scanners during a security check. In the medical environment, PwD undergo various examinations with ionizing radiation and strong EMF. In the occupational environment, not only strong EMF can occur, this can be combined with, for example, significant temperature exposure or increased ambient pressure (inside the protective suits of firefighters).
Medical devices used in diabetes therapy must function safely in most everyday situations and in many occupational situations. For safety, it is important to avoid environmental conditions that may generate inaccurate glucose measurements or incorrect insulin dosing. Therefore, medical devices must meet basic requirements and standards or they will not receive approval by the regulatory agency there (FDA) in the United States or CE mark in Europe and cannot be marketed. These requirements should ensure that the products have full functionality under defined everyday conditions.
The situation is different under occupational situations, such as for electric welders, auto assemblers, and mechanics who repair electric vehicles, where strong EMFs occur. The question often posed to physicians is whether a particular insulin pump or AID system will function safely under such conditions. If the answer is no, then they must be discarded and the PwD must transition to diabetes therapy with products unaffected by these conditions. In extreme cases, the question becomes whether the profession can be practiced by the PwD at all only with abandonment of advanced diabetes therapies.
After a general consideration of EMF, its influences and those of other factors in the private, medical and professional environment are considered. In addition, other aspects of the user environment will be discussed, like air pressure and humidity. One has to acknowledge though that the number of relevant publications about external physical and technical influences on medical devices for diabetes therapy is quite limited. The calculations provided below are our own and they are based on the equations provided.
Electromagnetic Fields
Instruction manuals of medical devices and other documents of the manufacturers refer to “electromagnetic emissions” and “electromagnetic immunity” of their products.1,2 Emissions concern any generation of EMF by the products themselves. Device immunity is important for estimating the influence of environmental EMF on a product’s function. By referring to the terms of use, manufacturers do not assume liability for their products if their function is disturbed by excessive EMF and users are harmed. However, the strength of EMF is difficult to assess for “laymen.” This is de facto true for almost all users, both PwD and diabetes team members. To characterize the applicability of medical devices for diabetes therapy in the presence of EMF, we give data for insulin pumps manufactured by Medtronic below as an example.1,2
According to the standards for electromagnetic compatibility (IEC standards) of medical devices, insulin pumps must operate reliably in the presence of electrostatic fields up to a field strength of 10 V/m (volts per meter). 3 Depending on the frequency of the field, this calculates minimum distances to be observed, which are listed in the manuals (eg, 30 cm from smartphones). Electric fields also induce a magnetic flux that can have a disturbing effect on insulin pumps. It is necessary to differentiate between the magnetic field strength, which an object feels in the field, measured in amperes per meter, (A/m) and the magnetic flux density (which is the magnetic induction, measured in tesla [T] or gauss [Gs]). The mathematical relationship between these quantities is: magnetic flux density = constant (magnetic permeability) x magnetic field strength. According to the IEC standard IEC 61000-4-8, an insulin pump must have electromagnetic immunity up to a magnetic field strength of 400 A/m over an exposure time of 60 seconds (for a short period over 3 seconds: 4000 A/m). 3 Converted into magnetic flux density, this means that the immunity is 5 x 10-6 Tesla = 0.05 Gauss. Insulin pumps are tested up to 6 x 10-4 Tesla = 6 Gauss, and guaranteed to function up to this value.
To illustrate the physical quantity Gauss, which is rarely used in everyday life, here are some examples: small magnets have a magnetic flux density of 1 Gs, a larger horseshoe magnet one of 10 Gs, the earth’s magnetic field in the middle latitudes of the earth is 0.3 to 0.5 Gs and in an MRI device (magnetic resonance imaging) this is between 5000 and 50,000 Gs. According to the IEC standard, no horseshoe magnet should be placed on an insulin pump and the pump must be removed before MRI examinations.
EMF in the Private Environment
The question arises, what other everyday conditions should be considered when using medical devices and do the EMFs from household appliances, loudspeaker magnets, WLAN routers, and so on. reach critical levels? Basically, it depends on how the field lines were designed. Induction stoves, for example, are unproblematic because the coils generating the magnetic field in these stoves are designed in such a way that the greatest magnetic field density is created exactly where the bottom of the pot is. EM vortex fields are created in the bottom, heating the food, frying it, and so on. Almost no stray field is generated and furthermore its strength decreases quadratically with distance toward the outside (like any field). This means that a distance of 10 to 20 cm is sufficient to prevent interference with medical devices such as an insulin pump, but a distance of 50 cm is safer. Neither an insulin pump nor the transmitter of a CGM system should be held directly over an active hotplate. The situation is comparable for microwave devices.
Information about the distance between medical devices for diabetes therapy and an EMF can be found in Table 1. These are minimum distances that should be kept from consumer products that generate EMF, larger distances always reduce the risk of interference. The minimum distance also depends on the frequency and energy of the source field. Smartphones operate—depending on the type—in a frequency range of 1.8 to 850 MHz, cordless phones in the household from 1.9 to 5.8 GHz. A distance of at least 30 cm must be maintained from these. WLAN networks in the EU operate at 2.4 GHz, and a minimum distance of 1 meter should be maintained from these because of the higher power.1,2 WLAN networks in the US (that adhere to the 802.11 standard) work over several distinct bands between 1000 MHz and 60 GHz.
Information on the Minimum Distance Between Sources of EMF (eg, Radio Frequency Transmitters in the Household) and, as an Example, an Insulin Pump (Here One From Medtronic MiniMed).
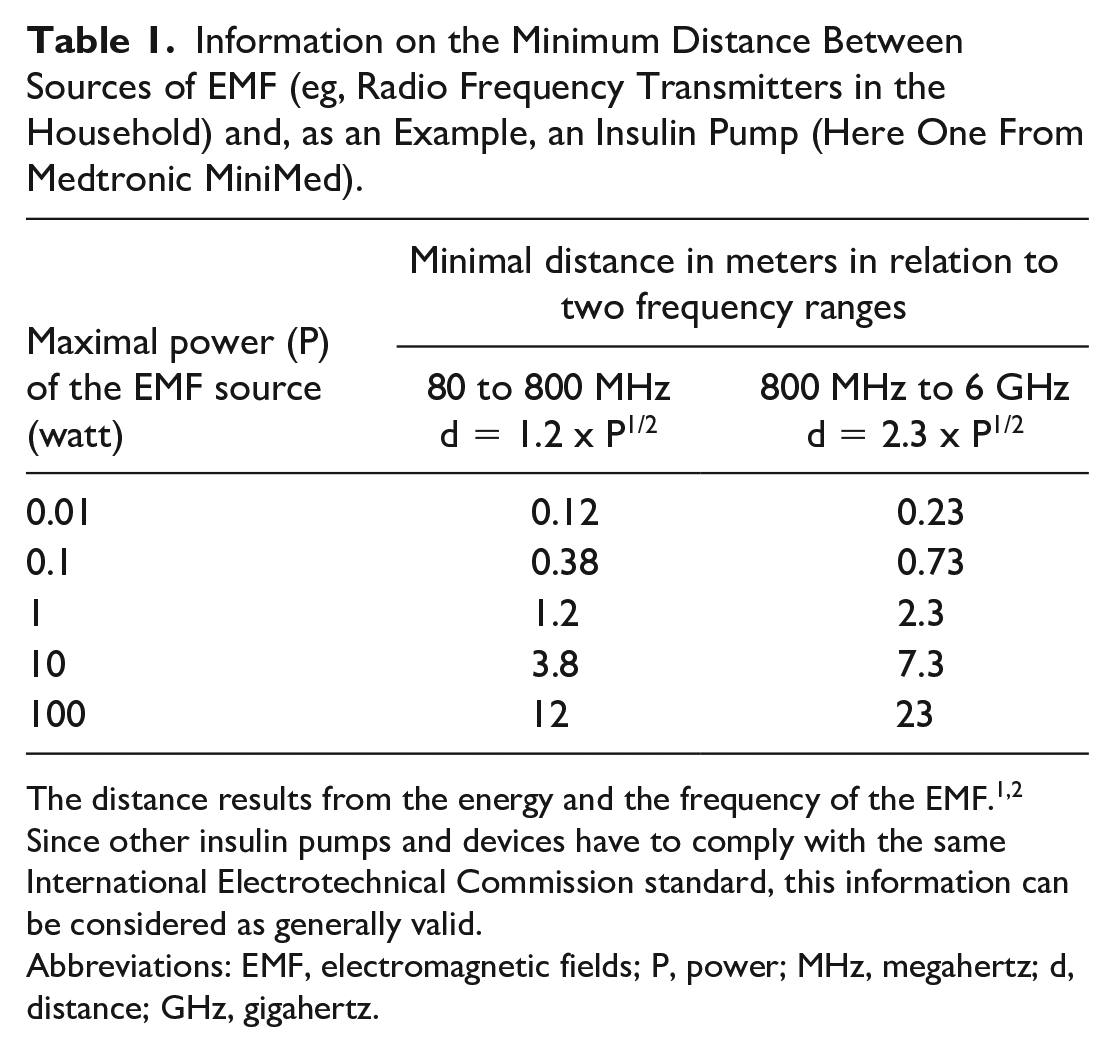
The distance results from the energy and the frequency of the EMF.1,2 Since other insulin pumps and devices have to comply with the same International Electrotechnical Commission standard, this information can be considered as generally valid.
Abbreviations: EMF, electromagnetic fields; P, power; MHz, megahertz; d, distance; GHz, gigahertz.
EMF in the Medical Environment
Exceptionally strong EMFs can occur during diagnostic imaging procedures in hospitals when such examinations are to be performed on PwD. For example, an insulin pump must never be placed in the vicinity of an MRI machine, which has an EMF of up to 50,000 Gauss, that is, 8400 times more than permissible. These values correspond to average values, both for the products themselves and for the external technology. Such strong EMF not only influence the proper function of such products, they can destroy them! Especially under such environmental conditions, the manufacturer’s instructions in the documentation for these products should be followed. In the presence of strong EMFs, the function of the transmitters of CGM systems is also not guaranteed. Protective measures against strong EMFs like shielding by metal foils (to establish a Faraday cage) or magnetic fields by ferroelectric foils, are not recommended, because the PwD has difficulties in estimating the effects of the fields and the real shielding protection achieved.
The situation is somewhat different with X-ray devices. X-rays themselves deposit little energy in a medical device due to their high penetrating power (see section “Interaction of medical devices with electromagnetic radiation”). For the medical imaging procedure, the X-ray tube is operated with an electrical voltage between 25 and 500 kV, depending on the device. In the electric field, which mainly builds up between the cathode and the anode, the electrons are accelerated from the cathode to the anode. At the anode, they are decelerated, which generates the X-rays. The resulting EMF could therefore influence an insulin pump, for example. The X-ray machines are well shielded so that, if possible, only the emitted X-ray beam leaves the equipment. The EMF generated by the energy supply of the X-ray tube is also shielded. Nevertheless, it is recommended to remove insulin pumps, and so on, before the examination. The situation is similar with computer tomography, which also uses X-rays, but which rotate in different directions around the object being examined.
EMF in the Professional Environment
A significant aspect for PwD is the practice of professions where strong EMFs occur, such as electric welders, electric car assemblers, and so on. Since the products should be used according to the manufacturer’s instructions, this may mean that they should be taken off for the period of exposure to stronger EMFs (eg, during an electric welding operation). Therapeutically, briefly taking off an insulin pump or disconnecting the transmitter in a CGM system is not problematic, which many users do anyway, for example, during a visit to the sauna, and for physical contact during sports. However, if the exposure affects the major part of the working day, it has to be decided whether the occupation should be practiced at all or whether therapy management with insulin pumps, CGM or AID systems has to be abandoned. It is also worth to mention that taking of the pump or CGM system just for the period of exposure is not possible with patch pumps or with modern CGMs (with either an integrated transmitter or with designs intended to prevent transmitter removal and sensor re-use).
To illustrate, a young man with type 1 diabetes who uses an AID system from Medtronic for his diabetes therapy wanted to work as a car mechanic and is expected to repair electric cars. He also has to charge the batteries at a fast charging station with a charging capacity of 40 to 120 kW (kilowatts). Using basic physical equations, the induction B of a current-carrying conductor can be calculated (according to Maxwell’s equations) to:
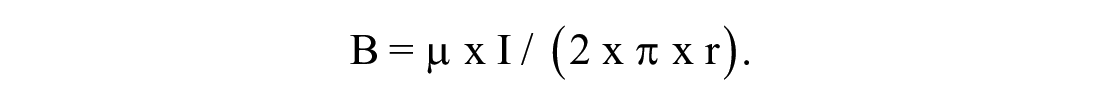
Here, I is the current strength, r the distance from the conductor and µ the magnetic field constant.
For the magnetic field strength H applies:
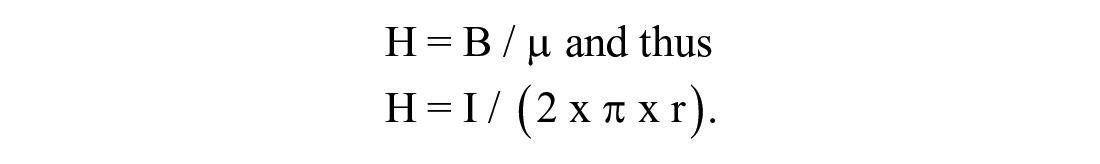
The magnetic field strength H at a distance of 10 cm from the current-carrying cable (which would therefore act on the AID system at this distance) is as follows
at a current strength of 30 A: 47.8 A/m (amperes per meter),
at a current strength of 50 A: 79.6 A/m.
At the charging power of the quick charging station and a voltage of 220 volts, an electric current of 182 to 545 A flows. This means, at a distance of 10 cm, the magnetic field strength is H = 290 to 867 A/m. According to the IEC standard IEC 61000-4-8, electromagnetic immunity must be provided up to a magnetic field strength H of 400 A/m for exposure over 60 seconds (briefly over 3 seconds: 4000 A/m). The PwD can carry out his professional activity, but he should not get too close to the insulated electric cable and, if he does, only for a short time. Larger distances, on the other hand, are not critical, at a distance of 1 m, the magnetic field strength is H = 30 to 87 A/m, at 50 cm, H= 58 to 174 A/m.
Generation of an EMF by the Medical Devices Themselves
If the medical devices themselves generate an EMF, they can affect sensitive electronic technology, e.g. in the work environment. For example, when transmitting data from a CGM system to an insulin pump coupled to it via radio interface (more modern pumps use Bluetooth), the electronic precision measurement technology in a laboratory could be disturbed. For this purpose, the electromagnetic immunity of this measurement technology must be determined from its manufacturer’s documentation. It is also theoretically conceivable that the communication of electronic components within an aircraft could be affected. For this reason, some (older) CGM systems are recommended to be switched off in aircraft, even if their low transmission power during data transmission is not expected to endanger flight safety. With modern CGM devices, most systems that are tested with respect to the FAA/RTCA DO-183 standard (https://global.ihs.com/doc_detail.cfm?&document_name=RTCA%20DO%2D183&item_s_key=00088339&item_key_date=131113), show compliance with airworthiness requirements. Therefore, this appears not to be an issue with such CGM systems.
Sometimes PwD fear that they themselves may be harmed by the energy emitted by a CGM transmitter. A comparison with the energy output when using a smartphone shows that the data transmission of a CGM system (with transmission of a glucose value every 5 minutes, ie, 288 times a day) corresponds to that of approx. 4 seconds of telephoning with a smartphone per day, that is, is extremely low.
Interaction of Medical Devices With Electromagnetic Radiation
In passenger aviation, further interactions of EMF and radiation with medical devices for diabetes therapy occur, for example, with full-body scanners at airport security checkpoints. There, X-rays are used to screen baggage, which may contain medical devices. On the surface, the situation is like that encountered in medical diagnostics. The key question is, what happens inside the medical devices when they come into contact with radiation, will their electronics be affected in such a way as to cause functional impairment or even failure of the device?
Basically, this depends on whether radiation damage of a relevant extent can occur in the device, especially in its electronics, and whether it is permanent. The extent of radiation damage depends in particular on how much energy per volume is deposited (ie, absorbed) during penetration in the product. This depends on the type of radiation and its energy (Table 2). Here, ionizing radiation may be dangerous, not only for a medical device, but also for organic material in the person wearing it.
Properties of Radiation and Its Ability to Produce Radiation Damage and Thus Destroy Medical Devices.
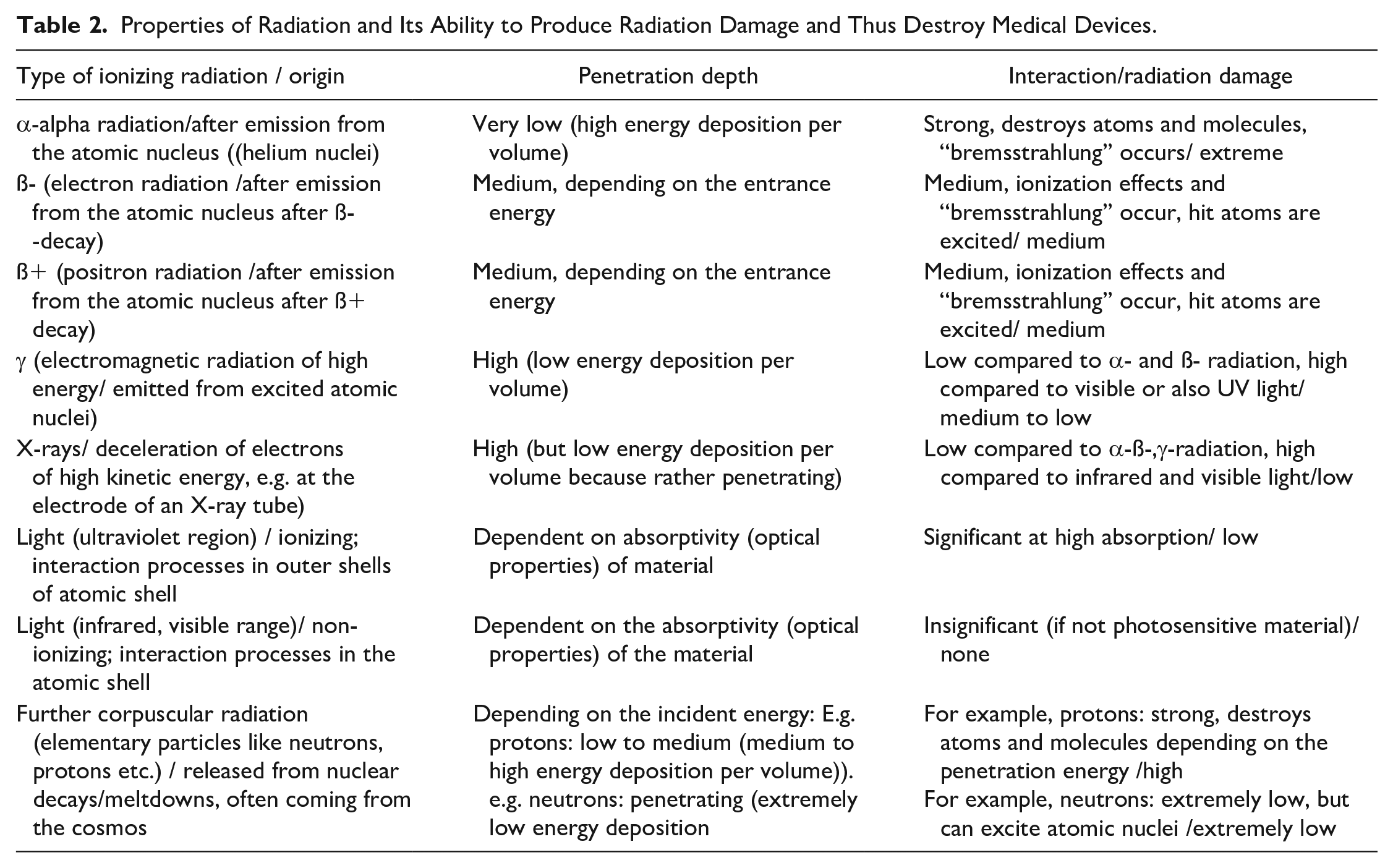
Ionizing radiation is produced after excitation or decays in the atomic nucleus and thus has an energy in the range of kilo-electron volts (keV) to mega-electron volts (MeV). This is thousands to millions of times higher than in processes in the electron shell, which, for example, result in the generation of light and are responsible for chemical reactions. The decisive factor is the extent to which electrons are knocked out of atoms or molecules by impact processes, leaving behind molecular remnants, that is,. radiation damage that does not repair itself (by recombination). This is especially the case with corpuscular radiation such as α-radiation, because its penetration depth into the material is very shallow, but all of the initial energy is deposited in the process. In contrast, other forms of corpuscular radiation such as ß—or ß+-radiation (electron or positron radiation from nuclear processes, respectively) penetrate further, passing through insulin pumps and similar large devices, but are absorbed only slightly and thus cause less radiation damage.
In contrast, gamma rays and X-rays are highly ionizing electromagnetic waves, similar to visible light, only much shorter wavelength and thus more energetic (visible light has wavelengths of 380 to 780 nm, X-rays from 0.001 nanometers [nm] to 10 nm, thus 50 to 50,000 times more energetic than light), gamma rays below 0.001 nm). While gamma rays originate from processes in the atomic nucleus, X-rays are produced by high-energy electron processes. Both types of radiation penetrate materials strongly, are absorbed only slightly, thereby depositing little energy as they pass through and causing only moderate radiation damage.
There have been anecdotal reports of PwD with type 1 diabetes having their insulin pump malfunction after passing through a full-body scanner at an American airport. Such scanners sometimes use X-rays in the low-energy range. There is evidence on the Internet that whole-body scanners operate with electromagnetic waves in the terahertz range 4 ; this corresponds to waves with a frequency of 1012 Hz (they belong to the microwave spectrum in the far infrared, different information is because of the different products used at airports). These waves penetrate biological tissues (otherwise they would not be used for this purpose), but leave little energy there and are non-ionizing, therefore they do not cause radiation damage. At most, there may be minor heating of insulin pumps, CGM systems and tissue. Therefore, scanning of PwD with actively used products is not expected to have a relevant impact on their function. The situation is similar for scanners that use radiation in the low-energy X-ray range.
As stated above, manufacturers of medical devices must test them for interaction with various radiation sources and EMF. If there is any doubt whether in the current situation these products are exposed to high levels of radiation, then the PwD should discard them. Caution should be exercised, especially when the products control insulin delivery, whether this is done by “classic” insulin pumps, where there are disturbances in their function, or by AID systems, where glucose levels measured by a CGM system to control insulin delivery. It can also happen that the medical devices emit alarms for no apparent external reason, that is, they exhibit a malfunction per se.
Influence of Other Factors on Insulin Pumps
PwD rely on insulin pumps having high dosing accuracy. This property of pumps is represented by manufacturers in so-called “trumpet curves,” which demonstrate that insulin delivery is reliable even at low doses in the range below 0.1 IU; this means that insulin of concentration U100 will reproducibly deliver a fluid volume of 0.001 ml. 5 In the clinical setting, an insulin pump pumps insulin from the reservoir via the infusion set into the patient’s subcutaneous adipose tissue. In so-called patch pumps, the visible infusion set is omitted and the insulin is applied directly. This creates an interconnected, self-contained “pump-body” system. The question is what happens when this system is subjected to rapid and significant changes in air pressure, as it is the case when flying.
Passenger airplanes have a cruising altitude of about 10,000 m. Inside the cabin, the air pressure is set to about 560 Torr (about 746 mbar; 74.6 hPa). This lower air pressure compared to the air pressure at sea level (approx. 760 Torr = 1013 mbar; 101.3 hPa) reduces the pressure gradient between the inside of the aircraft and the outside pressure, which means that the thickness of the outer wall of the aircraft can be kept thinner by design, which saves a considerable amount of weight. In an insulin pump, the air pressure is generally the same as in the environment, as there is always pressure equalization between the inside and outside because of the design. Otherwise, the pump would unintentionally deliver insulin if the external pressure dropped rapidly, that is, the PwD would be exposed to the risk of hypoglycemia. Since pressure equalization is rapid, it is basically irrelevant how high the pressure outside the pump is; there is no pressure gradient. This also applies to aircraft takeoff and landing, during which time rapid pressure changes occur in the cabin. This is also true when a fast elevator is used in a high tower.
However, a publication published in 2011 reported that there was an unintentional insulin delivery of 1.1-1.4 IU during aircraft takeoff. 6 However, this study did not examine the “total system,”, that is, with pumps connected to the subjects’ bodies, but rather the pump was “open” in a pressure chamber. This means that there is no counter pressure at the outlet at the tip of the needle of the infusion set because of the subcutaneous fatty tissue in which the needle sits in the real application. There is always a slight overpressure of about 10 mbar in the tissue compared to the air pressure at sea level (in the leg it is slightly higher than in the arm, for example). Even if the external air pressure changes, the counter pressure in the tissue is given. Consequently, there is no undesired insulin delivery in the normal case of use in an airplane.
Siphon Effect
The so-called siphon effect was reported 10 years ago, based on observations of infusion sets not connected to the patient. 7 In a laboratory experiment, infusion sets with a length of 80 to 110 cm were attached to different insulin pumps. The tip of the infusion set was then raised or lowered relative to the position of the insulin pump. Measurement of the basal amount of insulin delivered revealed differences depending on the position of the tip of the infusion set relative to the pump. This was interpreted as an insulin dosing error, the cause of which was said to be the siphon effect.
When the tip of the infusion set is raised or lowered relative to the pump, the change in height results in a change in the gravitational pressure on the tip, which corresponds to the magnitude
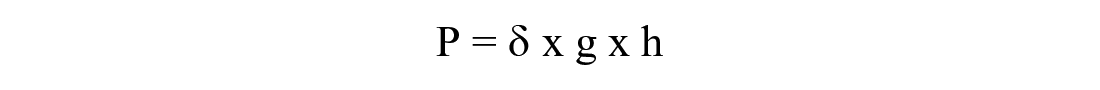
δ is the density of the flowing fluid, g is the acceleration because of gravity, h is the change in height. According to Bernoulli’s equation (this corresponds to the law of conservation of energy of classical mechanics for flowing media)
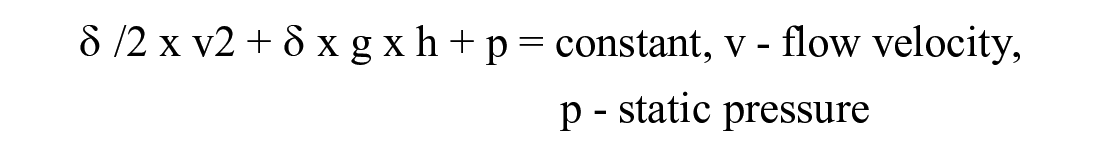
this leads to a change in the total pressure, which in the case of the open cannula tip results in the so-called siphon effect. Under real clinical conditions, however, the tip of the cannula is stuck in the subcutaneous fat tissue. As explained above, there is a counterpressure there that is several mbar higher than the external air pressure. The small pressure differences resulting from a maximum conceivable change in height (infusion tube lengths of 110 cm) cannot overcome this counterpressure of the tissue. Consequently, there is no siphon effect under real application conditions, that is, there are no inaccuracies in insulin delivery caused by this.
It is worth to mention the EN 60601-2-24 standard. This is intended for manufacturers to check for protection against over-infusion (free flow) conditions. With a tubed pump, the infusion set is used in combination with reservoirs; such reservoirs, at least in some examples, are designed to prevent free flow and work against gravity. This is in addition to the counter pressure provided by sub-cutaneous tissue.
Insulin Delivery During Diving
When diving, the PwD must not dive too deep for various reasons if insulin is supplied by a pump: Insulin pumps—according to their approval—are only waterproof up to a depth of 3.6 m. It is worth to mention that other pumps are rated as waterproof to a depth of 3 feet for up to 30 minutes (IPX7 rating). Furthermore, pumps claiming to be waterproof to IPX8 may vary in the depth to which they are tested, as testing conditions and duration are not strictly specified and are defined by the manufacturer.
If deeper diving is required, the pump must be stowed in a pressure-resistant dry box. In this case, the concern about hypoglycemia because of an unwanted insulin supply is usually unfounded, because the conditions during diving are exactly the opposite of those during flying because of the increase in pressure; that is, the external water pressure exerts considerable pressure on the subcutaneous fat tissue. As a result, the pressure at the tip of the infusion set is higher; therefore, at greater water depths (at 10 m depth, the air pressure is twice that of sea level), insulin is no longer infused into the body. During longer dives, in extreme cases, the pump will at some point give an occlusion alarm (= closure of the infusion set), in which case the infusion set is not actually closed, but the insulin backs up because of the back pressure, which leads to this error message being triggered. However, when the diver comes back to sea level, additional insulin might be delivered by the pump.
Influence of External Factors on the Quality of the Glucose Measurement
In SMBG, a low or a high ambient temperature can falsify the measurement results. According to the manufacturers, most blood glucose meters and test strips should be used at an ambient temperature of 10°C to 40°C during measurement. The relative humidity should be in the range of 10% to 90%. Thermistors and algorithms implemented in the measurement systems provide temperature compensation in many blood glucose meters to guarantee high measurement accuracy even with rapid temperature changes (e.g., visiting a restaurant after skiing), but patients should still wait at least 15 minutes for the temperature of the affected meters and strips to equalize before blood glucose measurement in such a case. 8 However, it is not only the environmental conditions at the time of the actual performance of a measurement that are important; it is also important to ensure appropriate environmental conditions when storing the test strips. International standards (DIN 15197) exist for the evaluation of the performance of SMBG systems; however, yet there are none for CGM systems.
In CGM systems, it is an advantage that the glucose sensor is placed in the subcutaneous adipose tissue, where “laboratory conditions” exist, that is, there are relatively well-defined temperature and pressure conditions. Studies with CGM systems in pressure chambers simulating pressure levels as in an airplane, at the height of mountain peaks (5,500 m), and at 30 m water depth (4 times the normal atmospheric pressure) showed that reliable measurement results are obtained under these conditions. 9 The situation is different with regard to direct pressure applied to the skin: For example, if the user is lying during sleep on the skin site where the glucose sensor has been pricked, pressure is exerted on the glucose sensor there or the skin blood flow is reduced in this area, this may cause the glucose concentration in the local interstitial fluid to drop and apparently low glucose values are indicated to the PwD. The glucose sensors of CGM systems should be stored within the range of conditions indicated on the product label provided by the manufacturer.
Any influence of external factors on the performance of CGM systems can be quite impactful with AID systems. To our knowledge no studies have been performed and published yet evaluating the impact of such factors on the performance of AID systems. Depending on the environment the patient is in, also insecure transmission of data should be considered.
Summary
The medical devices used by a PwD in their diabetes therapy tolerate electromagnetic fields, ionizing radiation, changes in pressure and temperature over a considerable range with no clinically relevant influence on their function. Manufacturers systematically explore these factors during product development to exclude safety risks. Otherwise, the products cannot be placed on the market. In a product’s operating system, information is provided for several parameters regarding the value ranges of the physical influencing factors for which the manufacturers guarantee safe functioning of the product. However, all this does not exclude with absolute certainty that in the daily use of the products “disturbances” will not result from environmental factors if these reach a strength outside the usual framework. More respective studies should be performed and published. The PwD must be suitably informed about this during the training for the use of the respective medical products.
Footnotes
Acknowledgements
We’d like to thank John Walsh, San Diego, CA for his help with this manuscript.
Abbreviations
AID, Automatic Insulin Dosing; CGM, continuous glucose monitoring; DT, diabetes technology; EMF, electromagnetic field; PwD, patient with diabetes; SMBG, self measurement of blood glucose.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.T. was scientific director of Medtronic Germany, manufacturer and distributor of insulin pumps and CGM systems. L.H. is a consultant to a number of diagnostic and therapeutic companies in the development of new products. He is a shareholder in the Profil Institute for Metabolic Research in Neuss, Germany.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
