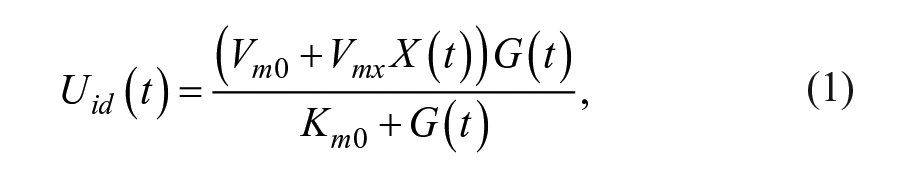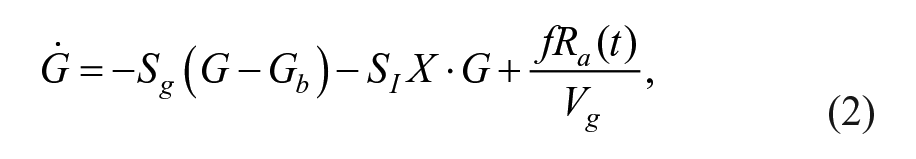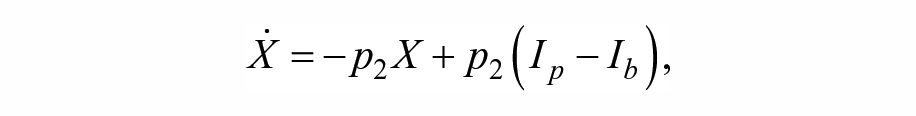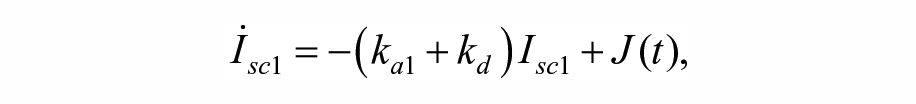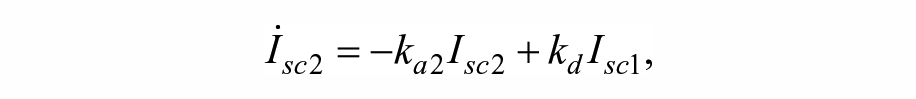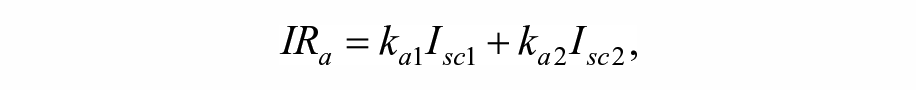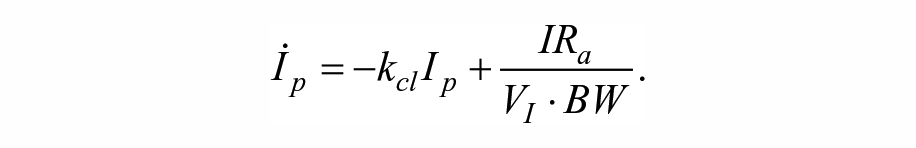Abstract
Background:
Model predictive control (MPC) has become one of the most popular control strategies for automated insulin delivery (AID) in type 1 diabetes (T1D). These algorithms rely on a prediction model to determine the best insulin dosing every sampling time. Although these algorithms have been shown to be safe and effective for glucose management through clinical trials, managing the ever-fluctuating relationship between insulin delivery and resulting glucose uptake (aka insulin sensitivity, IS) remains a challenge. We aim to evaluate the effect of informing an AID system with IS on the performance of the system.
Method:
The University of Virginia (UVA) MPC control-based hybrid closed-loop (HCL) and fully closed-loop (FCL) system was used. One-day simulations at varying levels of IS were run with the UVA/Padova T1D Simulator. The AID system was informed with an estimated value of IS obtained through a mixed meal glucose tolerance test. Relevant controller parameters are updated to inform insulin dosing of IS. Performance of the HCL/FCL system with and without information of the changing IS was assessed using a novel performance metric penalizing the time outside the target glucose range.
Results:
Feedback in AID systems provides a certain degree tolerance to changes in IS. However, IS-informed bolus and basal dosing improve glycemic outcomes, providing increased protection against hyperglycemia and hypoglycemia according to the individual’s physiological state.
Conclusions:
The proof-of-concept analysis presented here shows the potentially beneficial effects on system performance of informing the AID system with accurate estimates of IS. In particular, when considering reduced IS, the informed controller provides increased protection against hyperglycemia compared with the naïve controller. Similarly, reduced hypoglycemia is obtained for situations with increased IS. Further tailoring of the adaptation schemes proposed in this work is needed to overcome the increased hypoglycemia observed in the more resistant cases and to optimize the performance of the adaptation method.
Keywords
Introduction
Automated insulin delivery (AID) systems have been shown to be safe and effective in improving glycemic outcomes in individuals with type 1 diabetes (T1D). However, insulin dosing in T1D is complicated by several physiological and psychobehavioral aspects influencing insulin demand. Fluctuating insulin needs due to metabolic variability are mostly linked to changes of an individual’s insulin sensitivity (IS), driven by factors such as circadian rhythms, physical activity, psychological stress, and menstrual cycle.1,2 These time-varying insulin requirements represent an important challenge for fully closed-loop (FCL) systems. In this context, model predictive control (MPC) has become one of the most popular regulatory strategies for AIDs due to its flexibility and predictive ability.3-6 These algorithms rely on a model of insulin-glucose dynamics to compute an optimal control policy satisfying a set of constraints within a predefined time horizon at each sampling. Although the effectiveness of MPC-based AID systems has been evaluated in various clinical studies, there are still challenges related to parametric uncertainty and unmodeled phenomena that could affect the performance of MPC. 3 ,7-9 These challenges are mainly due to the setting of biased parameters that negatively affect the accuracy of the model,6,10,11 and the use of compact (oversimplified) models that only capture the basic dynamics and neglect complex interactions within the process.3,12 To overcome these aspects, several approaches include a disturbance input to account for unmodeled phenomena. These approaches use low-order population models.9,13 This provides a lumped measure of the overall model mismatch at each time instant, which increases the robustness of the controller. Alternatively, personalization strategies have been explored by modifying either the controller’s internal model6,9,14-20 and/or controller hyperparameters3,13,16,21-24 based on subject-specific clinical variables such as total daily insulin (TDI), correction factor (CR), or body weight (BW), or on the estimation of the transient dynamics and glucose variability, or model-individual mismatch. Furthermore, there is evidence that increasing model complexity in an MPC framework has no significant effect on controller performance, while adapting model parameters to a virtual patient population increases time in the normoglycemic range and decreases time in hypoglycemia. 25 In particular, online model identification to estimate IS and update the prediction model within the MPC has shown to reduce model-plant mismatch and improve glycemic control in situations where insulin action changes due to variations of insulin clearance or acute IS fluctuations due to exercise or during pregnancy.17-19, 26 It is worth noting that in the latter approaches, the MPC does not include a disturbance input. In this regard, there is evidence indicating that personalized models and disturbance inputs can be used to improve performance within a zone MPC framework.13,20 In addition, considering hybrid closed-loop (HCL) systems, different techniques were proposed to continuously track postprandial glucose dynamics and IS within an adaptive basal-bolus calculator, 18 showing an increase in time in target range, and the ability of this scheme to handle different parametric variations by solely adjusting IS. Similarly, in Hovorka et al 27 a technique was proposed for tracking IS in real time from CGM and insulin pump data for its use in a smart bolus calculator; this algorithm was clinically validated, showing a reduced postprandial hypoglycemia following an exercise-induced IS increase.
Given the advantages that model personalization and IS tracking have shown in closed-loop systems, and in particular for MPC-based systems, in this work we evaluate whether these benefits are observed within our AID system both in its HCL and in its FCL configurations. 28 Considering that this AID system uses a disturbance input to account for the differences between the model and the individual, we aim to evaluate whether tracking the current metabolic state of the individual and tailoring the controller to adapt to it achieves protection against hypoglycemia and hyperglycemia when the person is experiencing increased or decreased IS. To evaluate how this adaptation can affect closed-loop performance in terms of clinically relevant metrics, we leverage the Food and Drug Administration–accepted University of Virginia (UVA)/Padova T1D simulator 29 to (1) obtain estimates of IS for the adult in silico population under different conditions and (2) evaluate the impact of IS-based controller adaptation using a novel performance index.
Methods
Evaluation of the effect on performance of an IS-informed HCL/FCL was performed in silico using the 100-adult UVA/Padova simulator30,31 cohort in three steps: (1) modification of IS parameters in the simulator, (2) estimation of IS, and (3) performance analysis.
IS Variations in the Simulator
Modification of IS parameters related to insulin-dependent glucose utilization was made by multiplying each parameter by a factor
where
Estimation of IS
Estimates of IS were obtained by means of a simulated mixed meal glucose tolerance test. 32 For this, one-day simulations with a single meal with 80 g of carbohydrates (CHO) were performed, providing an insulin bolus determined based only on each subject’s CR. The estimation was performed based on a variation of the Subcutaneous Oral Glucose Minimal Model, 33 with a triangular submodel for the subcutaneous insulin transport infusion. The model equations are as follows:
State variables and parameters for the model are described in Table 1.
State Variables, Input Variables, and Parameters With Their Respective Units and Population Values.
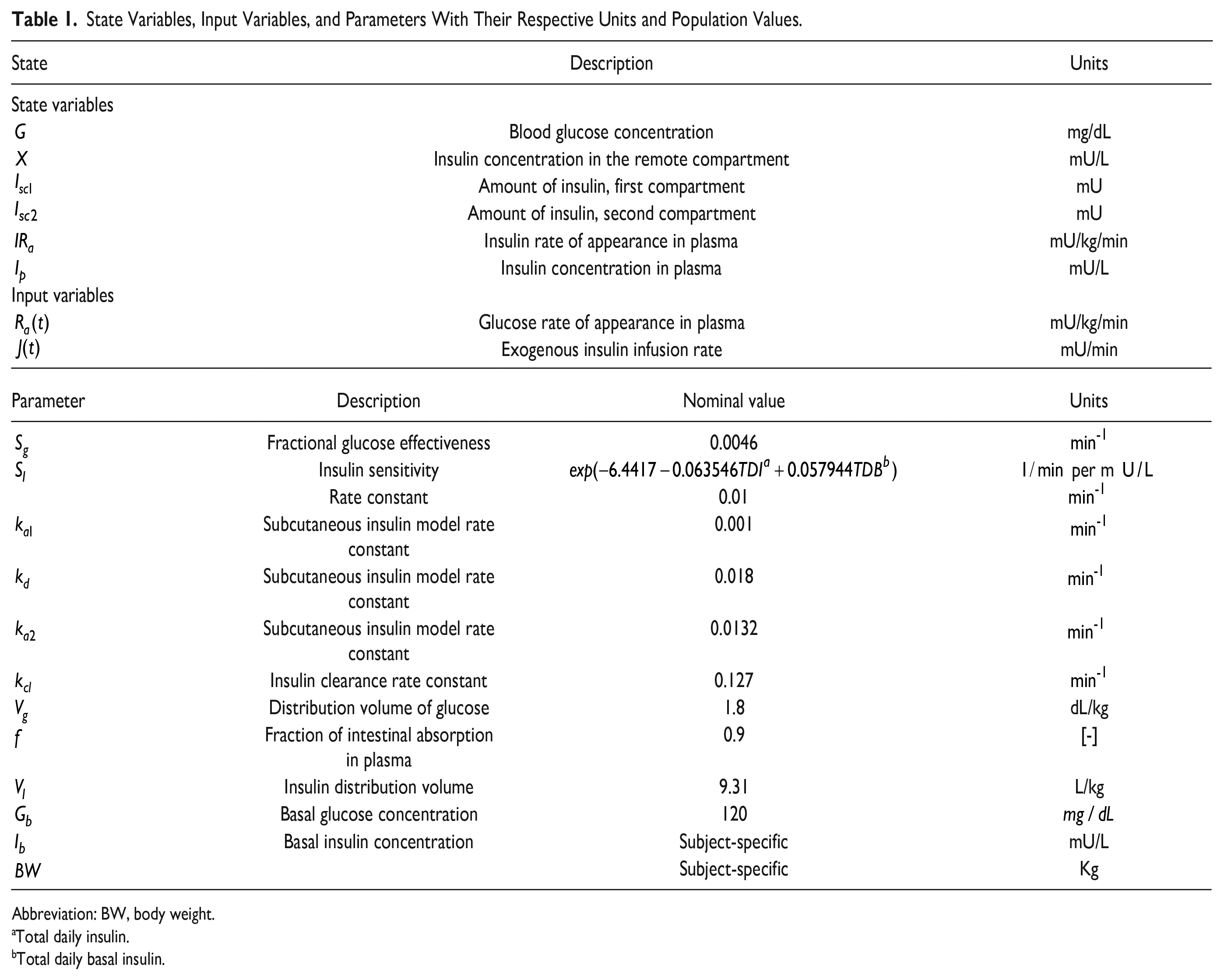
Abbreviation: BW, body weight.
Total daily insulin.
Total daily basal insulin.
Parameter
Strategy for informing IS to the AID system
This individualization is carried out by modifying the MPC prediction model and key parameters in the controller according to the estimated IS. A schematic representation of the AID system and the key parameters modified is presented in Figure 1.
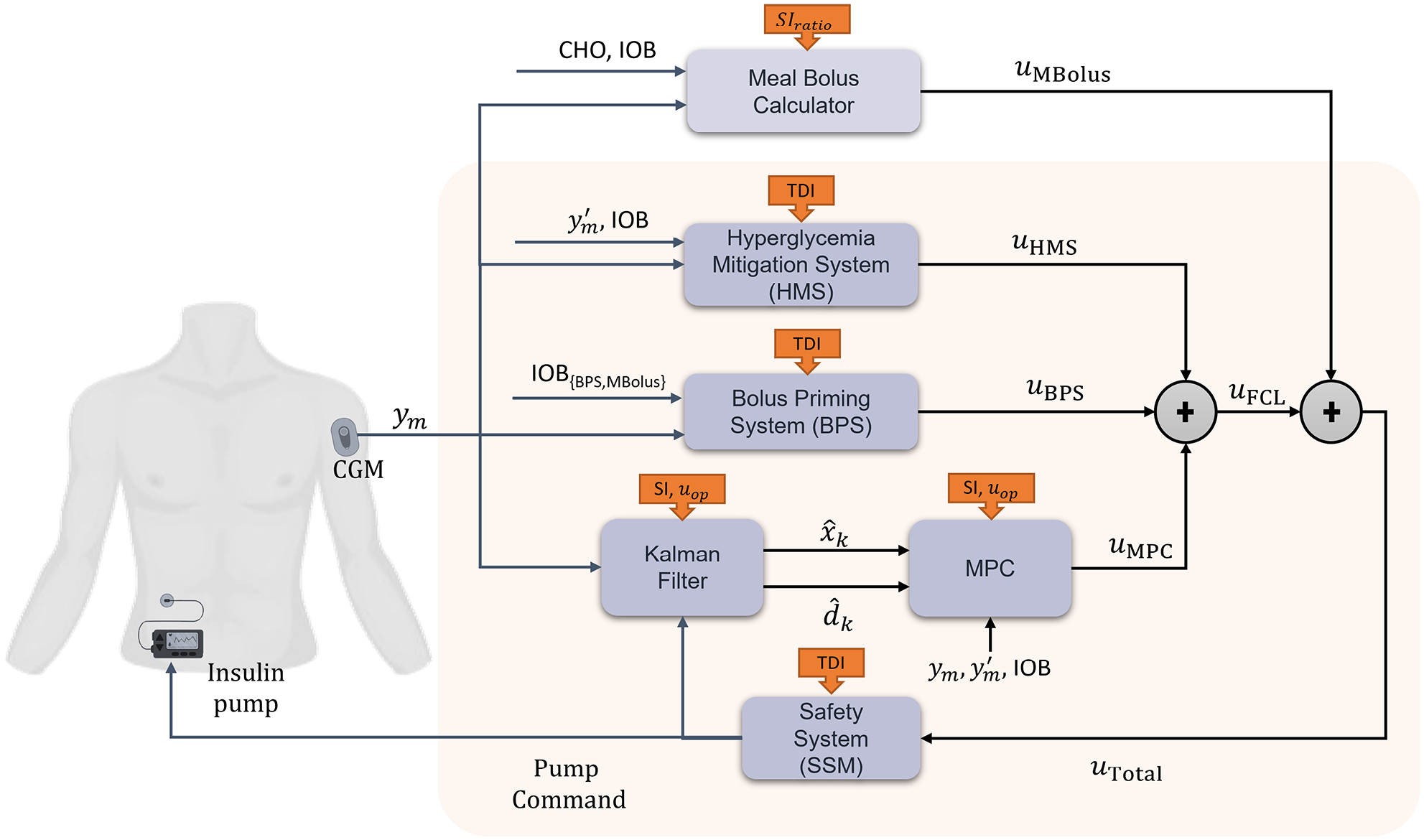
Schematic representation of the AID system, displaying the key systems components and the integration of the estimated IS values.
AID system
The UVA AID system is a system that integrates the following modules: (1) a safety system (SSM) to compensate for imminent hypoglycemia by attenuating the control action; (2) a Hyperglycemia Mitigation System (HMS) to compensate for prevailing hyperglycemia by delivering correction doses; (3) a Bolus Priming System (BPS) to mitigate abrupt positive disturbances; (4) a Kalman Filter (KF) to estimate both states and disturbance input; (5) a linear MPC algorithm that regulates the basal insulin based on the KF estimates; and (6) a Bolus Calculator (BC) to generate prandial insulin boluses. The HCL uses all the modules, while in the FCL configuration, the BC module is turned off to not require any meal announcements. The reader is referred to 3 a full description of each module.
Controller individualization
The proposed adaptation of the system considers modifications of the HMS, BPS, BC, and MPC modules. Overall, we modify key parameters within each module by a factor depending on the physiological state of the subject, defined as
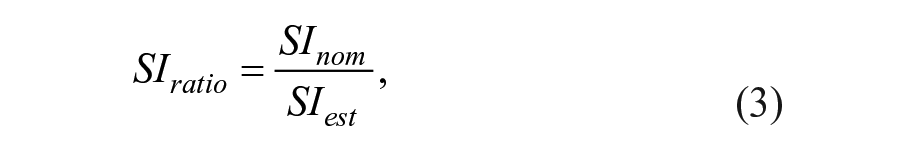
where
KF and MPC
In order to embed the metabolic model
1
into the AID linear MPC formulation, the meal subsystem in the model is disregarded (for both HCL and FCL configurations) and a disturbance input
To modify the action of the MPC based on IS, we consider two modifications: (1)
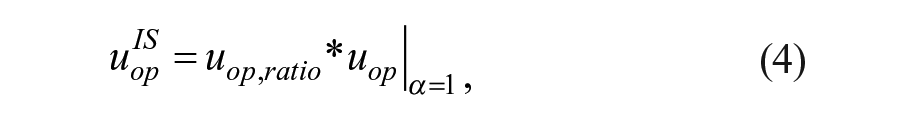
where
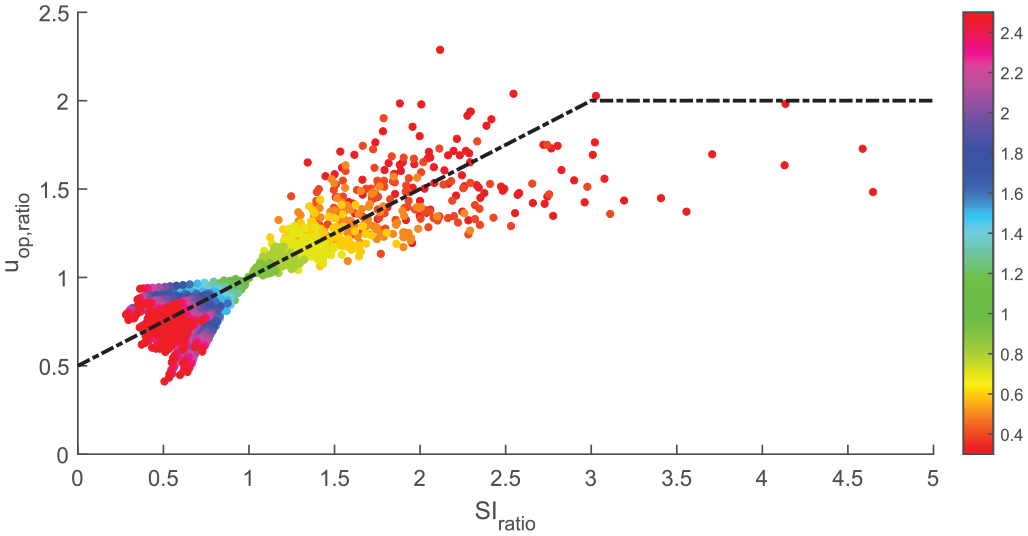
Relation with
It is observed that the modification of basal insulin needs
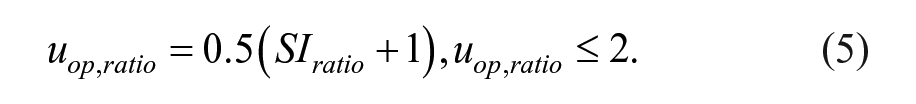
The correlation obtained for the linear definition is 0.8859 (P value < 10−4), indicating a significant, strong positive correlation. According to Equation (5), the operating point is increased as IS decreases, to compensate for the increased insulin requirements. Similarly, the operating point is decreased when insulin requirements are lower due to increased IS. However, the linear description offers a better fit for lower values of
HMS and BPS
In these modules, boluses are dependent on the subject’s TDI. For HMS, they are computed with CF estimates from the 1800-rule, that is, CF = 1800/TDI. 35 In turn, for BPS, they are computed as a percentage of the subject’s TDI. The integration of the IS estimate is thus made by computing TDI with the same scheme as the basal Operating point (OP). In this way, the TDI parameter for these modules is obtained as follows:
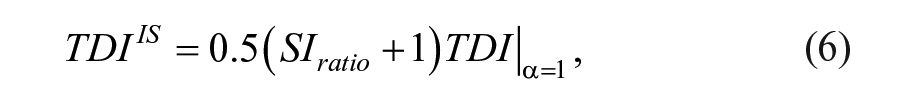
where
BC
This module is used only for HCL and requires a meal announcement. Using the estimated CHO content of the meal, boluses are computed according to the following:
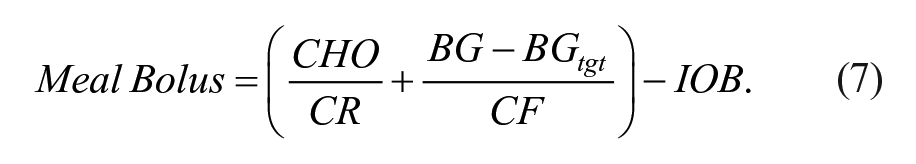
The IS-informed BC would then modify the amount obtained through
7
by a factor of
Performance Analysis of Personalized AID System
Performance of the HCL/FCL system when not informed (naïve) versus informed with changing IS levels was assessed using a novel performance metric penalizing the time outside the target glucose range. The index,
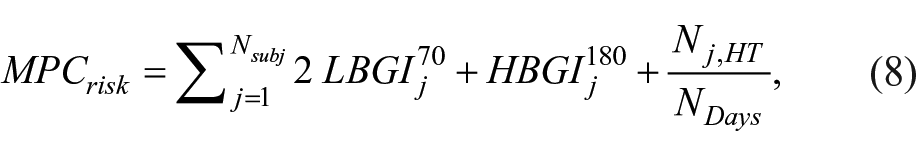
where
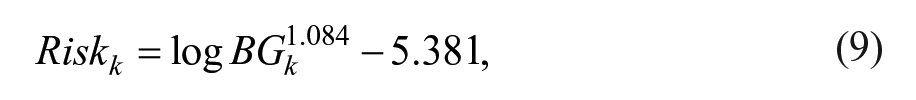
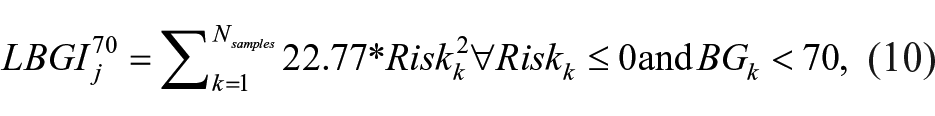
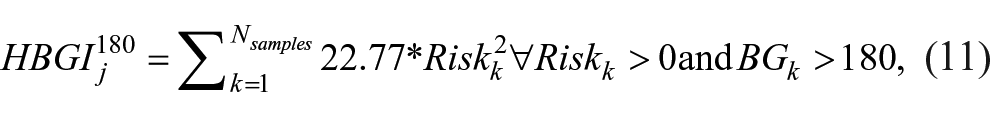
with
For this analysis, one-day simulations are performed for each value of
Three meals are provided at 7:00, 13:00, and 19:00 hours, with 80, 70, and 60 g of CHO respectively, considering that in FCL meals are unannounced. Intraday variability in IS and dawn phenomenon were not included. Hypoglycemia treatments (15 g CHO) were administered for BG lower than 60 g/dL, waiting 15 minutes before administering a new treatment, if needed.
Results
IS Estimation
Distribution of IS across the different values for
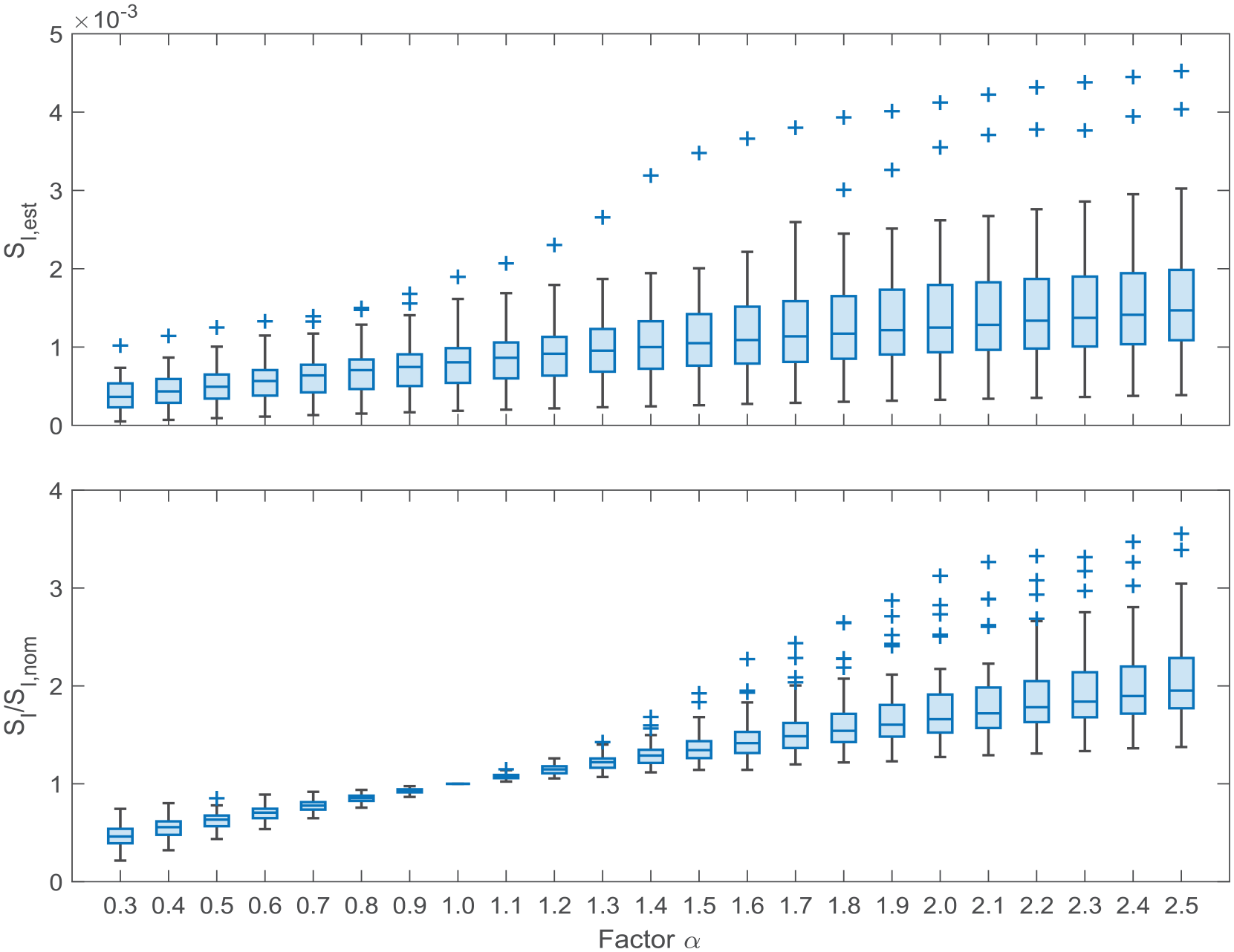
Top: Distribution of insulin sensitivity estimates across the different variation scenarios considered. Bottom: Ratio of insulin sensitivity with its value obtained at nominal conditions.
Performance Evaluation of Model and Controller Individualization
The variation of showing the effect of informing IS to the different modules of the AID system is presented in Figure 4.
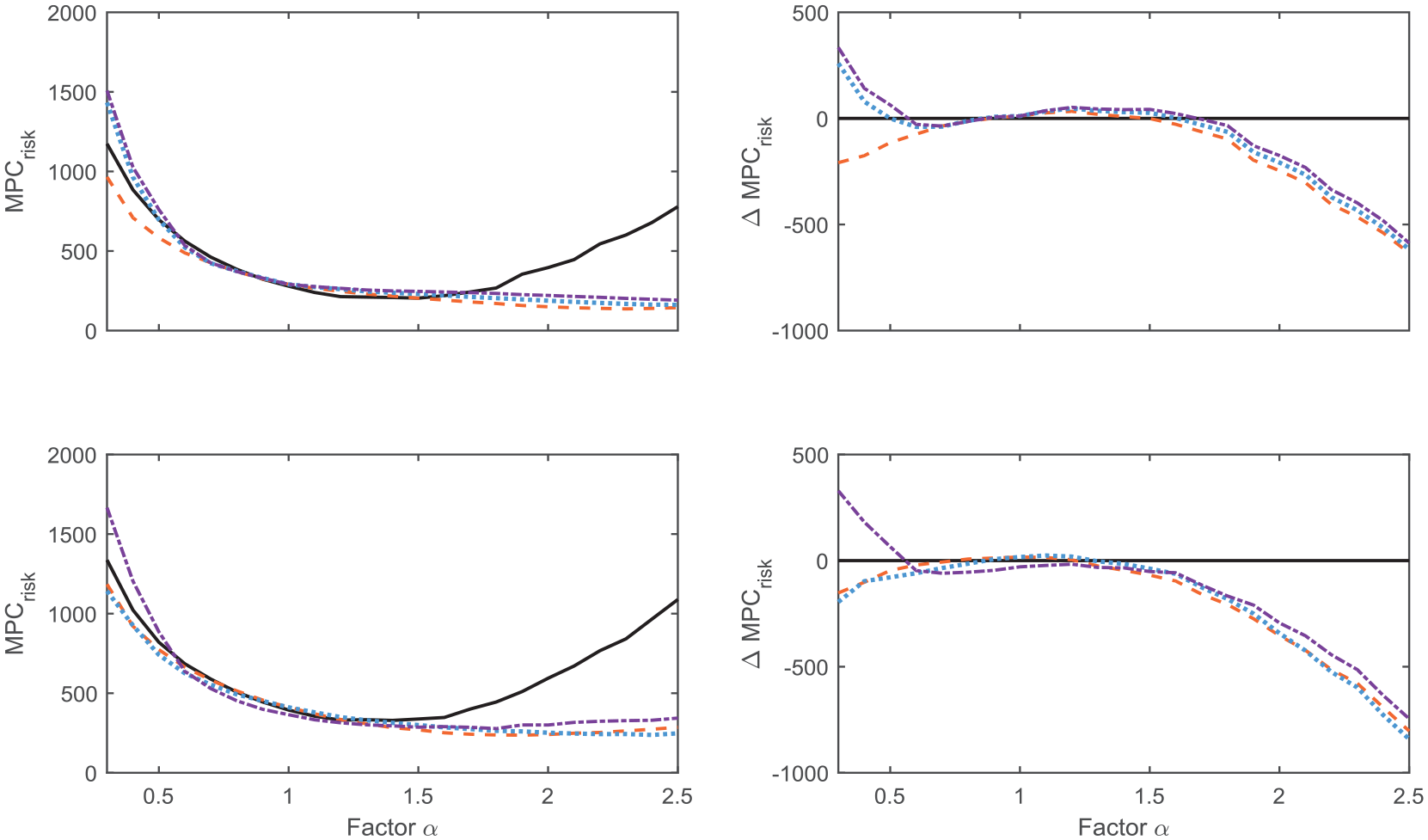
Performance index (left panels) and delta in performance with respect to the naïve controller (right panels) for the different stages of the adaptation strategy for the hybrid closed-loop (top) and fully closed-loop (bottom) systems. Solid black: Naïve (baseline) controller. Dashed orange: Model personalization with insulin sensitivity. Dotted blue: Model personalization with insulin sensitivity and individualized basal operating point. Dashed dotted purple: Model personalization with insulin sensitivity and individualized basal operating point and boluses.
The first observation is that solely informing the MPC prediction model improves the AID system’s performance, which is in line with findings that the quality of the prediction model is crucial to the performance of MPC algorithms.
39
According to this, improving the prediction model is also beneficial when using the disturbance input
Glycemic outcomes for the naïve and IS-informed HCL and FCL systems are presented in Figures 5 and 6, respectively. A comparable or increased TIR is obtained by the informed controller in all cases, indicating better average performance than the naïve controller. For the increased sensitivity cases
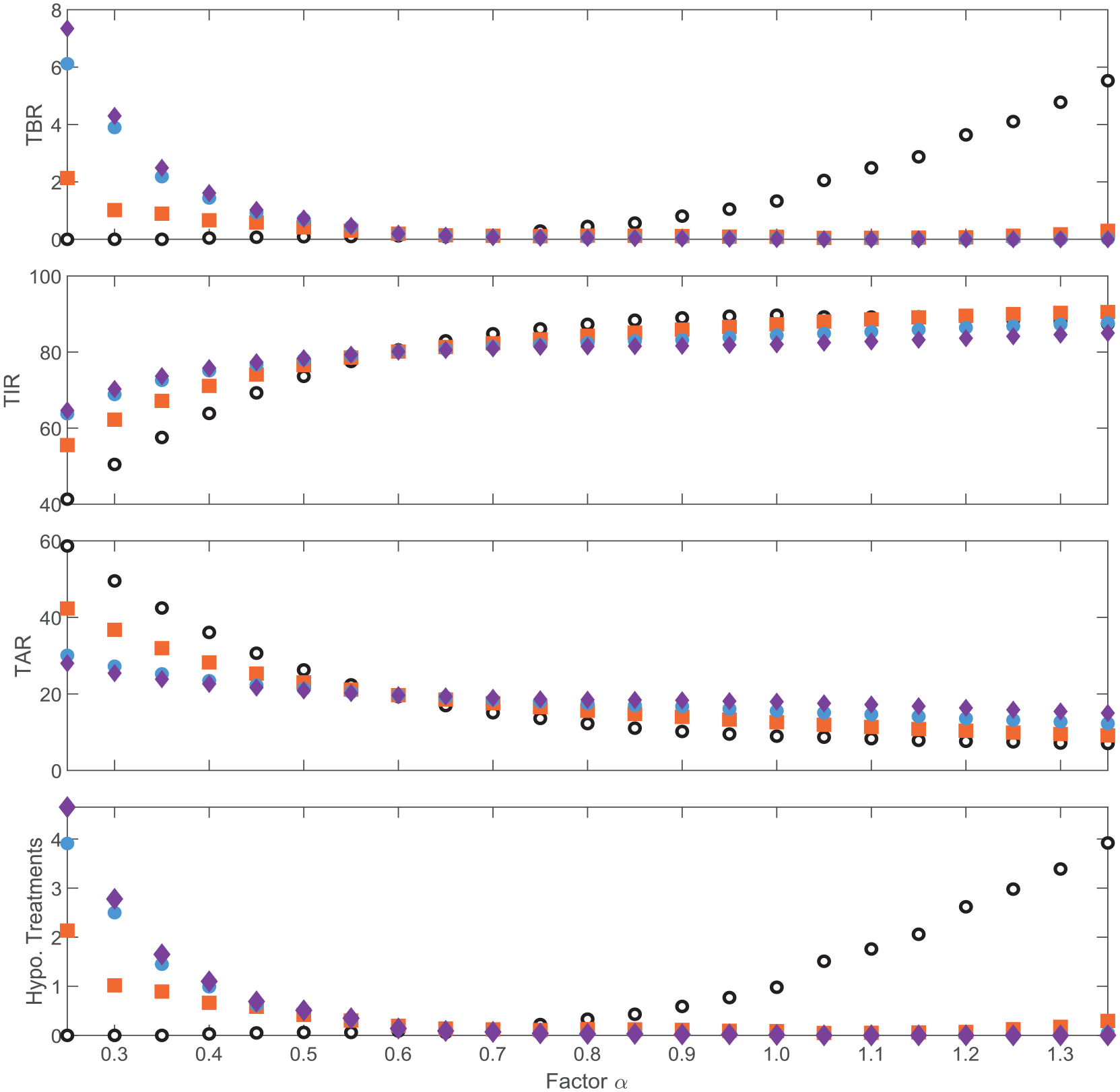
Mean outcome metrics for the hybrid closed-loop system: naïve (empty black circles), insulin sensitivity–informed model (filled orange squares), insulin sensitivity–informed model and basal operating point (blue filled circles) and insulin sensitivity–informed model, basal operating point, and boluses (purple diamonds).
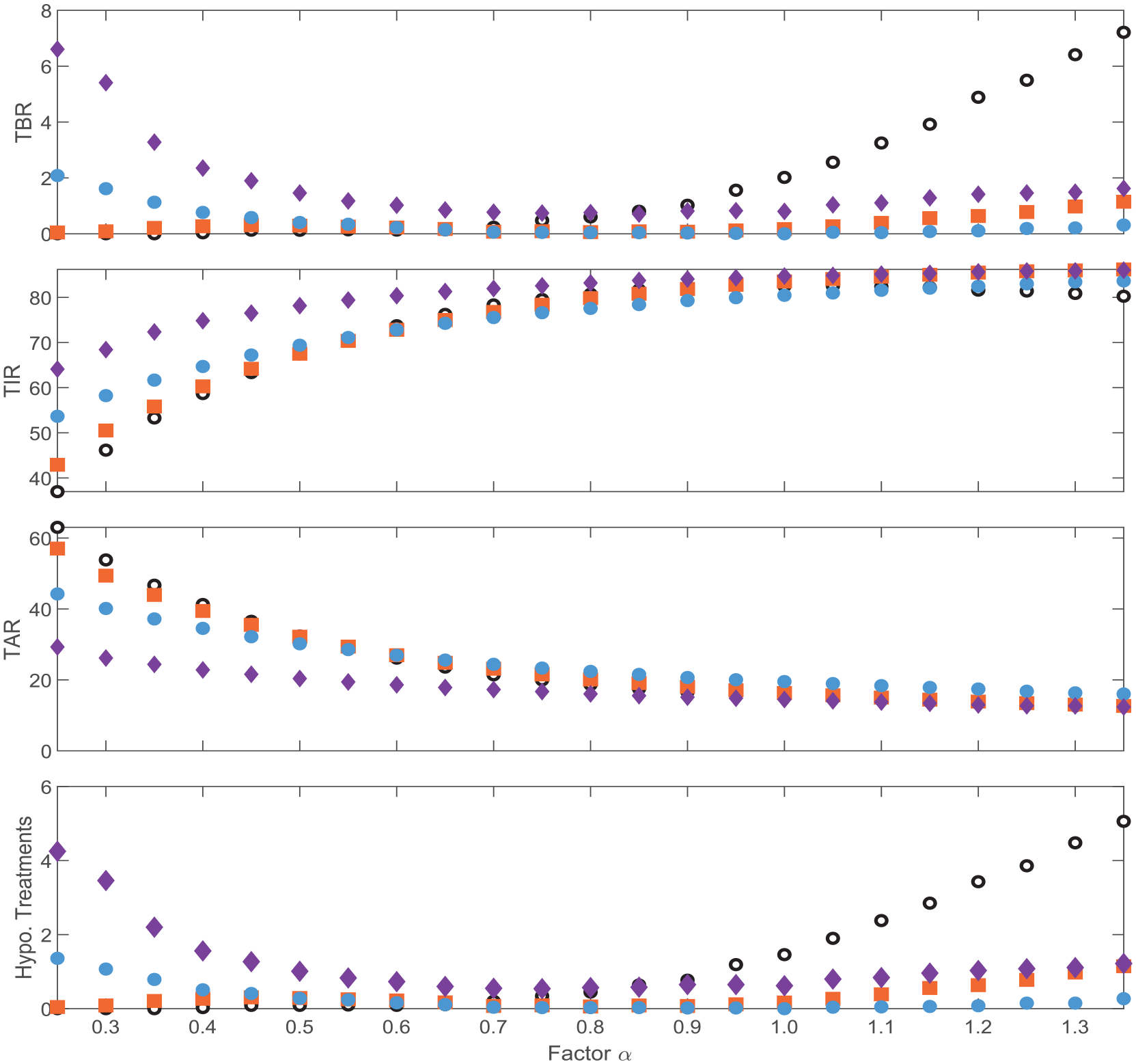
Mean outcome metrics for the hybrid closed-loop system: naïve (empty black circles), insulin sensitivity–informed model (filled orange squares), insulin sensitivity–informed model and basal operating point (blue filled circles) and insulin sensitivity–informed model, basal operating point, and boluses (purple diamonds).
Discussion
Insulin dosing is complicated due to several reasons such as user involvement to dose insulin for meals, education for carbohydrate counting to correctly dose insulin, and many other behavioral and metabolic factors that affect insulin needs. Such metabolic factors, as circadian rhythms, physical activity, psychological stress, menstrual cycle, and so on, vary greatly among the T1D population, but also among the same individual, mostly mediated by IS.
It has been shown that IS-informed bolus calculators improve outcomes for open loop and HCL therapy.1,10,40 In this work, we find evidence that this advantage might also be appreciated when using IS to update the MPC’s internal model, basal operating point, and boluses in closed-loop systems, noting that the feedback action of AID system provides a degree of robustness to increased sensitivity up to 50% in terms of hypo-protection. To be noted, this evidence was obtained without temporal changes in SI, but rather a constant value for the duration of the experiments. Further work involves assessing the effects of these system modifications within a time-varying framework and developing the method for estimating IS in real time within our FCL/HCL system. In this regard, we intend to extend the method used in Fabris et al, 1 to include the estimation of IS within the FCL/HCL AID system and optimize insulin dosing modulation while removing the necessity of having accurate meal information as an input to track IS in the FCL framework. Moreover, this integration would also require modifying the characterization of the disturbance input and adding the estimation of IS in the Kalman filter as a separate state, to track both acute (induced by physical activity, for example) and longer-term IS fluctuations (intraday and interday variations).
From the proposed experiments, it could be noted that adapting all modules of our AID system is beneficial for glucose control. When analyzing the value of the
The proposed performance index based on hypoglycemia and hyperglycemia risks allows obtaining a condensed representation of the glycemic control achieved by the system using a single index. Moreover, although it is well known that risks are good indicators of glycemic control, the number of hypoglycemic treatments was also included in the performance index to avoid unreliable TBR values, as overly aggressive controllers can provide lower TAR and TIR but at the expense of increased hypoglycemia treatments, which in turn is detrimental to the individual’s quality of life. It is worth highlighting that for more resistant cases, including the adaptation of the basal operating point did not modify the value for the
Finally, the controller’s tuning was optimized using
Conclusions
Feedback in AID systems provides a certain degree of tolerance to changes in IS. However, IS-informed systems improve glycemic outcomes, providing increased protection against hypoglycemia and hyperglycemia according to the person’s physiological state. This proof-of-concept analysis highlights the beneficial effects of integrating accurate IS estimates into AID systems. These findings underscore the importance of considering IS fluctuations in MPC-based AID algorithms, offering valuable insights for optimizing T1D management. Further research is warranted to explore real-time methods for continuously updating IS estimates and refining their integration into AID systems. Clinical trials are also needed to validate the efficacy and safety of IS-informed MPC algorithms in real-world settings.
Footnotes
Abbreviations
AID, automated insulin delivery; BG, blood glucose; BW, body weight; BC, bolus calculator; BPS, Bolus Priming System; CHO, carbohydrates; CR, correction factor; FDA, Food and Drug Administration; FCL, fully closed loop; HBGI, high blood glucose index; HCL, hybrid closed loop; HMS, Hyperglycemia Mitigation System; IS, insulin sensitivity; KF, Kalman filter; LBGI, low blood glucose index; MPC, model predictive control; SSM, safety system mechanism; SOGMM, Subcutaneous Oral Glucose Minimal Model; TAR, time spent in glycemic range above 180 mg/dL; TBR2, time spent in glycemic range above 250 mg/dL; TBR2, time spent in level 2 hypoglycemia; TIR, time spent in target glycemic range; TDB, total daily basal; TDI, total daily insulin; T1D, type 1 diabetes; UVA, University of Virginia.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.M-.V. receives research support and royalties through her institution from Dexcom. C.F. receives royalties from Dexcom and Novo Nordisk managed through her institution. M.D.B. receives research support through his institution from Tandem, Dexcom, and Novo Nordisk; M.D.B. received honorarium and travel compensation from Sanofi and Tandem; M.D.B. consults for Dexcom and Sanofi.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by Dexcom, Inc (San Diego, California) and National Institutes of Health under Grant R01-DK-129553.