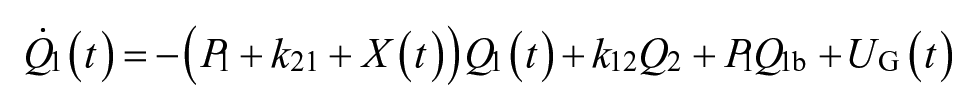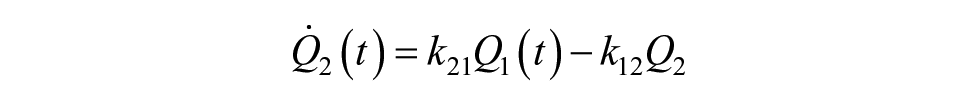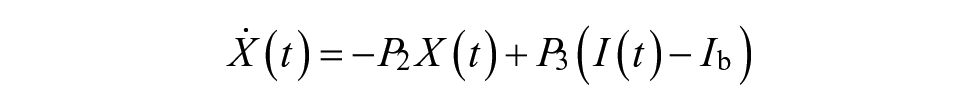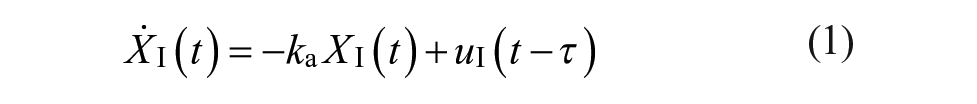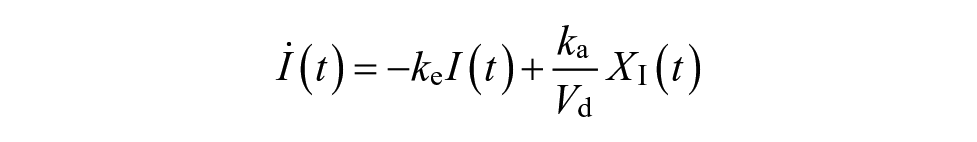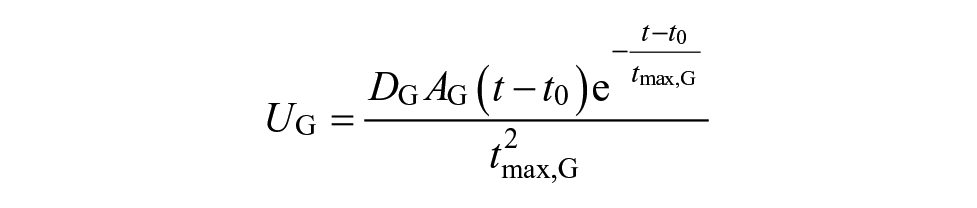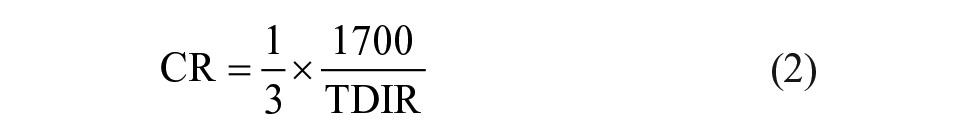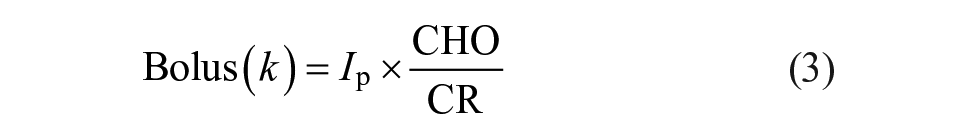Abstract
Background:
People with type 1 diabetes (T1D) have varying sensitivities to insulin and also varying responses to meals and exercise. We introduce a new adaptive run-to-run model predictive control (MPC) algorithm that can be used to help people with T1D better manage their glucose levels using an artificial pancreas (AP). The algorithm adapts to individuals’ different insulin sensitivities, glycemic response to meals, and adjustment during exercise as a continuous input during free-living conditions.
Methods:
A new insulin sensitivity adaptation (ISA) algorithm is presented that updates each patient’s insulin sensitivity during nonmeal periods to reduce the error between the actual glucose levels and the process model. We further demonstrate how an adaptive learning postprandial hypoglycemia prevention algorithm (ALPHA) presented in the previous work can complement the ISA algorithm, and the algorithm can adapt in several days. We further show that if physical activity is incorporated as a continuous input (heart rate and accelerometry), performance is improved. The contribution of this work is the description of the ISA algorithm and the evaluation of how ISA, ALPHA, and incorporation of exercise metrics as a continuous input can impact glycemic control.
Results:
Incorporating ALPHA, ISA, and physical activity into the MPC improved glycemic outcome measures. The adaptive learning postprandial hypoglycemia prevention algorithm combined with ISA significantly reduced time spent in hypoglycemia by 71.7% and the total number of rescue carbs by 67.8% to 0.37% events/day/patient. Insulin sensitivity adaptation significantly reduced model-actual mismatch by 12.2% compared to an AP without ISA. Incorporating physical activity as a continuous input modestly improved time in the range 70 to 180 mg/dL during high physical activity days from 84.4% to 84.9% and reduced the percentage time in hypoglycemia by 23.8% from 2.1% to 1.6%.
Conclusion:
Adapting postprandial insulin delivery, insulin sensitivity, and adapting to physical exercise in an MPC-based AP systems can improve glycemic outcomes.
Keywords
Introduction
Closed loop control for type 1 diabetes (T1D) also known as the artificial pancreas (AP) is an emerging control technology. The insulin delivery rate at each time interval is calculated using a control algorithm that considers current and past continuous glucose measurements (CGM). 1 A number of studies have shown that model predictive control (MPC) is an effective control strategy because it models delayed insulin kinetics.2-4 The first MPC algorithm for diabetes management was introduced by Parker et al 5 Model predictive control was further developed for single-hormone and dual-hormone APs with alternative mathematical models and different insulin delivery methods and timing intervals.6-10 Model predictive control performs optimally if the parameters of the mathematical models are defined accurately and are consistent with the characteristics of the person with T1D or plant. Model-plant mismatch may occur because the model parameters derived and used in the MPC are typically obtained from a population model or average across many patients. Model-plant mismatch may also occur if one of the parameters of the model (eg, insulin sensitivity) is estimated a priori (eg, using total daily insulin as an estimate), but this estimate happens to be inaccurate. This mismatch can degrade the performance of the controller. A number of other factors can cause the model-plant mismatch. First, the mathematical model in an MPC is inherently less complex than a human or a plant model that is designed to represent a human’s glucoregulatory system. Second, the MPC’s controller model does not have accurate knowledge of the process noise or the measurement noise. Boiroux et al6,11 incorporated the measurement noise and a time-varying filtered process noise into MPC’s mathematical model for reducing the mismatch. Furthermore, the physiology of each patient including the patient’s sensitivity to insulin or to carbohydrates may change over time, whereas the model in an MPC remains fixed, thereby creating further mismatch between the model and the plant if the model within the MPC does not adapt. One approach to improve control performance in automated insulin delivery (AID) systems is to try to identify parameters within the model to match the patient who is using the system. A natural parameter to try to match is the parameter representing the insulin sensitivity of the patient. This process of identifying a model parameter based on data collected during a test is known as system identification.12,13
In this paper, we extended the MPC developed by Resalat et al9,10 to make it adaptive and patient-specific to minimize the model-plant mismatch. Specifically, we use system identification to identify a parameter in the model that represents the patient’s sensitivity to insulin. We introduce the insulin sensitivity adaptation (ISA) algorithm, which updates the insulin sensitivity factor (ISF) within the MPC controller model at each nonmeal period. The ISF defined in the mathematical model of the MPC is used to calculate insulin amounts. If the ISF is initialized to a value that is too high for a patient, the MPC will presume that the patient needs less insulin and hyperglycemia can occur. Conversely, if the ISF is initialized to be lower than the patient’s ISF, then too much insulin will be dosed to the patient and hypoglycemia can occur. Enabling the ISF within the MPC to adapt based on the patient’s response to insulin is hypothesized to reduce the model-plant mismatch and optimize the performance of MPC.
While the ISA algorithm is designed to adapt and learn each patient’s response to insulin during nonmeal periods, it is also important to adapt insulin dosing in response to meals since each patient may respond differently to food intake. We have previously reported on an adaptive learning postprandial hypoglycemia prevention algorithm (ALPHA) that adjusts the insulin delivery after meals within a hybrid AID where meals are announced. 14 The ALPHA algorithm updates a postprandial aggressiveness factor based on prior postmeal glucose patterns that have been measured. For example, if postprandial hypoglycemia has been observed, ALPHA reduces the postprandial aggressiveness factor after future meals. The ALPHA algorithm may be used to determine the optimal postmeal insulin that should be delivered by an AID. In this paper, we show how ALPHA can be combined with the ISA algorithm to improve glycemic outcomes of an AID system.
In addition to adapting the model within the MPC to match the patient’s individual insulin sensitivity using ISA and carbohydrates using ALPHA, we have incorporated another form of adaptation whereby the MPC responds dynamically to exercise. Exercise can also be a major disturbance to a person with T1D. An AID that can respond in real-time to exercise is also critical since moderate aerobic exercise is known to cause hypoglycemia in people with T1D. 15 In the current paper, we show the benefit of including exercise as a continuous input into an AP system during free-living conditions as well as during scheduled exercise. And we show the benefit of using both ISA and ALPHA to adapt to patient-specific physiologies and improve time in target glucose ranges.
Other research teams have also used an adaptive approach to optimize the performance of MPC. Laguna Sanz et al 16 defined a trust index that indicated how closely the MPC model was able to predict prior glucose values based on the residuals across a prediction horizon. They updated the cost function of the MPC by adaptively changing an aggressiveness factor to change insulin dosing. They found that when implemented in a zone MPC, there was not a significant change in time in target range, but there was decreased time in hypoglycemia when using the adaptive algorithm. Toffanin et al 17 also introduced a run-to-run adaptive algorithm. They adaptively changed the bolus insulin and overnight basal insulin in a run-to-run design with MPC. They modified overnight basal insulin and daytime bolus insulin at each run showing increased time in the range of 11.39%. Toffanin et al adapted the insulin-to-carbohydrate ratio in response to postprandial glucose excursions. We have shown previously 14 that adapting postprandial insulin is more effective than adapting carbohydrate ratios when using the ALPHA algorithm.
Other work has been done to adapt the AP to exercise. Turksoy and Cinar showed that using an autoregressive model-based controller, exercise could be included as an input to the controller.18,19 Breton et al showed that incorporating heart rate into an AP, the rate of decline of glucose during exercise could be reduced. 20 Our group has also shown that incorporating automated exercise detection into both a single-hormone and a dual-hormone AP can help reduce time in hypoglycemia.21,22 For these prior studies, however, exercise levels above four metabolic equivalent of task (METs) were detected and an exercise adjustment algorithm was executed that turned off insulin and increased glucagon, but only during and shortly after the detected exercise event. 23
The primary contributions of this paper include the following. First, we introduce an ISA algorithm that may be used to identify an individual patient’s insulin sensitivity parameter within an MPC model during nonmeal periods. Second, we show how ISA and the previously published ALPHA algorithm can be used to adapt to both nonmeal and postprandial periods, respectively, and thereby improve glycemic performance. Third, we show how exercise metrics can be incorporated into a model within an MPC to enable the MPC to adapt to real-time exercise. We demonstrate improved performance in glycemic outcomes using real-world heart rate, accelerometry, and meal data to demonstrate how glycemic outcomes may be improved if exercise metrics are incorporated as continuous inputs into an MPC AID system.
Material and Methods
Controller Design
The schematic of the MPC used in this study is shown in Figure 1. The MPC used in this paper is a hybrid MPC which requires the user to announce meals to the system. The MPC consists of a process model (i.e. mathematical model), a reference trajectory, and an optimization tool to calculate the next insulin delivery rate. The process model is a mathematical description of glucose and insulin metabolism. It is defined by five differential equations in this study and is further described in Resalat et al. 10 The ISA algorithm updates the ISF of the process model in the MPC during nonmeal periods. A nonmeal period is defined as a minimum of three hours over which there are no meals or exercise events. The nonmeal periods are oftentimes during the night when the patient is sleeping. The ALPHA algorithm adapts postprandial insulin to help prevent meal-based excursions. The ALPHA algorithm can be used with any type of control algorithm, not just MPC. It only uses glycemic excursion information from prior meal events to adjust the postprandial insulin delivery using an adaptive aggressiveness factor. We used 99 statistically sampled virtual patients from the OHSU virtual patient population to represent patients with T1D (ie, the plant).9,24 The virtual patient population was generated based on a glucoregulatory model consisting of insulin kinetics and dynamics models and a glucose kinetics model. The parameters of the insulin dynamics model were statistically sampled to build a virtual population with different insulin sensitivities. As provided in Resalat et al, 25 the average total daily insulin requirement for these virtual patients of 44.3 closely matched a clinical data set that was used to generate the virtual patients. In general, the complexity of the process model should be less than the plant for better representing real-world control scenarios whereby the human is substantially more complex than the MPC process model. The plant in the OHSU virtual patient population is represented by eight differential equations, making it more complex than the MPC process model.
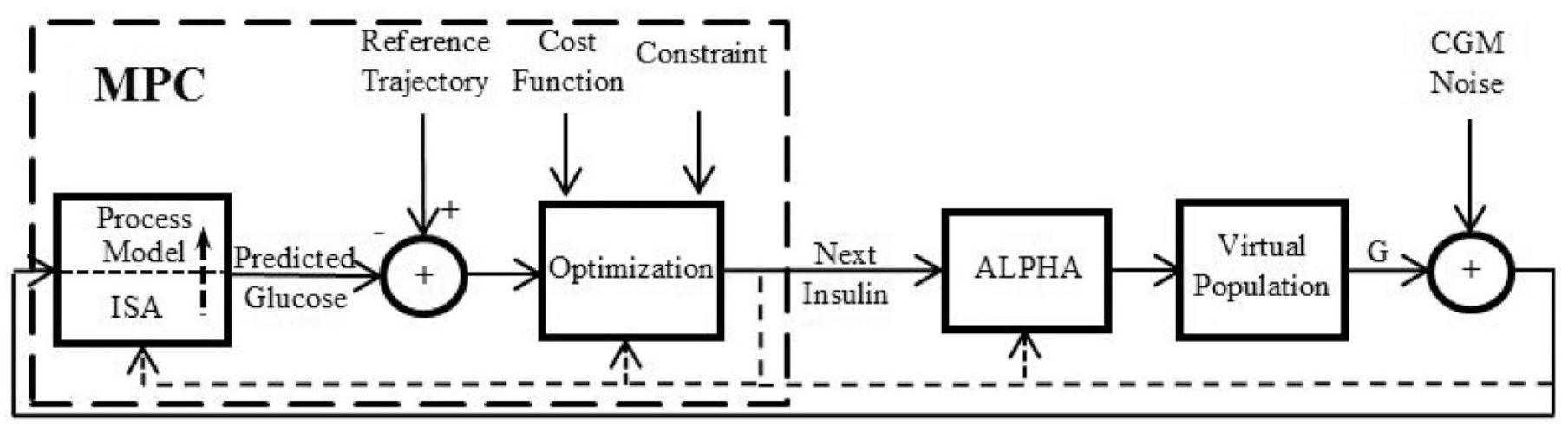
The schematic of the model predictive control. Adaptive learning postprandial hypoglycemia prevention algorithm modifies basal insulin during meal periods. Insulin sensitivity adaptation modifies basal insulin by updating insulin sensitivity factor of the process model during nonmeal periods.
The process model of the MPC consists of an insulin kinetics model, 26 an insulin dynamics model, and a glucose kinetics model 27 as described in Resalat et al. 10 It is presented with the following equations:
where Q1 and Q2 are glucose masses in accessible and nonaccessible compartments, respectively (mg/kg). The Q1 compartment is the glucose in plasma where it can be measured (i.e. accessible), while the Q2 compartment is the glucose in tissue where it cannot be measured (i.e. nonaccessible). P1 is the glucose effectiveness (min−1). X is the effect of insulin on blood glucose (min−1). P2 represents the decay rate of X (min−1). P3 shows the effective rate of insulin in plasma (min−2 per mU/L). The ratio between P3 (min−2 per mU/L) and P2 (min−1) represents the ISF. 10 Q1b and Ib are basal plasma glucose and insulin, respectively. k12 and k21 (min−1) are rate parameters describing glucose exchange kinetics, respectively. 27 XI(t) is the amount of insulin in the subcutaneous depot (mU/kg), I(t) is the plasma insulin concentration (mU/L), and u(t) is subcutaneous infused insulin (mU/kg/min). ke is the elimination rate of insulin (min−1), ka is the absorption rate of insulin (min−1), Vd is the insulin volume of distribution (L/kg), and τ is the time delay for injected insulin to be effective in the interstitial fluid (min), which was set to zero. 10 UG represents the glucose absorption rate from meals (mg/kg/min). tmax,G (min) is the time-to-maximum appearance rate of glucose in Q1, AG is the carbohydrate bioavailability (unitless), t0 is the meal announcement time (min), and DG is the estimated carbohydrate intake (mg/kg). DG is converted from grams to milligrams per kilograms to be compatible with Q1 in the glucose kinetics model.
The process model determines the predicted glucose levels over the prediction horizon (NP) which are compared with the reference trajectory. See supplemental Figure S1 to see an example of how the MPC model predicts glucose across the 300-minute prediction window. For glucose levels greater than the target value (Gt = 115 mg/dL), the reference glucose trajectory linearly approaches the target, while for glucose levels less than the target, they exponentially approach the target as shown in Resalat et al. 10 The time constant of the exponential term is comparably low to shut off insulin faster for low glucose levels. The constraint of basal delivery was set to 80 units/hour, enabling more aggressive basal control for high glucose levels.
Incorporating Exercise into the Model
We have previously described how we incorporate an exercise model into the MPC.10,25 This is briefly described in the Supplemental Material.
Identifying the Insulin Sensitivity During Nonmeal Periods
The insulin sensitivity factor defined as ISF =
Adapting Postprandial Insulin
We have previously developed ALPHA that can be used to adapt postprandial insulin dosing to improve glycemic control. ALPHA modifies the aggressiveness of the postprandial insulin delivery as follows. If postprandial glucose levels fall below 90 mg/dL, postprandial insulin delivery after the subsequent meal is reduced proportional to the difference between the minimum glucose level and 70 mg/dL. If glucose levels drop below 70 mg/dL, ALPHA shuts off postprandial insulin delivery. ALPHA modifies insulin delivery for 90 minutes after the meal announcement. ALPHA is further described in Resalat et al. 14
Evaluating ISA and ALPHA Under Real-World Meal Scenarios
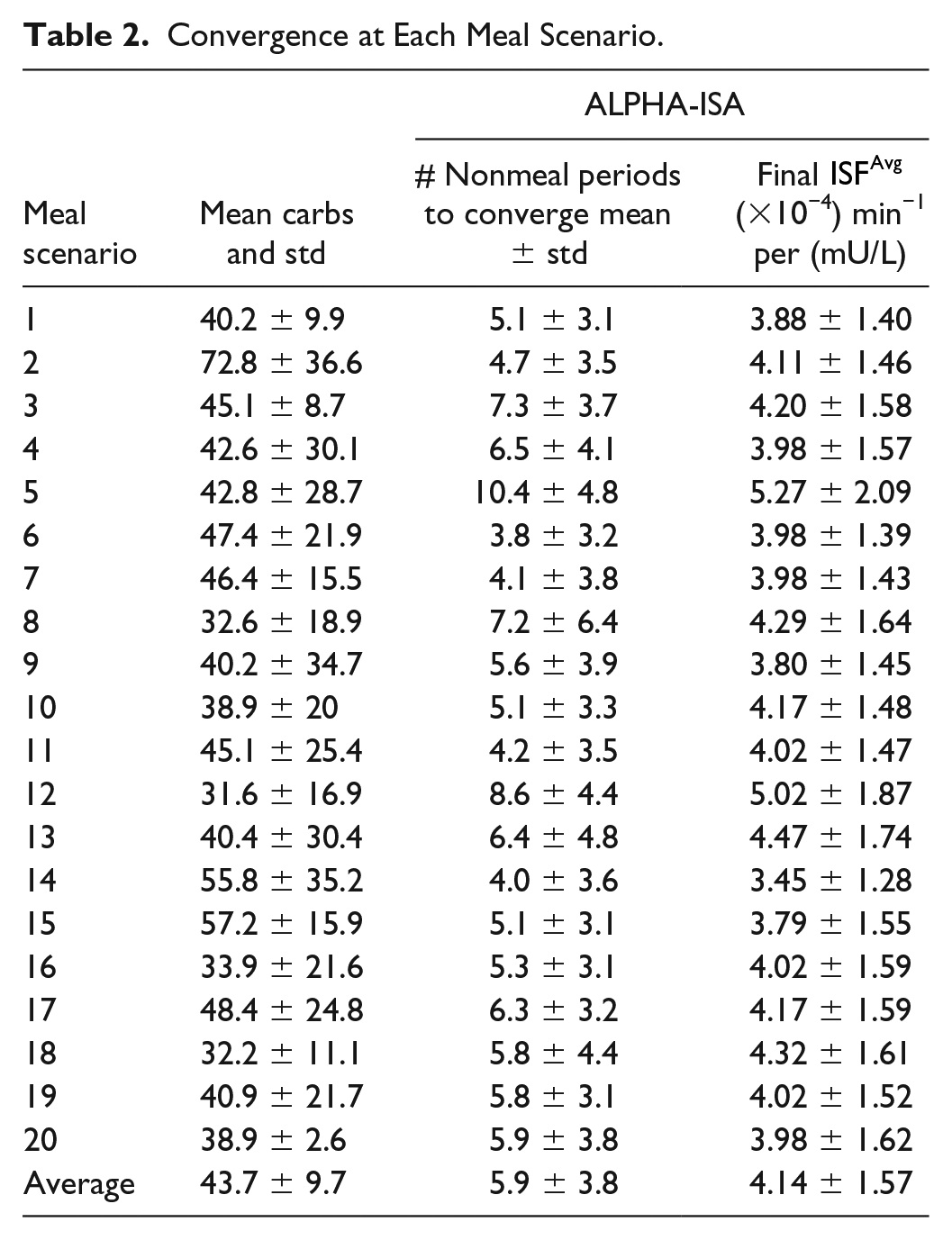
We used twenty meal scenarios from a four-day outpatient AP study. 21 Each meal scenario was extended to 28 days by replicating the original meal scenarios. This was done to investigate the convergence rate of the ISF in the ISA. Then, each meal scenario was given to the virtual patients who were then subjected to the following conditions: (1) the OHSU MPC controller (called AP), (2) the AP + ISA (called ISA), (3) the AP + ALPHA (called ALPHA), and (4) AP + ALPHA + ISA (called ALPHA-ISA). Since we used a hybrid AP whereby the meals were announced, we now describe how the meal boluses were calculated. The premeal bolus prior to adaptation by ALPHA is a function of the carbohydrate amount (CHO), the carbohydrate ratio (CR), and the percentage of the insulin bolus given prior to the meal (Ip). The CR is defined as 28
And the premeal bolus is calculated as
Since we know that people with T1D oftentimes do not correctly estimate their carbohydrate intake correctly, we challenged the simulations by introducing a randomly selected ±30% meal uncertainty that was applied to each carbohydrate intake as done by other groups. 29 We introduced ±30% circadian variability of ISFs for each virtual patient to represent intraday variability of the insulin sensitivity. 29 And finally, since each patient’s initial insulin sensitivity factor (ISF = P3/P2) was determined based on their total daily insulin requirement, we incorporated a ±30% variability of the initial total daily insulin requirement estimate to challenge the ISA algorithm.
Evaluating Physical Activity as a Continuous Input Using Real-World Exercise Data
We used the same four-day real-world meal and exercise scenarios acquired from a prior AP study as described above 21 to evaluate physical activity as a continuous input to the AP system. During this study, participants continuously wore a Zephyr patch that acquired heart rate and accelerometry data. We converted these data to METs using a method previously described by Zakeri et al. 30 And METs were used as an input to the AP as described further in the Supplemental Materials. Exercise (especially aerobic exercise) is known to relate to substantial drops in glucose and increases in hypoglycemia as has been reported in the past.21,22,31 In these past studies, we showed how heart rate and accelerometry can be used as metrics to estimate exercise that then relates to drops in glucose. Using the physical activity and meal data acquired from these study participants with T1D, we evaluated the performance of the exercise-enabled MPC algorithm compared with the nonexercise-enabled MPC algorithm during the entire study duration that included free-living, nonstructured exercise periods of time and also during structured exercise periods of time (ie, start of exercise until four hours after the exercise or until first meal). The structured exercise that was done by participants in this study was moderate intensity (60% of VO2 maximum) aerobic exercise, done for 45 minutes on a treadmill. This structured exercise was done in the clinic on day 1 and day 4 of the study. Participants were also instructed to perform moderate intensity exercise on their own at home during day 3 of the study. The participants were instructed to wear the Zephyr patch the entire time that they were awake and to take it off to recharge the battery at night while they slept.
Evaluation Metrics and Statistical Analysis
We evaluated percent time in target range (70-180 mg/dL) and percent time in hypoglycemia (<70 mg/dL) as the primary outcome measures. Secondary outcome measures were percent time in hyperglycemia (>180 mg/dL), root mean square error (RMSE) of the MPC model predicted output relative to the plant output across the 300-minute prediction horizon, total number of rescue carbohydrates required per day, low blood glucose index (LBGI), and high blood glucose index (HBGI). The paired statistical two-sample t-test was used to test statistical difference among AP, ISA, ALPHA, and ALPHA-ISA with significance level set to 0.05. A paired statistical two-sample t-test was also used to test the difference between the AP and the AP + exercise.
Results
Adapting Postprandial Insulin and ISF
We found overall that the ALPHA algorithm was effective at reducing time in hypoglycemia by 42.1% relative to the AP without ALPHA as shown in Table 1. However, this was at the expense of also reducing time in range from 81.5% to 79.2%. By including the ISA algorithm in combination with ALPHA, we were able to reduce the time in hypoglycemia with less impact on time in range. As shown in Table 1, the number of rescue carbohydrates per day was reduced by 67.8% when using ALPHA-ISA (0.37/day) compared with AP (1.15/day). Time in range changed from 81.5% (AP) to 79.4% (ALPHA-ISA).
Comparison Among Artificial Pancreas, Insulin Sensitivity Adaptation, Adaptive Learning Postprandial Hypoglycemia Prevention Algorithm, and ALPHA-ISA Over the Meal Scenarios.
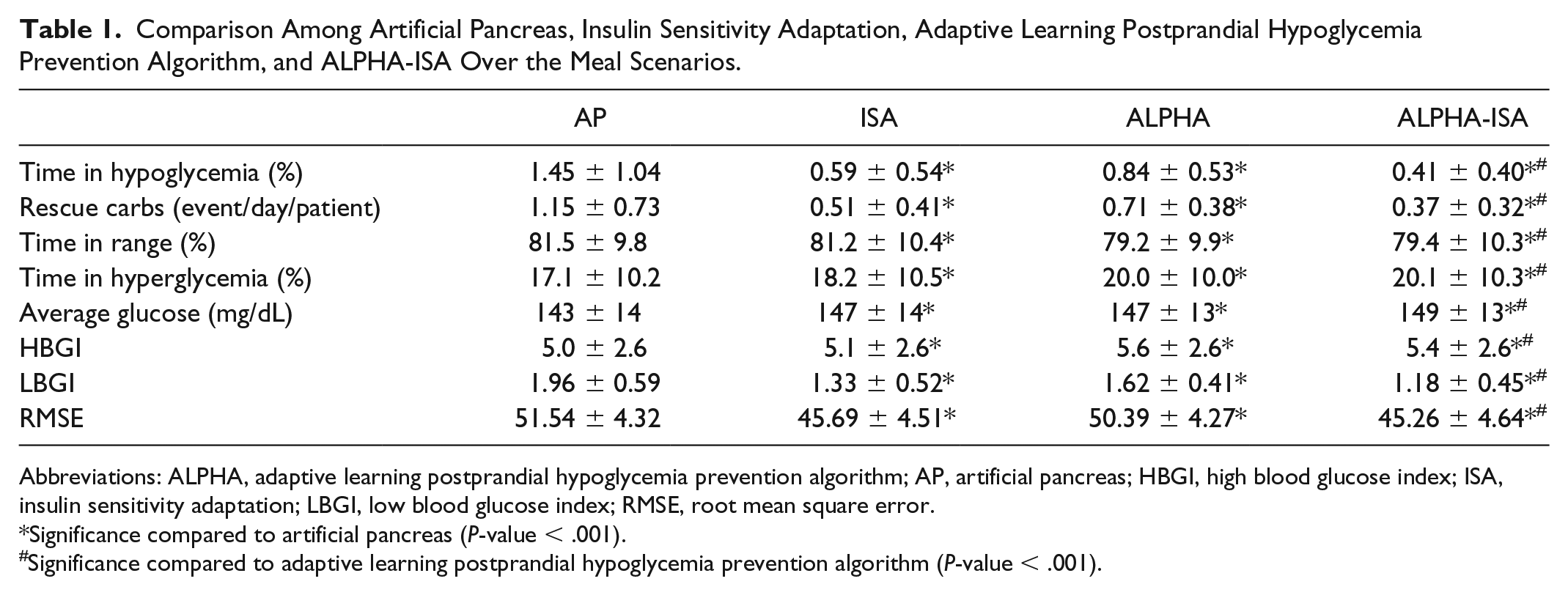
Abbreviations: ALPHA, adaptive learning postprandial hypoglycemia prevention algorithm; AP, artificial pancreas; HBGI, high blood glucose index; ISA, insulin sensitivity adaptation; LBGI, low blood glucose index; RMSE, root mean square error.
Significance compared to artificial pancreas (P-value < .001).
Significance compared to adaptive learning postprandial hypoglycemia prevention algorithm (P-value < .001).
The ISA algorithm was effective at reducing the model-plant mismatch, specifically during the overnight periods when the insulin sensitivity was adapted to each patient. Figure 2 shows how the ISA algorithm reduces model-plant mismatch during nonmeal periods. The RMSE values across all meal scenarios and subjects are shown in Table 1. It also shows the comparison among AP, ISA, ALPHA, and ALPHA-ISA over the virtual patients and meal scenarios for the entire experiment including meal and nonmeal periods. ALPHA-ISA significantly reduced the RMSE during meal and nonmeal periods from 51.5 mg/dL (AP) to 45.3 mg/dL (ALPHA-ISA) as shown in Table 1. Supplemental Figure S5 shows how the error increased for predictions further into the future. This figure also shows that ISA yielded a significant reduction in RMSE that was consistent across the prediction horizon.
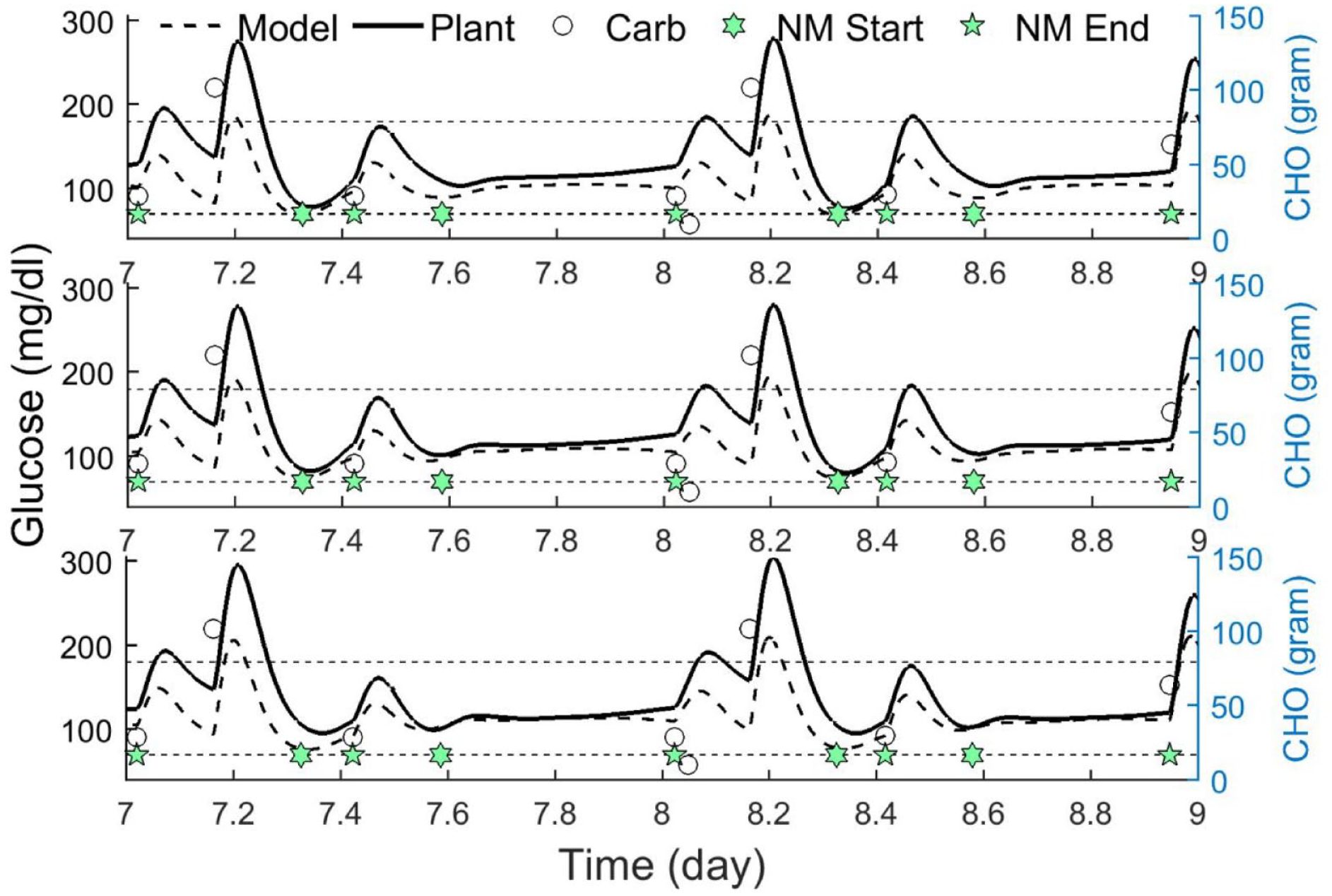
Performance of artificial pancreas (top), insulin sensitivity adaptation (middle), and ALPHA-ISA (bottom) for a representative subject in a representative meal scenario. Start time (NM start) and end time (NM end) of the nonmeal periods are shown with star-pentagon and star symbols, respectively.
Figure 3 compares the performance of AP with ALPHA-ISA over the virtual patients for one representative meal scenario (meal scenario 17). The lower panel shows the adaptation of
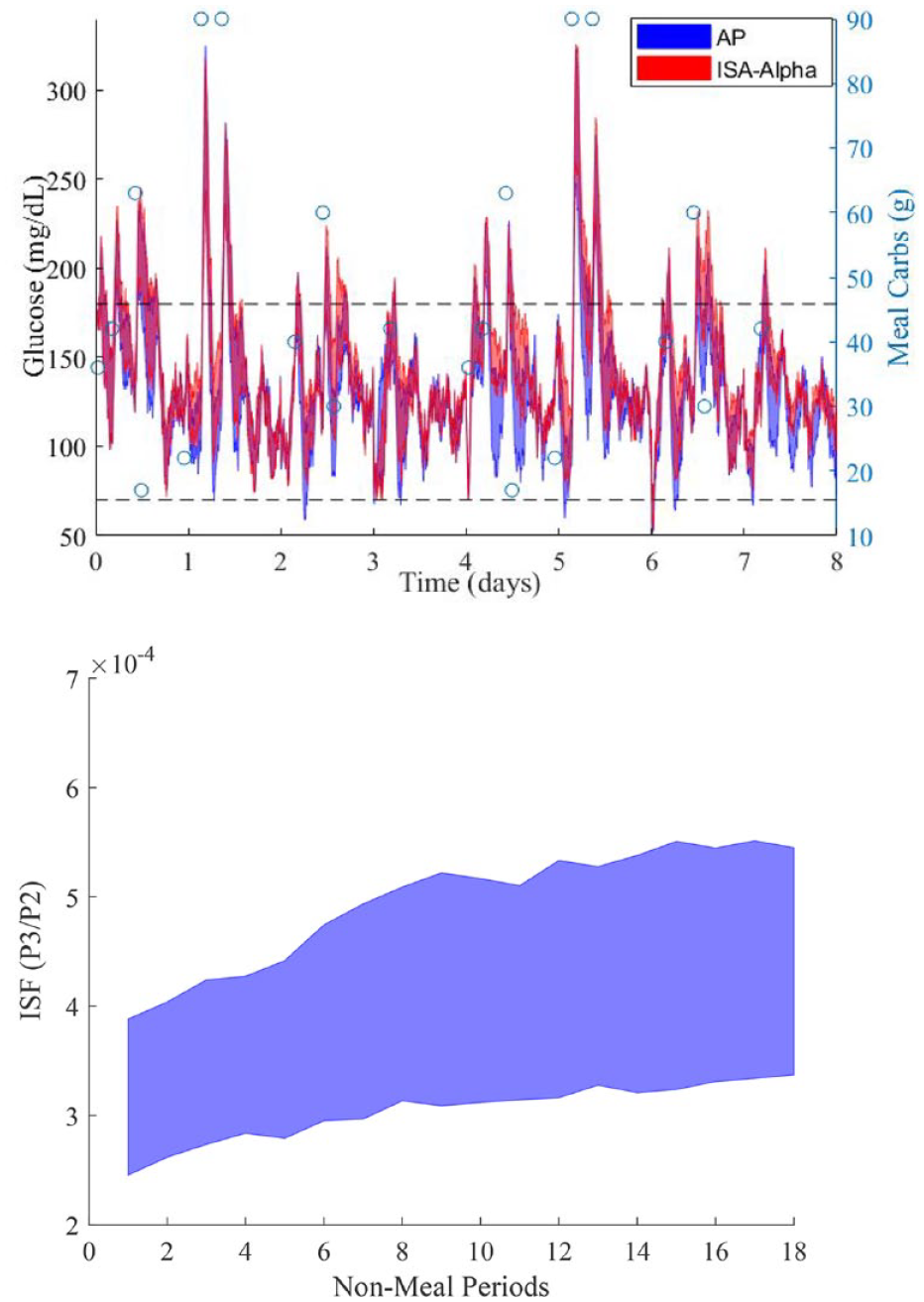
The top panel shows the interquartile range of the glucose profile across all subjects for meal scenario 17. “o” denotes the amount of carbohydrates consumed. Notice that insulin sensitivity adaptation helps prevent several hypoglycemia events as time goes by and adaptation takes place. The bottom panel shows the interquartile range of the ISFAvg over the nonmeal periods across the virtual patients also for meal scenario 17. Dashed lines represent hypoglycemia and hyperglycemia thresholds.
Convergence at Each Meal Scenario.
Results of Adapting to Exercise
Including exercise as a continuous input to the AP controller led to a 23.8% reduction in time in hypoglycemia and 25% reduction of number of rescue carbohydrates and led to a small increase of time in target range (84.4%-84.9%) as shown in Table 3. The amount of insulin used when exercise metrics were included as inputs was slightly lower at 42.9 units/day compared with when exercise metrics were not included (43.3). These results indicate that including exercise as a continuous input to the AP system may result in better glycemic outcome measures.
Comparison Between an Exercise-Enabled (AP-Ex) and a Nonexercise Enabled (AP).
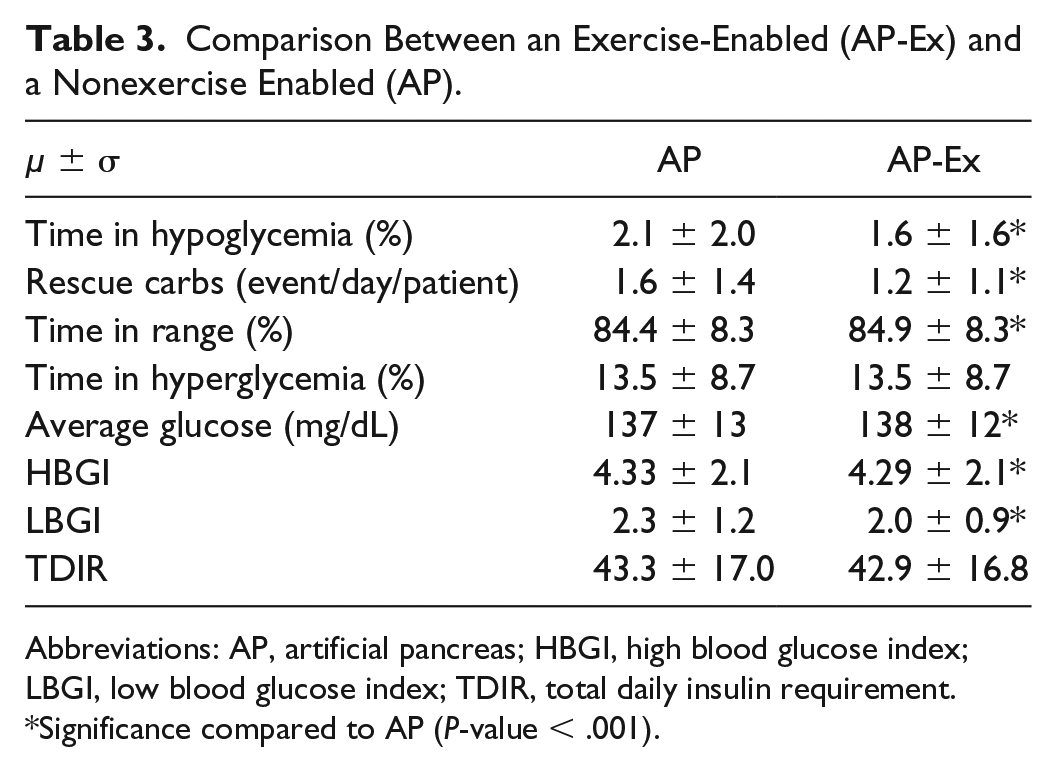
Abbreviations: AP, artificial pancreas; HBGI, high blood glucose index; LBGI, low blood glucose index; TDIR, total daily insulin requirement.
Significance compared to AP (P-value < .001).
Discussion
In this paper, we have demonstrated how adaptation of postprandial insulin, adaption of insulin sensitivity, and adaptation to physical activity and exercise as an input into an AP can yield improvements in glycemic outcomes in automated insulin delivery. As we also showed in a prior publication, 14 incorporating adaptive postprandial insulin delivery using ALPHA can reduce hypoglycemia and the need for rescue carbohydrates, but at the cost of reducing time in target range. By simultaneously incorporating an adaptive insulin sensitivity measure that adapts to each patient during nonmeal periods, we can still observe a substantial reduction in time in hypoglycemia while minimizing the reduction of time in target range.
While prior studies have shown a benefit of incorporating physical activity measures as an input to AP systems,9,18-22 there has not yet been a study showing how physical activity incorporated as a metric during free-living (ie, nonscheduled exercise) can yield improvements in glycemic outcomes compared with not including physical activity. These prior studies have shown that if insulin can be shut off early and also that optionally glucagon can be given in response to or in anticipation of exercise, exercise-induced hypoglycemia can be avoided. We were able to show that physical activity metrics (heart rate and accelerometry) may be incorporated as an additional input under free-living as well as scheduled exercise periods of time to yield improvements in glycemic control including moderate but statistically significant reduction of time in hypoglycemia, number of rescue carbohydrates, increased time in target range, reduction of time in hyperglycemia, and improvements in LBGI and HBGI. In the future, we plan to evaluate these adaptive control algorithms in a clinical study on people with T1D over long-term in-home studies to demonstrate the benefit of adaptation over time.
The ISA and ALPHA algorithms are run-to-run control approaches, meaning that there is adaptation after prescribed intervals of time. The algorithms used here are similar to the ones proposed by Toffanin et al. 17 There are several differences between the algorithms. First, in Toffanin et al, the premeal insulin was adapted by changing the carbohydrate ratio in response to postprandial periods of time. The ALPHA algorithm presented in the current paper instead adjusts the postprandial insulin based on prior postprandial glycemic excursions. We showed in Resalat et al 14 that adjusting the postprandial insulin was more effective at preventing postprandial hypoglycemia compared with adjusting the premeal insulin. Another difference between these two run-to-run algorithms is that Toffanin et al adapted an aggressiveness factor during the nonmeal periods, while in the current paper, we solved for the patient’s individual insulin sensitivity to reduce plant/model mismatch. Both algorithms have shown to be beneficial. In the Toffanin et al paper, time in range was substantially increased by 11.39% and no impact on hypoglycemia (since they observed very low hypoglycemia without adaptation). The ISA + ALPHA algorithm primarily showed a 71.7% reduction in time in hypoglycemia with a small reduction of time in range. While both papers imposed a similar variability in meal and insulin sensitivity during the experiments, it is not possible to do a fair comparison between the algorithms because (1) we used different virtual patient populations and (2) the real-world meal scenarios that we used in our work were different than the simulated meal scenarios used by Toffanin et al.
Other groups19,20 and our group22-23 have also described how to integrate exercise into an AP. Some of these such as Breton et al 20 and Jacobs et al 23 used fixed rules to determine when to shut down or shut off insulin based on exercise metrics such as heart rate. Others such as Turksoy et al, 19 Hajizadeh et al, 32 and the algorithm presented in the current paper use exercise as a continuous input. The Cinar group18,32 has taken a different approach with their multivariable MPC algorithm that adapts the model at every time point whereby exercise is one of many variables input into the model. No one has yet shown data indicating that an AP that adjusts for exercise using continuously collected physical activity data should perform better than an AP that does not include these data as additional inputs. Results presented in the current paper indicate that there is a small but significant improvement in time spent in target range and reduction in hypoglycemia when utilizing physical activity data as a continuous input to an AP compared with when the physical data are not included as shown in Table 3. Future clinical studies that we are planning may prove whether this is a beneficial approach to closed loop control.
In this paper, we have looked at how adapting insulin sensitivity and response to meals can work in tandem within an adaptive environment. And we have shown how incorporating exercise metrics as a continuous input can provide modest benefit in glucose control compared with not including exercise metrics. We have not yet shown how ISA, ALPHA, and continuous exercise perform together; however, our expectation is that they will complement each other. And we plan to evaluate this in the future within a clinical study. Insulin sensitivity adaptation and ALPHA have been designed currently to not adapt if exercise is detected during a nonmeal period or a postmeal period, respectively. In this way, the exercise event will not confound adaptations in insulin sensitivity or postprandial glucose response.
The primary limitation of this study is that it was done in silico. We purposely designed the MPC control model to be less complex than the plant model in the virtual patient population, making it more challenging for the MPC to adapt over time to match the plant. However, the human body is substantially more complex than the plant model in our virtual patient population. When adaptation is done in actual humans, the insulin sensitivity parameter in the MPC will adapt to account for all discrepancies between the human physiology and the MPC model. A further limitation inherent in simulation-based studies is that the statistical outcomes (ie, P-values) may be misleading. White et al showed that looking at P-values to determine significance within simulation studies can be misleading because P-values are determined by statistical power which “can be arbitrarily high in a simulation context, producing miniscule P-values regardless of the effect size”. 33 In this paper, we did not use an excessively large number of virtual patients in order to achieve a low P-value, rather a total of 99 virtual subjects was used. Furthermore, in addition to showing the P-value, we report on the effect size (ie, improvement in glycemic outcomes including time in range and time in hypoglycemia). A final limitation is that the model for exercise in the plant model is similar to the model for exercise in the MPC controller, and the model was designed for aerobic exercise. In the future, we will need to incorporate models for other types of exercise including resistance training and moderate/high intensity interval training.
Conclusion
This paper showed that incorporating exercise as a continuous input as well as adapting insulin sensitivity and postprandial insulin delivery in a MPC design can be helpful for improving glycemic outcomes. Implementing an adaptive algorithm that can learn each patient’s insulin sensitivity over time can help reduce the mismatch between the model within an MPC and an actual patient’s glucoregulatory system. This can then result in improved glycemic outcomes, specifically reduced time in hypoglycemia. Combining algorithms that adapt to insulin sensitivity and postprandial basal insulin delivery can further improve glycemic outcomes. And we have shown that integrating exercise metrics including heart rate and accelerometry data as continuous inputs into an AID system can also reduce hypoglycemia. Further testing is needed to verify these results in a clinical study.
Supplemental Material
Supplemental_Material_FINAL – Supplemental material for Adaptive Control of an Artificial Pancreas Using Model Identification, Adaptive Postprandial Insulin Delivery, and Heart Rate and Accelerometry as Control Inputs
Supplemental material, Supplemental_Material_FINAL for Adaptive Control of an Artificial Pancreas Using Model Identification, Adaptive Postprandial Insulin Delivery, and Heart Rate and Accelerometry as Control Inputs by Navid Resalat, Wade Hilts, Joseph El Youssef, Nichole Tyler, Jessica R. Castle and Peter G. Jacobs in Journal of Diabetes Science and Technology
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The guarantor of this research is Peter G. Jacobs who takes responsibility for the contents of this article. NR, WH, and PGJ developed the MPC design, the study design, wrote the manuscript, and supported the data collections. WH contributed to the tuning of the MPC model parameters and did the exercise analysis. NT and WH contributed to the in silico simulations. JRC and JEY contributed to the writing, study design, data collection, data analysis, and data interpretation. JRC and PGJ have a financial interest in Pacific Diabetes Technologies Inc., a company that may have a commercial interest in the results of this research and technology.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Institutes of Health (grant 1DP3DK101044), the JDRF (grant 2-SRA-2017-502-M-B), and by Oregon Clinical and Translational Research Institute UL1TR002369 from the National Center for Advancing Translational Sciences at the NIH.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.