Abstract

Glycated hemoglobin A (HbA1c) is an established tool for diagnosis, therapy monitoring, and prognosis assessment of diabetes mellitus. Therefore, a reliable and accurate HbA1c analysis is crucial. Various measurement methods are available for determining HbA1c. 1
The ion-exchange high-performance liquid chromatography (HPLC) is one of the most common methods in use. In addition to the HbA1c concentration, hemoglobinopathies can be detected. The enzymatic assay is a relatively new method for detecting HbA1c. 2 The advantages are in the direct automatic processing of whole blood patient samples and better cost-effectiveness. The disadvantage is that only a pure HbA1c concentration is determined photometrically and the Hb fractions are not separated and possible interferences (eg, hemoglobinopathies) cannot be recorded.
Abbott’s enzymatic assay has shown good traceability, precision, and comparability with gold standard methods such as HPLC when measured on the Abbott ARCHITECT device.2,3
The aim of this study was to evaluate for the first time the enzymatic HbA1c assay on Abbott’s latest generation analyzer, the Alinity c, on a large patient cohort and to compare it with Tosoh’s ion-exchange HPLC. For this we used anonymously leftover material from randomly selected 136 patients from the daily routine analysis. The ethylenediaminetetraacetic acid whole blood samples were analyzed directly, without storage, in parallel, on the three devices—Abbott Alinity c, Tosoh G8, and Tosoh G11. The imprecision was recorded on all three devices by measuring triplicates of two different assay-related HbA1c quality controls daily for five consecutive days, followed by within-run and total imprecision calculations according to the CLSI EP15-A3 protocol. Comparison was undertaken using Passing-Bablok regressions, Pearson correlation coefficients
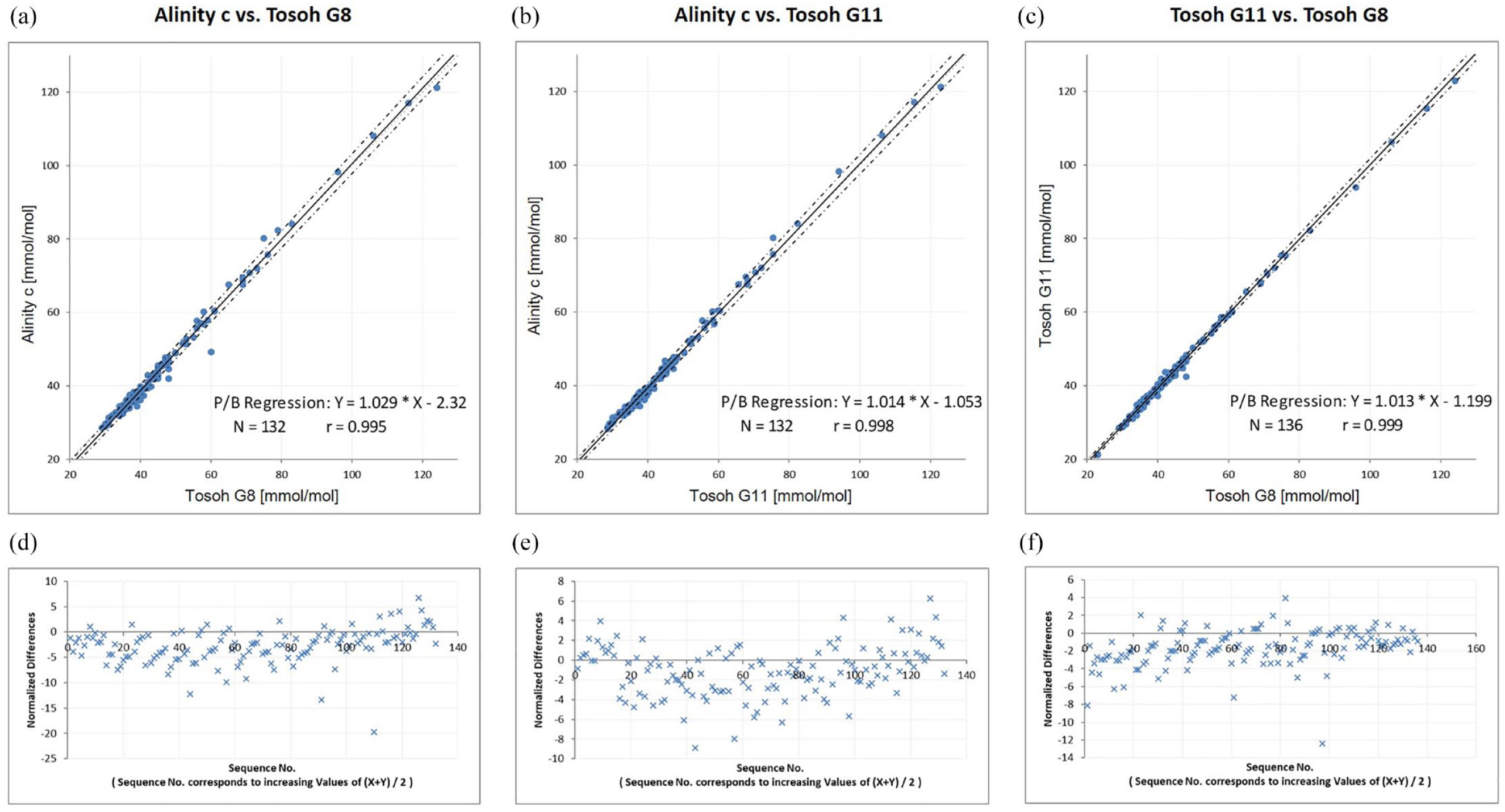
Passing-Bablok regression analysis (a-c) and Bland-Altman difference plots (d-f) on Alinity c, Tosoh G8, and Tosoh G11.
All three devices showed a good within-run and total imprecision with a coefficient of variation ≤2% for the Tosoh devices and <1% for the enzymatic Alinity assay in both control levels. The high accuracy and traceability of the enzymatic Abbott assay, which was already known for the ARCHITECT,
3
could now be confirmed for the Alinity c analyzer. In a direct comparison, a good regression of the Alinity c compared with the HPLC could be determined (Figure 1). Both in comparison with the Tosoh G8 (HbA1c Alinity c [mmol/mol] = 1.029 × HbA1c Tosoh G8 [mmol/mol] − 2.32;
We were able to confirm the good performance of the enzymatic Abbott assay on the Abbott analyzer Alinity c. The influence of possible interferences was not considered in this study. However, it could be shown recently that certain hemoglobinopathies can lead to an incorrect HbA1c determination), 4 which needs to be taken into account.
Footnotes
Acknowledgements
We thank Tosoh Bioscience for providing reagents, calibrators, control material, and support, as well as temporarily the Tosoh G11 analyzer for the study.
Abbreviations
HbA1c, hemoglobin A1C; EDTA, ethylenediaminetetraacetic acid; HPLC, high-performance liquid chromatography; CLSI, Clinical & Laboratory Standards Institute.
Author Contributions
Conceptualization, B.Z., A.E.; methodology, B.Z., A.E.; software, B.Z., A.E., A.F.; validation, B.Z., A.E., A.F.; formal analysis, B.Z., A.E.; investigation, B.Z., A.E.; resources, B.Z., A.E.; data curation, B.Z., A.E.; writing—original draft preparation, B.Z., A.E.; writing—review and editing, B.Z., A.E., A.F.; visualization, B.Z., A.E.; supervision, A.E.; project administration, B.Z., A.E. All authors have read and agreed to the published version of the manuscript.
Availability of Data and Materials
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to privacy and ethical reasons.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The study was conducted according to the World Medical Association Declaration of Helsinki and approved by the Ethics committee of the University Medical Center Göttingen (approval no. 27/11/19An).
Informed Consent
Informed consent was obtained from all subjects involved in the study. No clinical trial registry was necessary.
