Abstract
Background:
Monitoring of HbA1c is the standard of care to assess diabetes control. In Trinidad & Tobago (T&T) there are no existing data on the quality of HbA1c measurement. Our study examined the precision and accuracy of HbA1c testing in T&T.
Methods:
Sets of 10 samples containing blinded duplicates were shipped to laboratories in T&T. This exercise was repeated 6 months later. Precision and accuracy were estimated for each laboratory/method.
Results:
T&T methods included immunoassay, capillary electrophoresis, and boronate affinity binding. Most, but not all, laboratories demonstrated acceptable precision and accuracy.
Conclusions:
Continuous oversight of HbA1c testing (eg, through proficiency testing) in T&T is recommended. These results highlight the lack of oversight of HbA1c testing in some developing countries.
Diabetes is a growing concern amongst the worlds’ population with a projected 360 million people projected to be affected by 2030 and a worldwide prevalence of 4.4%. 1 In the Caribbean, and specifically Trinidad and Tobago (T&T), diabetes prevalence continues to rise with 14.2% of the population currently affected, more than twice the global average. In addition, 135.6 per 1000 adults are undiagnosed. 1 Large studies such as the Diabetes Control and Complications Trial (DCCT) and United Kingdom Prospective Diabetes Study (UKPDS) have established the determination of HbA1c as an indicator of diabetes control and a predictor of long complications.2,3 The clinical use of the HbA1c therefore requires assays harmonized to DCCT measurements and to internationally accepted standards. In recognition of this issue, developed countries have established national and international standardization programs (eg, NGSP, National Glycohemoglobin Standardization Program, and IFCC, International Federation of Clinical Chemistry) to ensure the clinical utility of HbA1c testing.4,5 HbA1c measurements should also be monitored through regular proficiency testing (PT) of individual laboratories. 6 In T&T, a developing country, no data exist on HbA1c precision and accuracy and there is no national PT program. Unreliable HbA1c determinations would have a negative impact on diabetes care, potentially increasing costs and complications.7-9 Therefore, Johns Hopkins Medicine International and the Diabetes Diagnostic Laboratory (DDL) at the University of Missouri, under the umbrella of the Trinidad and Tobago Health Sciences Initiative, performed 2 pilot studies to assess the accuracy and precision of HbA1c testing in T&T.
Methods
All T&T laboratory directors met as a group prior to the study and consented to participation. For each study, sets of 10 samples containing blinded duplicates were created from 5 whole-blood pools with HbA1c levels between 5.0 and 9.5% HbA1c. The DDL assigned values to each sample based on the mean of multiple analyses by 2 different NGSP secondary reference laboratories (SRLs); SRL9 using Tosoh G8 HPLC and SRL3 using Trinity ultra2. Samples were shipped to participating laboratories from the DDL; some sample sets were shipped fresh at 4°C, and some were shipped frozen on dry ice to accommodate different assay method requirements. Laboratories were instructed to analyze the samples upon receipt.
To assess within-day imprecision, the pooled estimate of the SDs between the duplicates (Sp) was calculated; 0.229 was the acceptable limit based on the current NGSP laboratory monitoring criterion. To assess accuracy, each laboratory’s results were compared to those assigned by NGSP. Reports were generated and distributed to all participating labs and discussed in a group setting which included a health official from T&T.
Six of 7 laboratories that participated in the first comparison study also participated in the second, representing all public laboratories and the 2 largest private laboratories. Nine laboratories participated in the second study, 2 of which analyzed the samples on 2 different methods making a total of 11 laboratories/methods. Methods included in the first comparison study were the Roche Tinaquant on Cobas Integra 400+, Cobas 6000, Hitachi 911, Alere Nycocard, and Sebia Minicap. The second comparison included the above methods with the exception of the Roche Tinaquant on Hitachi 911 and in addition the Abbott C8000, Siemens DCA Vantage, and Alere Afinion were included.
Results
Within-day imprecision, shown in Figure 1, was within acceptable limits (Sp < 0.229) for all but the laboratory using the DCA Vantage and 2 laboratories using the Nycocard. One of the laboratories using the NycoCard showed especially variable results with Sp > 0.7 and > 1.5 for the first and second comparison studies, respectively. Imprecision results for the laboratory using the DCA Vantage had an Sp just outside the acceptable limit. Results for each laboratory/method are shown in Figure 2 compared to the NGSP assigned values and the acceptable bias limit of ±6%. The first comparison study revealed bias results within 6% of the NGSP target value for all laboratories except for both laboratories using the Nycocard and 1 result for the laboratory using the Hitachi 911. Of note, laboratory 1A reported results in IFCC% in the first study; IFCC now recommends reporting IFCC results as mmol/mol Hb. Since all other HbA1c results in T&T are reported in NGSP%, these IFCC results were aligned to NGSP% using the master equation NGSP% = 0.915 (IFCC%) + 2.15, 10 for the data analyses. In the second comparison laboratory 1A reported results in NGSP%. Some results from both laboratories using the Nycocard, 1 laboratory using the Cobas 6000, the laboratory using the DCA Vantage, and 1 laboratory using the Afinion were outside of acceptable limits for the second comparison.
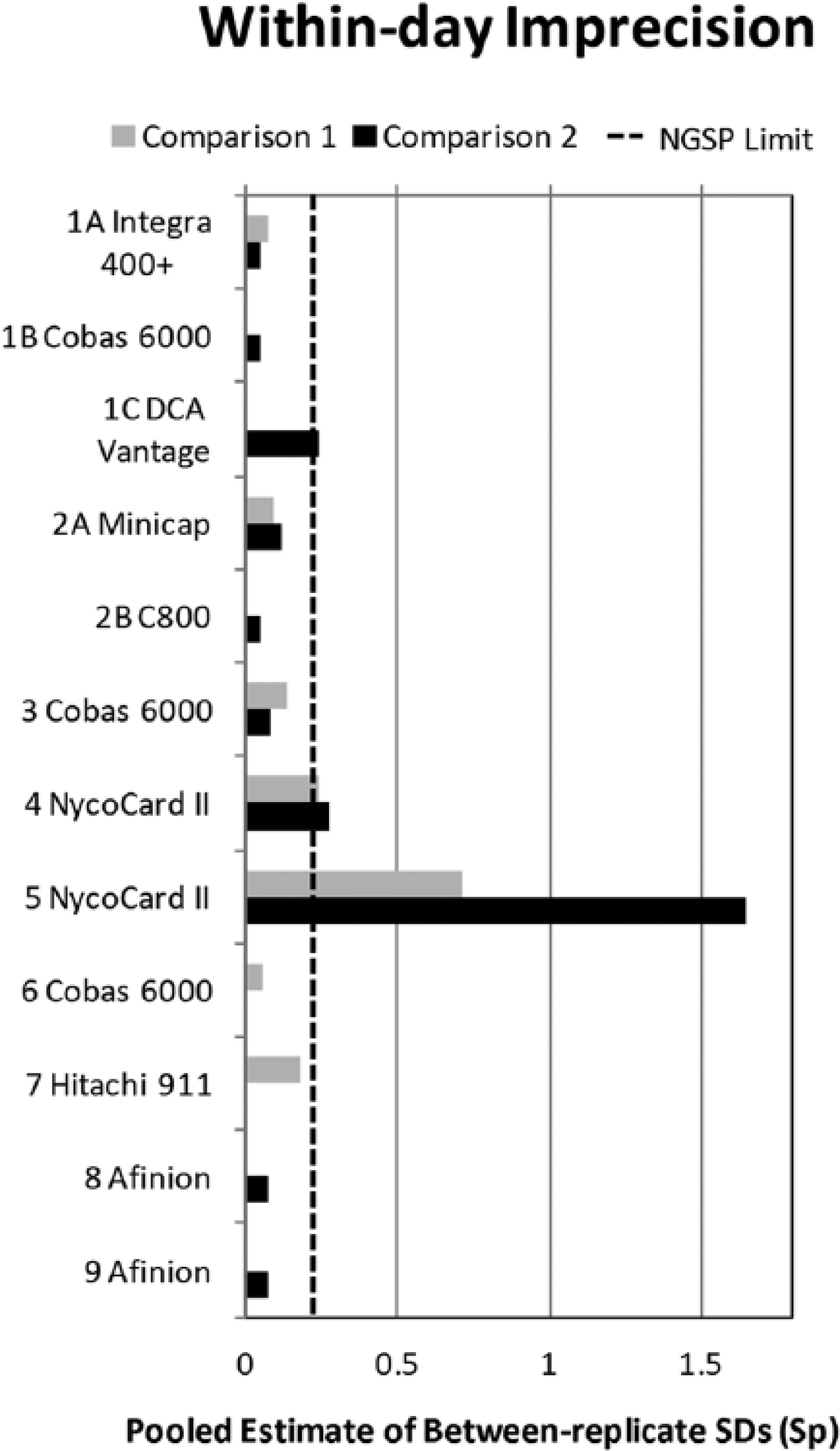
Within-day imprecision shown as the pooled estimate of between-replicate SDs (sp) for each laboratory (referenced by numbers 1-9) using each method (referenced by letter A or B). The dashed line is the NGSP network monitoring limit.
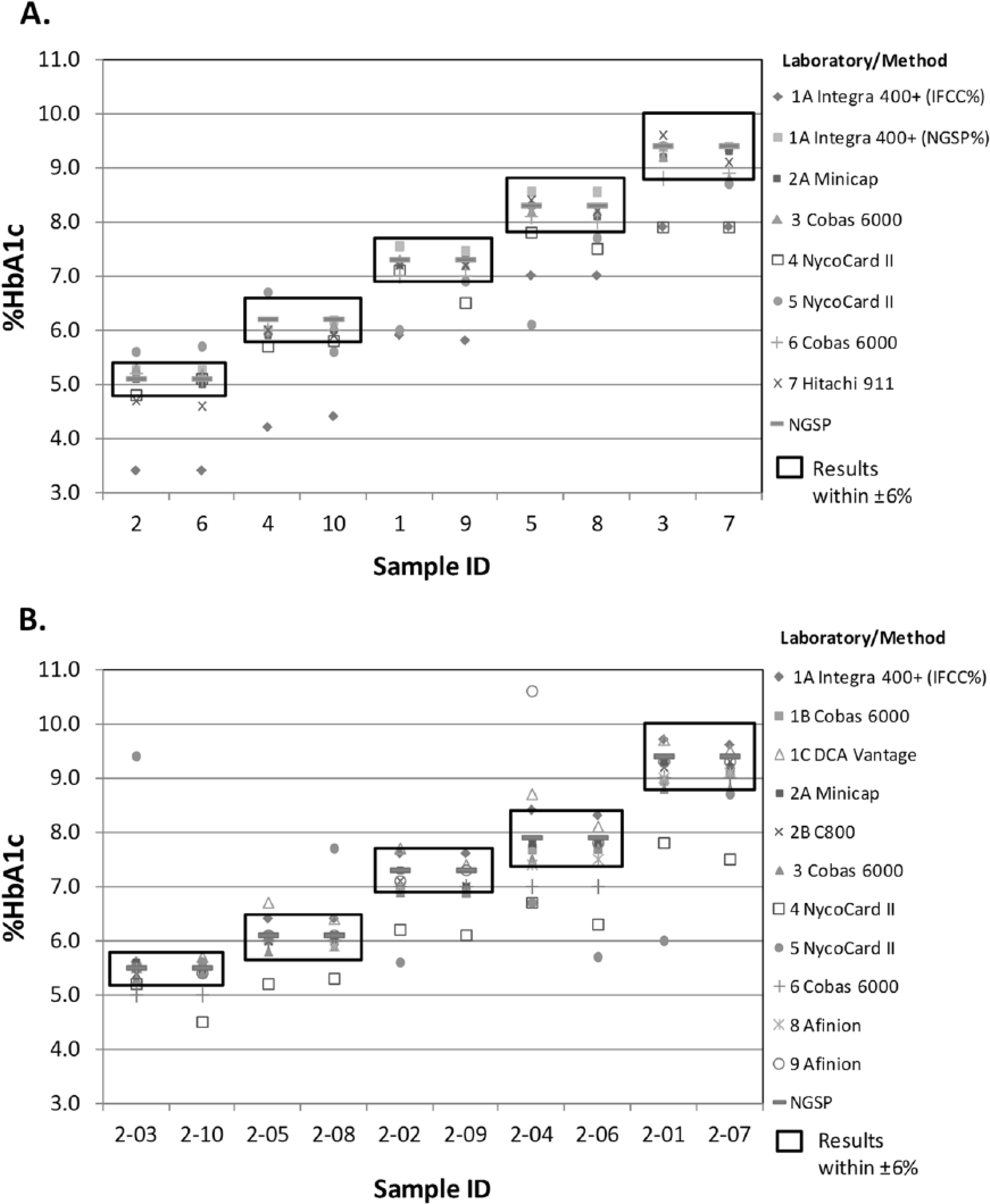
Bias results for the first (A) and second (B) comparison study. Each point represents a single result for each lab/method for each pooled sample. Replicate samples are adjacent to each other. The boxes represent ±6% of the NGSP assigned value for each sample pool.
Discussion
HbA1c is a posttranslational modification of HbA with the nonenzymatic addition of a glucose moiety to 1 or both NH2-terminal valine residues on the Hb beta-chains. 11 Its measurement can be directly related to average blood glucose over 90-120 days making it the cornerstone for monitoring glycemic control in patients with diabetes.12,13 While the burden of diabetes is felt worldwide, prevalence continues to rise at an alarming rate in the developing world. 13 Lack of access to quality care leads to a rise in complications, exponentially increasing the costs on overburdened health systems. 14 The results of this study have huge implications for diabetes care in the island of T&T. Two large public hospitals were relying solely on the point of care cartridge based Nycocard system for all their HbA1c values. Our precision testing revealed that these values were consistently outside of the NGSP acceptable limits and were poorly correlated with standards based on DCCT and UKPDS. Suboptimal performance has been described previously for this method. 15 This means that a large proportion of T&T’s diabetes population was not being managed optimally unbeknownst to clinicians. T&T has a large prevalence of diabetes complications, 16 and it is possible that these HbA1c measurements have contributed to these outcomes. These results argue strongly for regular PT testing within the T&T health system.
Prior to the NGSP in the United States, HbA1c testing showed a similar spread with values ranging between 4.0% and 8.1% on the same blood sample. 17 Once the NGSP was established HbA1c testing improved dramatically over time. 18 This study relied on the NGSP and proves that a regional partnership with an existing program is useful for smaller countries. One lab was able to change to a better HbA1c method. Another laboratory changed to NGSP% versus IFCC% reporting so that their results could be compared to results from other laboratories in T&T. While most laboratory methods performed well in both comparisons, several did not. The observed variations in results demonstrate a need for continued evaluation and improvement in HbA1c results in T&T.
In conclusion, further PT should be performed in T&T to ensure that the quality of HbA1c testing is sufficient to meet clinical needs. Practitioners in developing countries should be aware of the reliability of their HbA1c testing as deviations from international standards will lead to inadvertent errors in patient care.
Footnotes
Acknowledgements
The authors would like to acknowledge the cooperation of the government of Trinidad and Tobago. We would like to express our gratitude toward the lab directors and lab technicians of all laboratories involved.
Abbreviations
DCCT, Diabetes Control and Complications Trial; DDL, Diabetes Diagnostic Laboratory; IFCC, International Federation of Clinical Chemistry; NGSP, National Glycohemoglobin Standardization Program; PT, proficiency testing; SRL, secondary reference laboratory; T&T, Trinidad and Tobago; UKPDS, United Kingdom Prospective Diabetes Study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by Johns Hopkins International.
