Abstract
Background:
Methods:
An mvAID system is developed to supplement the glucose measurements with additional physiological signals from a wristband device, with the signals analyzed using artificial intelligence algorithms to automatically detect the occurrence of PA and estimate its intensity. This additional information gained from the physiological signals enables more proactive insulin dosing adjustments in response to both planned exercise and spontaneous unanticipated physical activities.
Results:
In silico studies of the mvAID illustrate the safety and efficacy of the system. The mvAID is translated to pilot clinical studies to assess its performance, and the clinical experiments demonstrate an increased time in range and reduced risk of hypoglycemia following unannounced PA and meals.
Conclusions:
The mvAID systems can increase the safety and efficacy of insulin delivery in the presence of unannounced physical activities and meals, leading to improved lives and less burden on people with T1D.
Keywords
Introduction
Hybrid closed-loop insulin delivery improves glycemic control in people with type 1 diabetes mellitus (T1D) compared with sensor augmented pump therapy, leading to clinically significant improvements in time in range, reduction in hemoglobin A1C, and lower risk of hypoglycemia.1 -5 Hybrid closed-loop control continuously adjusts basal insulin delivery in response to measurements of the blood glucose concentration (BGC) levels, with premeal boluses and exercise-related insulin dosing adjustments addressed through manual interventions by the user.6 -10 The burden on users to manually enter premeal boluses and modulate insulin dosing to accommodate physical activity (PA) limits the safety and efficacy of hybrid closed-loop insulin delivery.11 -13 Multivariable automated insulin delivery (mvAID) systems, also known as multivariable artificial pancreas, achieve better glycemic control than hybrid closed-loop control systems by supplementing the continuous glucose monitoring (CGM) measurements with real-time inputs of physiological signals received from wearable activity trackers to automatically detect the onset of PA and estimate its type and intensity, and mitigate its effects on glucose variations. 14
Recent advancements in automated insulin delivery (AID) systems have incorporated automation of mealtime boluses, with algorithms developed to analyze historical and current CGM measurements and consider the insulin-on-board to automatically detect the occurrence of meals and estimate the carbohydrate (CHO) consumption amounts.15 -18 The amount of CHOs consumed can also be estimated in real-time as the meal progresses and BGC rises in response to CHO intake.17,19 -23 Glucose-only AID systems can handle automation of mealtime boluses by analyzing the effects of meals on the CGM measurements, but they are ineffective in modulating insulin dosing in response to PA since the AID system only detects exercise with a significant delay when the BGCs decrease. 24 This limitation in handling PA can be addressed by incorporating additional physiological signals, like heart rate and energy expenditure, which can indicate the occurrence of PA and enable estimation of its intensity.
Physical activity presents a significant challenge to glycemic control for people with T1D. 25 Moderate-intensity PA can cause a precipitous decline in BGCs, which increases the risk of hypoglycemia. 26 Moreover, PA induces heightened insulin sensitivity and increased glucose disposal, which may persist for several hours following PA. Planned exercise may be managed by hybrid closed-loop control through early interventions initiated by the user in anticipation of the exercise, and timely reductions in insulin delivery well in advance of the exercise. In contrast to planned exercise, unplanned or spontaneous PA presents a major challenge to glycemic control because unanticipated PA cannot be preemptively accommodated by users of AID systems. A new generation of mvAID systems is proposed that incorporates additional physiological signals from a wristband device with artificial intelligence in real-time to automatically detect PA, identify its type and estimate its intensity, and modulate insulin dosing in response to concomitant evolving variations in PA-related glycemic dynamics.
This article presents a novel mvAID system that does not require any manual entries from users to handle the glycemic effects of planned and spontaneous physical activities and meals. The structure, modules, and algorithms encompassing the mvAID system are described in this article, and results evaluating the mvAID system in both simulation studies and pilot clinical studies are presented. The mvAID readily accommodates daily patterns of everyday life to improve glycemic control and is robust to unforeseen glycemic disturbances. Artificial intelligence powered predictive modules are developed and implemented on a mobile application, enabling the mvAID to proactively counteract the glycemic disturbances, including planned and spontaneous physical activities and meals. The mvAID system has multiple components, including modules to process the real-time streaming of the additional physiological signals to impute missing data from sensors, reconcile outliers, and eliminate artifacts in the physiological variables; estimate heart rate from wristband signals; estimate energy expenditure, a valuable indicator for the intensity of PA; estimate the plasma insulin concentration (PIC), a robust measure of the insulin circulating in the bloodstream; detect unannounced meals and estimate CHO consumption; optimize the insulin dosing with an adaptive model predictive control (MPC) formulation that predicts the future BGC trajectory profiles in response to the assessed PA levels, detected meals, and PIC; estimate the likely future risk of potential hypoglycemic and hyperglycemic episodes.27 -31 Deep neural networks are developed and trained on historical data to capture the daily activity behaviors and patterns of people with T1D. These patterns are used in predictive modeling and anticipating the future glycemic disturbances used by the MPC algorithm for more accurate insulin dosing. The safety and efficacy of the mvAID are demonstrated through simulation studies and the mvAID is translated to pilot clinical studies that validate the improvement in glycemic control over glucose-only AID systems.
Methods
Structure of mvAID System
Figure 1 depicts the framework of the mvAID system. The blue-colored blocks indicate modules that estimate glycemic disturbances in real-time,27,28,30,31 green-colored blocks are signal processing modules or algorithms used for real-time signal reconciliation,32,33 red-colored blocks represent the modules and algorithms in the feedback loop, namely, the system identification (modeling), MPC formulation, and safety constraint modules.
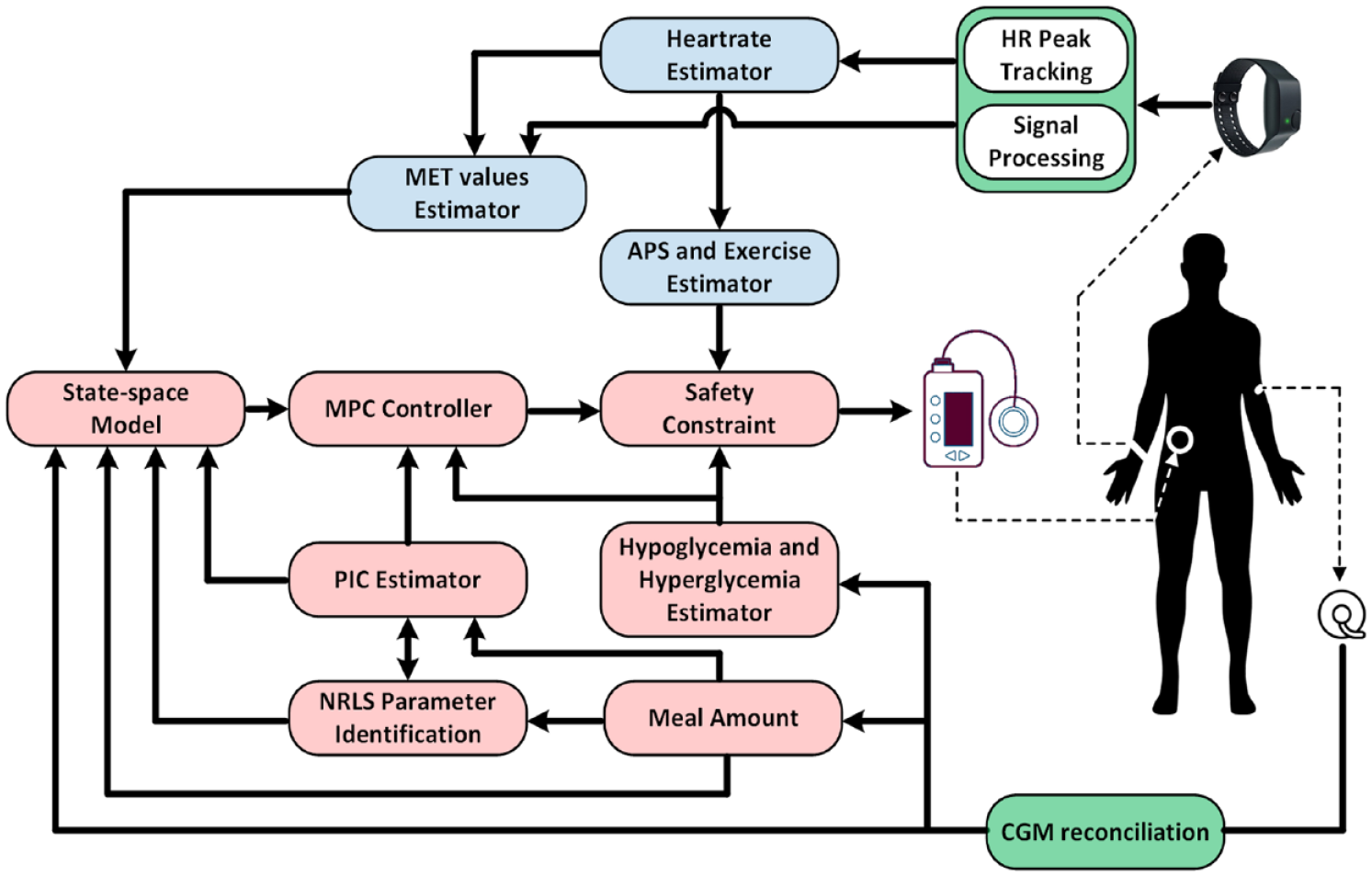
The schematic architecture of mvAID system.
Components of mvAID System
The closed-loop AID system consists of two main parts:
Feedback modules: Feedback modules are the main part of the control system, where the optimal insulin dose to administer is computed by the adaptive MPC algorithm based on CGM measurements and the past infused insulin data. The feedback module is made up of multiple sub-components: CGM signal reconciliation, PIC estimation, prediction of potential future hypoglycemia and hyperglycemia events, adaptation model parameter estimation using nonlinear recursive least squares (NRLS), safety constraints module, and adaptive MPC.34,35 Details on their implementation will be presented later.
Feedforward modules: The purpose of the feedforward modules is to further modify the controller output based on anticipated future occurrences of physical activities and meals. The feedforward modules estimate the disturbances affecting BGC dynamics using wristband signals. Examples of feedforward modules for PA include energy expenditure estimation through calculated metabolic equivalents of task (MET) values reflecting metabolic rate during activity and heart rate estimation.
The meal detection and the CHO intake amount estimation algorithms are predictor modules developed based on the CGM sensor signal and act similarly to feedforward modules. Hence, they are considered in a separate category, separate from feedback/feedforward modules.
Initialization of the mvAID System
The mvAID system consists of numerous modules that adaptively and recursively predict the variables of interest. Hence, initialization of these modules, variables, and predictive models is performed before computing the insulin dose suggestion by the MPC algorithm.
Initialization of the mvAID system is a crucial step that necessitates providing the demographic and insulin infusion routine information. The initialization module consists of a data-driven model with demographic information entered by the user as model input. The model outputs are time-varying parameters of the glucose-insulin dynamics whose values are calculated adaptively during the execution of mvAID. Initialization of physiological parameters in Hovorka’s model is imperative for estimating the PIC and the amount of CHO intake. To initialize the mvAID system, a data-driven approach is used where kernel partial least squares (KPLS) regression is implemented based on the pipeline model presented in Figure 2. Radial basis function kernel with 95% variance of input and output parameters (as listed in Table 1) are explained by latent vectors trained on data collected from 12 T1D subjects in open-loop clinical experiments.
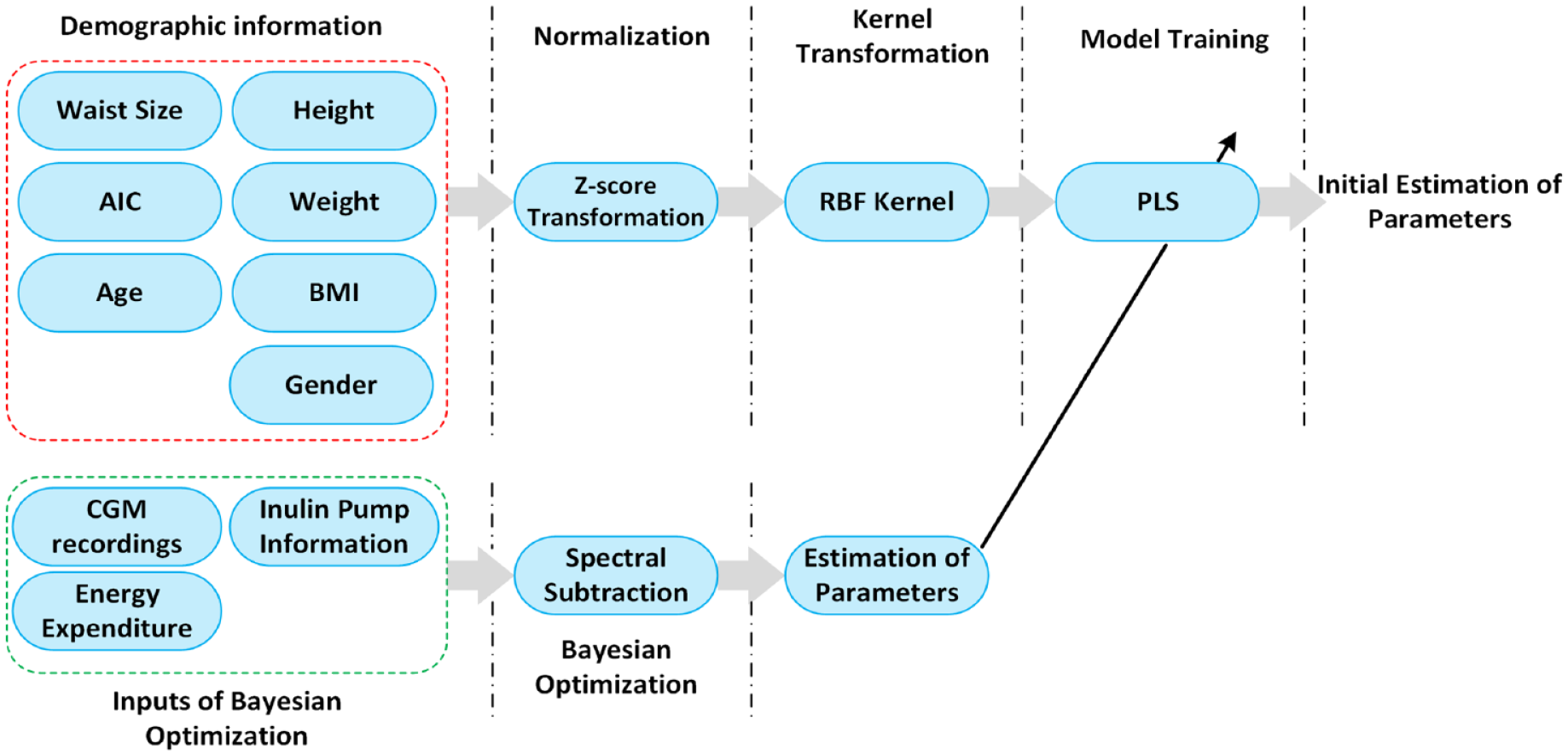
A pipeline for the initialization of physiological parameters in mvAID.
The List of Inputs and Predictor Outputs of the Partial Least Squares Model.
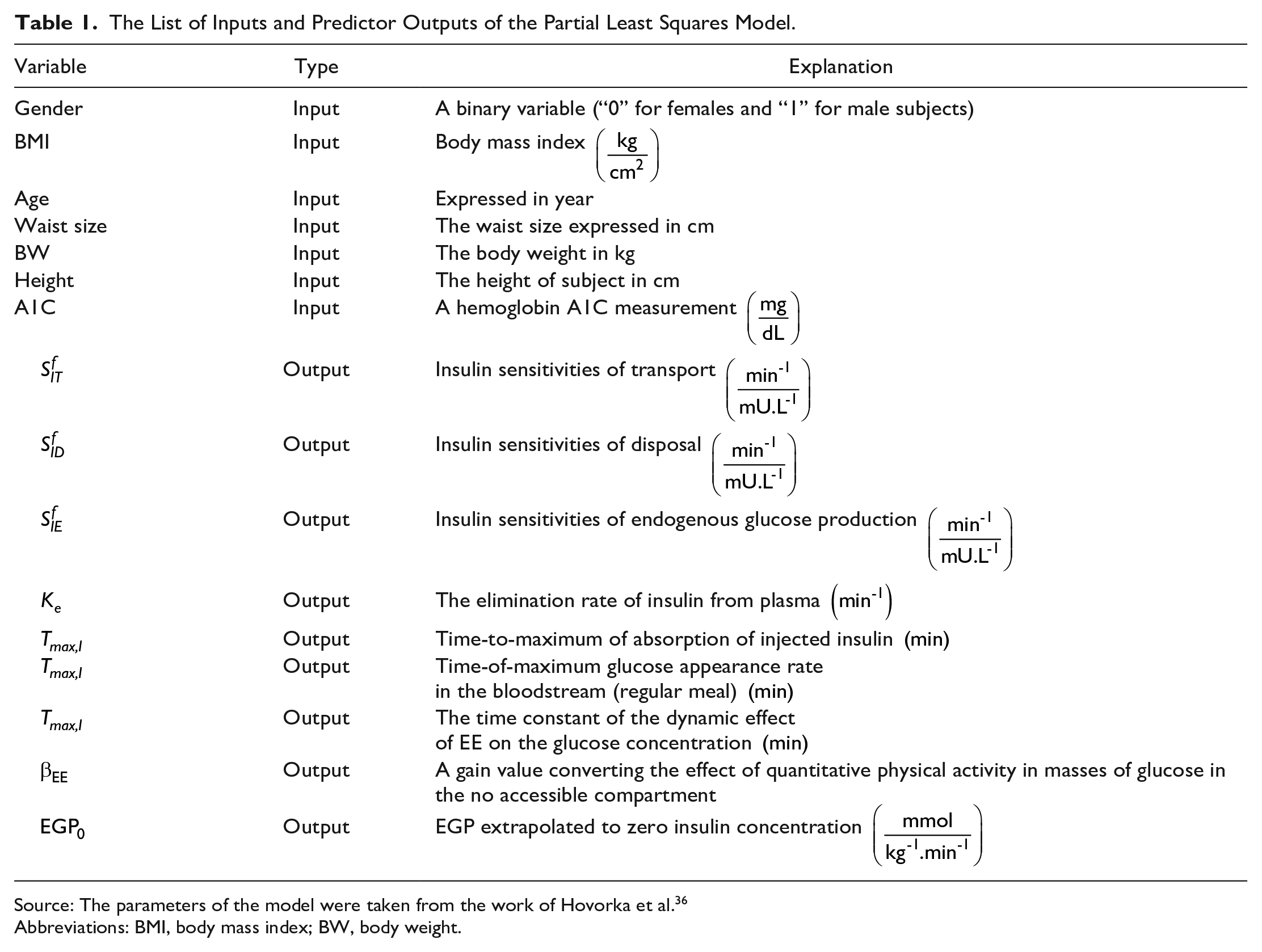
Source: The parameters of the model were taken from the work of Hovorka et al. 36
Abbreviations: BMI, body mass index; BW, body weight.
Estimating Disturbances Affecting BGC Regulation
Mitigating the effects of disturbances before they affect the outputs is the main responsibility of the feedforward blocks of the control loop. 37 In the mvAID system, feedforward modules translate raw physiological variable signals into meaningful features variables, usually to characterize disturbances and adjust the manipulated variables directly or indirectly.
Reconciliation and Processing of Wristband Physiological Signals
The interpretation of raw physiological signals and converting the measured variables data to informative estimates of disturbance variables requires denoising the raw physiological signals and performing feature engineering. Physiological signals are processed differently depending on disturbance variables. Heart rate plays a crucial role in predicting disturbances, such as PA and is a useful variable to estimate the effects of PA on the glycemic dynamics. Estimation of heart rate from blood volume pulse measurements requires denoising the raw blood volume pulse measurements, motion artifact removal, and designing an algorithm for tracking the frequency of heart rate peaks in real-time. The detailed process of estimating heart rate in real-time is provided by Askari et al.32,33
Reconciliation of Glucose Measurements
A closed-loop control feedback system relies heavily on BGC measurements captured by CGM sensors. Interruption in wireless connectivity between the CGM sensor and the mobile edge computing device where the AID system resides (cellphone or insulin infusion pumps) is considered a critical situation, where no monitoring of BGC is performed. Hence, temporarily predicting BGC values is vital for maintaining the functionality of the hypoglycemia/hyperglycemia detector, CHO intake and PIC estimators, and making control decisions. In this regard, up to six missing CGM measurements are predicted using a vector autoregressive (VAR) model with a recursive adaptation of model parameters. Predicted CGM values are replaced by cubic spline interpolated values after CGM connection is re-established.
Detecting PA and Quantifying Its Intensity With Energy Expenditure
The detection of PA occurs follows the preprocessing of the blood volume pulse signal for noise cancelation and artifact removal, and estimating heart rate values. A major obstacle to integrating multiple modules is implementing these modules on platforms with limited computational power. Prediction of energy expenditure through MET values is imperative to avoid the risk of hypoglycemia caused by PA and to have a more accurate model for use in the MPC algorithm. Combining the approaches presented in the work of Askari et al 33 for preprocessing the raw signals and using the simplified model developed in previous works,38,39 we develop and integrate an accurate estimation of MET values for automatically calculating an optimal amount of insulin in real-time. The developed MET estimation quantified the intensity of physical activities by measuring the last ten seconds of wristband signals, including three-axis accelerometer signals, skin temperature, blood volume pulse signal, and galvanic skin response. More details on the MET estimation module are provided in the work of Sevil et al. 39
The quantification of PA with MET values results in a continuous variable that can be applied directly to activate or inactivate the insulin pump or be utilized as an additional exogenous input to the linear state-space model employed by the MPC formulation. When the estimated MET values exceed a specific threshold value, a simple approach to ensure the safe regulation of BGC levels is to suspend the insulin delivery by the infusion pump. The estimation of MET values has been used directly in the formulation of the mvAID system. High values of estimated MET result in lowering BGC when performing low-intensity or moderate-intensity PA and this behavior has been considered in the extended exercise-glucose-insulin dynamic model.40,41
Real-Time Adaptive Estimation of PIC
Plasma insulin concentration is a variable whose value is critical for designing adaptive identification algorithms, predictive models, and safety constraint modules. Currently, there is no commercially available sensor for measuring PIC in real-time. Therefore, estimation of the PIC should be performed using adaptive approaches known as state observers. A nonlinear glucose-insulin dynamic model is integrated with an unscented Kalman filter (UKF) to predict the values of intermediate state variables including PIC. 42 To avoid degradation in the performance of the PIC estimator, the parameters of the glucose-insulin model must be updated in real-time. As a solution, an extended set of state variables is utilized for state estimation. In addition, a subset of parameters is adjusted in real-time by integrating an NRLS parameter identification algorithm. A parameter saturation function is considered to ensure that the model parameters take values inside a meaningful range and no stability issues occur. Details on the PIC estimation module are reported in the work of Hajizadeh et al. 34
Estimation of CHO Amount
A bolus insulin infusion is required if CHO intake is detected by the meal detection and CHO estimation module. The amount of CHO intake is necessary to estimate for handling unannounced meals, and the estimated CHO intake is used in the estimation of PIC with UKF and predicting the future glucose profile trajectory for predictive control.
The primary variable that provides real-time information on CHO intake is the CGM measurements. As part of the fuzzy model, several fuzzy logic rules are used to incorporate human knowledge into the estimation of the model output. The first fuzzy model is developed to detect meal events and triggering the second fuzzy model results in the estimation of meal amount. The structure of the fuzzy rules is comprehensively studied by Samadi et al.19,20 Given the list of fuzzy rules in previous studies, the meal flag is calculated by a fuzzy Sugeno model. Triangular fuzzy membership functions are used for both fuzzy models, and particle swarm algorithm is used to optimize the location of membership functions. Table 2 displays the comparison between total estimated CHO amount and their ground truth values. Figure 3 visualizes the CHOs consumed by a randomly selected subject to better illustrate the performance of meal estimator.
Total Predicted and Actual CHO Amounts for Each Subject.
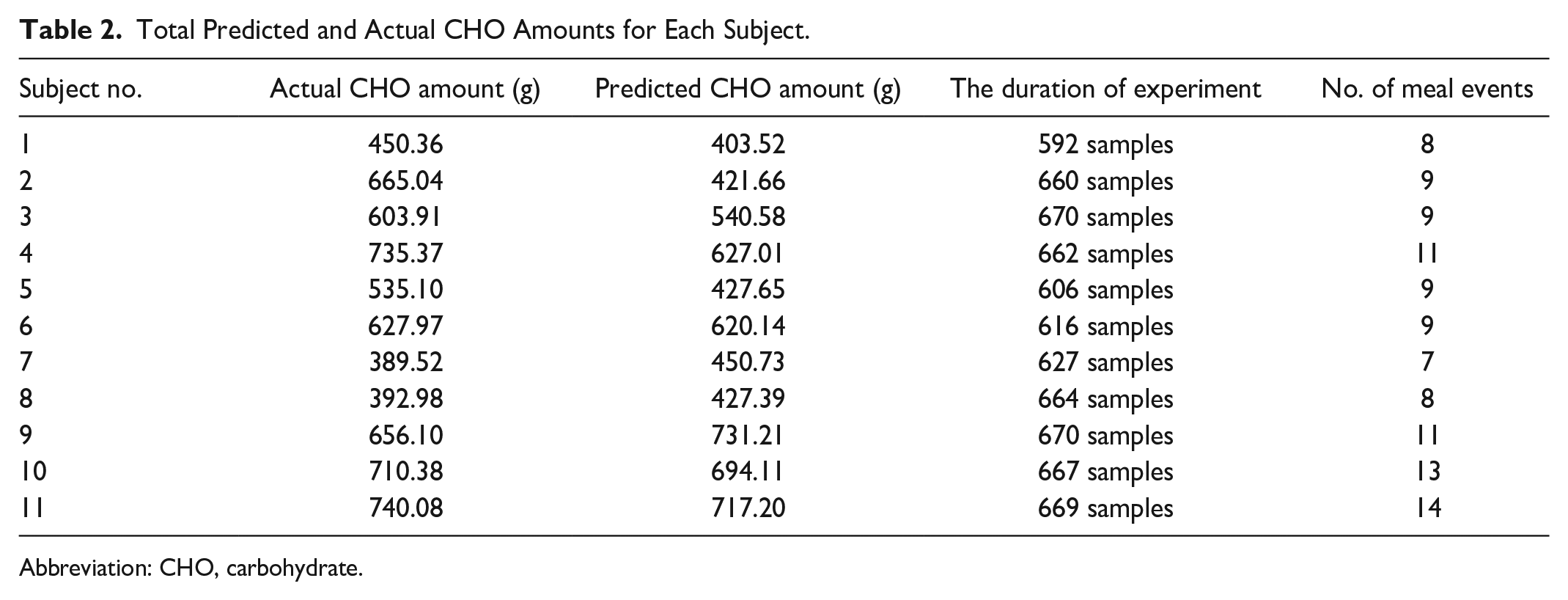
Abbreviation: CHO, carbohydrate.
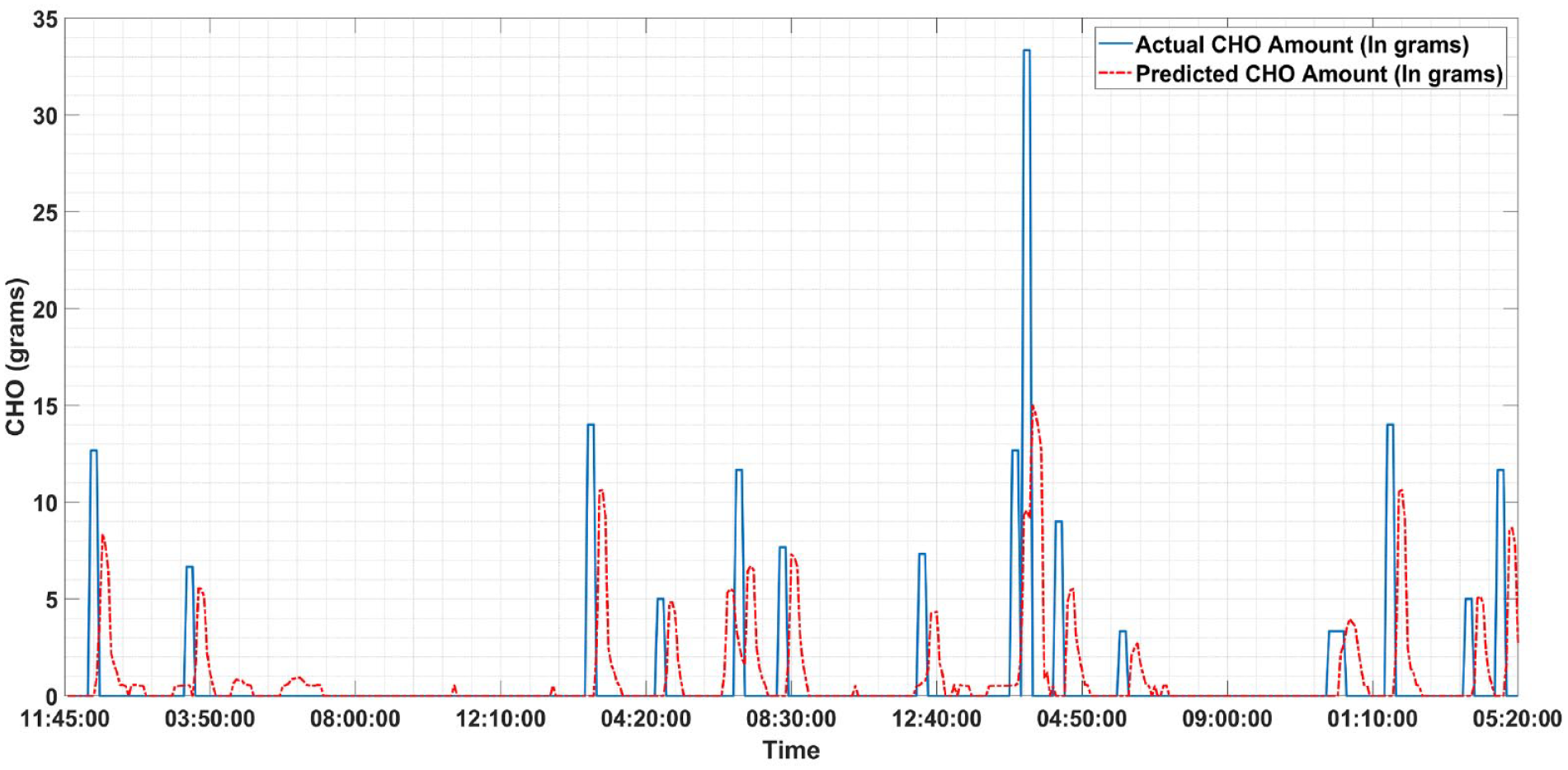
Prediction of carbohydrate consumption by a randomly selected subject: The actual amounts of carbohydrate intake recorded during the clinical experiment (solid blue line), the amounts of predicted carbohydrates (red dash-dotted line).
Risk Prediction of Potential Hypoglycemic and Hyperglycemic Events
An important feature of the safety module is the ability to predict in advance hypoglycemia and hyperglycemia for their early detection of potential occurrence, suspension of insulin delivery, or avoiding insulin overdosing.43,44 In addition, predicting potential hypoglycemia and hyperglycemia can be used to adjust the conservativeness or aggressiveness of the controller. Mamdani fuzzy logic systems were used to predict hypoglycemia and hyperglycemia as they are easy to incorporate human knowledge and leverage the glucose information extracted from the T1Ds.
The variation of the actual and estimated subcutaneous glucose concentration (mg/dL) is illustrated in Figure 4. The glucose concentration data are used in hypoglycemia and hyperglycemia prediction and in the MPC of the MPC.
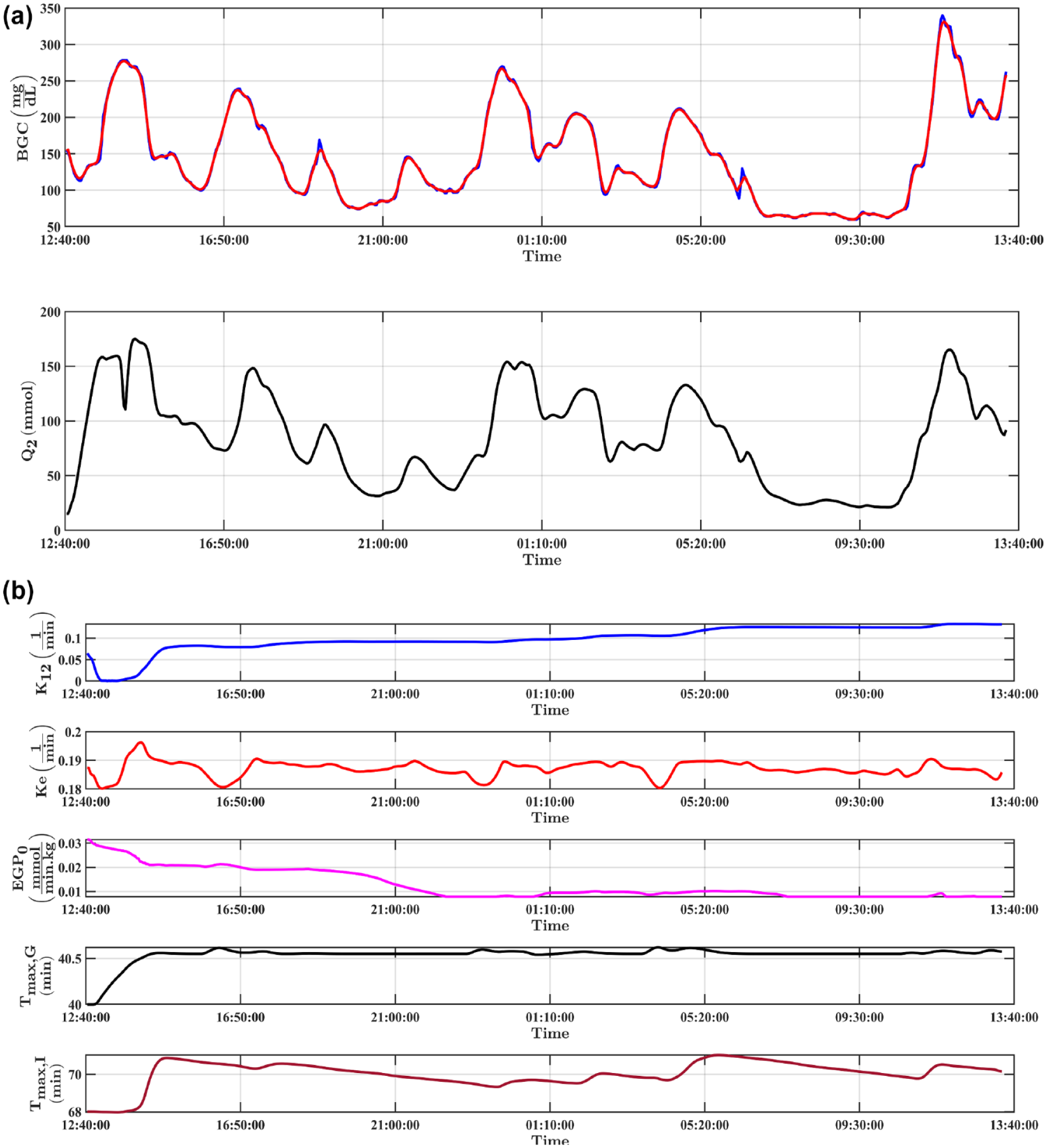
The variation of the actual and estimated subcutaneous glucose concentration (mg/dL). (a) The subcutaneously measured glucose concentration (blue), the subcutaneous glucose concentration estimated by UKF (red), the glucose amount in the peripheral tissue (mmol) (black). (b) The variation of adjustable parameters tuned with NRLS and UKF: The glucose transfer rate between accessible and non-accessible compartments (blue), the glucose elimination rate from plasma (red), time-of-maximum glucose appearance rate in the bloodstream (black), the time constant of the effect of energy expenditure on BGC reduction (magenta), and time-to-maximum insulin absorption (venetian red).
The potential hypoglycemia and hyperglycemia is predicted 30 minutes before the expected occurrence time. Although calculating the probability of hypoglycemia and hyperglycemia events is quite useful for automatically adjusting the aggressiveness of the MPC algorithm, converting the probabilities to the label of events is necessary for use in the safety module. The simulation results for the prediction of both hypoglycemia/hyperglycemia events are depicted in Figure 5.
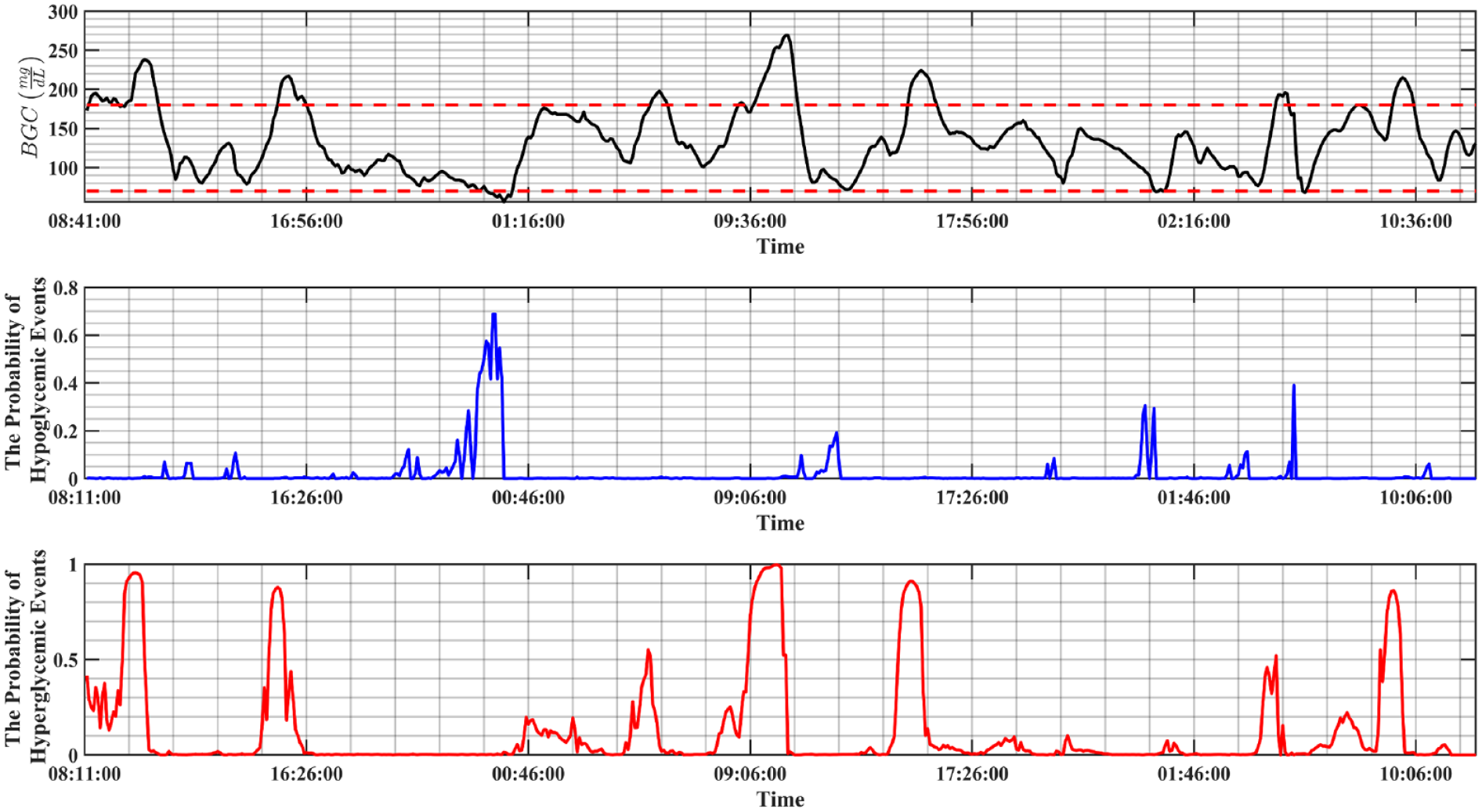
The prediction of hypoglycemic/hyperglycemic event predictor for a randomly selected subject with T1D (subject no. 11). The variation of the BGC (black line), the probability of hypoglycemic events calculated half an hour prior to the actual events (blue), the probability of hyperglycemic events calculated half an hour prior to the actual occurred events (red).
Adaptive Model Predictive Control System
The closed-loop control algorithm is the main module for determining the optimum amount of insulin to be infused in real-time. The manipulated variable is calculated using adaptive MPC. A constrained quadratic optimization problem is used to summarize the MPC formulation, which calculates the optimal path of manipulated variables in the control horizon and the resulting glucose concentration from trajectories in the prediction horizon. To avoid the effect of model drift and uncertainties in the model formulation, only the first value of the controller action is usually implemented by the mvAID and the sequence of computations is repeated every 5 minutes as new CGM data are reported. A major strength of MPC is accommodating constraints in the optimization problem. Constraints can be applied to the magnitude and rate of the change of input, output, and state variables and they appear in the form of equality and inequalities. More details on the MPC formulation used in this study are given in previous publications. 45
Safety Considerations
The safety constraint model is the last module to be executed before administering the amount of insulin. The main purpose of this module is to avoid any risky scenarios, such as suggesting insulin during PA or hypoglycemic episodes and avoiding any fast CHO suggestion during hyperglycemic periods. The safety constraint module evaluates several criteria on the probability of hypoglycemic/hyperglycemic episodes, the insulin dosage, and the detection of PA periods. Algorithm 1 summarizes the safety constraint module.
The safety constraint module for final adjustment of the control action and suggesting fast CHO amount to avoid hypoglycemic episodes.

Results
Simulation Results
The multivariable glucose-insulin-physiological variable simulator (mGIPsim) is used for the in silico evaluation of the mvAID system.40,46 The multivariable glucose-insulin-physiological variable simulator is based on an extended version of a glucose-insulin dynamic model, 36 and it has a total of 20 virtual patients with T1D with unique metabolic and physiologic characteristics and behaviors. The multivariable glucose-insulin-physiological variable simulator generates several other variables, such as heart rate and energy expenditure, as output variables for use with in silico testing of multivariable control algorithms designed to automatically handle glycemic disturbances, such as physical activities (Table 3). Each virtual subject follows a daily behavior with stochastic uncertainty according to predefined distributions for the meal timing and amounts and PA timing and intensity. The simulations are conducted for seven days in duration, with the figures showing a week in duration with glycemic events aligned at the same time to make it easy to discern the response to physical activities and meals.
Scheduling of Meals and Physical Activities Scenarios for the Closed-Loop Simulation.

The primary inputs of the glucose-insulin dynamic model include insulin, CHO intake, and MET values, and the BGC measured by CGM sensor is the output. Figure 6 depicts the variation of insulin amounts calculated by UKF. The variation of PIC and insulin amount in two compartments
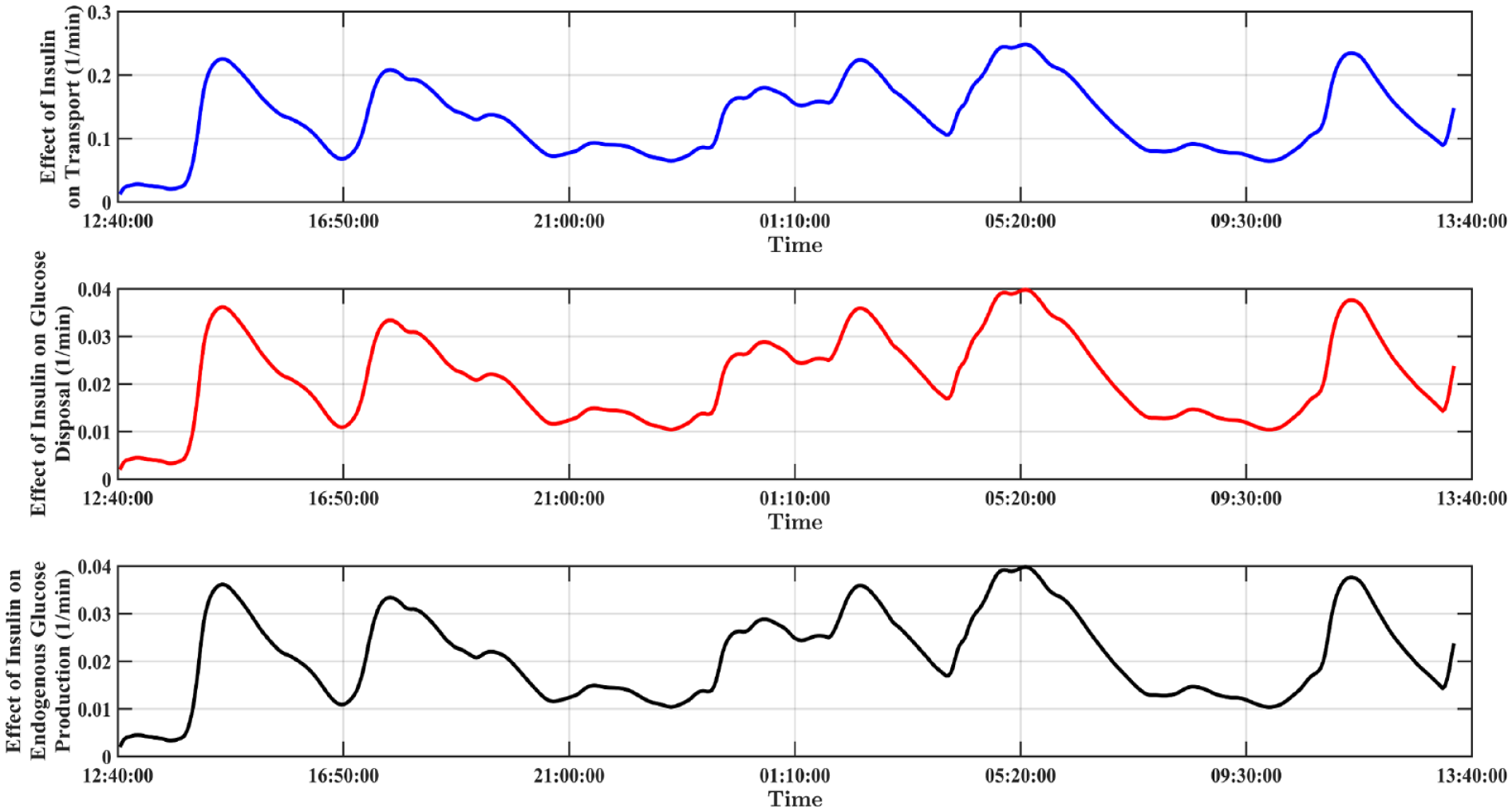
The variation of intermediate state variables estimated by UKF. Predicted amount of insulin in the first compartment
The parameter adaptation corresponding to the simulation and estimation of state variables is presented in Figure 4b. The transport rate,
Figure 7 illustrates the results of a closed-loop simulation study of ten subjects with unannounced inducement of PA and CHO intake based on meal and PA descriptions described in Table 3. There are no hypoglycemia events and average BGC exceeds 200 mg/dL only once in seven days.
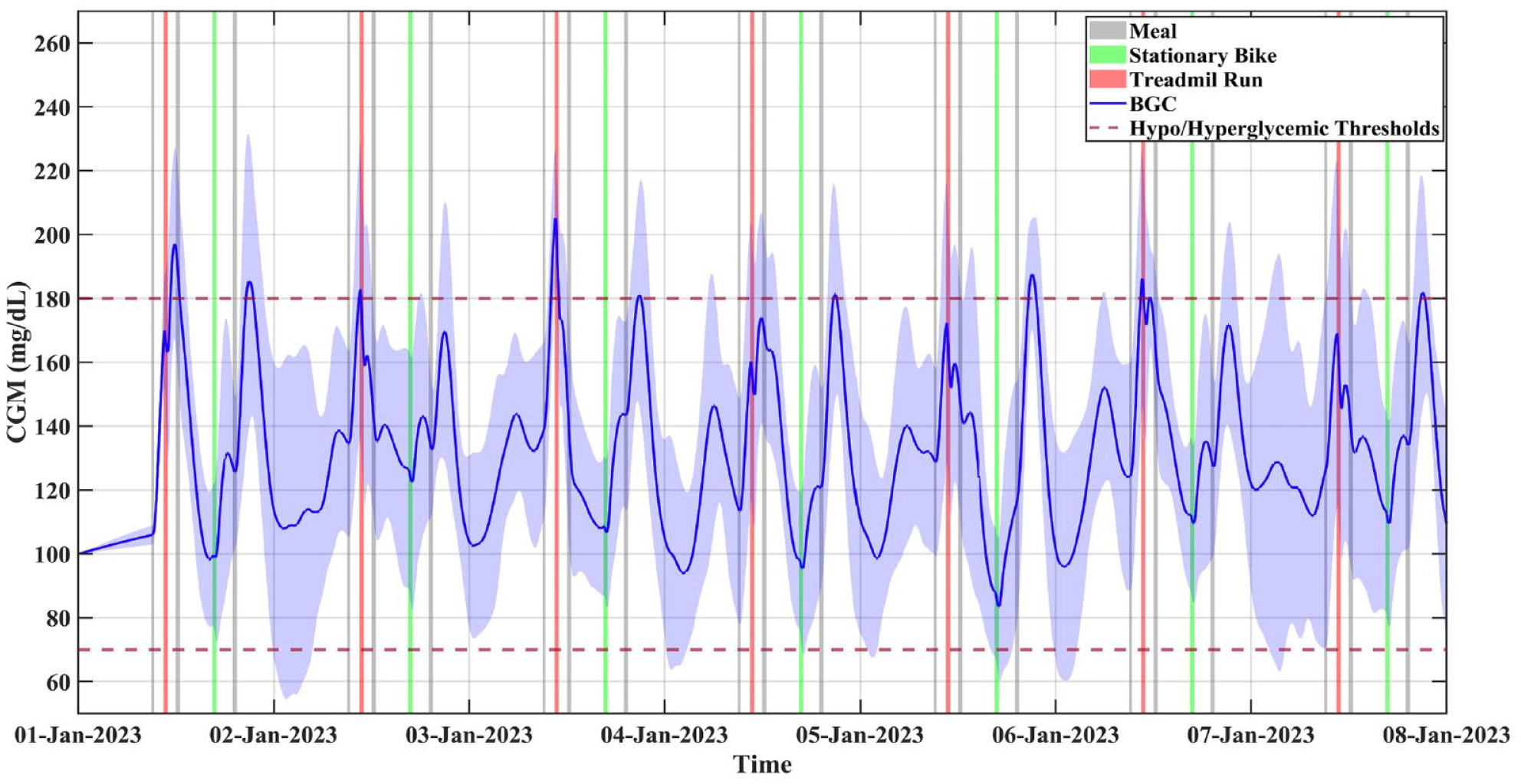
A closed-loop simulation study of ten subjects with unannounced inducement of PA and carbohydrate intake.
Exploratory Clinical Experiments
Exploratory clinical experiments are conducted to evaluate the safety and efficacy of our mvAID system. The exploratory study was conducted at the College of Nursing of the University of Illinois Chicago, and the study was approved by the Institutional Review Board (#2018-0989). The subjects were screened and after giving written informed consent completed questionnaires, performed vitals/physiological measurements and hemoglobin A1c test, and maximal exercise stress test. A typical closed-loop experiment consisted of physical activities (treadmill run or stationary bike) and meals. The exercise and CHO intake are unannounced to the mvAID. The exploratory study involves three clinical experiments, with one experiment conducted in a crossover fashion where the glucose-only single-variable is compared with the mvAID (Figure 8). The experiments included meals and medium-intensity PA (treadmill run and stationary bike) without any announcements to the mvAID system. The closed-loop clinical experiments of the mvAID system involved two subjects with T1D (one male and one female), with ages between 35 and 45 years, bodyweight 70 – 110 kg, total daily insulin of 35-80 U, and insulin-to-CHO ratio of 6.5-7 g/U. The mvAID system is operated similar to actual use in free-living conditions, without announcements of meals and PA to the system (Figures 8 and 9).
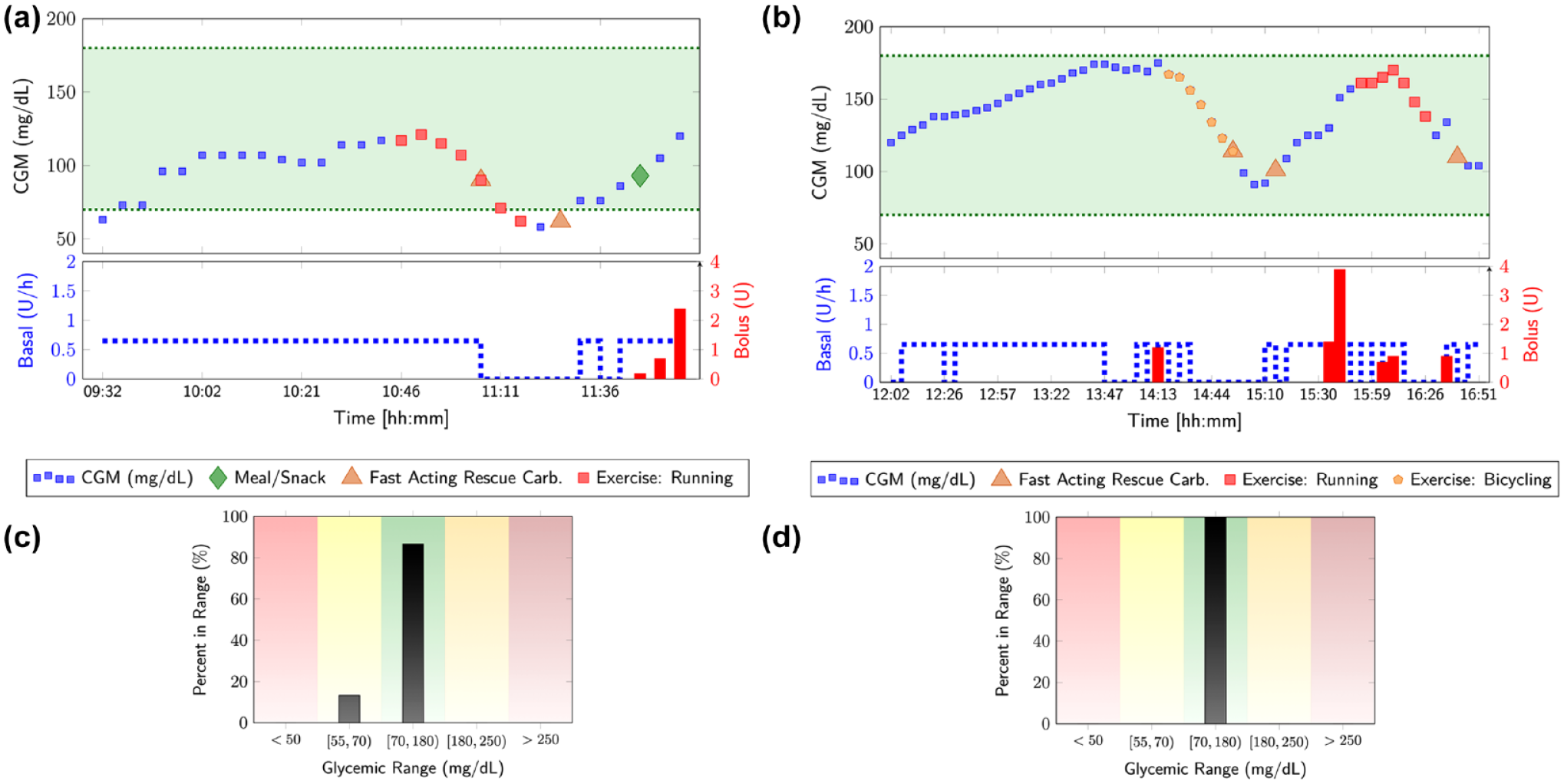
Crossover clinical experiment involving unannounced meals and physical activities with (a) glucose-only AP used in the morning and (b) mvAID used in the afternoon, showing the CGM measurements and administered basal and bolus insulin, with various glycemic events, such as meals, snacks, and physical activities indicated. The time spent in various glycemic ranges are summarized in (c) for the glucose-only AP and (d) for the mvAID system.
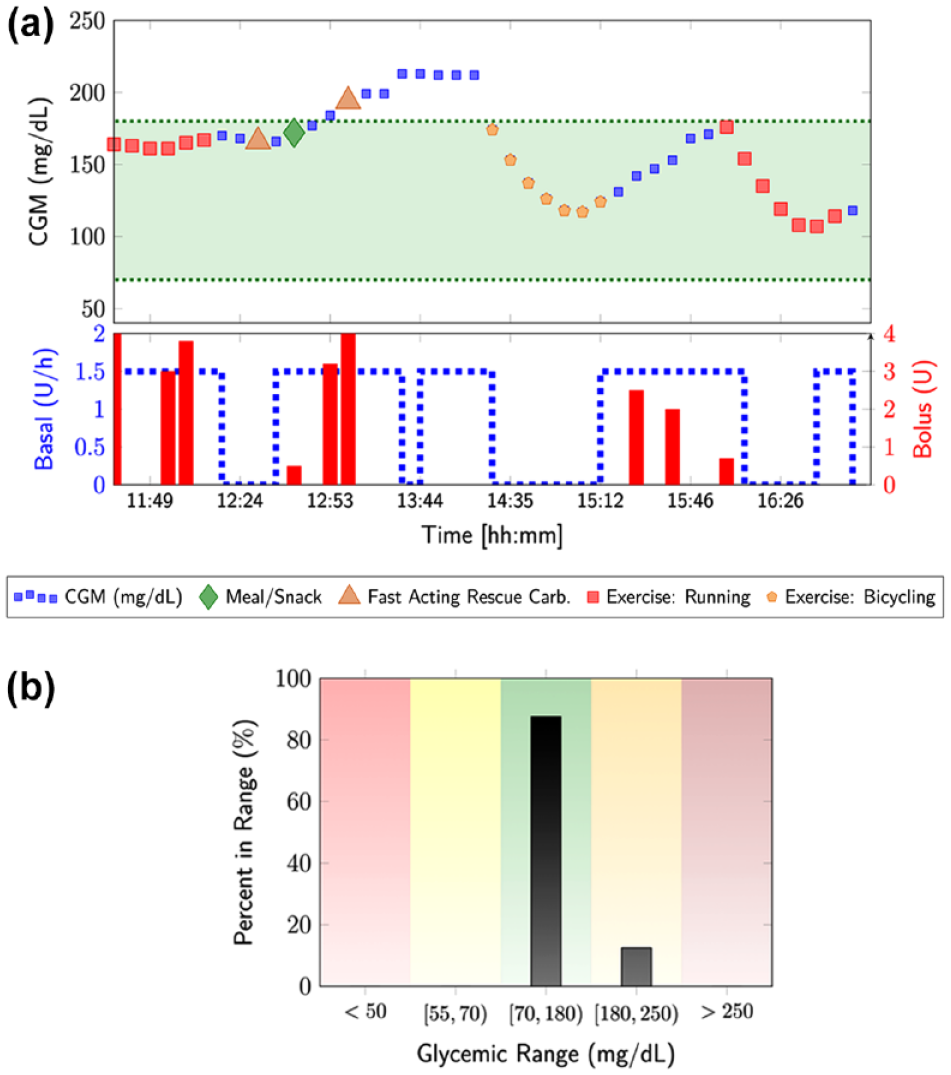
Clinical experiment involving unannounced meals and physical activities with the mvAID; (a) the CGM measurements and administered basal and bolus insulin, with various glycemic events, such as meals, snacks, and physical activities indicated and (b) the time spend in various glycemic ranges.
Crossover experiment: Glucose-only versus multivariable AID systems
A crossover experiment was conducted with the morning period involving closed-loop control with glucose-only (single-variable architecture) AP and the afternoon period involving mvAID that integrates additional physiological signals with the glucose measurements (Figure 8). The results show that the glucose-only AP is not able to detect the occurrence of the exercise until the BGC drops, thereby increasing the risk of hypoglycemia. In contrast, the mvAID detects the occurrence of the PA and evaluates its intensity using estimates of energy expenditure and incorporates this information in predictions of the future glucose trajectory profile to proactively modulate insulin dosing, which reduces the risk of hypoglycemia.
Experiment with multivariable AID system
A subject used the mvAID system during a full day of activities, including two bouts of exercise and meal consumption that were unannounced to the system. The pilot study results again showed the mvAID proactively detect the PA and reduced the basal insulin infusion rate to mitigate the glycemic effects of the PA. The first rescue carb followed by a meal prevented the drop in glucose levels after the morning run, but the combined effect of the two rescue carbs and meal caused a mild hyperglycemia that disappeared with the stationary bike PA in the afternoon. No insulin bolus was given after 1
Discussion of Results
This study showed the effectiveness of using additional physiological signals from a wristband device to handle unannounced glycemic disturbances in real-time. Results of previous in silico studies demonstrate that closed-loop control of BGC can be improved by integrating daily behavior and activity pattern information. Although planned exercise may be handled by considering historical trends and patterns, the capabilities of techniques that rely only on historical information are limited. Rather, the historical information can be synergistically incorporated with real-time estimation of disturbances, such as PA from additional physiological signals reported by the wristband device, which is more reliable approach for proactive control of BGC levels.
Developed modules include the multistep predictor of hypoglycemia/hyperglycemia, the safety constraint modules, CHO estimator modules, the state-space system identification module, and PIC estimator module, which are integrated to complete the feedback-feedforward features of the mvAID. The stability analysis of the system was studied using symbolic and numerical computations. A preliminary one-week in silico study of the proposed mvAID indicates more than 83% time in range calculated over all ten subjects. On average, a few number of hypoglycemic/hyperglycemic episodes were obtained as well by calculating < 4% and > 13% time-below-range and time-above-range, respectively.
The crossover clinical experiment (Figure 8) shows the controller performance effectiveness in the presence of different unannounced disturbance scenarios, including treadmill run, stationary bike, and CHO consumption. The closed-loop clinical experiment indicates the safety and efficacy of the mvAID and the improvement in performance over the single-variable AID system. The single-variable (CGM) AID system is not able to proactively take safe actions to mitigate the effects of unannounced disturbances, and as a result the AID system is more aggressive in infusing insulin and less capable in avoiding the occurrence of hypoglycemic events following unannounced PA. In contrast, the mvAID demonstrates well-controlled BGC because of the ability of automatically detecting and handling unannounced PA through the incorporation of additional physiological signals. One limitation of this work is the small number of exploratory clinical experiments conducted, though the pilot study was useful in demonstrating the safety and efficacy of the mvAID. Future studies will evaluate the mvAID in randomized controlled trials.
More research work in the future will be required to tailor the aggressiveness of the mvAID to individual subjects. The adaptive modeling of glucose-insulin dynamics and the adaptive MPC algorithm mean that performance will improve over time. This is a limitation of the current work, as the exploratory study had short duration that did not enable the testing and evaluation of the mvAID over long-term outpatient experiments, though the in silico simulation studies show the long-term performance of the adaptive learning approach and the mvAID for long time durations. The other improvements relate to the longer duration step-ahead prediction of BGC while the stability analysis of the state-space model is guaranteed.
Conclusion
An mvAID system that supplements the glucose concentration measurements with additional physiological signals from a wristband device to automatically handle unannounced PA is developed and successfully translated to exploratory clinical study. The in silico evaluation of the mvAID system and the exploratory clinical study involving unannounced PA and meals illustrate the safety and efficacy of the mvAID to handle unannounced glycemic disturbances, reduction of hypoglycemia risk during PA, and increase of time in range.
Footnotes
Acknowledgements
Financial support from NIH grants R01DK135116, 1R01DK130049 and 1DP3DK101075, and JDRF grants 2-SRA-2017-506-M-B and 3-APF-2022-1134-A-N made possible through collaboration between JDRF and The Leona M. and Harry B. Helmsley Charitable Trust are gratefully acknowledged.
Abbreviations
AID, automated insulin delivery; BGC, blood glucose concentration; CGM, continuous glucose monitoring; CHO, carbohydrate; KPLS, Kernel partial least squares; MDI, multiple insulin injection; MET, metabolic equivalents of task; MPC. model predictive control; mvAID, multivariable insulin delivery; NRLS, nonlinear recursive least squares; PA, physical activity; PIC, plasma insulin concentration; RNN, recurrent neural network; SAP, sensor augmented pump; T1D, type 1 diabetes; UKF, unscented Kalman filter.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH grants R01DK135116, 1R01DK130049 and 1DP3DK101075, and JDRF grants 2-SRA-2017-506-M-B and 3-APF-2022-1134-A-N made possible through collaboration between JDRF and The Leona M. and Harry B. Helmsley Charitable Trust.
