Abstract
Background:
The use of continuous subcutaneous insulin infusion (CSII) therapy in pregnancies affected by pregestational diabetes mellitus (DM) has generated mixed outcome data worthy of further investigation. This systematic review and meta-analysis aims to evaluate clinical outcomes associated with CSII versus multiple daily injections (MDIs) in pregnant persons with pregestational DM.
Methods:
A predefined, systematic, librarian-assisted search of MEDLINE (PubMed), Embase, Cochrane Library, Scopus, ClinicalTrials.gov, and World Health Organization International Clinical Trial Registry Platform (published from 2010 to 2022) yielded 3003 studies describing pregnancy outcomes associated with CSII and/or MDI for pregestational DM. The primary exposure was mode of insulin administration, with cesarean delivery and neonatal hypoglycemia as the primary maternal and neonatal outcomes, respectively. Secondary outcomes included hypertensive disorders of pregnancy, first and third-trimester glycemic control, large-for-gestational age (LGA) neonate, preterm birth, neonatal intensive care unit admission, need for respiratory support, hyperbilirubinemia, 5-minute Apgar <7, shoulder dystocia, and perinatal mortality. We calculated pooled odds ratios (OR) with 95% confidence intervals (CI) using random-effects models.
Results:
Among 39 eligible studies, 39% of the 5518 pregnancies included were exposed to CSII. Odds of cesarean delivery were higher with CSII (20 studies: 63% vs 56%, odds ratio [OR] 1.3 [95% confidence interval (CI) 1.2-1.5]), but we did not identify a difference in the odds of neonatal hypoglycemia (23 studies: 31% vs 34%, OR 1.1 [95% CI 0.9-1.5]). Among secondary outcomes, only the odds of LGA (20 studies: 47% vs 38%, OR 1.4 [95% CI 1.2-1.6]) were higher in individuals using CSII versus MDI.
Conclusions:
Use of CSII (vs MDI) for pregestational DM in pregnancy is associated with higher odds of cesarean delivery and delivery of an LGA neonate. Further evaluation of how CSII use may influence neonatal size and delivery route is warranted.
Keywords
Introduction
Approximately 35% of new cases of diabetes mellitus (DM) diagnosed each year in the United States occurs in reproductive-aged pregnancy-capable persons, and the prevalence of both type 1 (T1DM) and type 2 diabetes mellitus (T2DM) in this population has continued to increase globally in recent years.1-4 Pregestational DM, encompassing both T1DM and T2DM, affects approximately 1% to 2% of all pregnancies.1,3 Pregnant persons with pregestational DM, especially with suboptimal glycemic control (hemoglobin A1c ≥ 6.5%), carry higher risk of preterm birth (PTB), cesarean delivery (CD), hypertensive disorders of pregnancy (HDP), progression of long-term diabetic complications, and severe perineal lacerations.1,3,5 Their neonates experience higher rates of large-for-gestational age (LGA), shoulder dystocia, neonatal hypoglycemia (typically defined as blood glucose less than 40 mg/dL during the first 24 hours of life), neonatal intensive care unit (NICU) admission, respiratory distress syndrome (RDS) or transient tachypnea of the newborn (TTN), hyperbilirubinemia, congenital anomalies, and intrauterine fetal demise (IUFD).3,5
The state of insulin resistance in pregnancy is driven by placental hormones that increase in concentration throughout pregnancy. As insulin resistance accelerates with advancing gestational age, significantly higher doses of insulin are required after the first trimester of pregnancy to mitigate hyperglycemia.1,3,6 To reduce maternal and neonatal morbidity associated with both hyperglycemia and severe hypoglycemic events, intensive surveillance and medication titration to maintain euglycemia is required throughout pregnancy.3,6 As the gold standard for treatment of pregestational DM in pregnancy, insulin has traditionally been administered with multiple daily injections (MDI) of both basal and prandial bolus insulin. 6 Recently, however, increasing numbers of pregnant persons with pregestational DM enter pregnancy using continuous subcutaneous insulin infusion (CSII) therapy. With the technology rapidly advancing, CSII therapy administers a continuous basal infusion that can be programmed at varying rates throughout the day, calculates prandial bolus doses with matched insulin to carbohydrate ratios, and may interface with a continuous glucose monitor (CGM) to provide a personalized, automated system.5,7-9
Although both MDI and CSII allow for frequent insulin dose titration in pregnancy, data from observational cohort studies, randomized controlled trials (RCT), and previous meta-analyses comparing these methods, including with and without the use of CGM, suggest inconclusive evidence regarding the optimal antenatal insulin administration modality.5,10 A 2016 Cochrane Review concluded data from available trials were insufficient to recommend CSII versus MDI in pregnancy, and called for more research using the latest pump technology and insulins to assess perinatal outcomes. 11 Therefore, we conducted a contemporary systematic review and meta-analysis of randomized and observational studies to evaluate maternal and neonatal outcomes associated with CSII versus MDI in pregnant persons with pregestational DM.
Methods
We used a prespecified methodology according to guidelines for Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA, Supplemental Appendix 1) and Meta-analyses of Observational Studies in Epidemiology (MOOSE, Supplemental Appendix 2).12,13 The study protocol was registered with PROSPERO (#CRD42022313357) prior to search performance.
Information Sources and Search Strategy
A medical librarian searched published literature for records discussing pregestational diabetes (ie, T1DM or T2DM), insulin administration modality (CSII or MDI), and maternal and neonatal outcomes. The librarian created search strategies using a combination of keywords and controlled vocabulary in Medline (1946 to present), Embase.com (1947 to present), Scopus (1823 to present), Cochrane Database of Systematic Reviews (CDSR), Cochrane Central Register of Controlled Trials (CENTRAL), Clinicaltrials.gov (1997 to present), and World Health Organization International Clinical Trial Registry Portal (WHO ICTRP). All search strategies were completed initially in March 2022, and 3003 results were exported to EndNote (Clarivate, Philadelphia, PA). Of these, 1292 records were removed after using the de-duplication processes described by Bramer et al, 14 after which 1,711 unique records remained. An additional 12 records were identified in ClinicalTrials.gov and eight in WHO ICTRP. A manual search of bibliographies of relevant articles was also performed. All references were exported to an Excel workbook for review. Fully reproducible search strategies for each database can be found in Supplemental Appendix 3.
Eligibility Criteria and Study Selection
Two investigators (AYD and NYX) independently screened abstracts and articles pertaining to mode of insulin administration and perinatal outcomes and extracted data from each study. The corresponding author for each study was contacted via email in an attempt to obtain missing data for primary and secondary outcomes. Discrepancies in coding required agreement between authors (SAF, JH, AYD, and NYX) to be considered resolved.
Studies were included if they reported on pregnant persons with pregestational DM who received treatment with insulin, either by CSII or MDI, throughout pregnancy, and reported on the maternal or neonatal outcomes of interest described below. Studies were excluded if they represented a conference proceeding or abstract only, included nonhuman, animal fetuses, represented an ongoing study or study results were not available, were not available in English or Spanish, were a review article, or included duplicate data previously reported in another publication. In addition, studies were excluded from the meta-analysis if they did not report raw data for the included maternal and/or neonatal outcomes of interest.
Data Extraction
The primary exposure was mode of insulin administration. As a significant contributor to maternal morbidity, CD was chosen as the primary maternal outcome. The primary neonatal outcome was neonatal hypoglycemia (defined in most studies as blood glucose <40-45 mg/dL, 2.2-2.6 mmol/L, or requiring treatment with intravenous glucose infusion). Secondary outcomes included pregnancy loss under 20 weeks’ gestation (spontaneous abortion, or induced abortion or ectopic pregnancy in select studies), gestational age at delivery, PTB (prior to 37 weeks’ gestation), gestational weight gain (GWG), HDP (including gestational hypertension or preeclampsia, with or without severe features), birthweight (in grams), SGA (birthweight <10th percentile), LGA (birthweight >90th percentile), macrosomia (birthweight >4000g), shoulder dystocia, NICU admission, 5-minute Apgar <7, RDS or TTN, hyperbilirubinemia or jaundice, congenital anomalies, perinatal death (either IUFD or neonatal death), and other delivery complications (including severe perineal lacerations, maternal infection, other neonatal birth injury).
For each study, when data were available, we extracted mean or median maternal age at delivery, maternal gravidity and/or parity, pregnancy planning status, body mass index (BMI), history of long-term hypertension, duration of pregestational DM diagnosis, complications of pregestational DM (nephropathy with microalbuminuria or macroalbuminuria, retinopathy, neuropathy, and diabetic ketoacidosis in pregnancy), hemoglobin A1c (prepregnancy and in the first and third trimesters of pregnancy), severe hypoglycemia (eg, having an episode of hypoglycemia during pregnancy that required another person’s aid to achieve recovery), total daily dose of insulin per trimester, and type of basal and prandial insulin. Each of the aforementioned variables was described by the number of pregnant persons who received CSII versus MDI.
Data Synthesis
Meta-analysis was performed using the metan add-on program in Stata version 16.1 (StataCorp, College Section, TX, USA). Two-by-two contingency tables were created to compare the presence or absence of each adverse maternal or neonatal outcome stratified by mode of insulin administration. We calculated pooled odds ratios (OR) with 95% confidence intervals (CI). Random-effects models were used to account for clinical heterogeneity between studies, even when statistical heterogeneity was not evident. Forest plots were created to visually assess both effect size and identify outliers. For the primary maternal and neonatal outcomes, we also performed a sensitivity analysis including only those studies in which MDI or CSII were specifically initiated preconception and continued for the duration of pregnancy without treatment crossover.
We estimated heterogeneity across studies and tested its significance using the Higgins I2 statistic and Cochrane’s Q test. I2 of 50% was considered evidence of significant heterogeneity. Publication bias was evaluated visually using funnel plots for the primary maternal and neonatal outcomes, in addition to any significant secondary outcomes, and asymmetry was tested statistically using Egger’s test. This study was deemed exempt from Institutional Review Board review at Northwestern University.
Assessment of Risk of Bias and Quality of the Evidence
Quality assessment to determine risk of bias of included studies was also performed using the Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) tool. 15 Based on seven domains that could introduce potential biases, the ROBINS-I tool classifies whether observational studies have overall low, moderate, serious, or critical risk of bias in their estimates of the comparative effectiveness of interventions. Studies were excluded from the meta-analysis if they were at serious or critical risk of bias based on the ROBINS-I tool.
Finally, we assessed the quality of the evidence for key maternal and neonatal outcomes of interest assessed within the meta-analysis using the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) approach. 16 Certainty of the evidence for each outcome was determined to be very low, low, moderate, or high quality based on the following five domains that could downgrade study quality: risk of bias, imprecision, inconsistency, indirectness, and publication bias. Certainty of the evidence could be rated up for having a large magnitude of effect, presence of a dose-response gradient, or if all residual confounding would decrease the magnitude of effect. 17
Results
Study Selection
The search yielded 3003 articles published from 1970 to 2022. Six prospective observational cohort studies (including two secondary analyses of randomized controlled trials evaluating CGM therapy) and 33 retrospective cohort studies published from 2010 to 2022 met inclusion criteria and were selected for the systematic review (Figure 1 and Table 1).8,18-55 In aggregate, the 39 studies included in the systematic review included 7528 pregnancies.
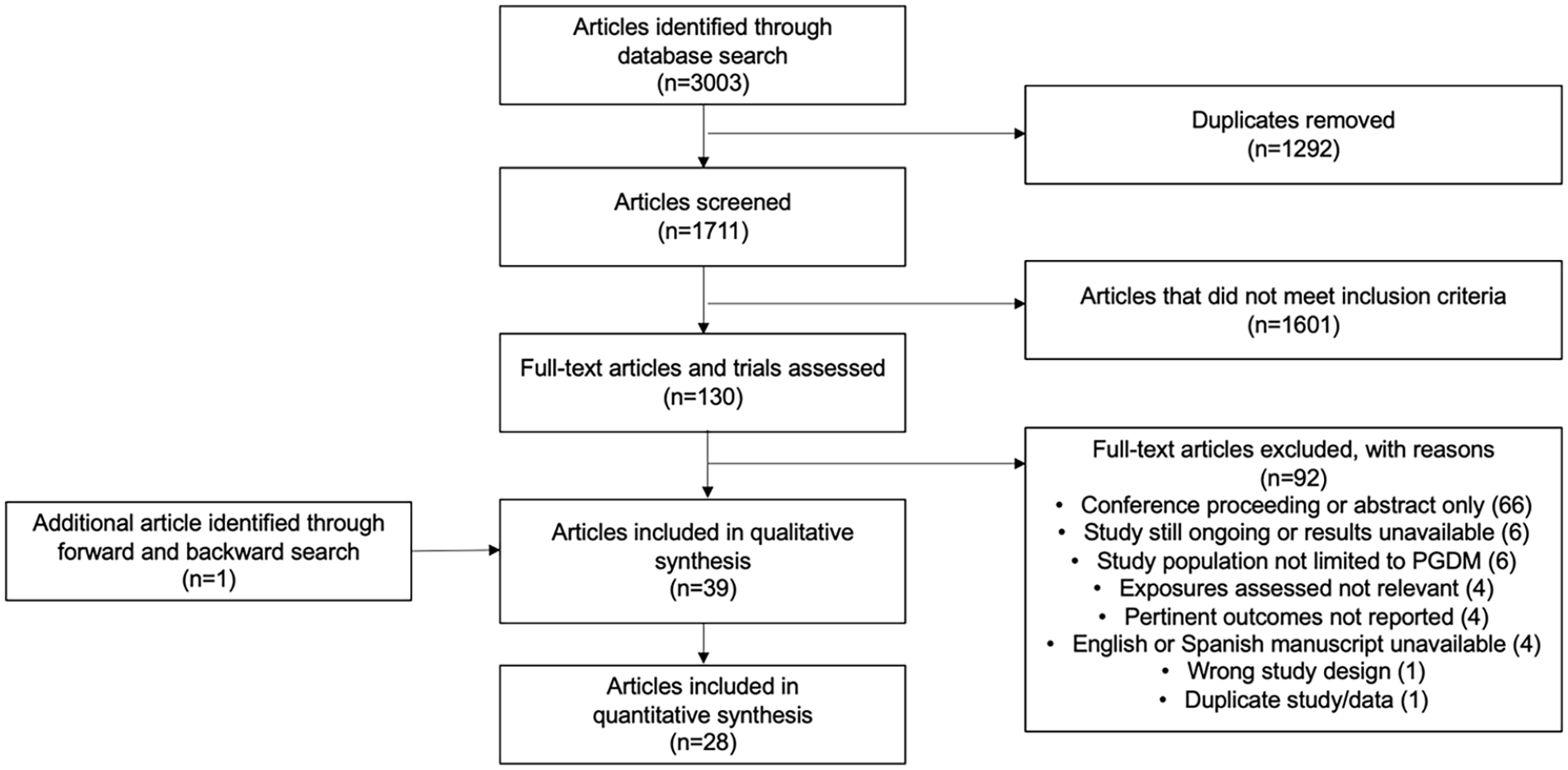
Flow chart of the literature review.
Baseline Study Characteristics.
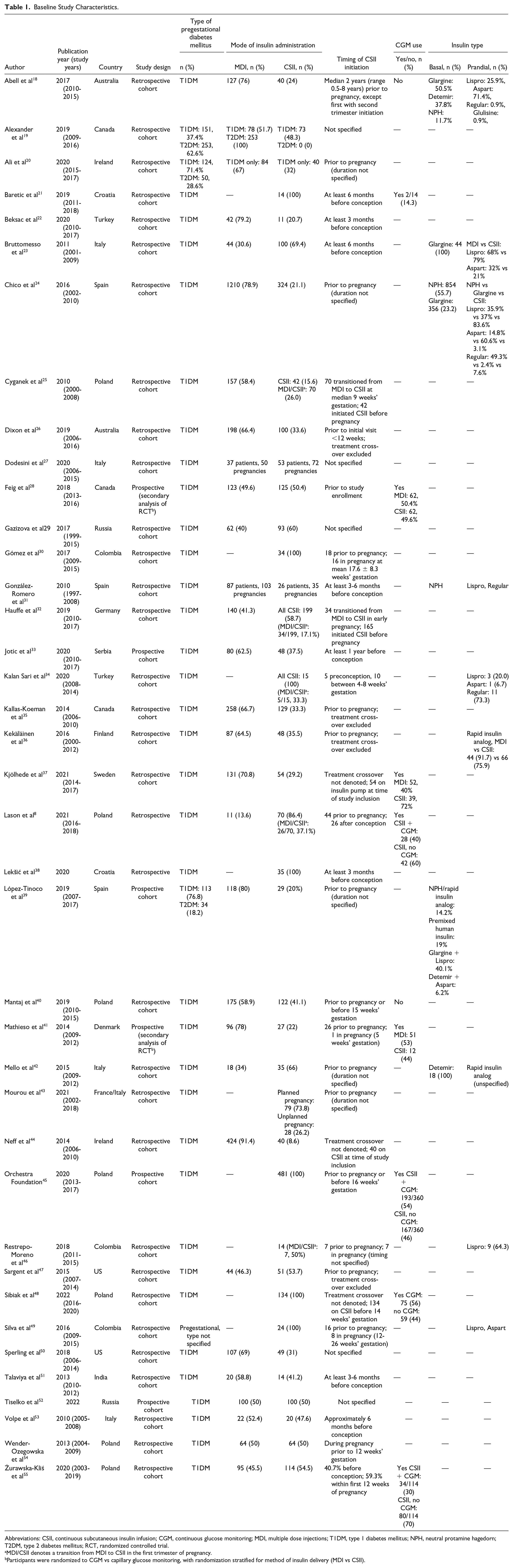
Abbreviations: CSII, continuous subcutaneous insulin infusion; CGM, continuous glucose monitoring; MDI, multiple dose injections; T1DM, type 1 diabetes mellitus; NPH, neutral protamine hagedorn; T2DM, type 2 diabetes mellitus; RCT, randomized controlled trial.
MDI/CSII denotes a transition from MDI to CSII in the first trimester of pregnancy.
Participants were randomized to CGM vs capillary glucose monitoring, with randomization stratified for method of insulin delivery (MDI vs CSII).
Study Characteristics and Systematic Review
Among included studies, 35 evaluated only individuals with T1DM, while four studies also included those with T2DM.19,39,49,56 A total of 3196 (42.5%) pregnancies employed CSII therapy, while 4332 (57.5) used MDI. Eight studies reported on concomitant use of CGM.8,21,28,37,41,48,55,45 Ten studies reported on type of basal and/or prandial insulin used, typically neutral protamine hagedorn (NPH) insulin, glargine, or detemir for basal insulin and lispro, aspart, or regular insulin for prandial insulin (Table 1).18,23,24,31,34,36,39,42,46,49 Maternal characteristics with respect to maternal age, parity, BMI, long-term hypertension, complications related to pregestational DM, and glycemic control are available in Supplementary Tables 1 to 3. Maternal age and BMI appear similar between users of CSII and MDI, though in many studies, those using CSII had pregestational DM for more years and higher rates of microvascular complications than those using MDI. In addition, 12 studies reported on tobacco use,18,24,26,28,30,32,35-37,41,50,55 four studies reported on preconception folic acid intake,24,26,28,56 six studies reported on individuals having a history of diabetic neuropathy,26,28,42,46,52,45 two studies reported on need for hospitalization for glycemic control,22,26 seven studies reported on change in hemoglobin A1c from the first to third trimester,27,28,42,45,46,48,55 and four studies reported on the change in daily insulin dose from the first to third trimester.18,21,36,55
Tables 2 and 3 detail obstetric outcomes and other outcomes related to neonatal size (eg, birthweight, SGA, LGA, macrosomia, and shoulder dystocia) according to insulin administration modality, with 32 studies reporting on frequency of CD as the primary maternal outcome of interest. For secondary obstetric outcomes (Table 2), 17 studies reported on pregnancy loss prior to 20 weeks’ gestation, 26 studies reported on PTB (including five studies reporting on PTB prior to 34 weeks’ gestation), 22 studies reported on GWG, and 24 studies reported on HDP. For the outcomes related to neonatal size (Table 3), 32 studies reported mean or median neonatal birthweight, while 34 studies specifically reported if neonatal birthweight met criteria for SGA, LGA, and/or macrosomia. In addition, 11 studies reported on shoulder dystocia.
Maternal and Other Obstetric Outcomes.
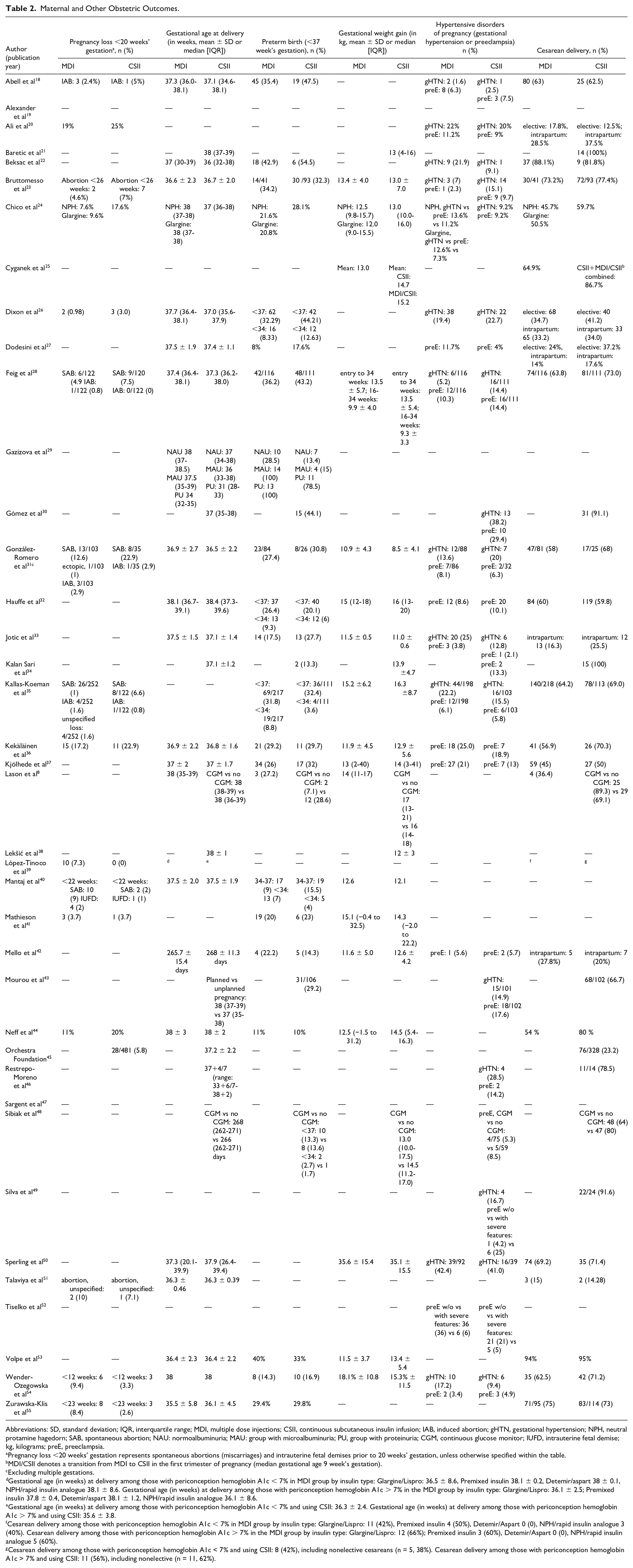
Abbreviations: SD, standard deviation; IQR, interquartile range; MDI, multiple dose injections; CSII, continuous subcutaneous insulin infusion; IAB, induced abortion; gHTN, gestational hypertension; NPH, neutral protamine hagedorn; SAB, spontaneous abortion; NAU: normoalbuminuria; MAU: group with microalbuminuria; PU, group with proteinuria; CGM, continuous glucose monitor; IUFD, intrauterine fetal demise; kg, kilograms; preE, preeclampsia.
Pregnancy loss <20 weeks’ gestation represents spontaneous abortions (miscarriages) and intrauterine fetal demises prior to 20 weeks’ gestation, unless otherwise specified within the table.
MDI/CSII denotes a transition from MDI to CSII in the first trimester of pregnancy (median gestational age 9 week’s gestation).
Excluding multiple gestations.
Gestational age (in weeks) at delivery among those with periconception hemoglobin A1c < 7% in MDI group by insulin type: Glargine/Lispro: 36.5 ± 8.6, Premixed insulin 38.1 ± 0.2, Detemir/aspart 38 ± 0.1, NPH/rapid insulin analogue 38.1 ± 8.6. Gestational age (in weeks) at delivery among those with periconception hemoglobin A1c > 7% in the MDI group by insulin type: Glargine/Lispro: 36.1 ± 2.5; Premixed insulin 37.8 ± 0.4, Detemir/aspart 38.1 ± 1.2, NPH/rapid insulin analogue 36.1 ± 8.6.
Gestational age (in weeks) at delivery among those with periconception hemoglobin A1c < 7% and using CSII: 36.3 ± 2.4. Gestational age (in weeks) at delivery among those with periconception hemoglobin A1c > 7% and using CSII: 35.6 ± 3.8.
Cesarean delivery among those with periconception hemoglobin A1c < 7% in MDI group by insulin type: Glargine/Lispro: 11 (42%), Premixed insulin 4 (50%), Detemir/Aspart 0 (0), NPH/rapid insulin analogue 3 (40%). Cesarean delivery among those with periconception hemoglobin A1c > 7% in the MDI group by insulin type: Glargine/Lispro: 12 (66%); Premixed insulin 3 (60%), Detemir/Aspart 0 (0), NPH/rapid insulin analogue 5 (60%).
Cesarean delivery among those with periconception hemoglobin A1c < 7% and using CSII: 8 (42%), including nonelective cesareans (n = 5, 38%). Cesarean delivery among those with periconception hemoglobin A1c > 7% and using CSII: 11 (56%), including nonelective (n = 11, 62%).
Outcomes Related to Neonatal Size.
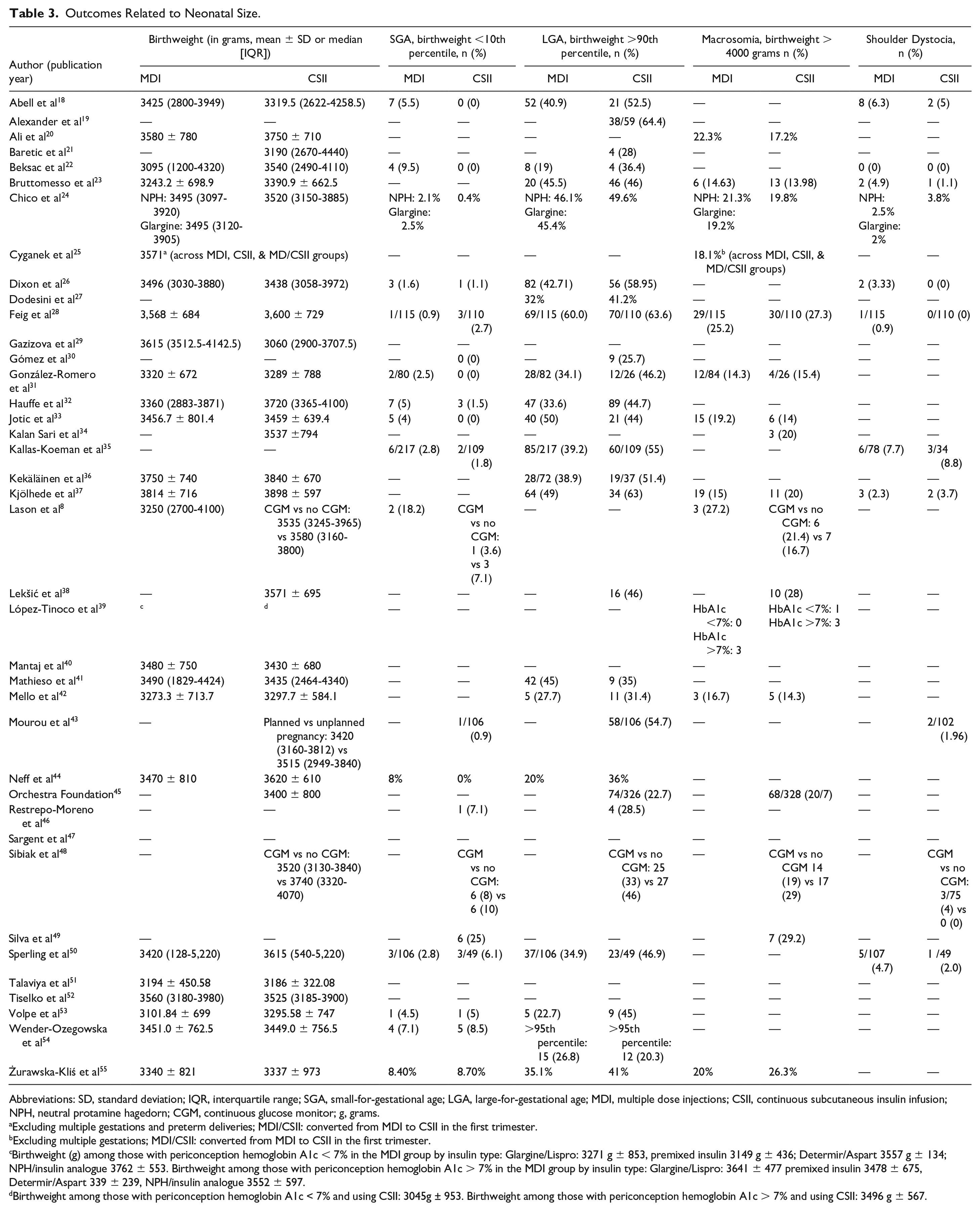
Abbreviations: SD, standard deviation; IQR, interquartile range; SGA, small-for-gestational age; LGA, large-for-gestational age; MDI, multiple dose injections; CSII, continuous subcutaneous insulin infusion; NPH, neutral protamine hagedorn; CGM, continuous glucose monitor; g, grams.
Excluding multiple gestations and preterm deliveries; MDI/CSII: converted from MDI to CSII in the first trimester.
Excluding multiple gestations; MDI/CSII: converted from MDI to CSII in the first trimester.
Birthweight (g) among those with periconception hemoglobin A1c < 7% in the MDI group by insulin type: Glargine/Lispro: 3271 g ± 853, premixed insulin 3149 g ± 436; Determir/Aspart 3557 g ± 134; NPH/insulin analogue 3762 ± 553. Birthweight among those with periconception hemoglobin A1c > 7% in the MDI group by insulin type: Glargine/Lispro: 3641 ± 477 premixed insulin 3478 ± 675, Determir/Aspart 339 ± 239, NPH/insulin analogue 3552 ± 597.
Birthweight among those with periconception hemoglobin A1c < 7% and using CSII: 3045g ± 953. Birthweight among those with periconception hemoglobin A1c > 7% and using CSII: 3496 g ± 567.
Table 4 reports on additional neonatal outcomes, with 31 studies reporting on neonatal hypoglycemia as the primary neonatal outcome of interest. For secondary neonatal outcomes, 21 studies reported on NICU admission, eight studies reported on 5-minute Apgar scores < 7,18,22,24,26,32,37,50,55 two studies reported on neonatal cord gases,24,54 14 studies reported on RDS or TTN, 17 studies reported on hyperbilirubinemia or jaundice, 25 studies reported on congenital anomalies, and 22 studies reported on perinatal death. Lastly, supplementary Table 4 demonstrates the seven studies that reported on other delivery complications (eg, severe perineal lacerations, maternal infection, and/or birth injury).
Additional Neonatal Outcomes.
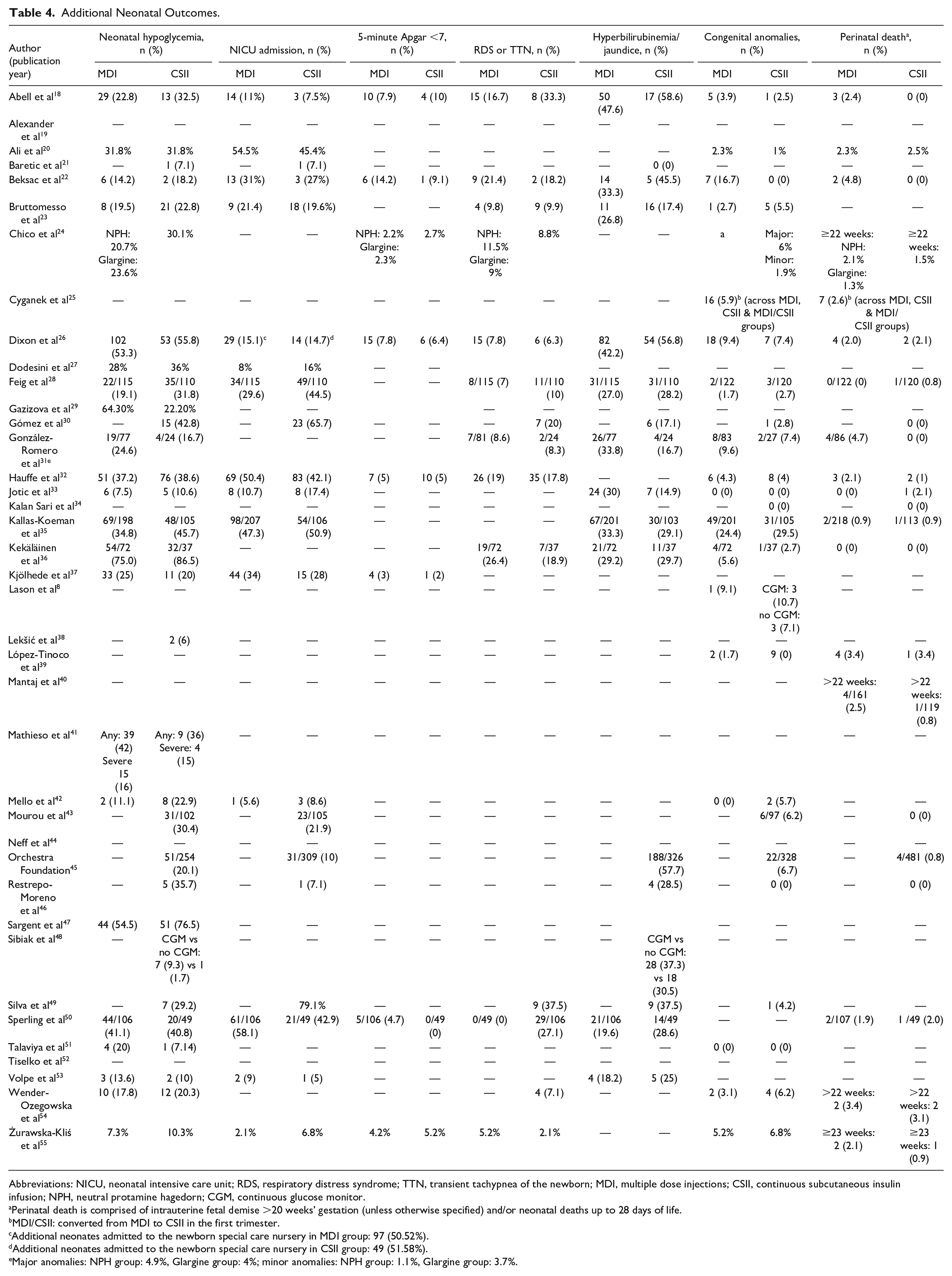
Abbreviations: NICU, neonatal intensive care unit; RDS, respiratory distress syndrome; TTN, transient tachypnea of the newborn; MDI, multiple dose injections; CSII, continuous subcutaneous insulin infusion; NPH, neutral protamine hagedorn; CGM, continuous glucose monitor.
Perinatal death is comprised of intrauterine fetal demise >20 weeks’ gestation (unless otherwise specified) and/or neonatal deaths up to 28 days of life.
MDI/CSII: converted from MDI to CSII in the first trimester.
Additional neonates admitted to the newborn special care nursery in MDI group: 97 (50.52%).
Additional neonates admitted to the newborn special care nursery in CSII group: 49 (51.58%).
Major anomalies: NPH group: 4.9%, Glargine group: 4%; minor anomalies: NPH group: 1.1%, Glargine group: 3.7%.
Meta-Analysis and Synthesis of Results
Eleven studies reported on maternal and neonatal outcomes associated with CSII, but did not include an MDI comparator arm, and thus were not included in the meta-analysis.19,21,30,34,38,39,43,45,46,48,49 The remaining 28 studies compared perinatal outcomes according to each insulin administration modality among 5518 pregnant persons (3282 [59.5%] MDI vs 2,236 [40.5%] CSII) with pregestational DM and were quantitatively synthesized in the meta-analysis.8,18,20,22-29,31-33,35-37,40-42,47,50-55
For the primary maternal outcome, CSII therapy was associated with greater odds of CD (20 studies: 63.24% vs 56.49%, OR 1.33 [95% CI 1.15-1.54]; Figure 2), with no significant heterogeneity between studies (I2 = 0.0%, P = .58). We confirmed the robustness of this positive association between CSII and CD in our sensitivity analysis that excluded studies in which participants had crossover between the insulin administration modalities during pregnancy (12 studies: 64.8% versus 59.1%, OR 1.29 [95% CI 1.09, 1.53]; I2 = 0.0%, P = .99, Supplementary Figure 1).
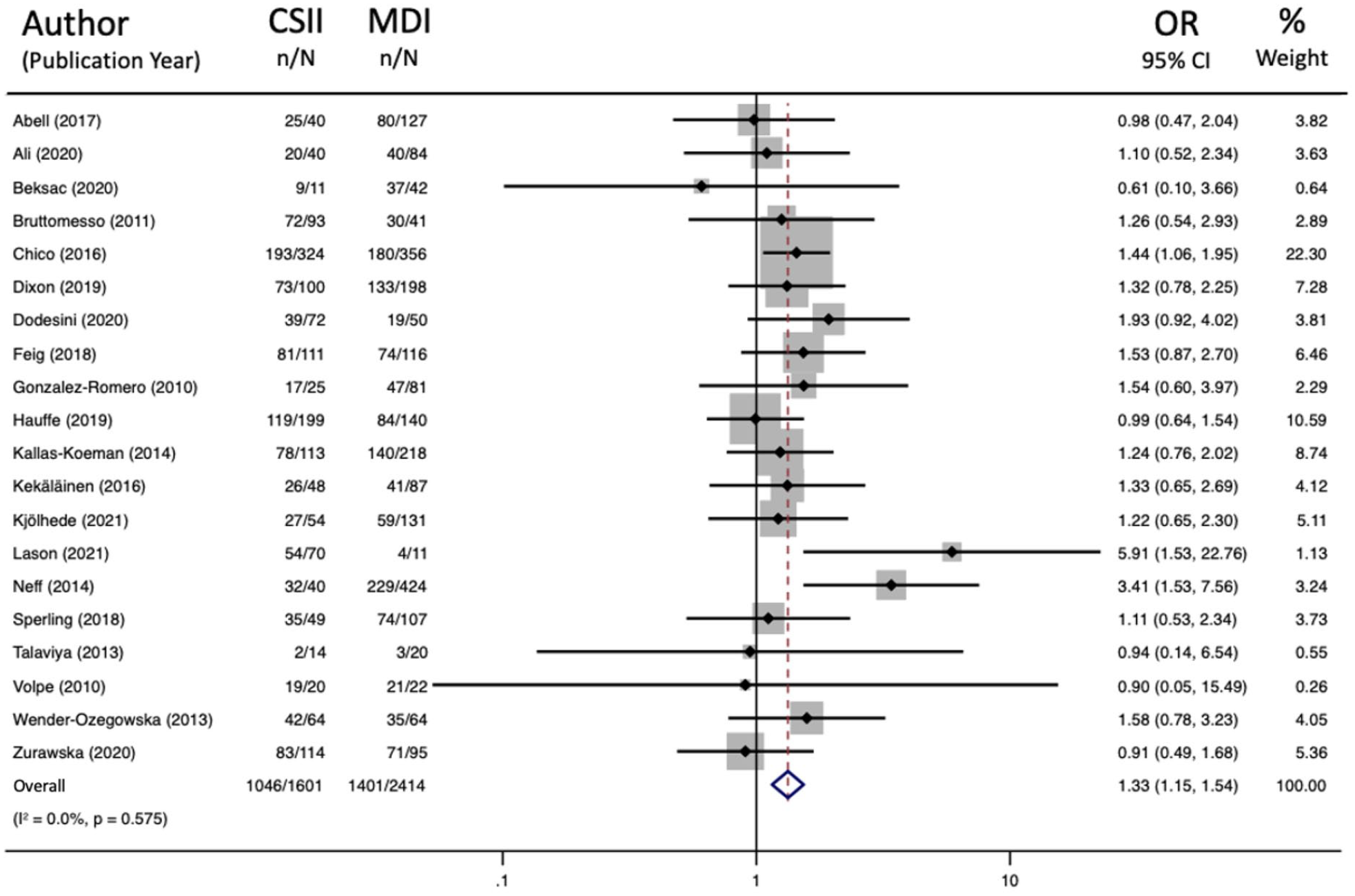
Forest plot for cesarean delivery.
For the primary neonatal outcome, CSII therapy was associated with similar odds of neonatal hypoglycemia (23 studies: 31.02% vs 32.60%, OR 1.14 [95% CI 0.99-1.45]; Figure 3) as MDI, although with significant heterogeneity between studies (I2 = 51.8%, P = .002). After performing our sensitivity analysis of studies in which the respective insulin administration modality was initiated preconception and continued throughout pregnancy without treatment crossover, we identified that CSII therapy was associated with greater odds of neonatal hypoglycemia (15 studies: 35.7% vs 31.9%, OR 1.33 [95% CI 1.11-1.59]; Supplementary Figure 2), with no significant heterogeneity between studies (I2 = 0.0%, P = .58).
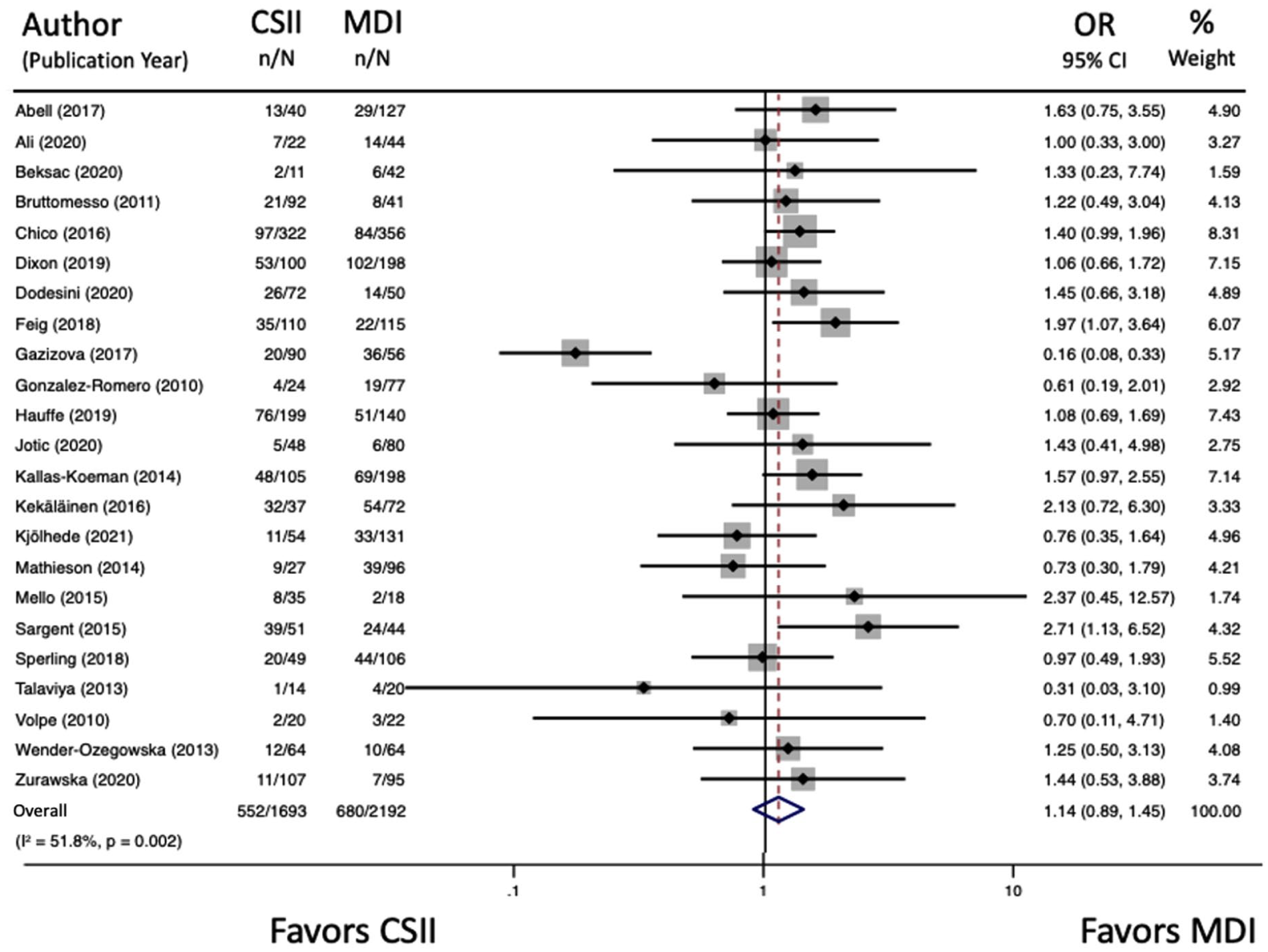
Forest plot for neonatal hypoglycemia.
Regarding the secondary outcomes assessed, the odds of PTB (22 studies: 27.42% vs 24.32%, OR 1.08 [95% CI 0.87-1.33]; I2 = 43.5%, P = .016, Supplementary Figure 3), odds of preeclampsia (15 studies: 9.99% vs 10.66%, OR 0.90 [0.69-1.17]; I2 = 4.7%, P = .40, Supplementary Figure 4), and standardized mean difference (SMD) in GWG (15 studies: 0.12 [95% CI −0.14, 1.22]; I2 = 91.8%, Supplementary Figure 5) were similar with CSII and MDI, respectively. However, CSII therapy was associated with greater odds of LGA (20 studies: 46.66% vs 37.47%, OR 1.35 [95% CI 1.18-1.55]; Supplementary Figure 6) when compared with MDI, with no significant heterogeneity between studies (I2 = 0.0%, P = .63). To test the robustness of this finding, we performed an additional post-hoc sensitivity analysis excluding studies in which participants had crossover between the insulin administration modalities during pregnancy, and the positive association between CSII and LGA remained significant (12 studies: 49.3% vs 41.8%, OR 1.34 [95% CI 1.13-1.58]; I2 = 0.0%, P = .52, Supplementary Figure 7).
The odds of all other secondary neonatal outcomes were similar with the use of CSII and MDI (Supplementary Figures 8–13). Finally, glycemic control in the third trimester, as assessed by hemoglobin A1c, was similar among both insulin administration modalities (Supplementary Figures 14–15), although first trimester glycemic control was slightly better with CSII compared to MDI use (SMD −0.45 [95% CI −0.90, −0.01]).
We did not identify evidence of publication bias with respect to CD and its association with mode of insulin administration (Egger’s P = .73; Figure 4). We also did not identify evidence of publication bias with respect to neonatal hypoglycemia (Egger’s P = .24; Figure 5) or LGA (Egger’s P = .82; Supplementary Figure 16), according to mode of insulin administration.
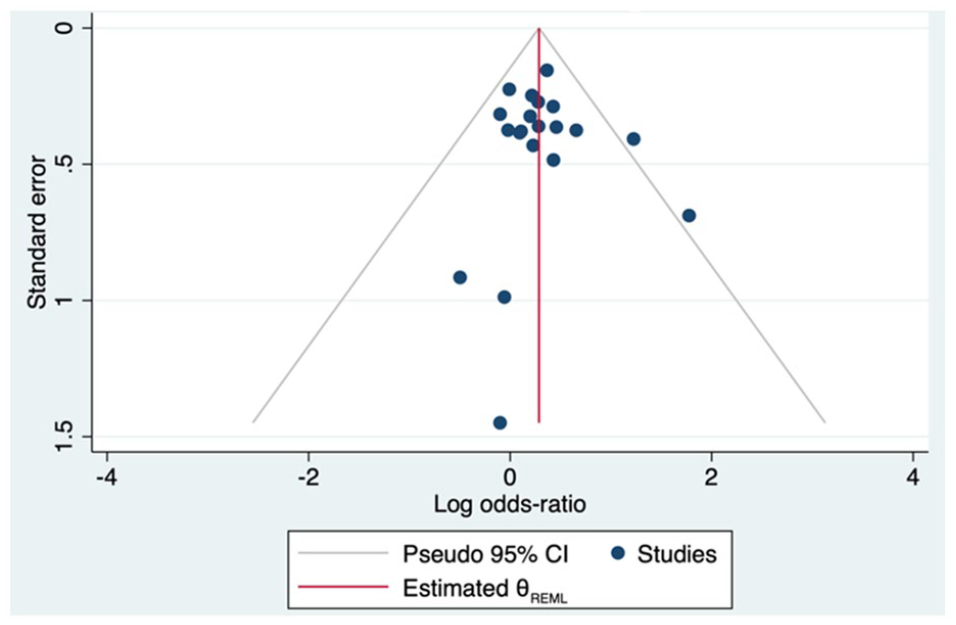
Funnel plot of publication bias for cesarean delivery.
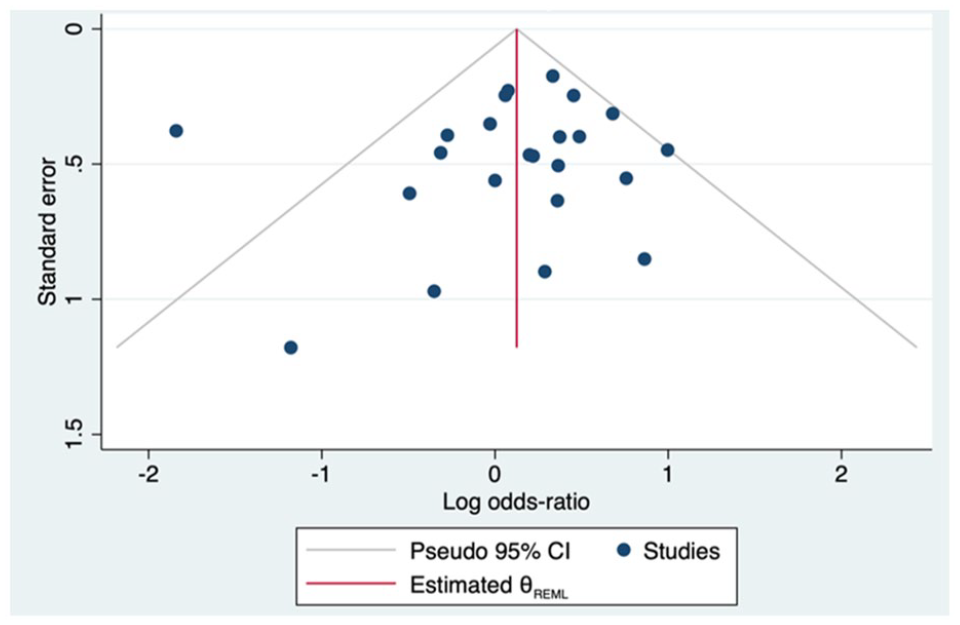
Funnel plot of publication bias for neonatal hypoglycemia.
Risk of Bias of Included Studies and Certainty of Evidence Assessment
Quality assessment with the ROBINS-I tool identified 31 observational studies to have moderate risk of bias, with four studies at serious risk of bias (Supplementary Table 5). Study quality was downgraded most commonly due to limited ability to control for confounding factors or evidence of selection bias, largely due to the observational nature and small sample sizes. Four studies were classified as having low risk of bias.24,28,40,41 In the certainty of evidence assessment using the GRADE approach (Table 5), the quality of evidence was determined to be low or very low for all outcomes assessed in the meta-analysis, largely due to risk of bias, inconsistency, and imprecision among studies that reported on these key outcomes.
Summary of Meta-Analysis Findings and Certainty of Evidence Assessment.
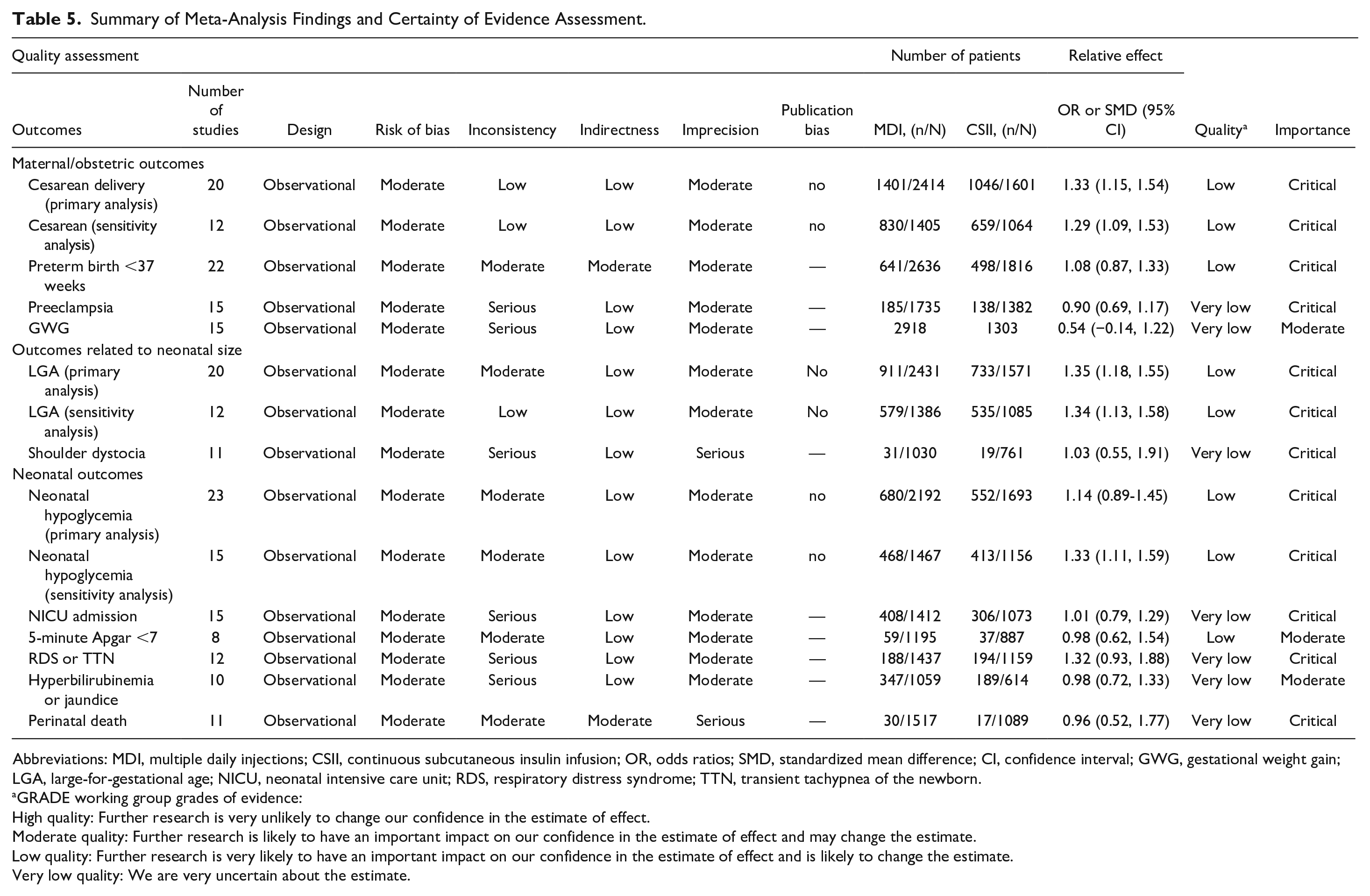
Abbreviations: MDI, multiple daily injections; CSII, continuous subcutaneous insulin infusion; OR, odds ratios; SMD, standardized mean difference; CI, confidence interval; GWG, gestational weight gain; LGA, large-for-gestational age; NICU, neonatal intensive care unit; RDS, respiratory distress syndrome; TTN, transient tachypnea of the newborn.
GRADE working group grades of evidence:
High quality: Further research is very unlikely to change our confidence in the estimate of effect.
Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate.
Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate.
Very low quality: We are very uncertain about the estimate.
Discussion
Main Findings and Comparison With Existing Literature
In this large, contemporary systematic review and meta-analysis of pregnant persons with pregestational DM, we found that CSII use was associated with higher odds of CD and delivery of an LGA neonate, and a modest improvement in first trimester hemoglobin A1c, compared to that observed with MDI use. We did not detect a difference between outcomes of CSII versus MDI use with respect to neonatal hypoglycemia overall, but we did identify higher odds of neonatal hypoglycemia among the subgroup of study participants who initiated CSII preconception. We did not detect a difference between outcomes of CSII versus MDI use for the other secondary neonatal outcomes.
A 2016 Cochrane review of five small RCTs comparing CSII versus MDI therapy in 154 pregnancies did not identify a difference between CD, LGA, or other neonatal morbidity outcomes. 11 However, the 2016 review included three studies from the 1980s, one from 1993, and the most current study was from 2005. Our results are more similar to those of a 2018 systematic review and meta-analysis of 47 studies of 7824 pregnancies complicated by T1DM that also identified that CSII use was associated with a higher risk of infants born LGA, although this study did not comment on CD. 5 Rys et al 5 also did not identify a significant difference between insulin administration modalities and neonatal hypoglycemia risk. This earlier meta-analysis by Rys et al incorporated RCTs and observational studies, but included abstracts and manuscripts published from 1988 to 2018. Studies in our analysis followed patients predominately from 2000s and 2010s while Rys et al included data also from the 1980s to 1990s. Thus, our meta-analysis provides a more current and comprehensive update, during a time of rapidly advancing CSII pump technology, including implementation of closed-loop systems, and rapid growth in the use of CSII among pregnancy-capable individuals.
Both our meta-analysis and that of Rhys et al 5 identified a similar degree of improvement in first trimester glycemic control with CSII over MDI use, with a 0.45% reduction in hemoglobin A1c, but an apparent lack of difference between CSII and MDI use with regard to overall glycemic control by the third trimester of pregnancy. Whether this improvement in early pregnancy glycemic control can be attributed to better glycemic control achieved by CSII over MDI alone, or due to greater engagement in pregnancy planning and periconceptional care among CSII users, remains unclear from our analysis.57-62 It is also important to consider that disparities in insurance payer status may underlie these improvements observed in early pregnancy glycemic control among CSII users. Compared to those with public insurance, privately insured pregnant persons are more likely to have access to and insurance coverage for CSII, have planned pregnancies, and access to primary and preconception care to optimize their glycemic control. Heightened awareness and motivation for behavior modification and diabetes self-management during pregnancy to reduce risks to the fetus, however, likely contribute to the similar third-trimester glycemic control regardless of mode of insulin administration. 63
We postulate that the association between CSII and CD identified herein may be due to the higher incidence of LGA identified in CSII users. Aside from suspected macrosomia greater than 4500 g as an indication for CD in pregnant people with pregestational DM, pregnant people with suspected LGA have longer labor duration, higher rates of dysfunctional labor and labor arrest diagnoses and, consequently, higher risk of intrapartum cesarean deliveries.64-66 The positive association of CSII therapy with delivery of an LGA neonate may be explained by improved placental function throughout pregnancy as a result of improved first trimester glycemic control.5,32,67,68
Pregnancy-specific challenges such as frequent insulin dose adjustment, day-to-day variability in prandial insulin pharmacokinetics, timing of pre-meal bolusing, and complex carbohydrate-to-insulin ratios with CSII therapy—all of which may be even more challenging if health literacy is inadequate for effective CSII use—lead to prandial hyperglycemia and greater time above range among CSII users in the latter half of pregnancy, as was demonstrated in the CONCEPTT trial. These aforementioned variables that contribute to the complexity of CSII use may ultimately contribute to fetal hyperinsulinemia, overgrowth, and resultant LGA compared to MDI users.28,69-71 However, the available data do not elucidate the relative contributions of these challenges with CSII therapy to the greater risk of LGA. Similarly, the available data do not account for the contribution of these complex CSII variables to fetal hyperinsulinemia and associated neonatal hypoglycemia risk in those who initiate CGM prior to pregnancy.
Strengths, Limitations, and Future Areas of Study
Our study has several strengths. We included a large representative, contemporary, international sample of 7528 pregnancies affected by pregestational DM, following a predefined protocol and comprehensive search strategy to limit selection bias. While prior systematic reviews have included only pregnant people with T1DM or excluded studies based on a specific insulin type, our systematic review included pregnancies affected by either T2DM or T1DM that used a variety of insulin types.5,72 Funnel plots generated also do not suggest publication bias affecting analysis of our primary outcomes. We analyzed a variety of clinically important maternal and neonatal outcomes, in addition to reporting on key outcomes with respect to timing of CSII initiation and measures of glycemic control according to mode of insulin administration.
However, the results of our meta-analysis should be interpreted with caution in the clinical setting, as inherent to all meta-analyses, our results are limited by the respective limitations of the primary studies included. Our analysis includes predominately retrospective cohort studies, which carry inherent limitations, risk of bias, and low quality of evidence compared to prospective or clinical trial designs. Missing data for secondary outcomes of interest and the heterogeneous methods of reporting data between studies limited data synthesis, beyond our limited ability to report on patient-level characteristics or control for confounding factors in this study-level systematic review (for example, indications for CD, or other confounders that may contribute to CD). While several studies included only participants in their CSII cohorts who had initiated CSII prior to pregnancy, a small proportion transitioned from MDI to CSII during early pregnancy (in some cases due to poor glycemic control with MDI, or per clinical practice protocol in others), which may confound the differences observed in the clinical outcomes between each treatment group. However, we attempted to control for this variation in timing of CSII initiation in our sensitivity analyses. Finally, we were unable to analyze outcomes based on use of CGM either with CSII or MDI, or the use of advanced insulin pump features (for example, low glucose suspend function, extended bolus function, etc.), and thus were unable to account for the influence of these technologies on pregnancy outcomes.
Future studies should further evaluate the role of CGM with CSII and MDI use, in addition to advanced pump features, in achieving euglycemia and thereby affecting pregnancy outcomes. In addition, future studies should evaluate the timing of initiation and role of CSII versus MDI in reducing glycemic variability to improve time in range, including prevention of prandial hyperglycemia, in mitigating adverse pregnancy outcomes. 71
Conclusions
In summary, the results of our systematic review and meta-analysis of contemporary observational studies and RCTs evaluating CSII and MDI use in pregnant people with pregestational DM suggest that CSII use is associated with higher odds of cesarean and delivery of an LGA neonate compared to that observed with MDI use, along with a modest decrease in first trimester hemoglobin A1c. Further evaluation of how CSII use may alter fetal growth trajectory and neonatal size—specifically LGA—and thereby influence route of delivery, is warranted. Our meta-analysis also highlights the need for additional, larger RCTs to further evaluate pregnancy outcomes according to timing of CSII initiation periconceptionally and mode of insulin administration with and without CGM. Finally, given the predominance of data available on CSII use in T1DM, but increasing use of CSII in those with T2DM, additional high-quality data on CSII use in pregnant people with T2DM would further strengthen our understanding of how CSII influences pregnancy outcomes in the broader population with DM.
Supplemental Material
sj-docx-1-dst-10.1177_19322968231186626 – Supplemental material for Continuous Subcutaneous Infusion Versus Multiple Daily Injections of Insulin for Pregestational Diabetes in Pregnancy: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-dst-10.1177_19322968231186626 for Continuous Subcutaneous Infusion Versus Multiple Daily Injections of Insulin for Pregestational Diabetes in Pregnancy: A Systematic Review and Meta-Analysis by Stephanie A. Fisher, Jingtong Huang, Ashley Y. DuBord, Nicole Y. Xu, Molly Beestrum, Charlotte Niznik, Andrea M. Yeung, Kevin T. Nguyen, David C. Klonoff and Lynn M. Yee in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-2-dst-10.1177_19322968231186626 – Supplemental material for Continuous Subcutaneous Infusion Versus Multiple Daily Injections of Insulin for Pregestational Diabetes in Pregnancy: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-2-dst-10.1177_19322968231186626 for Continuous Subcutaneous Infusion Versus Multiple Daily Injections of Insulin for Pregestational Diabetes in Pregnancy: A Systematic Review and Meta-Analysis by Stephanie A. Fisher, Jingtong Huang, Ashley Y. DuBord, Nicole Y. Xu, Molly Beestrum, Charlotte Niznik, Andrea M. Yeung, Kevin T. Nguyen, David C. Klonoff and Lynn M. Yee in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-3-dst-10.1177_19322968231186626 – Supplemental material for Continuous Subcutaneous Infusion Versus Multiple Daily Injections of Insulin for Pregestational Diabetes in Pregnancy: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-3-dst-10.1177_19322968231186626 for Continuous Subcutaneous Infusion Versus Multiple Daily Injections of Insulin for Pregestational Diabetes in Pregnancy: A Systematic Review and Meta-Analysis by Stephanie A. Fisher, Jingtong Huang, Ashley Y. DuBord, Nicole Y. Xu, Molly Beestrum, Charlotte Niznik, Andrea M. Yeung, Kevin T. Nguyen, David C. Klonoff and Lynn M. Yee in Journal of Diabetes Science and Technology
Supplemental Material
sj-docx-4-dst-10.1177_19322968231186626 – Supplemental material for Continuous Subcutaneous Infusion Versus Multiple Daily Injections of Insulin for Pregestational Diabetes in Pregnancy: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-4-dst-10.1177_19322968231186626 for Continuous Subcutaneous Infusion Versus Multiple Daily Injections of Insulin for Pregestational Diabetes in Pregnancy: A Systematic Review and Meta-Analysis by Stephanie A. Fisher, Jingtong Huang, Ashley Y. DuBord, Nicole Y. Xu, Molly Beestrum, Charlotte Niznik, Andrea M. Yeung, Kevin T. Nguyen, David C. Klonoff and Lynn M. Yee in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
This work was presented as a poster at the Society of Maternal Fetal Medicine 43rd Annual Pregnancy Meeting, February 6 to 11, 2023; San Francisco, CA.
Abbreviations
BMI, body mass index; CD, cesarean delivery; CDSR, Cochrane database of systematic reviews; CENTRAL, Cochrane central register of controlled trials; CI, confidence interval; CSII, continuous subcutaneous insulin infusion therapy; DM, diabetes mellitus; GRADE, grading of recommendation, assessment, development and evaluation; GWG, gestational weight gain; HDP, hypertensive disorders of pregnancy; IUFD, intrauterine fetal demise; LGA, large-for-gestational age; MDI, multiple daily injections; NICU, neonatal intensive care unit; OR, odds ratio; preE, preeclampsia; RCT, randomized controlled trial; RDS, respiratory distress syndrome; ROBINS-I, risk of bias in nonrandomized studies; SMD, standardized mean difference; T1DM, type 1 diabetes; T2DM, type 2 diabetes; TTN, transient tachypnea of the newborn; WHO ICTRP, World Health Organization International Clinical Trial Registry Portal.
Data Sharing
Data collection and extraction forms, and data used for all analyses, are available upon request from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SAF, CN, MB, LMY have no funding sources relevant to this article to disclose. JH, AYD, NYX, AMY, KTN, DCK have received funding from Diabetes Technology Society.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
