Abstract
Keywords
Type 2 diabetes (T2DM) individuals are at higher risk of developing erectile dysfunction (ED). A lower prevalence of neuropathy and ED is observed in subjects with better glycemic control. 1 Apart from targeting HbA1c and mean glucose concentrations, reducing the glycemic variability (GV) is the key to successful management of diabetes and associated complications.
A parallel arm, open-label, randomized, controlled study (ClinicalTrials.gov identifier NCT01468519) was conducted in T2DM men with ED (n = 20) to assess whether better glycemic control achievable with the continuous subcutaneous insulin infusion (CSII) relative to multiple daily injection therapy (MDI), 2 would result in improved ED and diabetic peripheral neuropathy. Subjects were randomized (1:1) to CSII (intervention) or MDI (control) arms. All subjects were on insulin, oral hypoglycemic agents (OHAs), and a statin. Concomitant therapies were continued and antihypertensives were used as required. Modifications to medication, diet, and exercise were prescribed based on their structured self-monitoring of blood glucose (sSMBG) values, professional continuous glucose monitoring (P-CGM, at 0 and 3 months of study) reports, and other comorbidities.
At six months, CSII arm showed increased International Index of Erectile Function scores (addresses the relevant domains of male sexual function, 3 +7.87, P = .0037). More subjects in the CSII than the MDI arm achieved reductions in ED severity 3 (Figure 1) and were listed under the “No ED” category (6 vs 1, P = .0108). Those in the CSII arm achieved lowered HbA1c levels (−0.87%, P = .0036), PHQ-9 scores (represents the severity of depression, 4 −1.33, P = .0017), and hsCRP levels (a biomarker of inflammation and risk factor for ED, −0.53 mg/l, P = .0110), whereas those in the MDI arm had increased HbA1c levels (+0.76%, P = .0227). Total cholesterol, triglycerides, low-density, and high-density lipoproteins showed no significant changes in both the treatment arms.
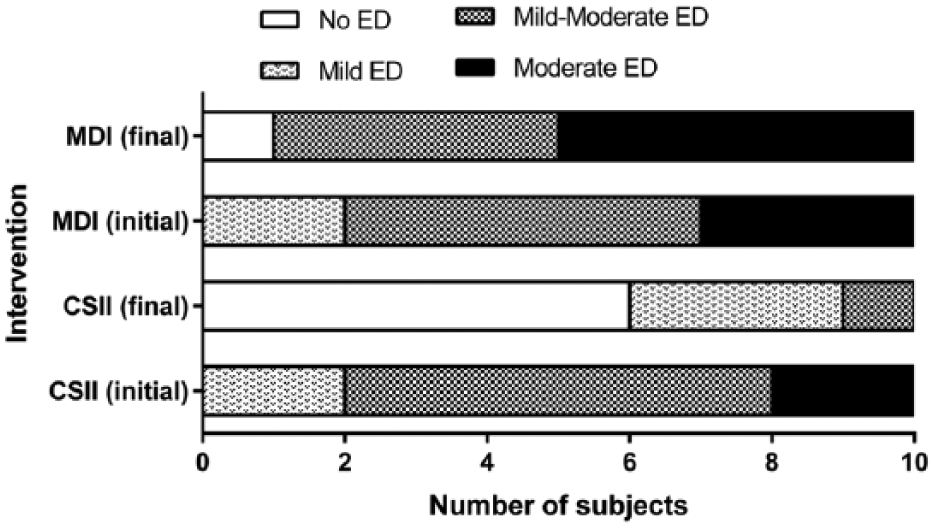
Severity of ED (based on the IIEF-5 scores) as reported by the subjects in the CSII and MDI arm at the start and end of study.
Vibration perception threshold analysis 5 demonstrated that CSII arm achieved improvements in the severity of neuropathy. Subjects in the mild, moderate, and severe categories were 40%, 10%, and 50% at the start of study vs 50%, 20%, and 10% at 6 months, respectively. In the MDI arm, the extent of neuropathy worsened (30%, 50%, and 20% at the start of study vs 20%, 30%, and 50% at 6 months). Global assessment question (GAQ) responses revealed that majority in the CSII than in the MDI arm (7 vs 3) believed their treatment improved their erections (P = .0248). Of the GAQ positive responders, majority in the CSII than the MDI arm (71.5% vs 66.67%) exhibited improvements in their neuropathy severity.
Our results indicate that CSII fine tunes the glycemic status and imparts a positive influence on sexual function of T2DM males. A P-CGM analysis performed at the study end, to substantiate whether it was the reduction in GV upon CSII use, which contributed to these improved outcomes, could have added more value to the study. However, at this point it can only be speculated that these benefits achieved with CSII, could be due to the maintenance of glycemic control with minimal GV, thereby reducing the chances of oxidative stress, accumulation of advanced glycation end products, and endothelial dysfunction, 6 the chief mediators of ED. A structured and systematic CSII therapy regimen could therefore deliver better effects, resulting in improved sexual functioning.
Footnotes
Abbreviations
CSII, continuous subcutaneous insulin infusion; ED, erectile dysfunction; GAQ, global assessment question; hsCRP, high sensitivity C-reactive protein; IIEF-5, abridged version of the International Index of Erectile Function Questionnaire; MDI, multiple daily injections; P-CGM, professional continuous glucose monitoring; PHQ-9, Patient Health Questionnaire–9; sSMBG, structured self-monitoring of blood glucose.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the conduct of study was partly supported by Roche Diagnostics. The data are owned by the investigators, and Roche Diagnostics was not involved in the conducting and monitoring of the trial, in data analysis, or in the publication process. ClinicalTrials.gov identifier: NCT01468519, ![]() .
.
