Abstract
Background:
Continuous subcutaneous insulin infusion (CSII) therapy is a valuable option especially for people with type 1 diabetes. Although insulin infusion sets (IIS) are essential components of most insulin pump systems, only few studies have been conducted on their performance and safety. In this study 2 IIS with soft cannulas were compared.
Methods:
In an open-label, randomized, crossover, multicenter 8-week study 80 type 1 diabetic subjects on CSII (20-74 years, mean 46.5 ± 12.9 years, 58.8% male) were enrolled to evaluate the use of 2 IIS. The Accu-Chek® FlexLink Plus (FL Plus) and the Accu-Chek® FlexLink infusion set (FL) were used 4 weeks each under real-life conditions at home. All patients had to record details about every IIS change in a diary. Insertion-related pain was assessed by means of a visual analogue scale (VAS).
Results:
The mean pain noted during insertion was similar for both IIS (VAS 6.1 ± 6.4 mm for the FL Plus and 5.8 ± 6.3 mm for the FL, P = .92). 20.0% of the 895 IIS changes with FL Plus and 16.0% of the 854 with FL were unplanned. During the 4243 patient days of the study, 13 patients had adverse events. Among these, 2 serious adverse events occurred. Both serious events and 1 nonserious adverse event were considered as related to the IIS. Of the patients, 18.7% had kinked cannulas. Most patients were satisfied with both IIS.
Conclusion:
Both IIS are safe and effective. The 2 IIS did not differ from each other with respect to pain observed during insertion.
The use of insulin pumps in individuals with diabetes has continuously grown with about 500,000 patients currently treated worldwide.1,2 Continuous subcutaneous insulin infusion (CSII) therapy has been evaluated in numerous clinical studies3-13 whereas only limited data are available for insulin infusion sets (IIS). 14
IIS vary in design and material, in particular of the cannula (steel or Teflon®), but all need to be inserted in the subcutaneous tissue manually or by means of an auto insertion device. Insulin is infused subsequently via the cannula that remains in the subcutaneous tissue after a guiding needle has been removed immediately after insertion. 14 To secure the cannula in place throughout the use of the IIS, the IIS has to be fixed appropriately to the skin by means of adhesive tape.
The recommended wear time for IIS is generally 2-3 days.15,16 During longer wear times, glycemic control deteriorates independent of the cannula material used, that is, steel and Teflon cannulas showed similar behavior. 17 Other issues observed frequently with IIS are infusion site problems (like infections and contact allergies), kinking (only observed with Teflon cannulas) or occlusions. As the relatively small insulin depot in the subcutaneous tissue is absorbed within hours, hyperglycemia and diabetic ketoacidosis can develop relatively rapidly when insulin delivery is interrupted.18,19 Patient compliance with CSII can be impaired by IIS issues, for example, pain associated with the insertion of the IIS, dermatological complications as well as handling aspects.20,21
The aim of this study was to evaluate 2 IIS under real-life conditions focusing on pain perception during insertion.
Methods
Study Design
This was an open-label, randomized, cross-over, multicenter study to evaluate 2 IIS, the Accu-Chek® FlexLink Plus infusion set (FL Plus) and its predecessor the Accu-Chek® FlexLink infusion set (FL) (Roche Diabetes Care, Mannheim, Germany) during use at home.
Study Conduct
The study was conducted by 5 sites in Germany from October 2013 to June 2014. A total of 80 patients were recruited and randomized in a 1:1 ratio to one of the infusion sets for the first 4-week period. Thereafter patients switched to the other IIS for the second 4-week period. For randomization, a list was created for each of the 5 sites with a block size of 4 subjects. The study was approved by the competent authority and the local ethics committee. The study was registered at ClinicalTrials.gov (Identifier: NCT02103595) and was conducted in accordance with the Declaration of Helsinki and ISO 14155.
Patients
Main inclusion criteria for study participation were diabetes mellitus type 1 or type 2 and usage of CSII for at least 3 months and an age of at least 18 years. The main exclusion criteria were a significantly impaired awareness of hypoglycemia, history or high risk of ketoacidosis during CSII, and any medication interfering with the assessment of pain.
Using a paired t-test, a sample size of 80 patients was calculated, providing a 2-sided significance level of 5%, a power of 80% to detect 0.9 mm difference in VAS, and allowing for 12% drop outs. Calculations were based on the assumption that for FL a mean VAS of 8.5 mm and a standard deviation of 10.8 mm would be observed and for FL Plus a mean VAS of 7.6 mm and the same standard deviation.
Devices Information
The soft cannula IIS studied have been on the market since 2010 (FL Plus) and 2001 (FL), respectively. In both devices, the soft Teflon cannula is inserted into the subcutaneous tissue at a 90° angle with the help of an introducer needle (stainless steel) which is removed after insertion. A transfer set connects the head set to the insulin reservoir in the pump by means of a female Luer connector (Figure 1). The head set consists of the connector plate that is placed on the skin of the patient, integrating the soft cannula and the adhesive plaster for fixation. For both IIS, an insertion device is available (Accu-Chek LinkAssist Plus and Accu-Chek LinkAssist, respectively). The patients were free to decide whether to use the insertion device or to insert manually. During the study, only cannulae of 8 mm length, and tubings of 70 cm (FL), respectively 80 cm length (FL Plus) were used. Users were instructed to replace the IIS every 3 days or more often if necessary.

FL Plus and FL (left), cannulas (middle), and respective insertion devices (right).
The FL Plus is an improved version of the FL IIS. The main differences between the catheters are related to a different geometry of the cannula (single step increase of the outer cannula diameter for FL and multiple steps for FL Plus which could lead to a more gradual tissue expansion during insertion in the latter case), introducer needle (hollow needle for FL and solid needle for FL Plus) and the patient protection (exposed introducer needle after insertion for FL and self-contained needle for FL Plus).
Study Objectives
The primary study objective was to evaluate the perception of pain during cannula insertion. Pain was assessed by the patient on a 100 mm VAS scale shortly after every insertion over each 4-week period. Secondary objectives included parameters of device usage (rate and reasons for IIS replacements, incidence of kinking, preferred insertion method, safety) and satisfaction parameters (7-point Likert scale for overall device satisfaction, device preferences and device recommendation).
All insertions were documented via a patient diary. These data were used to determine planned or unplanned changes, method of insertion and incidence of kinked cannulas. To assess infusion site problems, itching, redness, swelling, heat, and pain were recorded during short examinations at the baseline visit, during the fourth visit after 4 weeks and during the sixth visit after 8 weeks. The investigators had to classify any reaction as minor, moderate or severe. Adverse events (AE) and serious AE were documented during all visits. AEs were coded using MedDRA with details for type, primary body system organ class and preferred term.
To evaluate overall device satisfaction, patients were asked to record to which extent they agree with the statement “In general I am very satisfied with the infusion set” (7-point Likert-type scale from completely agree = 1 to completely disagree = 7) after the two 4-week periods. At Visit 6 each patient was asked which of the 2 IIS he or she preferred for further use and if he or she would recommend FL Plus to other insulin pump patients.
Statistical Methods
Data were analyzed by Biometrical Practice, BIOP AG, using version 9.2 of the SAS statistical package. Continuous data were reported in mean ± standard deviation (SD), minimum, and maximum. Categorical data were reported in frequencies and percentages. To compare the VAS values, a paired t-test with a 5% 2-sided significance level was used.
Results
Disposition and Demographics
Seventy-three of the 80 patients that were screened and randomized completed the study. Three patients withdrew consent, 4 patients dropped out due to noncompliance with study procedures. All patients had diabetes type 1. Duration of diabetes was 26.1 ± 11.5 years (min 4.0 years, max 54.0 years) (Table 1). Patients had been on CSII therapy for 12.8 ± 7.8 years (1.0 to 29.8 years). HbA1c at baseline was 7.6 ± 1.0% (5.9% to 11.1%). Patients used different insulins for their therapy: 75% used insulin lispro, 17.5% insulin aspart, 5% insulin glulisine and 2.5% regular human insulin. At baseline subjects were treated with a mean total dose of 52.8 U insulin divided into 22.8 U basal dose and 30.0 U bolus dose. The number of daily boluses amounted to 4.5. The duration of the 2 study periods was 2054 patient days for FL and 2189 patient days for FL Plus.
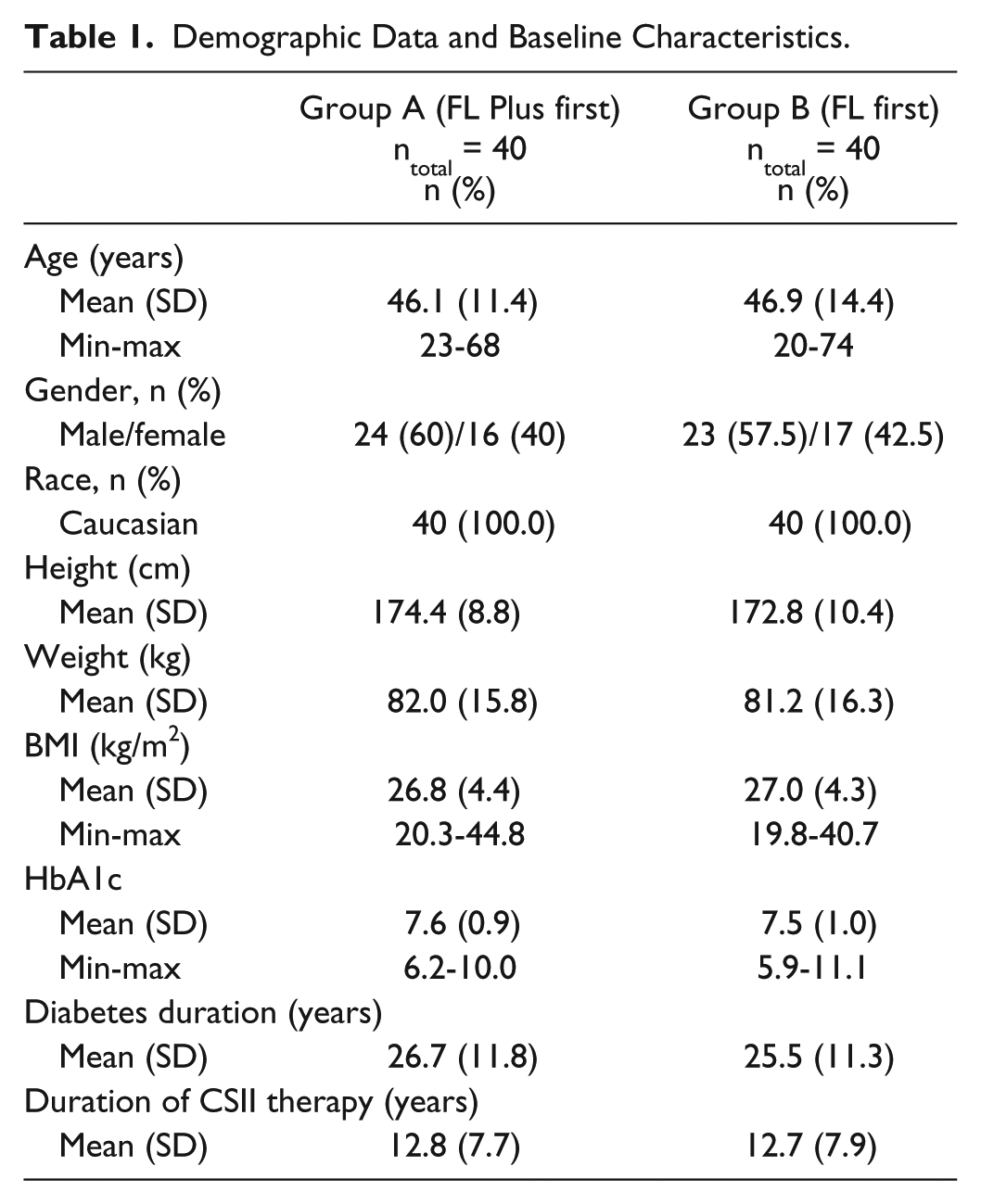
Demographic Data and Baseline Characteristics.
Pain Perception
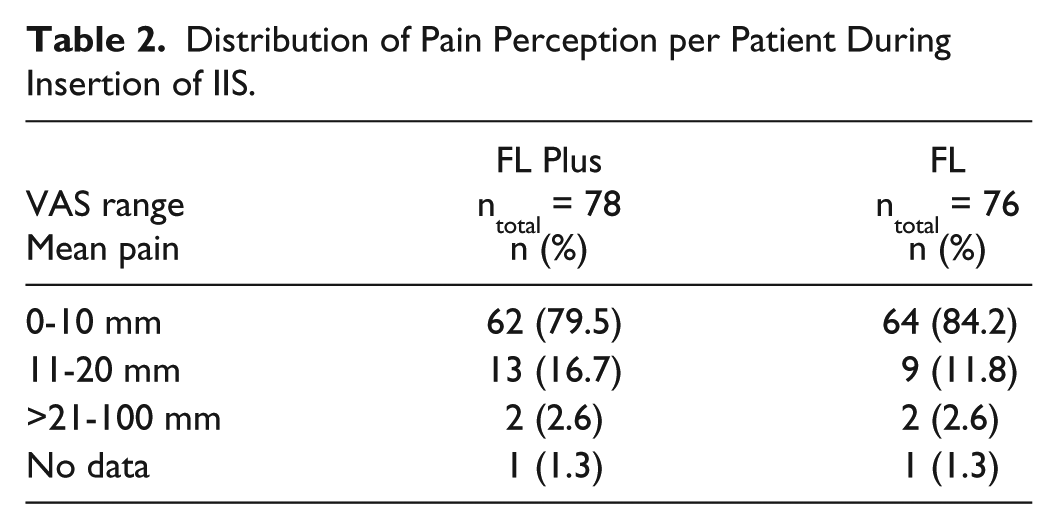
The null hypothesis of the study “no difference in the perception of pain between the infusion sets” could not be rejected after evaluation of the VAS values. Pain perception with either IIS was similar in both groups: 6.1 ± 6.4 mm (mean ± SD) in FL Plus group versus 5.8 ± 6.3 mm in the FL group (P = .92) (Table 2). There was no difference between the mean of the worst pain each patient reported: 17.4 mm with FL Plus and 17.0 mm with FL, respectively.
Distribution of Pain Perception per Patient During Insertion of IIS.
Device Usage
60.2% of the patients in the FL Plus group reported to use the inserter for IIS insertions and 34.1% did it manually (5.7% were inconsistent or did not report). 58.7% of the patients in the FL group used the inserter, 36.2% did it manually (5.1% were inconsistent or did not report).
Of the 1971 infusion sets used, the insertions of 1749 were correctly documented in the subject diary: 895 with FL Plus and 854 FL. In total, 78.5% of the replacements with FL Plus and 82% of the replacements with FL were planned according to patients’ individual rhythms. A total number of 316 IIS changes were unplanned: 20% with FL Plus and 16% with FL. In all, 65 patients with FL Plus and 52 patients with FL performed 1 or more unplanned changes.
The most frequent reasons for unplanned changes were related to insulin delivery issues, for example, kinked cannulas and occlusion alarms (Table 3). Discomfort, infusion site inflammation, or detached adhesive were less frequently given as reason for unplanned changes. Other reasons for unplanned changes as reported in the patients’ diaries were diverse, including error in blood glucose measurement, dropped insulin pump, pulled-out insulin catheter and cartridge replacement.
Reported Reasons for Unplanned Infusion Set Changes (More Than 1 Answer Was Allowed).
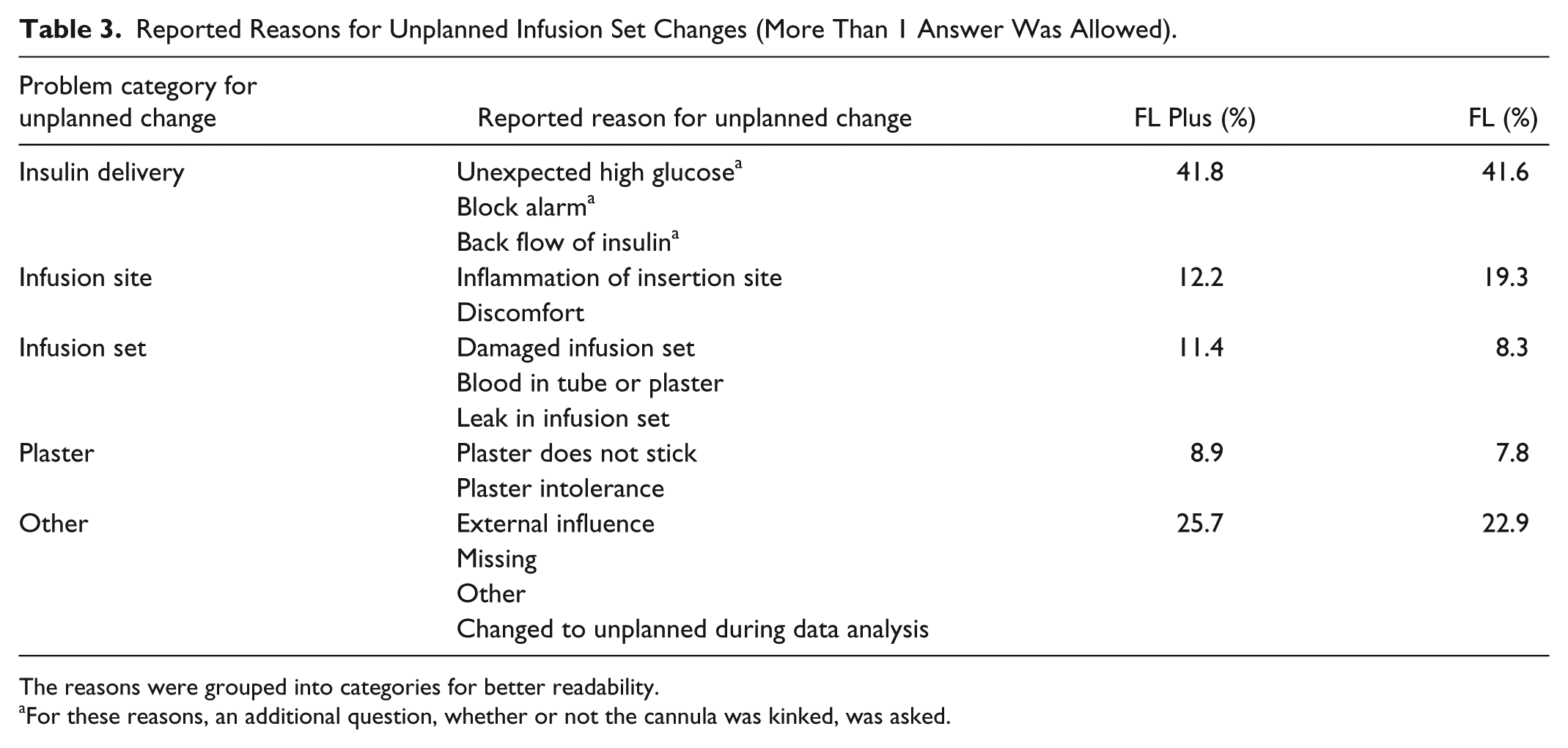
The reasons were grouped into categories for better readability.
For these reasons, an additional question, whether or not the cannula was kinked, was asked.
The mean time since the previous IIS change was 2.58 days for FL Plus and 2.51 days for FL. Most changes were done 3 days after previous insertion for both IIS.
For unplanned changes due to issues in insulin delivery, patients were asked if the cannula was kinked. The majority of all patients (81.3%) had no kinked cannulas during the study with any of the IIS, 9 patients had one or more kinked cannulas with FL Plus and 6 patients with FL. 7 patients with FL Plus and 6 patients with FL were not able to determine if cannulas had kinked and recorded “not sure”. The calculated incidence of kinked cannulas was 1 of 100 FL Plus cannulas and 0.8 of 100 FL cannulas.
Safety
Thirteen patients reported AEs. The AEs occurred in 6 patients while wearing FL Plus and 9 with FL. Three AEs were regarded as being related to usage of IIS. AEs with hyperglycemia/ketosis occurred in 1 patient of each IIS group and contact dermatitis occurred in 1 patient with FL Plus. Two serious AEs were observed: one with FL Plus (a case of appendicitis), and one with FL (a symptomatic ketosis short after study start that may be attributed to a kinked cannula after manual insertion). In the latter case the patient had an episode of high blood glucose levels, ketones with polyuria and polydipsia. After self-administration of insulin she recovered.
The investigators classified potential local reactions at the insertion sites in all patients, where most patients did not experience any itching, redness, swelling, heat, pain or any other adverse symptoms. With FL Plus a minority of patients experienced minor symptoms like itching (n = 3), redness (n = 4), swelling (n = 5) and pain (n = 1); with FL also a few patients experienced minor (itching n = 2; redness n = 7; swelling n = 6, heat n = 1; pain n = 2) and moderate symptoms (itching n = 1; swelling n = 1, heat n = 1).
Treatment Satisfaction
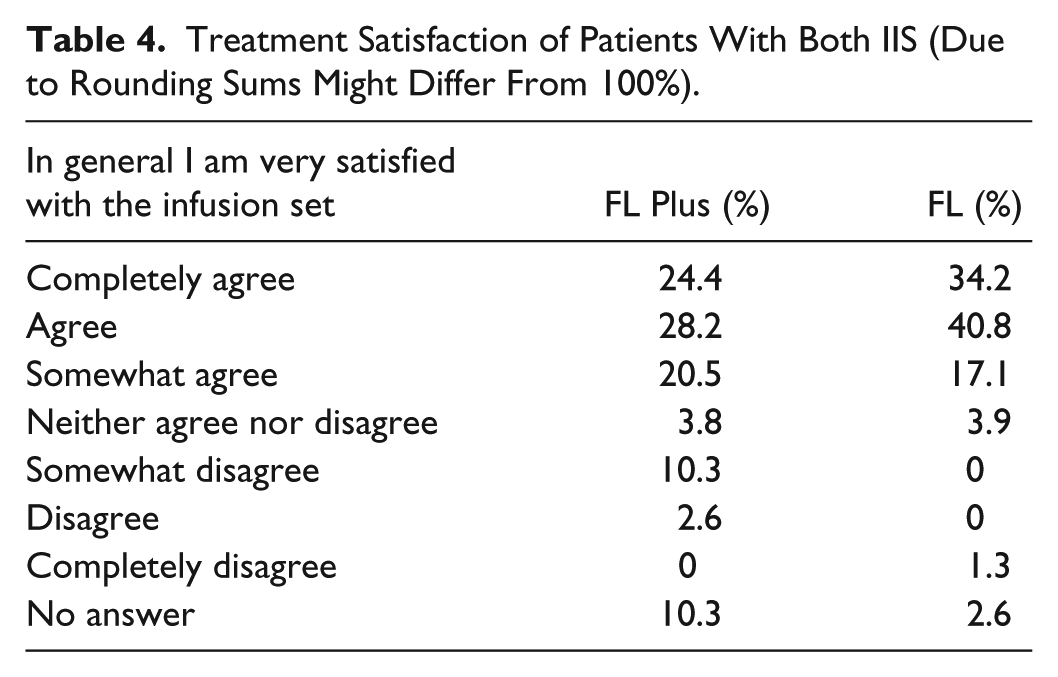
Patients were asked at the end of each 4-week-period, how much they agreed with the statement “In general I am very satisfied with the infusion set” (7-point Likert scale). The results for both IIS groups are listed in Table 4. Treatment satisfaction was high in both IIS with about 7 and 9 respectively out of 10 patients agreeing partially or completely with the mentioned statement.
Treatment Satisfaction of Patients With Both IIS (Due to Rounding Sums Might Differ From 100%).
At the last study visit all patients were asked which IIS they would prefer in the future: 40% of the individuals would prefer FL Plus, 40% would prefer FL, 10% had no preference and the remaining 10% provided no answer. Of the patients, 58.8% would recommend using FL Plus to other pump patients, whereas only 22.5% would not.
Discussion
Publications about IIS originating from clinical investigations are rather scarce. Insertion-site related problems such as infections or pain during insertion were reported to be a main reason for CSII patients to discontinue pump therapy, however only limited data are available in this regard.21,22
The study reported here compared 2 IIS using soft cannulas in a randomized cross-over study under real-life conditions. Pain perceived on insertion was very low in both groups. There was no significant difference between both IIS regarding this primary outcome measure. The observed VAS values were similar to results obtained in a previous study of FL. 23
The 2 IIS tested in this study differed in geometry of the cannula and the introducer needle. The results showed no difference in pain perception that could be attributed to different tissue expansion during insertion. But as pain perception was very low already in FL, there was only little space for improvements. An effect of the self-contained introducer needle in FL Plus on patient safety, that is, fewer needle stick injuries, was not systematically assessed and would have required a larger study.
The IIS change pattern in this study was not predetermined; patients were advised to change their IIS in accordance with their individual rhythm while following the manufacturer’s recommendation to change at least every third day. The majority of reasons for unplanned IIS changes could be attributed to IIS characteristics that may theoretically be influenced by further product improvements, for example, with regard to insulin delivery or adhesive tape. But for many of these problems, no solutions within the design of the infusion set alone are feasible or they would require design changes that would severely impact on the usability and acceptance of the systems. However, in as much as 25% of the unplanned changes, other problems such as pulled-out catheters appeared that cannot be attributed to the specific IIS. Adequate training in the handling of the IIS might help to improve these.
A recently reported randomized cross-over study in 24 individuals with type 1 diabetes compared predefined IIS change patterns: every 2 days versus every 4 days. 24 Longer wearing time leads to a significantly higher number of catheter-related events (290 versus 495 events for 2 and 4 days wearing time, P < .05 and 750 versus 934 treatment-related events for 2 and 4 days wearing time, P < .001), supporting the recommendation not to use IIS longer than a period of 2 to 3 days. There were more unscheduled IIS changes in the 4-day group than in the 2-day group (n = 143 versus n = 73). More problems regarding the adhesive tapes were observed in the 4-day group, and more cases of soft cannula bending in the 2-day group.
It has to be noted that differences between that study and our study such as fixed versus variable IIS wearing times or evaluation of the IIS after removal by the investigator versus the patient make a comparison of the results difficult. Nevertheless it appears that in both studies problems regarding insulin delivery were the main reasons for unplanned or unscheduled IIS changes.
As reported before with other Teflon cannulas, kinking can be an issue. 17 The frequency of kinking observed in our study is relevant, but rather low (order of 1 in 100 cannulas inserted); 81.3% of all patients did not have any kinked cannula. Compared with other reported data for IIS kinking with 64.1% of patients affected, the percentage of individuals who reported kinking in this study was substantially lower than in a recent patient survey. 19 This may be attributed to the shorter observation period, the different way of observing (patient diary versus survey) and potentially the products used. Furthermore, in this study kinking was the reason for a very small percentage of unplanned IIS changes and only rarely led to clinically relevant AEs like hyperglycemia (n = 1/ FL Plus) or ketosis (n = 1/ FL). Thus, both IIS appear to be safe in daily use.
Finally treatment satisfaction with both IIS was high, with about 6 out of 10 patients recommending FL Plus.
Conclusions
In this study relevant patient and device parameters were assessed during a long follow-up period with a relatively large number of recruited patients. Insertion of both IIS was associated with minimal pain. The differences in the design of the introducer needle and soft cannula between the FL and FL Plus infusion set have no impact on pain perception. Both IIS are effective and safe. Rates of unplanned changes of IIS as well as kinked cannulas were similar with both infusion sets. A relevant part of the unplanned changes could not be related to the IIS design. Treatment satisfaction with FL and FL Plus infusion set was high in experienced CSII users.
Footnotes
Acknowledgements
We thank Michael Pickering (BIOP Biometrical Practice, Basel, Switzerland) for statistical analysis of and support for the study and this publication. ACCU-CHEK, ACCU-CHEK FLEXLINK and ACCU-CHEK LINKASSIST are trademarks of Roche. © 2016 Roche Diabetes Care.
Abbreviations
AE, adverse event; CSII, continuous subcutaneous insulin infusion; FL, Accu-Chek FlexLink infusion set; FL Plus, Accu-Chek FlexLink Plus infusion set; IIS, insulin infusion set; SD, standard deviation; VAS, visual analogue scale.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GF is general manager of the IDT (Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany), which carries out clinical studies on the evaluation of BG meters and medical devices for diabetes therapy on its own initiative and on behalf of various companies. GF/IDT have received speakers’ honoraria or consulting fees from Abbott, Bayer, Berlin-Chemie, Becton-Dickinson, Dexcom, LifeScan, Menarini Diagnostics, Novo Nordisk, Roche Diabetes Care, Sanofi, and Ypsomed. SA, AF, GK, and KP have no conflicting interests. TK and BP are employees of Roche Diabetes Care, Mannheim, Germany.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Roche Diabetes Care, Germany, provided financial support for this study.
