Abstract

Type 1 diabetes (T1D) conveys risk for cardiovascular disease (CVD). 1 The American Diabetes Association (ADA) recommends screening blood pressure (BP), weight, and low-density lipoprotein (LDL) in pediatric patients with T1D. 2 Diabetes providers, parents, and adolescents with T1D endorse difficulty enacting lifestyle changes. 3 There may be opportunities to promote behavior change using digital health and extrinsic rewards. 4 This three-month prospective randomized controlled pilot/feasibility trial in adolescents with T1D and elevated CVD risk evaluated an intervention motivating heart-healthy behaviors using a smartphone application (app), team challenges, and financial incentives.
We recruited 44 adolescents, 14 to 19 years old with T1D duration ≥ 6 months. Participants were English-speaking, had access to a smartphone, and demonstrated ≥1 CVD risk factor: dyslipidemia, overweight/obesity, prehypertension/hypertension, or HbA1c ≥ 8%. Study protocol received institutional review board approval (#2017-34) and Clinicaltrials.gov registration (#NCT03531021). Informed consent/assent was obtained before beginning study procedures.
Participants were randomized 1:1 to intervention or control arms, stratified by age (14-16, 17-19). The psychoeducational/behavioral intervention, developed by an interdisciplinary team, was based upon the Health Belief Model 5 and included two modules: “Hypertension and Lipid Knowledge” and “Heart-Healthy Goals.” Intervention participants completed modules at study start and control participants at study conclusion. Participants downloaded a step-tracking app onto smartphones. Intervention participants completed team challenges in the app, accruing points toward financial rewards. Intervention participants received remote check-ins regarding app use and goal completion. Control participants received matching correspondence reminding them of study visits. At baseline and study conclusion (three months), participants completed the Adolescent Food Habits Checklist (adapted) (AFHC) 6 and provided biomedical data (HbA1c, LDL, body mass index [BMI], BP). At study’s end, participants completed a 15-item study satisfaction survey with 13 intervention-specific questions.
Of 116 approached, 44 adolescents (30% male, 88% white) enrolled. Mean age was 17.0 ± 1.6 and T1D duration 8.7 ± 3.3 years; 82% used an insulin pump and 63% used a continuous glucose monitor. Ninety-one percent (n = 40, 20 intervention, 20 control) completed the study. Intervention participants completed 85% of remote visits (three, six, and nine weeks). At check-ins, most intervention participants (18/22, 82%) reported an increase in at least one heart-healthy behavior described in their goals at some point during the study. Half the intervention participants (n = 11) completed ≥50% of the challenges.
Intervention participants reported favorable satisfaction, especially regarding learning new information, having increased confidence in making healthy choices, eating more healthy foods, and enjoying study participation (Table 1). There were no substantial changes in body weight, BP, lipid levels, or HbA1c over three months. The AFHC scores improved significantly in both groups (intervention: 2.9 point increase, P = .008; control: 2.0 point increase, P = .02).
Intervention Group Satisfaction Survey Results.
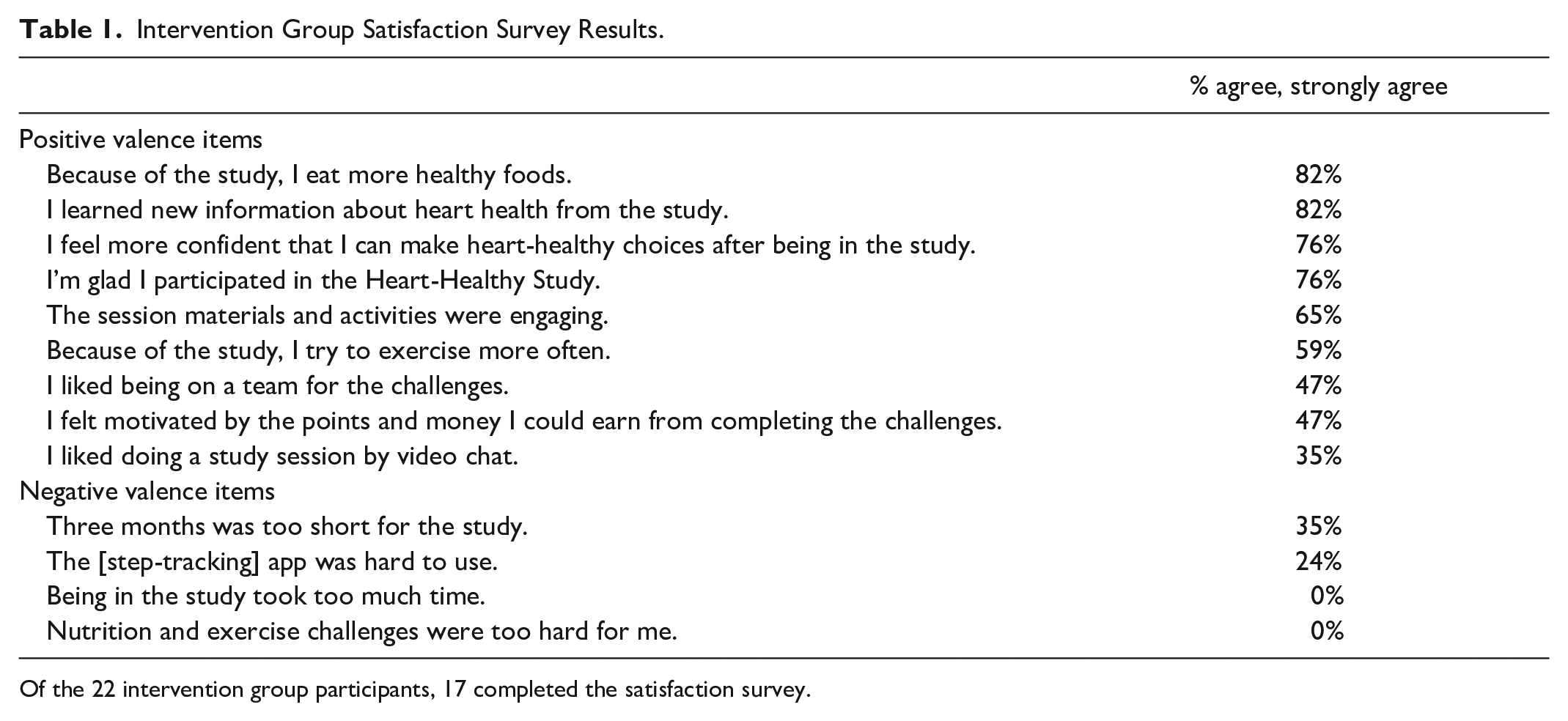
Of the 22 intervention group participants, 17 completed the satisfaction survey.
This study demonstrates feasibility and acceptability of a psychoeducational and motivational intervention with app-based team challenges and financial incentives to improve heart-healthy behaviors in adolescents with T1D and elevated CVD risk. Further research in larger, diverse samples of adolescents can help assess the impact of such interventions on CVD risk factor reduction and other diabetes outcomes.
Footnotes
Abbreviations
BMI, body mass index; BP, blood pressure; CVD, cardiovascular disease; HbA1c, hemoglobin A1c; LDL, low-density lipoprotein; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Katz is currently an employee and stockholder of Eli Lilly and Company. Her contribution to this work was completed during her employment at the Joslin Diabetes Center. There are no other conflicts of interest to report.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Heart, Lung, and Blood Institute (K23HL125976 [to Michelle Katz]) and the National Institute of Diabetes and Digestive and Kidney Diseases (P30DK036836 [to Joslin Diabetes Center] and K12DK094721 [to Lori Laffel and Tara Maxwell]) of the National Institutes of Health; the Katherine Adler Astrove Youth Education Fund; the Maria Griffin Drury Pediatric Fund; and the Eleanor Chesterman Beatson Fund.
Ethical Approval
This study was approved by the institute’s Committee on Human Studies and registered on clinicaltrials.gov. Consent and assent, as appropriate, were obtained prior to study procedures.
