Abstract
The introduction of continuous glucose monitoring inaugurated a new era in clinical practice by shifting the characterization of glycemic control from HbA1c to novel metrics. The one that gained widespread attention over the past decades was glycemic variability (GV), which typically refers to peaks and nadirs of blood glucose measured over a given time interval. GV can be dichotomized into two main categories: short-term and long-term. Short-term GV reflects within-day and between-day glycemic oscillations, and its contribution to diabetic complications remains an enigma. In this review, we summarize the available data about short-term GV and its possible association with both microvascular and macrovascular complications, evaluating different pathogenic mechanisms and demonstrating nonpharmaceutical, as well as pharmaceutical, therapeutic interventions.
Keywords
Introduction
The Diabetes Control and Complications Trial (DCCT) Research Group, using glycated hemoglobin (HbA1c) as the key parameter of their study, proved that tight glycemic control successfully prevents the development of microvascular complications among patients with insulin-dependent diabetes. 1 Two years later, a report estimated the connection between HbA1c levels before and during the DCCT trial and the incidence of retinopathy in diabetes patients of both groups (intensive and conventional therapy). It was shown that retinopathy could not be entirely interpreted just from the use of HbA1c, as there were intensively treated patients with well-controlled HbA1c levels who showed deterioration of retinopathy, while other conventionally treated patients with elevated HbA1c levels demonstrated no progression after nine years of monitoring. 2 Even though HbA1c remains the leading point of reference for glycemic control, it does have certain limitations and may not be the ideal biomarker for diabetes and its complications. It depicts the mean glycemia over the past two to three months, without giving any information about hypoglycemic or hyperglycemic episodes, glucose fluctuations, and glycemic profile of the patients. Conditions that determine red blood cell turnover, such as hemoglobin variants, iron deficiency, and drug use, affect its accuracy and confuse clinicians and their therapeutic options. 3
The emerging use of continuous glucose monitoring (CGM) devices into clinical practice shed light into new aspects of glycemic control such as glycemic variability (GV). GV has been established as an important metric to define glycemic management. It typically refers to the peaks and troughs of blood glucose measured over a given time interval. It can be split into short term, representing within-day (intraday) and between-day (interday) glycemic variations, and long term, usually determined by visit-to-visit HbA1c, visit-to-visit fasting plasma glucose (FPG) GV, and visit-to-visit postprandial glucose GV. 4 The most frequently used metrics of short-term intraday GV are the standard deviation (SD), which is the variation around the mean glucose concentration, and the coefficient of variation (CV), defined as SD divided by the mean glucose concentration. 5 It should be emphasized that SD is extremely affected by the mean glucose level; a patient with a higher mean glucose level will have higher SD, and thus, CV is preferable and should be considered the primary measure of variability as it corrects and balances SD. 6 Moreover, CV is a metric relative to the mean, which makes it a better reflection of hypoglycemic fluctuations than the SD alone. 7 Another traditional measure of intraday variability is the mean amplitude glycemic excursion (MAGE). This describes the average of blood glucose values above one SD from the 24-hour mean blood glucose level, translated as the mean differences from peaks to troughs. 8 The mean of daily differences (MODD) is considered the best metric of interday GV and is calculated based on the differences between two glucose values measured at the same time within a 24-hour period. 9 Continuous overall net glycemic action (CONGA) is another short-term GV metric estimated by the differences between glucose levels calculated at regular time intervals and then on the SD of these differences. 10 In addition, CGM devices have enabled the use of time-related GV metrics such as time in range (TIR) and ambulatory glucose profile (AGP). TIR is a measurement of the time a patient spends within the target blood glucose range, which, for most, is between 3.9 and 10 mmol/L and can provide a more accurate picture of glycemic control. Nevertheless, time spent outside the target range also needs to be quantified separately to assess any trends toward hyperglycemia and hypoglycemia. 11 AGP is a visual tool that presents the distribution of glucose measurements for a certain period, usually 14 days, in a graphical format. Because the median glucose level and interquartile ranges are provided, they can be used to assess between-day GV at given time intervals (Table 1). 12 Another aspect worth mentioning is the glucose pentagon model (GPM), a composite metric of glycemic management consisting five parameters including HbA1c, SD, mean glycemia, duration of hyperglycemic status, and urea under the curve for hyperglycemia, which seems to have greater prognostic value of developing diabetes-related late complications than HbA1c. 13 Vigersky et al 14 altered the GPM, creating the comprehensive glucose pentagon (CGP) by introducing new axes—CV, intensity of hyperglycemia and hypoglycemia, and time out of range, forming a totally glucose-centric metric that can replace HbA1c. CGP, the potential of which remains to be validated in clinical trials, provides a short-term analysis of glycemic monitoring and points out the component of glycemic control that is more prone to imbalance. 14
The Main Short-term GV Metrics.
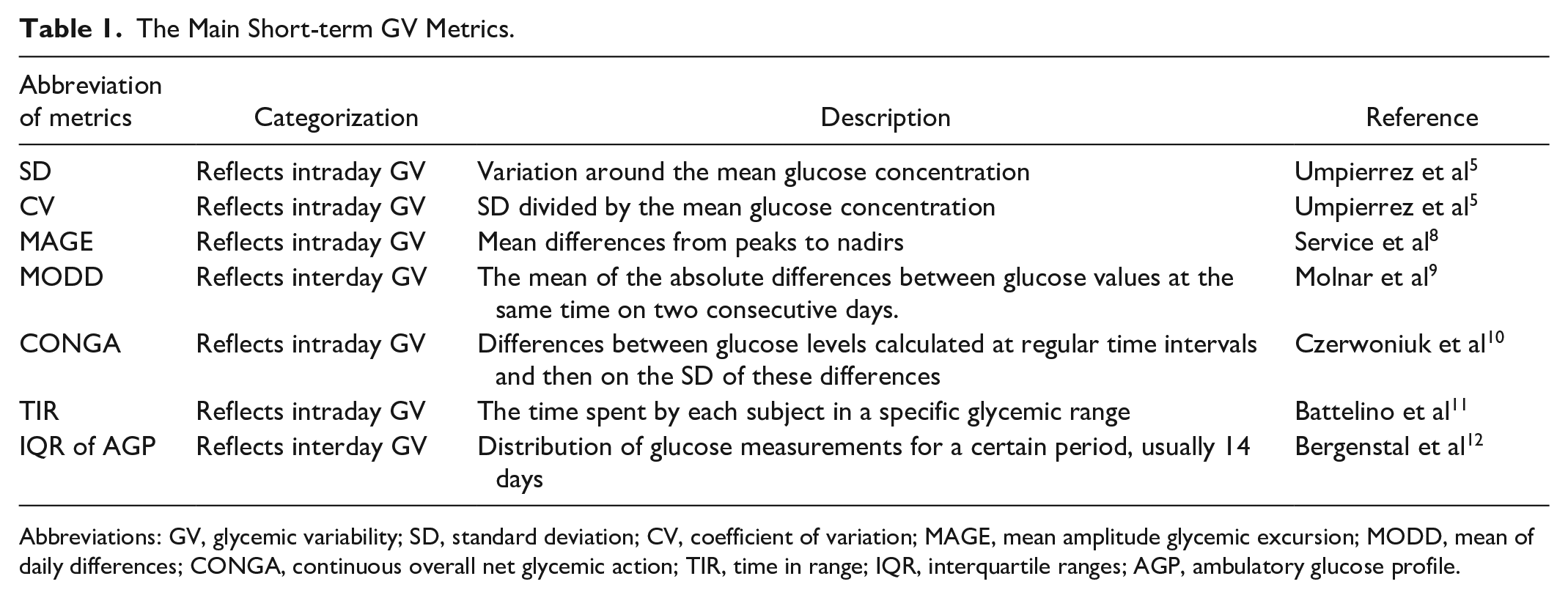
Abbreviations: GV, glycemic variability; SD, standard deviation; CV, coefficient of variation; MAGE, mean amplitude glycemic excursion; MODD, mean of daily differences; CONGA, continuous overall net glycemic action; TIR, time in range; IQR, interquartile ranges; AGP, ambulatory glucose profile.
In this review, we summarize the findings about short-term GV, and we delineate the currently available information on its association with microvascular and macrovascular complications in patients with diabetes.
Pathogenic Background
The way that GV associates with diabetic complications seems to be a conjugation of different pathogenic mechanisms (Figure 1).
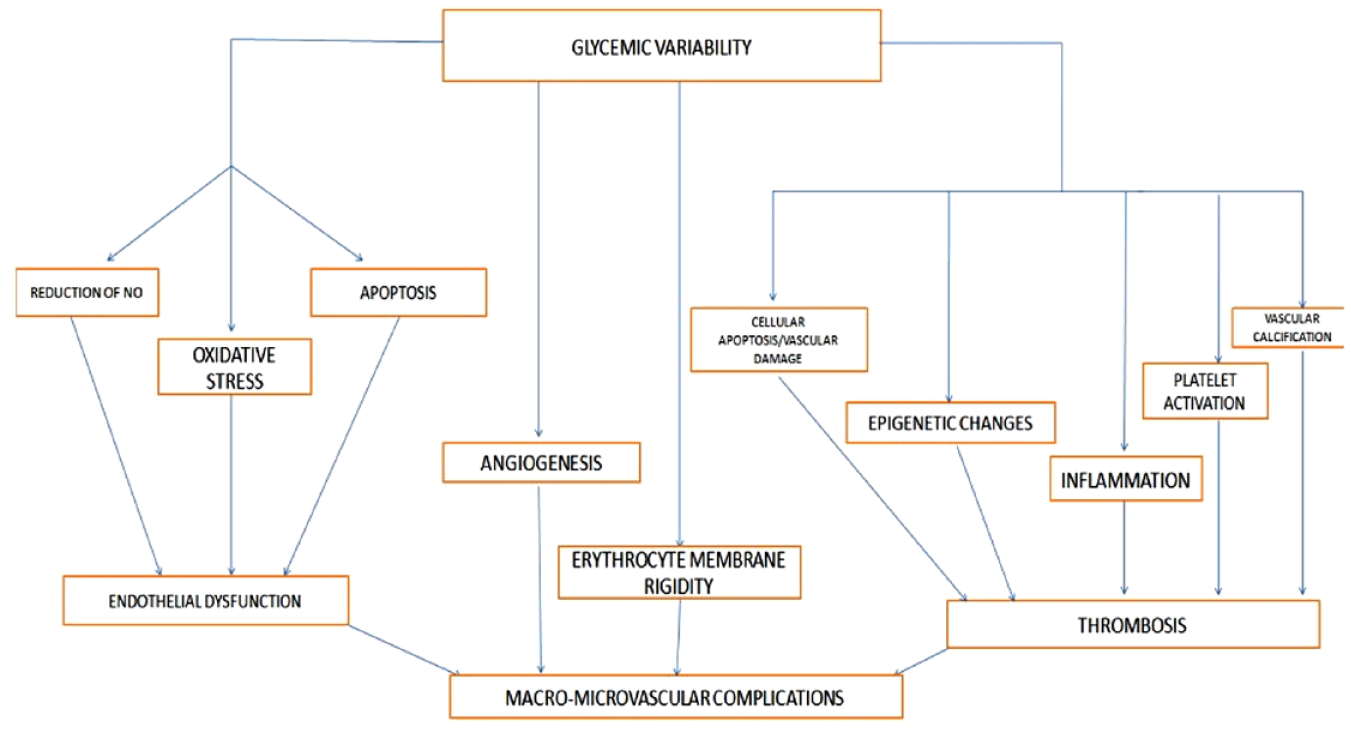
Pathogenic background of diabetic macrovascular and microvascular complications.
It is known that hyperglycemia generates oxidative stress by overproduction of the superoxide radical in mitochondria. 15 However, except chronic hyperglycemia, glycemic oscillations provoke endothelial dysfunction through oxidative stress. Ceriello et al 16 in their study demonstrated that glycemic spikes always have an impact on endothelial integrity and that high GV, over a period of 24 hours, is more damaging than stable excess of glucose. Short-term hyperglycemic spikes are associated with long-lasting epigenetic changes in the promoter of the nuclear factor KB subunit p65 in endothelial cells, leading to high p65 gene expression. This increase in p65 expression led to elevated expression of proatherogenic genes such as macrophage chemoattractant protein-1 (MCP-1) and vascular cell adhesion molecule-1 and could be inhibited by reducing mitochondrial superoxide production. 17 Risso et al 18 proved that human umbilical vein endothelial cells (HUVECs) were more prone to apoptosis when they were exposed to oscillating glucose than stable glycemic status. This result was enhanced when Quagliaro et al 19 demonstrated that exposure of HUVECs to oscillating high glucose levels results in reactive oxygen species (ROS) overproduction through a protein kinase C–dependent activation of nicotinamide adenine dinucleotide phosphate oxidase, inducing cellular apoptosis and vascular damage. Another group studied the relationship between stable and intermittent hyperglycemia and expression of vascular endothelial growth factor (VEGF) in human retinal endothelial cells, estimating at the same time the potential role of ROS. They demonstrated that HERCS exposed to stable hyperglycemia have increased cell proliferation, expression of VEGF, and production of oxidative markers. These outcomes were enhanced in cells that were exposed to fluctuating high glucose levels, rather than stable high levels, indicating that GV may be more damaging than stable hyperglycemia. 20 Monnier et al 21 came to the same conclusion showing that the urinary excretion rates of 8-iso-prostanglandin F2-alpha (PGF2a), a marker of oxidative stress, were found strongly correlated with MAGE but not with typical markers of mean glucose exposure, such as HbA1c, FPG, and so on. Another study exhibited that better glycemic control in terms of FPG and day-to-day GV led to the reduction of oxidative stress in patients with type two diabetes mellitus (T2DM). 22 Furthermore, Colomo et al 23 assessed for the first time the relationship between GV and oxidative stress in children with type one diabetes mellitus (T1DM). They measured oxidative stress using urinary secretion of 8-iso-PGF2, and they estimated GV as CV, MAGE, and SD, evaluating the patients for five days and proving that there was a significant correlation between oxidative stress and parameters of GV. 23 A correlation between GV and oxidative stress was also found in T2DM remitters after a bariatric surgery. 24 Except from oxidative stress, GV induces endothelial dysfunction by reducing the synthesis of nitric oxide, the vascular relaxing factor, in HUVECs via phosphoinositide-3-kinase/AKT1/endothelial nitric oxide synthase signal pathway. 25 Cell proliferation, angiogenesis, and migration functions in HUVECs were more seriously impaired by greater GV, which also provoked autophagy, with miRNA-1273g-3p array being the cornerstone of this injury. 26 Endothelial protection against hyperglycemia-induced metabolites can also be inhibited by the suppression of key enzymes due to glycemic oscillations. 27 Vascular calcification and aortic stiffness are other parameters related to short-term GV. A higher SD was correlated with a higher endoplasmic reticulum (ER)-induced apoptotic process; higher levels of 78-kDa glucose-regulated protein, caspase-3, Wnt1, galectin-3, and bone morphogenetic protein-2; and higher vascular calcification in patients with T2DM and unstable angina. 28 Snell-Bergeon et al 29 came to the same conclusion for men with T1DM. Based on the Maastricht study, Foreman et al 30 proved that higher daily glucose oscillations and lower TIR are associated with a higher carotid-femoral pulse wave velocity, which is a classic measure for aortic stiffness. It is known that repeated hypoglycemia induces platelet activation via prothrombotic mechanisms. 31 Aside from hypoglycemia-induced platelet activation, Nusca 32 et al were the first to prove a positive relationship between short-term GV and platelet reactivity, introducing a possible prognostic role of GV in rendering patients more prone to thrombosis.
The relationship between GV and inflammatory pathways has also been demonstrated. Hyperglycemia is associated with elevated levels of tumor necrosis factor alpha, interleukin-6, and cluster of differentiation 11B in monocytes, but this activation was stronger in conditions of oscillating high glucose levels. 33 The fluctuations in glucose level provoked M1 polarization of macrophages through the Toll-like receptor 4-interferon regulatory factor 5 pathway and also led to the expression of matrix metalloproteinase-9 which is associated with diabetic complications. 34 In human retinal pericytes, intermittent but not stable hyperglycemia triggered ER stress by enhancing the expression of activating transcription factor 4 and C/EBP homologous protein which are key factors of ER stress-related inflammation and cell death. The elevation of MCP-1 during oscillating, hyperglycemic conditions confirmed the role of GV in inflammation. 35 In mouse peritoneal macrophages, GV enhanced the IL-18 levels which are known to be related to diabetic atherosclerosis, via the c-jun N-terminal kinase pathway. 36 Sun et al 37 in their study demonstrated that GV led to overproduction of ROS, which correlates to elevated levels of inflammatory and proinflammatory adipokines and specifically plasminogen activator inhibitor-1 (PAI-1), MCP-1, and IL-18 and decreased levels of anti-inflammatory and antiatherogenic adipokine adiponectin, indicating a possible connection between GV, metabolic syndrome, and obesity. The simultaneous impact of short-term GV on both oxidative stress and chronic inflammation has also been discovered by Chang et al, 38 who estimated that MAGE was correlated with markers of oxidative stress but also with high-sensitivity C-reactive protein (hs-CRP). Moreover, fluctuating glucose provoked neural apoptosis and inflammatory response in diabetic female rats. 39 Furthermore, patients without diabetes with a coronary artery disease who had higher MAGE demonstrated higher hs-CRP and lower reactive hyperemia index, a measure of endothelial dysfunction. 40 The impact of GV on angiogenesis may indicate another missing link between GV and vascular complications. The number of circulating, endothelial progenitor cells (EPCs), which are bone marrow–derived cells responsible for endothelial repair and revascularization, had also a negative association with short-term GV,41,42 while reduction in MAGE, using continuous, subcutaneous insulin infusion, has led to an increase of EPCs in patients with T1DM. 43
Lastly, Rodrigues et al 44 not only proved the link between short-term GV and oxidative stress but also revealed a possible connection between glycemic oscillations and erythrocyte membrane rigidity. A higher membrane rigidity was noticed in the subgroup with higher CV, suggesting that greater GV influences the erythrocyte membrane nature, making it more rigid to hypo-osmotic lysis. These findings implicate a potential role of these cells in diabetic complications, as changes in membrane firmness make them more rigid and prevent their traversal through atherosclerotic vessels, commonly found in diabetes cases, 44 a phenomenon that may contribute to ischemic diabetic complications.
Short-term GV and Hypoglycemia
Numerous studies support the significance of short-term GV in relation to the risk of hypoglycemia. A reanalysis of the DCCT data set by Kilpatrick et al 45 has demonstrated that a 1-mmol/L rise in daily SD of blood glucose, independent of HbA1c levels, leads to a 1.07-fold increased risk of a first hypoglycemic event and even higher for subsequent events in insulin-treated T1DM patients. Another study investigating T1DM patients has shown that short-term GV measured using various indices including CV, MAGE, MODD, and CONGA was considerably higher and correlated to an increased number of hypoglycemic events. 46 Using CGM devices, Klimontov and Myakina demonstrated that, unlike HbA1c, CONGA2, mean absolute glucose, and MAGE could predict nocturnal hypoglycemia in insulin-treated T2DM patients. 47 A similar study has also shown that T2DM patients on insulin who experienced hypoglycemic events had significantly higher SD of glucose levels while there was no difference between HbA1c values in the hypoglycemic and nonhypoglycemic groups. 48 Moreover, two subsequent studies by Monnier et al 49 using CGM have demonstrated that the SD of the mean glucose levels in T1DM and insulin-treated and non-insulin–treated T2DM patients was significantly higher as the frequency of hypoglycemic events increased. 50 The most recent study by Monnier et al 51 has also defined a lower threshold of CV of glucose (36%), which, if exceeded, leads to a significant rise in the incidence of hypoglycemic events, irrespective of the type of diabetes and treatment received. Overall, short-term GV seems to play an important role in predicting hypoglycemia and should be considered during the management of diabetes.
Short-term GV and Macrovascular Complications
Glycemic Variability and Coronary Syndromes
The importance of short-term GV and the reason why it seems to attract the interest of more and more clinicians lie in its possible association with both macrovascular and microvascular diabetic complications. Back in 2007, Mita et al 52 studied the effect of glycemic swings on atherogenesis, demonstrating that glucose fluctuations accelerate macrophage adhesion to endothelium and the development of atherosclerotic plaques, irrespective of cholesterol levels. It is known that the most usual reason of acute coronary syndrome (ACS) is a thrombosis provoked by the rupture of a vulnerable coronary plaque. In their study, Xhang et al 53 investigated the relationship between short-term GV and culprit plaque fragility. They recruited elderly patients with non-ST segment elevation ACS, recording short-term glycemic parameters for 72 hours (MAGE, LAGE, MODD, postprandial glucose excursions) and assessing criminal plaques with coronary angiography and virtual histology intravascular ultrasound. They found out that GV metrics were negatively associated with percent fibrous volume but positively associated with percent necrotic volume and calcium volume, indicating that increased GV is correlated with increased culprit plaque vulnerability. 53 However, GV does not only affect culprit atherosclerotic lesions but it also seems to accelerate the development of nonculprit lesions, with MAGE being an independent predictor of their rapid progression. 54 Moreover, in another study, daily glucose swings were correlated with the development of lipid-rich plaques involving thin-cap fibroatheroma in coronary artery disease patients that are under statin or other lipid-lowering drugs. 55
Gerbaud et al 56 were the first to prove that higher glucose fluctuations are an independent predictor of midterm major adverse cardiovascular events in patients with T2DM experiencing an ACS. In patients hospitalized directly after an acute myocardial infarction (AMI), GV adversely affects myocardial salvage 57 and wall motion severity at the infarct area, increasing the final infarct size after myocardial reperfusion therapy, 58 as well as triggers left ventricular remodeling in the chronic phase. 59 The role of GV has also been studied by Lazzeri et al 60 who proved that glucose oscillations in patients with a cardiogenic shock after ΑΜΙ constitute an independent predictor of mortality. Acute glucose swings after transcatheter aortic valve implantation are correlated with major adverse events 30 days after the procedure, while hypoglycemia is not associated with this outcome. 61 T2DM patients, with elevated levels of intraday GV, demonstrate higher 10-year risk of cardiovascular disease estimated using the Framingham risk score. 62 Interestingly, increased MAGE was an independent predictor of contrast-induced nephropathy in patients with diabetes who underwent a PCI after AMI. 63
Glycemic Variability and Arrythmias
Except for ACS, glucose fluctuations may be the missing link between diabetic heart and arrhythmias. GV in patients with ACS not only increases the risk of major cardiovascular and cerebrovascular events in a 30-day follow-up but also triggers the incidence of atrial fibrillation during hospitalization. 64 Sertbas et al 65 proved that GV is associated with prolonged QTc duration and QTc dispersion, constituting a risk factor for life-threatening ventricular arrhythmias. In 2019, a multicenter retrospective study demonstrated that increased postoperative GV assessed with SD, MAGE, and CV for 24 hours after coronary artery bypass grafting constitutes a risk factor for atrial fibrillation. 66 Furthermore, the impact of GV on arrhythmias is affected by age. Higher levels of GV recorded in elderly make them more prone to atrial premature beats, couplets of atrial premature beats, atrial tachycardia, and ventricular premature beat. 67
Glycemic Variability and Stroke
The interplay between short-term GV and stroke is another point worth mentioning. Yoon et al 68 calculated the GV of patients with diabetes admitted with an acute ischemic stroke in the first three days. The study demonstrated that a higher GV, measured using the J-index, was associated with worse cardiovascular outcomes at three months. Dichotomization of patients according to their initial glucose levels did not result in significantly different cardiovascular outcomes, excluding the effect of hyperglycemia on GV. 68 A similar study reported a positive association between short-term GV and poor functional outcomes, based on the modified Rankin Scale, at three months in patients who had an acute ischemic stroke. This association remained significant irrespective of the number of glucose measurements and hypoglycemic events, as well as after stratification of patients according to their HbA1c levels on admission. 69 A study by Hui et al 70 established that early neurological deterioration (END), measured using the National Institute of Health Stroke Scale, in patients with T2DM and stroke, was associated with higher short-term GV calculated using SD and MAGE. Interestingly, HbA1c was not independently associated with END. 70 In another study using CGM devices, it was reported that increased short-term GV, measured during the first 72 hours of acute ischemic stroke, was associated with death or dependency at three months. Moreover, there was a significant association between short-term GV and END in the first seven days after the stroke. 71 Furthermore, a relationship has been found between increased short-term GV and infarct volume growth during the initial postischemic period, using a CGM device. 72 A recent article by Kim et al 73 has demonstrated a positive relationship between time-related short-term GV in the first three days following successful endovascular thrombectomy and worse functional outcomes in the first three months in patients admitted with an acute ischemic stroke. Additionally, the probability of a symptomatic intracerebral hemorrhage was significantly higher in the patients with elevated short-term GV. 73
Short-term GV and Microvascular Complications
Glycemic Variability and Neuropathy
The main microvascular complications in diabetes include diabetic retinopathy (DR), diabetic nephropathy, and diabetic peripheral neuropathy. 74 Avogaro and Fadini 75 in their review pointed out that microangiopathy can also include impairments in other organs such as brain, heart, and even EPCs in bone marrow. The role of short-term GV in diabetic microangiopathy still remains unclear.
Short-term GV could be an independent risk factor for diabetic peripheral neuropathy (DPN), as there is a correlation between MAGE and axonal loss of the medial, plantar nerve. 76 MAGE was also associated with excitability markers of modified motor and sensory axonal functions in patients with T1DM. 77 Furthermore T2DM patients with established DPN and well-controlled HbA1c demonstrated higher values of GV than patients without DPN. 78
Another chronic microvascular diabetes-related complication, cardiac autonomic neuropathy (CAN), seems to be associated with short-term GV in patients with both T1DM and T2DM. CGM parameters of short-term GV were related to CAN in patients with T1DM, while time spent in severe hypoglycemia was an independent predictor for CAN. 79 Higher SD was correlated with more serious CAN in patients with T1DM and severe systolic fall of blood pressure as a response to standing, indicating that GV plays a role in sympathetic dysfunction. 80 Furthermore, in adults with newly diagnosed T2DM, MAGE was a considerable CGM metric for discovering CAN. 81
On the other hand, short-term GV does not affect erectile function and the risk for premature ejaculation (PJ) in patients with T1DM. Caruso et al 82 enlisted for their study 112 young adults with T1DM and assessed sexual disorders by using appropriate questionnaires. Men with diabetes and high GV presented a similar incidence of sexual malfunctions with patients with low GV. 82 Moreover, in another study, MAGE and SD of blood glucose had no association with PJ, with GV demonstrating a significant correlation with PJ only in the hypoglycemic status. 83 More studies are required to prove the role of short-term GV in the prevalence of sexual disorders.
Glycemic Variability, Nephropathy, and Retinopathy
Short-term GV seems not to be associated with nephropathy as well. Short-term GV in patients with T2DM who experienced unexplained large glycemic oscillations have not raised the possibility of albuminuria. 84 Kilpatrick et al 85 using data from the DCCT trial failed to prove a relationship between short-term GV and microangiopathies including both retinopathy and nephropathy. Afterward, several studies confirmed their findings.86 -88 However, evidence exists to suggest the possible association between retinopathy and short-term GV. In a study of 2013, in patients with T1DM and T2DM, it was found that short-term GV, measured by CONGA and SD, was a potential risk factor for the development of DR; however, significance was lost after a multivariate regression analysis. 89 Last but not least, CONGA but not HbA1c, was correlated with retinal inner nuclear layer impairment, indicating a possible role of intraday GV in the function of muller glia and consequently to retinal neurodegeneration. 90 The majority of studies that investigated and proved the impact of GV on retinopathy and kidney diseases referred to long-term GV, underlying the need for more data about the role of short-term GV. MAGE has also been associated with cognitive impairment in older patients with T2DM, 91 indicating a possible role of GV in blood-brain barrier alterations and dementia.
Short-term Glycemic Variability and Therapeutic Interventions
Glycemic Variability and Nonpharmaceutical Interventions
The use of real-time CGMs (rt-CGMs) among patients with T1DM led to greater ameliorations in short-term GV metrics than self-monitoring of blood glucose, regardless of the way that insulin was delivered. 92 Also, in comparison with intermittently scanned CGM, rt-CGMs had induced greater improvements in CV and SD in patients at high risk of hypoglycemia. 93 Other nonpharmaceutical interventions that seem to affect GV are lifestyle and dietary changes. Moderate-intensity aerobic exercise before breakfast, in adults with dawn phenomenon, lowered short-term glucose oscillations throughout the day. 94 Munan et al 95 in their meta-analysis proved that short-term (less than two weeks) exercise improved both 24-hour glucose concentrations and MAGE. A low-carbohydrate high-fat breakfast decreased postprandial glycemic oscillations, premeal hunger, and sugar cravings. 96 However, neglecting breakfast led to postprandial hyperglycemia after lunch and dinner and, as a consequence, to worse glycemic control and greater GV. 97 Lower but sufficient carbohydrate consumption accompanied by protein calories (15% of the daily calorie intake) is necessary in order to achieve a better glycemic control in patients with T1DM (Table 2). 98
Nonpharmaceutical and Pharmaceutical Interventions.
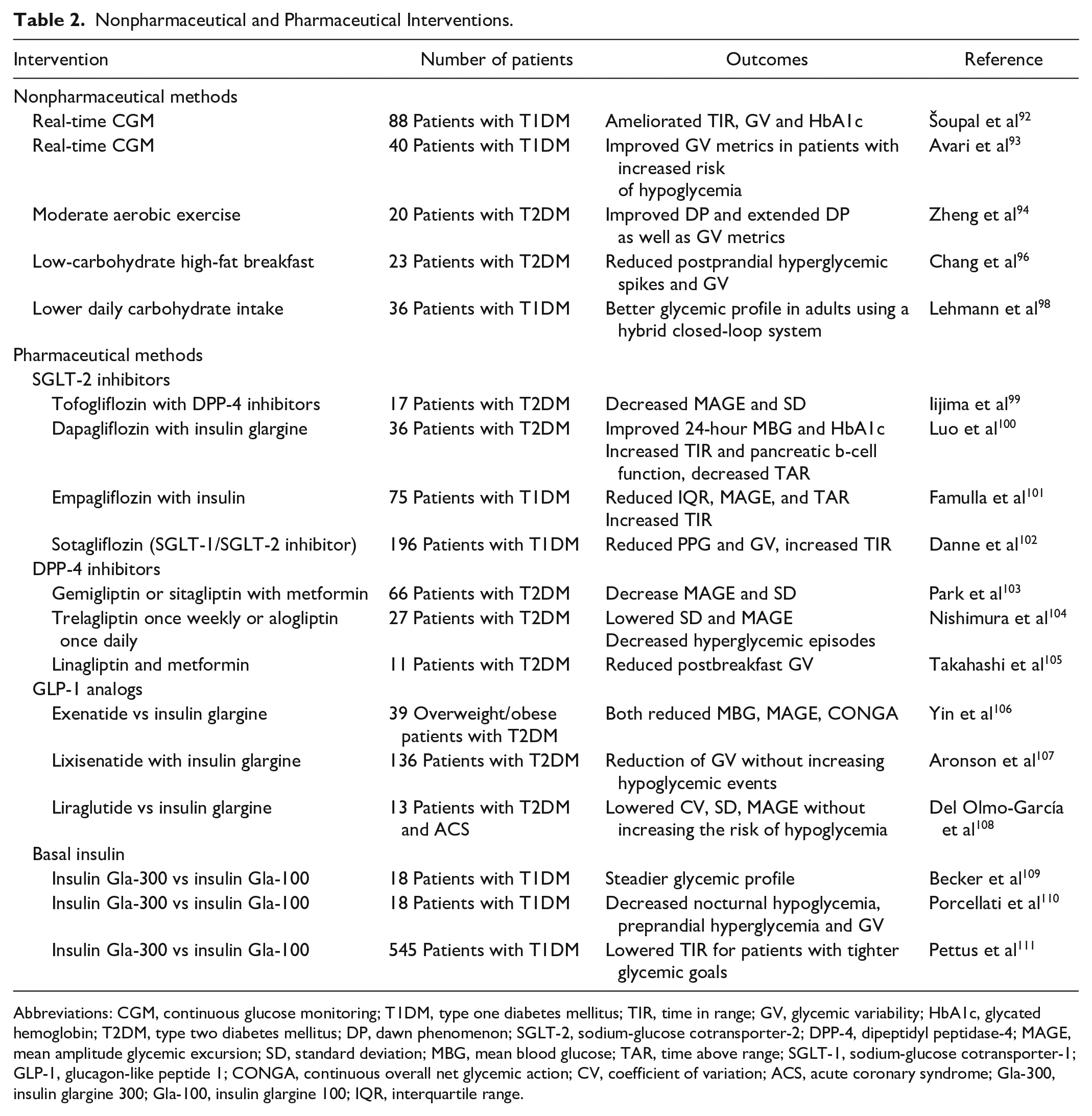
Abbreviations: CGM, continuous glucose monitoring; T1DM, type one diabetes mellitus; TIR, time in range; GV, glycemic variability; HbA1c, glycated hemoglobin; T2DM, type two diabetes mellitus; DP, dawn phenomenon; SGLT-2, sodium-glucose cotransporter-2; DPP-4, dipeptidyl peptidase-4; MAGE, mean amplitude glycemic excursion; SD, standard deviation; MBG, mean blood glucose; TAR, time above range; SGLT-1, sodium-glucose cotransporter-1; GLP-1, glucagon-like peptide 1; CONGA, continuous overall net glycemic action; CV, coefficient of variation; ACS, acute coronary syndrome; Gla-300, insulin glargine 300; Gla-100, insulin glargine 100; IQR, interquartile range.
Glycemic Variability and Pharmaceutical Interventions
Glucose-lowering agents that minimize glycemic fluctuations also exist, with combinations being usually the most effective. The combination of tofogliflozin, a sodium-glucose cotransporter 2 inhibitor (SGLT-2) with dipeptidyl peptidase 4 inhibitors (DPP-4) among adults with T2DM lowered MAGE and SD, while tofogliflozin alone increased TIR regardless of DPP-4 administration. 99 Short-term GV and TIR in patients under insulin glargine were improved after dapagliflozin was added, without increasing the risk of hypoglycemic events. 100 Empagliflozin in doses of 2.5 mg, 10 mg, and 25 mg among patients with T1DM, who already were under insulin therapy, significantly improved GV and TIR compared with placebo, mostly during night and early morning hours. 101 Danne et al 102 using sotagliflozin, a dual SGLT-1 and SGLT-2 inhibitor, came to the same conclusion. Patients who received gemigliptin or sitagliptin demonstrated fewer glycemic oscillations than patients treated with glimepiride, 103 while administration of trelagliptin once weekly or alogliptin once daily improved short-term GV and reduced blood glucose levels. 104 Moreover, in Japanese patients with poor glycemic control, the combination of low-dose metformin and linagliptin, even if it did not change GV parameters, ameliorated post-breakfast glycemic swings, compared to high-dose metformin. 105 Injectable, glucose-lowering therapies seem to have an impact on short-term GV as well. Both insulin glargine and exenatide, without a significant difference, improved GV in obese and overweight patients with poor glycemic control. 106 The combination of insulin glargine and lixisenatide in patients with T2DM reduced GV to a greater extent than glargine or lixisenatide alone, without increasing hypoglycemic episodes. 107 In patients with ACS, suboptimal administration of liraglutide (0.6 mg daily) during hospitalization decreased more CV, SD, and MAGE compared with insulin glargine, despite the fact that this difference was not statistically significant. 108 Pharmacokinetic (PK) and pharmacodynamic (PD) profiles of insulin glargine-300 (Gla-300) were steadier than those of insulin glargine 100 (Gla-100), creating in this way a more stable glycemic status. 109 Propitious differences in PK/PD profiles and endogenous glucose production have been recorded with the evening-dose of Gla-300, and as a consequence, patients demonstrated lower nocturnal insulin activity, greater insulin activity in the afternoon, and lower intraday PK/PD variability. Thereby, nocturnal hypoglycemia, preprandial hyperglycemia, and short-term GV can be decreased in patients with T1DM administered with Gla-300. 110 Pettus et al 111 in their study demonstrated that Gla-300 and Gla-100 affected TIR and GV equally, but for patients with tighter control (HbA1c < 7.5 at the end of the study), Gla-300 provoked greater improvements in TIR (Table 2).
Conclusions
GV and particularly short-term GV can be fairly considered as an HbA1c-independent risk factor for diabetic complications. Although there is rising evidence and plethora of data emphasizing the possible role of short-term GV in diabetic morbidity, considerable fields of research still remain unexplored or unclear, indicating the need for more studies. Perceiving the precise role of short-term GV in diabetic complications can be an additional useful tool in clinicians’ hands, guiding them to targeted and personalized therapeutic interventions.
Footnotes
Abbreviations
ACS, acute coronary syndrome; AGP, ambulatory glucose profile; AMI, acute myocardial infarction; CAN, cardiac autonomic neuropathy; CGM, continuous glucose monitoring; CGP, comprehensive glucose pentagon; CONGA, continuous overall net glycemic action; CV, coefficient of variation; DCCT, the diabetes control and complications trial; DP, dawn phenomenon; DPN, diabetic peripheral neuropathy; DPP-4, dipeptidyl peptidase-4; DR, diabetic retinopathy; END, early neurological deterioration; EPCs, endothelial progenitor cells; ER, endoplasmic reticulum; FPG, fasting plasma glucose; Gla-100, insulin glargine 100; Gla-300, insulin glargine 300; GLP-1, glucagon-like peptide 1; GPM, glucose pentagon model; GV, glycemic variability; HbA1c, glycated hemoglobin; HUVECs, human umbilical vein endothelial cells; MAGE, mean amplitude glycemic excursion; MBG, mean blood glucose; MCP-1, macrophage chemoattractant protein-1; MODD, mean of daily differences; PD, pharmacodynamic; PGF2a, 8-iso-prostanglandin F2-alpha; PJ, premature ejaculation; PK, pharmacokinetic; PPG, postprandial glucose; ROS, reactive oxygen species; rt-CGMs, real-time CGMs; SD, standard deviation; SGLT-1, sodium-glucose cotransporter-1; SGLT-2, sodium-glucose cotransporter-2; T1DM, type one diabetes; T2DM, type two diabetes; TAR, time above range; TIR, time in range; VEGF, vascular endothelial growth factor.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
