Abstract
Background:
We evaluated the effect of meloxicam on insulin lispro pharmacokinetics and glucose pharmacodynamics over 10 days of continuous subcutaneous insulin infusion (CSII) at one infusion site in people with type 1 diabetes (T1D).
Method:
This phase 1, randomized, double-blind, single-center, two-way crossover study enrolled adults with T1D for ≥1 year on stable CSII for ≥3 months. Participants randomly received U100 insulin lispro and LY900027 (U100 insulin lispro + 0.25 mg/mL meloxicam). Primary end points were area under the insulin lispro curve from 0 to 5 hours (AUCIns.0-5h) after bolus administration prior to a mixed-meal tolerance test (MMTT) and maximum observed concentration of insulin lispro (CIns.max) on days 5, 7, and 10, versus day 3 (baseline).
Results:
A total of 20 participants were randomized. Insulin absorption was accelerated for insulin lispro and LY900027 from days 1 to 7. The AUCIns.0-5h was significantly lower on day 10 versus day 3 for LY900027 (−19%) and insulin lispro (−14%); the AUCIns.0-5h did not differ significantly between treatments. The CIns.max increased with LY900027 and insulin lispro (by ~14%-23% and ~16%-51%) on days 5, 7, and 10 versus day 3. The CIns.max of LY900027 was ~14%-23% lower than insulin lispro CIns.max on days 7 and 10 (P ≤ .0805). Accelerated insulin absorption and a modest loss of total insulin exposure led to a loss of MMTT glycemic control at later time points.
Conclusions:
The pharmacokinetics of insulin changed over catheter wear time even when an anti-inflammatory agent was present. Postprandial glycemic control was adversely affected by the accelerated insulin absorption and decreased insulin exposure.
Keywords
Introduction
Recent years have seen a surge in innovations for the enhancement of insulin pump therapy. Among the latest of these advancements is the extension of the safe and viable wear time of insulin infusion sets beyond the standard 2 to 3 days. Multiple clinical studies have now demonstrated that up to 75% of insulin pump users may safely use a single infusion set for up to 7 days without experiencing unexplained hyperglycemia.1 -4 The reasons for unexplained hyperglycemia remain unknown, although several hypotheses propose local inflammation of the subcutaneous tissue at the infusion site as a likely cause.5 -11
Continuous glucose monitors (CGM) are also long-dwelling, subcutaneously placed devices augmenting pump therapy, which have faced challenges in maintaining patency over time due to localized inflammation.12 -14 Multiple efforts to temper such inflammation through the use of locally administered anti-inflammatory agents, such as corticosteroids, have been reported with some success.15 -18 The localized environments surrounding CGM and issues governing their patency may differ somewhat from cannulas designed for continuous subcutaneous insulin infusion (CSII). If localized inflammation indeed contributes to unexplained hyperglycemia at CSII time points beyond 7 days,5 -11 then it is reasonable to hypothesize that delivery of an anti-inflammatory agent to the site of insulin infusion may aid in deferring its onset.
Clinical studies evaluating extended-wear infusion sets reported thus far have been conducted as outpatient studies, with data collected from CGM and insulin pump downloads.1 -4 Such study designs are particularly useful for demonstrating safety and efficacy in a real-world scenario. However, CGM and pump downloads tell only a portion of the story of how insulin infusion rates and glycemic control evolve over time. Missing from the scientific literature thus far is an in-depth evaluation of how insulin pharmacokinetics (PK) and glucose pharmacodynamics (PD) evolve during extended wear. Here, we report the results of the first study evaluating insulin lispro PK and glucose PD over the course of 10 days of CSII at a single infusion site in people with type 1 diabetes (T1D; NCT04161976). In addition, we hypothesized that an anti-inflammatory compound added to an insulin formulation could prolong the catheter wear time while maintaining the insulin lispro PK and PD properties over time. We therefore examined whether the addition of the nonsteroidal anti-inflammatory drug (NSAID) meloxicam to the insulin lispro formulation could improve infusion site patency over time.
Methods
Study Design
This was a phase 1, randomized, double-blind, single-center, two-way crossover study conducted at Profil, Neuss, Germany, between December 2019 and August 2020. After providing informed consent and confirming eligibility, participants were randomized to 1 of 2 treatment sequences: U100 insulin lispro + 0.25 mg/mL meloxicam (LY900027) followed by U100 insulin lispro or U100 insulin lispro followed by LY900027 delivered through CSII. The 0.25 mg/mL dose of meloxicam was chosen based on studies in pigs, which showed that the addition of this dose of meloxicam to the U100 or U200 formulation of insulin lispro maintained the glucose response to insulin when an infusion site was used for >3 days.
The two 10-day treatment periods were separated by 7 to 21 days (Supplemental Figure 1). Participants were in-clinic for 1 night from day −1 to day 1, and day 2 to day 3, and then from the evening of day 4 until day 10 of each treatment period. The PK and PD of insulin were assessed by frequently sampled insulin PK profiles and a breakfast mixed-meal tolerance test (MMTT) at baseline on day 1, and then again on days 3, 5, 7, and 10.
Details of ethics approval are summarized in the supplemental methods.
Study Population
Enrolled participants were those aged between 18 and 64 years with T1D for ≥1 year on stable CSII for at least 3 months, with a total daily insulin dose of >0.4 to ≤1.5 U/kg/day and a glycated hemoglobin of ≤9.0%. Exclusion criteria included the following: known allergies to adhesives, NSAIDs (eg, meloxicam), insulin lispro, insulin glulisine, related compounds, or any components of the study drugs. The full list of inclusion and exclusion criteria are in Supplemental Table 1.
Treatment
A standardized CSII soft Teflon catheter with a 6-mm needle (Medtronic QuickSet) was inserted on day −1 of each treatment period, and patients underwent an overnight stabilization phase with intravenous insulin glulisine or glucose infusion. A fixed basal rate of CSII was administered from −1 hour until the end of the MMTT. Participants continued their individual CSII basal rate pattern using the Medtronic 640G insulin pump (Northridge, CA, USA) during the entire study, including the meal test days, unless safety issues arose; during the MMTT, the basal rate was changed to a single hourly rate based on the participant’s individual basal needs from at least 1 hour before the start of the MMTT until the end of the meal test period (300 minutes), and then changed back to the individual basal rate after completion of the meal test.
Study drugs were administered as standard single-wave bolus doses on days 1, 3, 5, 7, and 10 after catheter insertion, using a Medtronic MiniMed 640G pump with 1.5 U/min speed, and glucose monitored using the Dexcom G6 GCM system (San Diego, CA, USA). The bolus doses of study drug were individualized per participant to account for the carbohydrate content of the standardized meals, while participants were inpatients, and meals consumed outside of the test meals. The bolus doses administered prior to the MMTT for both insulin lispro and LY900027 were the same on all MMTT days for individual subjects. Bolus dose modification was allowed once after the MMTT on day 1 to further optimize the bolus dose if necessary.
The catheter inserted on day −1 stayed at the same catheter insertion-site location until completion of day 10 or until a change of catheter became necessary. Study drugs (insulin lispro and LY900027) were continuously infused throughout the 10-day treatment period. Treatment was stopped in the event of the following: catheter failure due to an unexplained hyperglycemic episode, with plasma glucose (PG) >250 mg/dL not responding to correction bolus administered through subcutaneous insulin infusion or any adverse event (AE) deemed possibly or probably related to study drug infusion, which is considered unacceptable by the participant and/or the investigator, and which requires change of the infusion catheter. If catheter failure occurred during the first treatment period, participants were permitted to undertake the second treatment period after the required washout time.
End Points
The primary end points were area under the insulin lispro curve from 0 to 5 hours (AUCIns.0-5h) after bolus administration prior to the MMTT on days 5, 7, and 10, and maximum observed concentration of insulin lispro (CIns.max) on days 5, 7, and 10, compared with the day 3 baseline.
Secondary end points included both PK and PD end points. The PK end points were AUCIns.0-0.5h, AUCIns.0-1h, AUCIns.0-2h, and AUCIns.0-4h after bolus administration prior to a MMTT on days 1, 3, 5, 7, and 10; AUCIns.0-5h on day 1 and time to maximum observed serum insulin concentration (tIns.max) on days 1, 3, 5, 7, and 10. The PD end points were as follows: incremental area under the PG curve above baseline from 0 to 5 hours (ΔAUCPG.0-5h) after bolus infusion on days 1, 3, 5, 7, and 10; maximum PG excursion (ΔPGmax), after bolus infusion on days 1, 3, 5, 7, and 10; and daily insulin requirements, including total daily insulin dose (basal and bolus), total daily prandial insulin doses, total daily basal dose, and number and total daily dose of correction bolus doses. Insulin PK and glucose PD profiles were presented as unadjusted data and baseline-adjusted data, where the baseline value was subtracted from all postdose measurements. Other end points are summarized in Supplemental Table 2.
Assessments
Treatment compliance
The study staff administered all doses of study drugs at the trials site, with administered doses recorded in a case report form. During the outpatient period between days 1 and 4, participants documented all administrations of study drugs and any hyper- or hypoglycemic episodes in a diary, which was checked at all visits to the study site.
Pharmacokinetic/pharmacodynamic assessments
The details on the PK and PD assessments performed are summarized in the supplemental methods.
Safety
Adverse events occurring until the follow-up visit were considered as treatment-emergent. The local tolerability at the catheter infusion site was evaluated by means of the following assessments: spontaneous pain, pain on palpation, itching, redness, edema, and induration/infiltration. As per the protocol, only reactions where the severity or duration of the changes seen were more severe or prolonged than would be expected following catheter removal were recorded as AEs. Each of these assessments was reported on a scale of 0 (none), 1 (mild), 2 (moderate), and 3 (severe). In case of catheter infusion site reactions (ISRs), detailed evaluation was performed by a blinded trial physician.
Statistical Analysis
The planned sample size was 20 patients so that 14 patients would complete the trial. Based on an analysis of internal Lilly trial data on multiple-dose insulin lispro, the expected 90% confidence interval (CI) for within-patient comparison in the logarithm of AUCIns.0-5h of serum insulin lispro was 0.87-1.15 for 14 patients.
The full analysis set and the safety analysis set were defined as all randomized participants, while the modified full analysis set was defined as all participants who completed the study. The PK end points were calculated based on standard noncompartmental methods using Phoenix WinNonlin (Certara L.P., Princeton, NJ, USA, Version 8.1). Furthermore, SAS for Windows (SAS Institute Inc., Cary, IN, USA, Version 9.4) was employed for all other statistical calculations and all analyses. All available data were included in data listings and tabulations. No imputation of values for missing data was performed.
In general, continuous variables were presented using descriptive summary statistics, including arithmetic mean (mean), coefficient of variation (CV), geometric mean (GeoMean) and geometric CV (GeoCV), median, minimum (Min), maximum (Max), and SD.
Mixed-effect models were performed between LY900027 and insulin lispro as well as between days within each treatment using SAS procedure PROC MIXED (see supplemental methods for details). Least-squares means (LSMs), their CIs, and P values were generated. Values were considered statistically significant at P < .1.
Results
Study Population
A total of 24 participants were screened and 20 participants were randomized. Eighteen participants completed the study, with two participants discontinuing the study prematurely (reported as “not willing to continue study” or for “personal reasons”).
The baseline characteristics of the participants in the full analysis set are summarized in Supplemental Table 3.
Insulin Pharmacokinetics
Mean insulin profiles showed accelerated absorption for both insulin lispro and LY900027 with increasing catheter wear time. Increases in insulin lispro concentrations within the first hour assessment period became more pronounced with progressing catheter wear time from day 1 to day 7 in both the unadjusted and adjusted concentration profiles (Figure 1; Supplemental Figure 2).
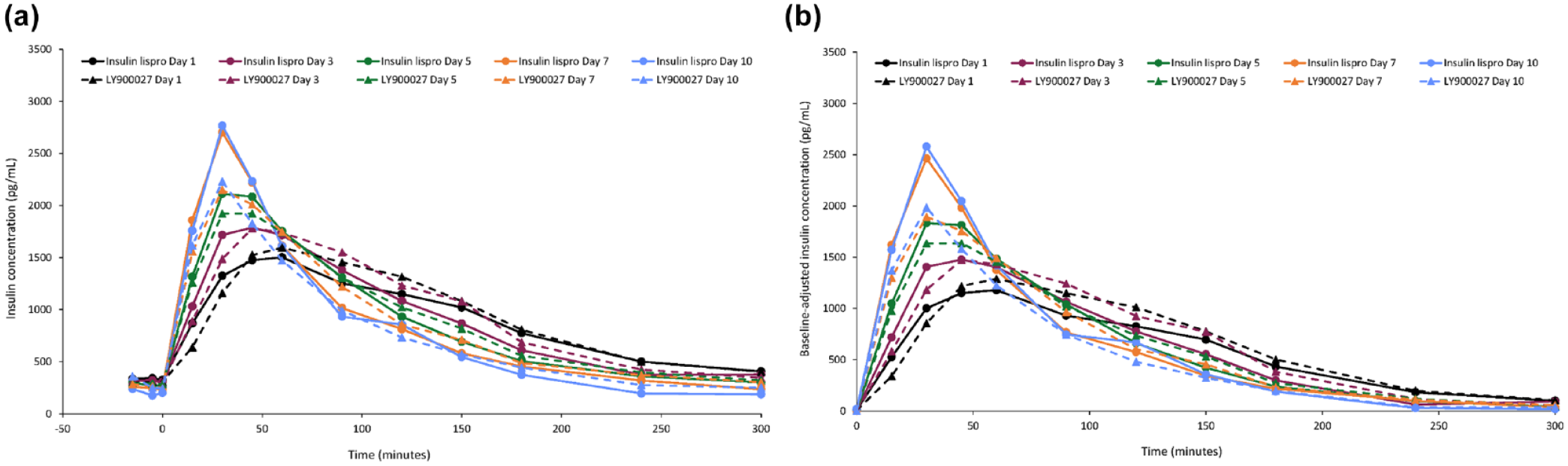
Mean insulin concentration-time profiles after bolus administration of study drugs prior to a standard mixed meal: (a) Unadjusted profile and (b) Baseline-adjusted profile.
The within-treatment comparison for overall post-meal exposure (AUCIns.0-5h) after the MMTT showed similar exposure on days 5 and 7 versus day 3, and a significantly lower exposure on day 10 versus day 3 (19% and 14% lower for LY900027 and insulin lispro, respectively; Table 1). For the baseline-adjusted insulin exposure parameters, ΔAUCIns.0-5h values for LY900027 were significantly lower than those for insulin lispro only on day 10 (0.831 vs. 1.025; P = .011) but were similar on day 3 to day 7. CIns.max increased compared with the day 3 baseline, being approximately 14% to 23% higher for LY900027 and 16% to 51% higher for insulin lispro on days 5, 7, and 10 versus day 3 (Table 1). There was a significantly (P < .1) shorter tmax within treatments due to faster absorption on days 5 (LY900027 only), 7 (LY900027 and insulin lispro), and 10 (LY900027 and insulin lispro) versus day 3 (Table 1).
Within- and Between-Treatment Comparisons of Primary and Selected Secondary Pharmacokinetic End Points in the Modified Full Analysis Set.
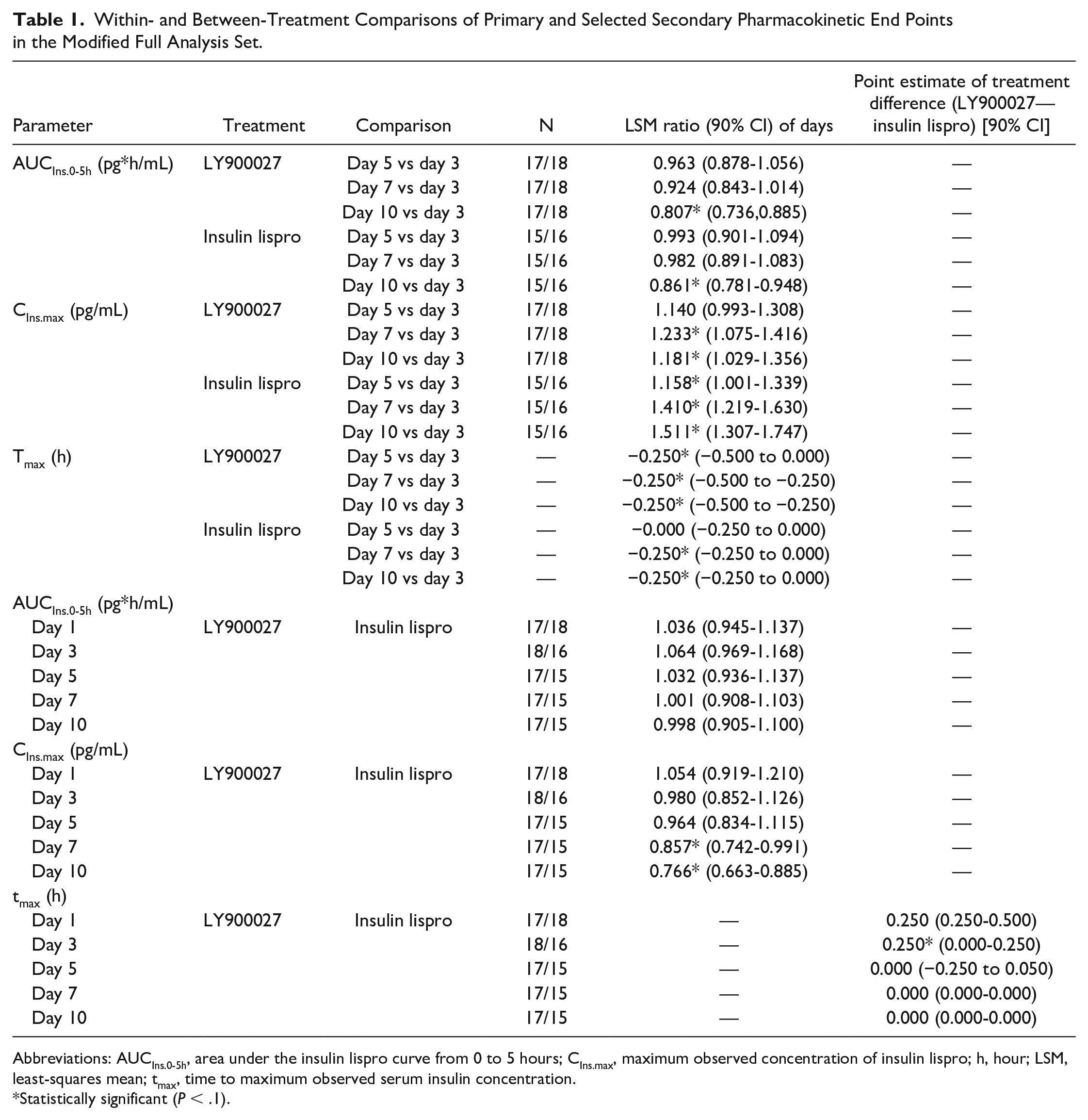
Abbreviations: AUCIns.0-5h, area under the insulin lispro curve from 0 to 5 hours; CIns.max, maximum observed concentration of insulin lispro; h, hour; LSM, least-squares mean; tmax, time to maximum observed serum insulin concentration.
Statistically significant (P < .1).
In the between-treatment comparison, AUCIns.0-5h did not differ significantly between LY900027 and insulin lispro on any of the assessment days (Table 1). Insulin exposure during the 0- to 2-hour and 0- to 4-hour intervals on all assessment days was also not different between LY900027 and insulin lispro. The CIns.max of LY900027 was approximately 14% to 23% lower than that of insulin lispro toward the end of the treatment period (days 7 and 10: ≤0.0805). There was no difference between treatments during the first half of the treatment period with shorter catheter wear time (days 1-5; P ≥ .5261). The tmax for LY900027 was reached by ~15 minutes later than insulin lispro on days 1 and 3 (statistically significant based on P values; P ≤ .0596) but not on days 5, 7, and 10 (Table 1).
Glucose Pharmacodynamics
The mean unadjusted and adjusted glucose profiles showed insufficient postprandial glucose control from day 5 onward (Figure 2). Overall, within-treatment glucose excursions over 5 h (AUCPG.0-5h and ΔAUCPG.0-5h) and maximum PG excursions (PGmax and ΔPGmax) increased over time for both LY900027 and insulin lispro (Table 2). The increases on days 7 and 10 were statistically significant versus day 3. There were no statistically significant between-treatment differences in PG end points. Supplemental Figures 3 and 4 show the PG curves for the first 90 minutes of the time course.
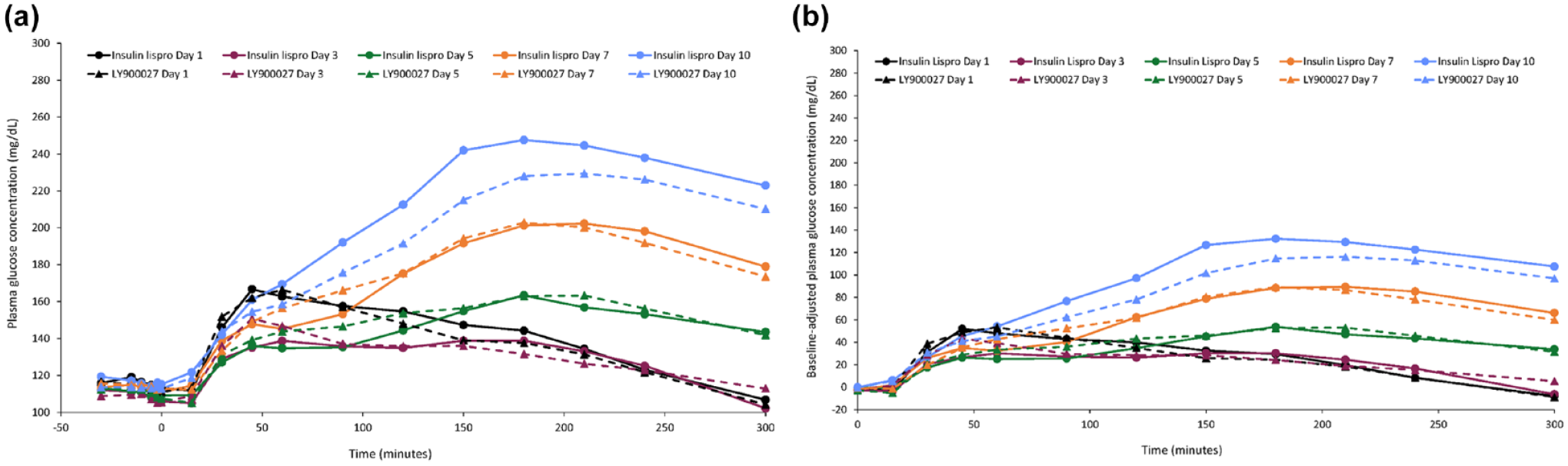
Mean plasma glucose concentration-time profiles after bolus administration of study drugs prior to a standard mixed meal: (a) Unadjusted profile and (b) Baseline-adjusted profile.
Unadjusted and Baseline-Adjusted Within-Treatment Comparisons of Plasma Glucose End Points in the Modified Full Analysis Set.
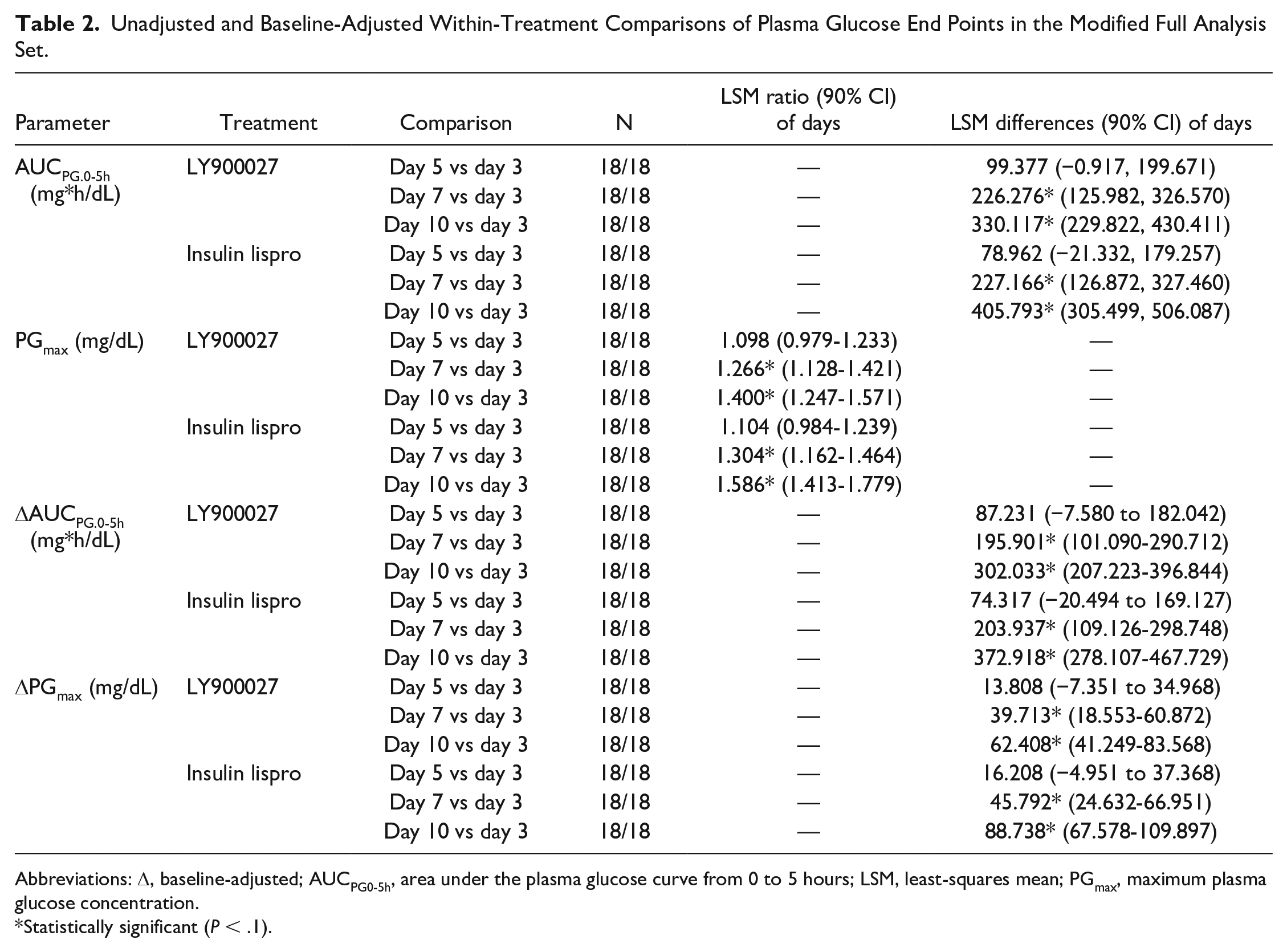
Abbreviations: Δ, baseline-adjusted; AUCPG0-5h, area under the plasma glucose curve from 0 to 5 hours; LSM, least-squares mean; PGmax, maximum plasma glucose concentration.
Statistically significant (P < .1).
Insulin Dose
Supplemental Figure 5 shows the insulin dose trend across days for total daily insulin dose, prandial insulin dose, basal dose, and the number of bolus correction doses. Basal insulin rates for each participant remained unchanged throughout the study, by design. For both LY900027 and insulin lispro, total daily insulin dose increased by approximately 10% to 15% from day 3 to day 9, primarily driven by increased prandial bolus dose and increased frequency of correction bolus doses. The within-treatment differences of total insulin doses were statistically significant for days 6, 8, and 9 with LY900027, and days 6, 7, 8, and 9 with insulin lispro (Table 3). Overall, insulin requirements between LY900027 and insulin lispro were similar and not statistically significantly different.
Within-Treatment Comparison for Total Daily Insulin Dose in the Modified Full Analysis Set.
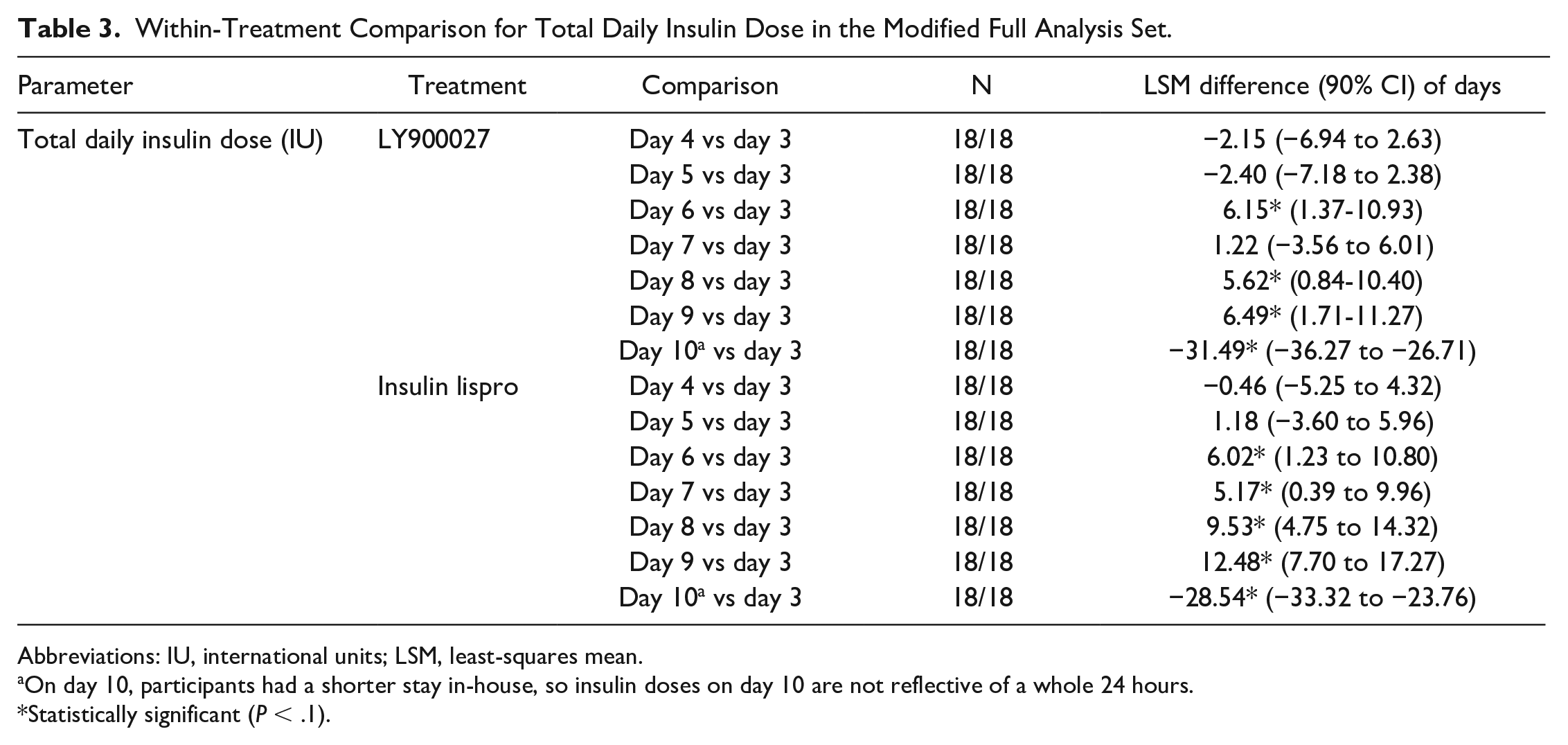
Abbreviations: IU, international units; LSM, least-squares mean.
On day 10, participants had a shorter stay in-house, so insulin doses on day 10 are not reflective of a whole 24 hours.
Statistically significant (P < .1).
Catheter Wear Time
There were no catheter failures attributed to the prolongation of usual catheter wear time to >3 days. Three catheter failures occurred prior to day 3, 1 before starting the first treatment period (visit 2, day −1), 1 due to unexplained hyperglycemia with noticed leakage of insulin in the outpatient period before the second treatment period (visit 3, day −1), and 1 due to a protracted hyperglycemic episode in the evening of visit 3, day −1. As, in all three cases, the catheter was removed prior to day 3, patients were allowed to continue in the study, 2 patients restarting at a rescheduled visit, and 1 patient with a newly inserted catheter.
Hyperglycemic Episodes
During the treatment period, 33 unexplained hyperglycemic episodes (defined as PG >250 mg/dL) were recorded. None met the criteria for an AE (ie, clinically significant hyperglycemia) and all could be resolved without premature termination of a treatment period. Sixteen of the unexplained hyperglycemic episodes occurred in 8 participants (44.4%) during treatment with LY900027 and 17 in 9 participants (50.0%) during treatment with insulin lispro. Two episodes occurred in the outpatient period (close to visit 3, day −1 or visit 2, day 4), and 1 was documented with polyuria.
Continuous Glucose Monitoring
There was no change in 24-hour average glucose over time as measured by CGM (Supplemental Figure 6). In addition, 24-hour average glucose was similar between LY900027 and insulin lispro (Supplemental Table 4). Percentage of time in range also did not change over time with insulin lispro and LY900027 (Supplemental Table 4).
Safety and Tolerability
At least 1 treatment-emergent AE (TEAE) was experienced by 13 participants (68.4%) after dosing with LY900027 and 17 participants (85.0%) after dosing with insulin lispro. The most frequently reported TEAEs were ISRs (similar between-treatment groups: 63.2% with LY900027 and 75.0% with insulin lispro), headache (10.5% with LY900027 and 5.0% with insulin lispro), and infusion site complications, phlebitis (5.3% with LY900027 and 5.0% with insulin lispro), and thrombophlebitis (5.3%) with LY900027.
With regard to pain associated with infusions, the majority of participants had a pain visual analogue score (VAS) of <10/100 mm; there were 8 occurrences of a VAS pain score >10/100 mm (2 in the LY900027 treatment period and 6 in the insulin lispro treatment period). Median VAS scores were similar between treatments.
There was no between-treatment difference in hypoglycemic events. Most hypoglycemic events were mild (level 1), some were level 2, and there were no severe hypoglycemic events reported. No significant clinical laboratory or vital signs findings were reported.
Discussion
This study demonstrated accelerated absorption for both insulin lispro and LY900027 that became more pronounced as catheter wear time progressed up to day 7, with no substantial loss of insulin bioavailability prior to day 10. However, the accelerated insulin absorption led to a loss of MMTT glycemic control in the later part of the time action profile following the meal, due to the left-shifted rapid time-action profile of the insulin, no longer covering the late carbohydrate absorption of a mixed meal. In contrast, glucose values during the early postprandial period were lower as a result of acceleration of insulin absorption from day 3 to day 10. These PK and PD data identify a potential cause of the previously unexplained hyperglycemia in patients treated with CSII, which interestingly is not a significant reduction in insulin bioavailability but an acceleration of the insulin PK profile leading to a mismatch in the insulin coverage for the entire postprandial duration following a mealtime bolus up to day 7. The combination of accelerated insulin PK profile and reduced bioavailability further enhanced the loss of MMTT glycemic control on day 10. Acceleration of insulin pharmacodynamic effect has been reported in the literature by other groups,5,11,19,20 sometimes referred to as the “Tamborlane effect,” but this is the first report of PK and PD data up to 10 days of catheter wear displaying progressive acceleration of insulin absorption, resulting in suboptimal postprandial glucose control.
There was no change in the basal insulin dose during the trial as per the design, with only a modest increase (up to 15%) in total prandial insulin dose requirement, and no cases of catheter failure due to the prolonged wear time; of note, the study was not powered to detect differences in this end point. No safety signals of concern were reported.
The presence of meloxicam did not appear to significantly affect the results, with no consistent differences in PK and PD parameters, insulin dose requirement, or safety parameters from day 1 to day 10 between LY900027 and insulin lispro. The CGM data showed similar average glucose and TIR with LY900027 and insulin lispro throughout the 10-day study period.
Catheter failure was expected in this trial based on clinical experience reported in the literature, especially after day 4 or 5; because of this, study participants were required to be inpatients under medical supervision. Surprisingly, no occurrences of catheter failure were reported by participants up to 10 days of use. Moreover, all cases of unexplained hyperglycemia with PG >250 mg/dL were effectively treated with correction insulin bolus doses administered using the same catheter and insulin pump. Prolongation of catheter wear time did not lead to a complete loss of glycemic control but resulted in impaired glucose control, which could be managed with additional correction doses. Thus, it is advisable that patients who choose to use their catheter for a longer period of time should be aware of the findings reported here to safely control their glycemic levels.
A study that investigated the PK and PD of insulin with an extended-wear insulin infusion set in patients with T1D 2 has several key differences from this study, including a shorter wear time (7 days) and evaluation of PD under clamp conditions versus the use of a meal test to mimic a real-life scenario as in this study.
A limitation of this study is the lack of a diverse population, with participants being 90% men and 100% white, although this would not be expected to affect the study conclusions. A strength of this study is the crossover design, the collection of PK data, and the use of an MMTT, all of which enable a robust assessment of the utility of meloxicam in altering catheter wear time. However, the absence of catheter failure due to unmanageable injection site reactions, or uncontrolled hyperglycemia, is surprising. Frequent but mild injection site reactions were seen, and hyperglycemia was also apparent; however, both events were of similar frequency and severity between treatments, and neither led to discontinuation of the catheter. Of note, participants were inpatients after day 3, and therefore had more access to medical monitoring and advice than would be possible with standard outpatient care. Immediate access to inpatient care may have improved glycemic control and provided more reassurance to participants if they developed episodes of hyperglycemia or mild injection site reactions, enabling them to complete the 10-day wear time.
Conclusion
This study demonstrated that the PK of insulin changed over catheter wear time even in the presence of an anti-inflammatory agent in participants with T1D treated with CSII. A modest loss of total insulin exposure combined with an accelerated insulin PK profile had a significant deleterious impact on postprandial glycemic control.
Supplemental Material
sj-docx-1-dst-10.1177_19322968221145200 – Supplemental material for Evaluation of Insulin Lispro Pharmacokinetics and Pharmacodynamics Over 10 Days of Continuous Insulin Infusion in People With Type 1 Diabetes
Supplemental material, sj-docx-1-dst-10.1177_19322968221145200 for Evaluation of Insulin Lispro Pharmacokinetics and Pharmacodynamics Over 10 Days of Continuous Insulin Infusion in People With Type 1 Diabetes by Parag Garhyan, Edward Pratt, Oliver Klein, Susanne Famulla, Eric Zijlstra, Amy Lalonde, Monica Swinney, Christof Kazda and Eyal Dassau in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors would like to acknowledge Sheridan Henness and Francisco López de Saro (Rx Communications, Mold, UK) for medical writing assistance with the preparation of this manuscript, funded by Eli Lilly and Company.
Abbreviations
ΔAUCIns/PG.0-5h, incremental area under the insulin lispro/PG curve from 0-5 hours; ∆PGmax, maximum PG excursion; AE, adverse event; AUCIns/PG.0-xh, area under the insulin lispro/PG curve from 0-x hours; BMI, body mass index; CGM, continuous glucose monitoring; CIns.max, maximum observed concentration of insulin lispro; CSII, continuous subcutaneous insulin infusion; CV, coefficient of variation; GeoMean, geometric mean; GeoCV, geometric CV; HbA1c, glycated hemoglobin; ISRs, infusion site reactions; LLOQ, lower limit of quantification; LSM, least-squares mean; Max, maximum; Min, minimum; MMTT, mixed-meal tolerance test; NSAID, nonsteroidal anti-inflammatory drug; PD, pharmacodynamics; PG, plasma glucose; PK, pharmacokinetics; T1D, type 1 diabetes; TEAE, treatment-emergent adverse event; tIns.max, time to maximum observed serum insulin concentration; TiR, time in range; ULOQ, upper limit of quantification; VAS, visual analogue score.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Parag Garhyan, Edward Pratt, Amy Lalonde, Christof Kazda, and Eyal Dassau are employees and shareholders of Eli Lilly and Company. Monica Swinney was an employee of Eli Lilly and Company at the time the study was conducted. Oliver Klein, Susanne Famulla, and Eric Zijlstra are employees of Profil.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Eli Lilly and Company.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
