Abstract

Continuous subcutaneous insulin infusion (CSII) therapy has been shown to effectively improve glycemic control and quality of life in type 1 Diabetes Mellitus (DM1) patients1,2; however, its prescription requires careful consideration of factors such as patient adherence, understanding, and technical proficiency. 3 Ensuring uninterrupted insulin delivery makes any failure in the CSII system potentially life-threatening. 4 Despite advances in technology for treating DM1, adverse events (AEs) remain common, affecting at least 40% of patients annually. 5
The present study was conducted at the Diabetes Center Insulin Pump Ambulatory, Federal University of São Paulo, to identify AEs associated with batteries from different brands used in CSII devices. Semi-structured interviews via video call were conducted with 118 patients using the device for at least six months. The collected data were systematically categorized into five areas: user interface, alert system, software and connectivity, durability, and electrical system, with the latter being the primary focus of this study on battery performance. 6
The primary device manufacturers involved were Roche, Medtronic, and Tandem Diabetes Care, with Medtronic recommending AAA Energizer alkaline batteries and Roche recommending AA Powerone batteries. While Roche users consistently adhered to these recommendations, Energizer users occasionally deviated from the guidelines. Educating users on the importance of following battery recommendations could help prevent related issues. Among Roche users, 54% reported that Powerone batteries lasted up to 60 days, whereas 30% of Energizer users with continuous glucose monitoring (CGM) sensors observed a battery lifespan of up to 30 days (Figure 1). No significant correlations were found between demographic variables and the occurrence of AEs across different age groups, suggesting that user profiles do not strongly influence AEs. Instead, battery-related AEs appear to be more closely associated with hardware malfunctions. Reported AEs include persistent or premature low battery warnings, often occurring after replacing with a new or non-recommended battery. Other key issues involve battery lot defects, reduced insulin delivery due to low battery alerts, device restarts upon battery replacement, and rapid battery level drops after replacement. Additional problems include error messages indicating the battery has not been replaced, automatic data formatting after prolonged periods without battery power, and structural issues such as loose or stuck battery compartment lids, and oxidation in the battery compartment.
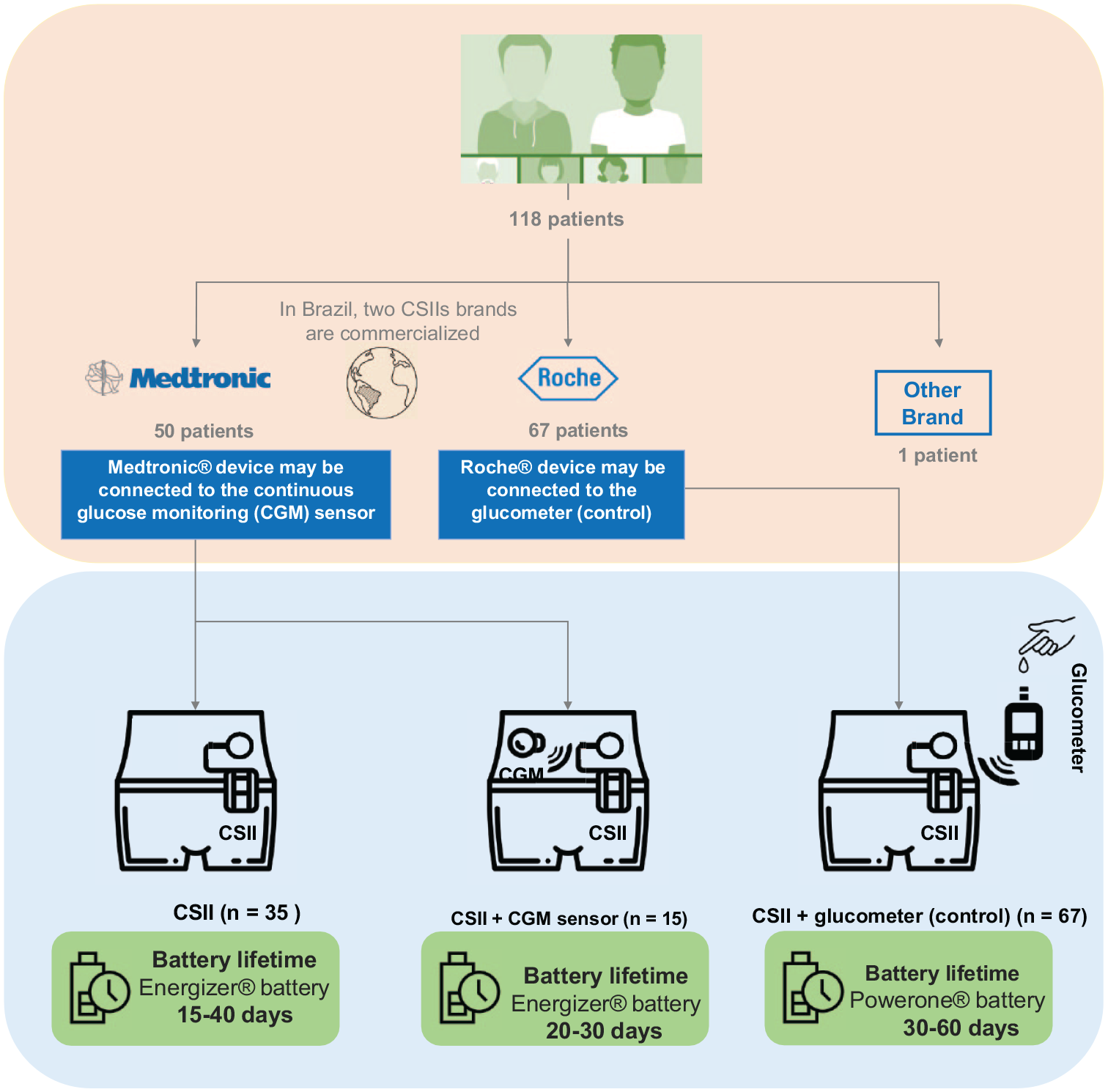
Comparative analysis of Powerone and Energizer battery life in Roche and Medtronic CSIIs.
In this study, AEs were examined across a diverse population, including children, adolescents, and adults. Key findings revealed that battery-related issues with CSIIs are common, particularly across different Medtronic device models, highlighting the need for personalized recommendations. These results point to critical patient safety concerns in DM1 treatment and emphasize the importance of ongoing monitoring, safety protocols, and patient education. Incorporating these insights into clinical practice, regulatory frameworks, and device manufacturing is essential to improving the safety and effectiveness of CSII therapy.
Footnotes
Acknowledgements
Patients with DM1 and staff of the Diabetes Center Insulin Pump Ambulatory, Federal University of São Paulo for their unrestricted support in data collection.
Abbreviations
AEs, adverse events; CGM, continuous glucose monitoring; CSII, Continuous Subcutaneous Insulin Infusion; DM1, diabetes mellitus Type 1.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Coordination for the Improvement of Higher Education Personnel—Brazil (CAPES, Funding Code 001), São Paulo Research Foundation (FAPESP No. 2018/22094-0), and the National Council for Scientific and Technological Development (CNPq 308721/2020-9).
