Abstract
Background:
Initiation of continuous subcutaneous insulin therapy (CSII) in type 1 diabetes requires conversion of a basal insulin dose into a continuous infusion regimen. There are limited data to guide the optimal insulin profile to rapidly achieve target glucose and minimize healthcare professional input. The aim of this pilot study was to compare circadian and flat insulin infusion rates in CSII naïve adults with type 1 diabetes.
Methods:
Adults with type 1 diabetes commencing CSII were recruited. Participants were randomized to circadian or flat basal profile calculated from the total daily dose. Basal rate testing was undertaken on days 7, 14 and 28 and basal rates were adjusted. The primary outcome was the between-group difference in absolute change in insulin basal rate over 24 hours following three rounds of basal testing. Secondary outcomes included the number of basal rate changes and the time blocks.
Results:
Seventeen participants (mean age 33.3 (SD 8.6) years) were recruited. There was no significant difference in absolute change in insulin basal rates between groups (P = .85). The circadian group experienced significant variation in the number of changes made with the most changes in the morning and evening (P = .005). The circadian group received a greater reduction in total insulin (−14.1 (interquartile range (IQR) −22.5-12.95) units) than the flat group (−7.48 (IQR −11.90-1.23) units) (P = .021).
Conclusion:
The initial insulin profile does not impact on the magnitude of basal rate changes during optimization. The circadian profile requires changes at specific time points. Further development of the circadian profile may be the optimal strategy.
Introduction
The use of insulin pump therapy in type 1 diabetes (T1D) is associated with improvements in overall glucose, exposure to hypoglycemia and quality of life. 1 Initiation of insulin pump therapy in people with T1D requires conversion of a basal insulin dose, given as once or twice daily long-acting insulin, to a continuous basal infusion regimen. This conversion may be based only on the basal insulin dose, or total daily insulin dose, and may result in a flat basal insulin profile or an initial variable basal rate.
Initial variable basal rates aim to replicate circadian changes in insulin requirements and are derived from total basal insulin in adults older than 24 years, and from weight in adults aged 18 to 24 years. Initial rates were developed from 63 people with T1D using insulin pump therapy over 14 years of age and have been assessed against a flat basal rate in a small randomized controlled trial with 12 participants. Mean glucose was lower in the circadian basal rate group with particular differences noted in the early morning when the increase in glucose was more pronounced in the flat basal rate group. 2
In 50 people with T1D treated with insulin pump therapy, HbA1c was lower in those with lower basal rates at midnight, and in those with higher basal rates in the afternoon, suggesting a benefit of circadian patterns. 3 In 33 people with T1D older than 16 years, basal rate distribution established at the commencement of pump therapy did not alter over 6 months. 4 However, a 6-month cross-over study of circadian rates and oligophasic basal rates showed no difference in HbA1c. 5
In a retrospective analysis of 339 inpatient 24-hour fasting basal rate tests showed hyperglycemia despite increased basal rates at dawn (01:00 until 07:00 hours) with a smaller rise in basal requirements in the afternoon, and a flat basal profile in the hours between 2100 and 0100, and 1000 until 1400. 6
In clinical practice, following initiation of insulin pump therapy, basal rates are personalized to capillary blood or continuous interstitial fluid glucose monitoring (CGM) data. Initial basal rates may be established as flat or circadian based on physiological and observational data.4,7 In adults with T1D starting insulin pump therapy, there are limited data to guide the optimal insulin profile to rapidly achieve target glucose and minimize healthcare professional input.
We hypothesize that circadian insulin infusion rates in continuous subcutaneous insulin infusion (CSII) naïve people with T1D are closer to optimal basal rates, compared with a flat initial rate, and present data from a clinical pilot study assessing the impact of circadian and flat basal rates implemented with insulin pump therapy.
Methods
Participants with T1D who had been assessed and screened for insulin pump therapy were recruited from clinics at Imperial College Healthcare NHS Trust, London. Participant information sheets were given to potential subjects, either in person in clinic, or via post with a participant invitation letter. A diagram illustrating patient flow through the study is shown in Figure 1.
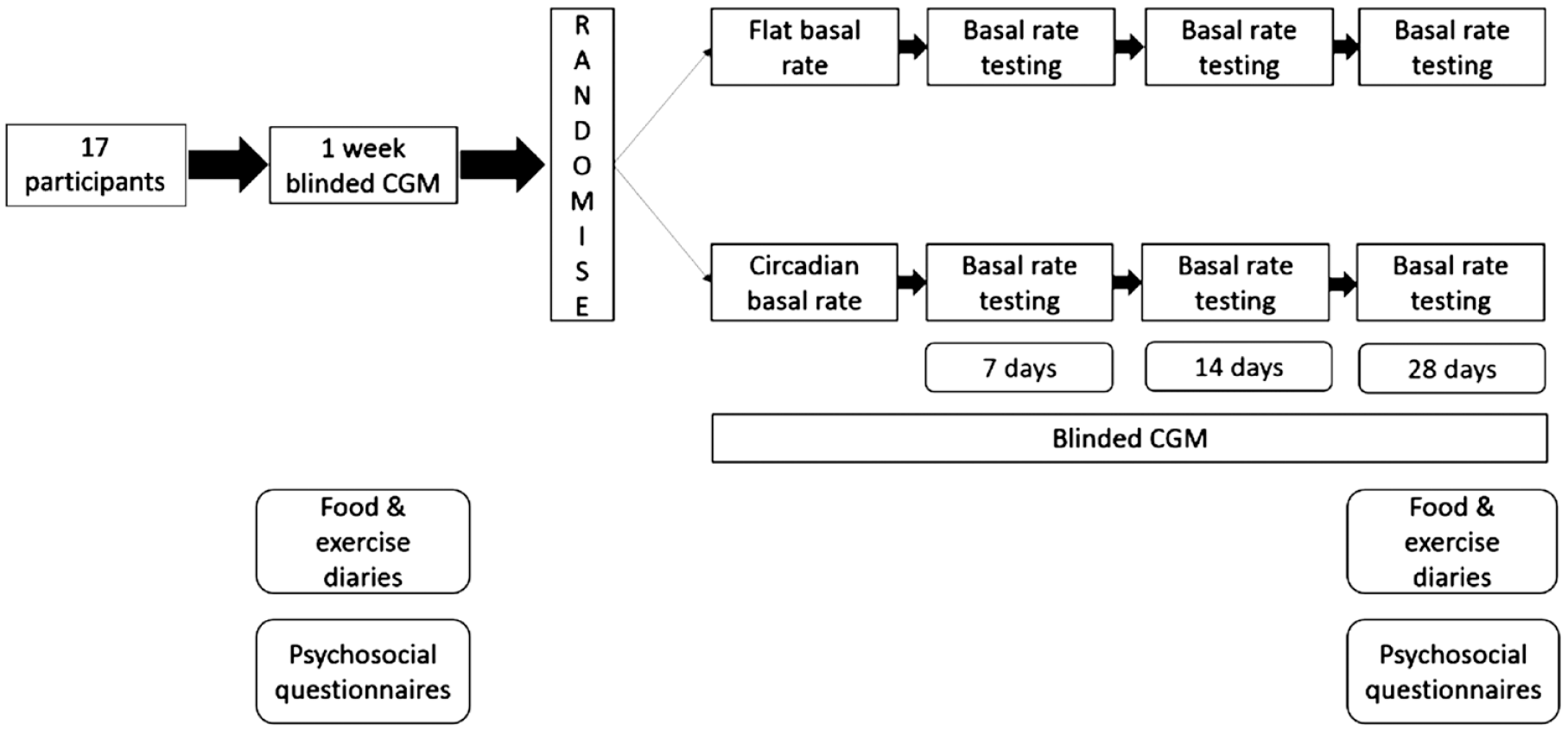
Diagram illustrating study design and patient flow.
Inclusion criteria included being an adult older than 18 years, a diagnosis of T1D for >1 year using an intensified insulin regimen with a decision made to commence insulin pump therapy in line with the NICE TA151 guidelines, 8 which states that CSII is recommended as a treatment option for those who have been unable to achieve target HbA1c levels with multiple daily injections (MDIs) without the person experiencing disabling hypoglycemia, which has an adverse impact on their quality of life. All participants had received structured education in the previous 3 years, had an HbA1c ≤ 75 mmol/mol (9%), a stimulated c-peptide <200 pmol/L and had no severe hypoglycemia (defined as needing third party assistance) in the previous year. Participants with previous use of insulin pump therapy were excluded as were night or shift workers, people who were pregnant or planning pregnancy, who were breastfeeding, who were enrolled in other clinical trials, who had an active malignancy or were under investigation for malignancy, and people with recurrent severe hypoglycemia Addison’s disease, coeliac disease, gastroparesis, autonomic neuropathy and who exercised a concomitant use of GLP-1 analogues and gliptins. Any participant who was excluded at screening was replaced by another participant.
No formal power calculation was undertaken for this pilot study of the effect of circadian basal rates.
At screening, blood samples were taken to measure HbA1c, glucose, kidney function, serum C-peptide, coeliac antibodies and thyroid function test. A urine sample was taken to test for pregnancy in women of childbearing age. A medical and medication history was taken. A validated questionnaire was used to screen for gastroparesis. 9 A range of insulin pumps were demonstrated (Medtronic Minimed 640G, Northridge, California; Roche Accu-Chek Combo and Roche Accu-Chek Insight, Mannheim, Germany) and the advantages and risks of insulin pump therapy, as well as the features of the available devices were explained to ensure a fully informed decision was made about commencing insulin pump therapy and about the type of pump suiting them best.
At least 24 hours after this appointment, potential participants were contacted to see whether they want to continue onto insulin pump therapy, and if so, whether they would like to take part in the trial, or whether they would prefer to start insulin pump therapy with their usual care team.
After obtaining an informed consent, participants were taught to insert and replace a Dexcom G4 (Dexcom, San Diego, CA) sensor for use with blinded CGM. Participants wore a sensor for seven days prior to pump initiation, and continuously after pump initiation until the end of the study. They were asked to keep a food and exercise diary during the pre-pump week, and during week 4 of insulin pump therapy. Participants completed questionnaires to measure awareness of hypoglycemia (Gold), fear of hypoglycemia (HFS-ii), general quality of life (WHOQoLBref), diabetes distress (DDS-ii) and diabetes quality of life (DQOL), which were repeated at the end of the study.
Participants were randomized to a flat basal rate or circadian rate and then initiated pump therapy in pairs within the same randomization group. Participants took half their usual basal insulin the night before commencement of pump therapy or excluded the morning dose if they were on twice daily basal insulin. They were taught how to use their insulin pump by a diabetes-specialist dietitian trained in insulin pump therapy. Participants learned to set up the pump, insert the cannula and to adjust the settings. Both arms received 75% of their pre-pump total daily insulin dose via CSII, of which half was given via basal insulin. These basal rates were calculated in advance ready for programming at the CSII initiation visit. The circadian basal rate was calculated using the Roche slide calculators, based on the total daily basal dose.
Participants received structured information on self-management including correction of high and low blood glucose levels and were instructed to return to insulin injection by pen device in case of pump failure. The use of temporary basal rates during exercise and activity was standardized. Participants were given the researcher’s contact details for queries in working hours, and a 24-hour product support number in case of pump failure.
Participants were asked to record their blood glucose levels using capillary blood glucose testing a minimum of four times per day – before meals, before bed and if they felt unwell. These results were reviewed by the researcher via telephone on day 1, 2 and 4 of pump therapy, and insulin pump bolus settings (insulin:carbohydrate ratio) were adjusted by 10% in the event of two values being <4 mmol/L within 4 hours of a meal, or >12 mmol/L 4 hours after the meal, in the absence of a precipitating factor.
Participants undertook a series of fasting basal rate testing commencing on day 7, 14 and 28 of insulin pump therapy. Participants were asked to refrain from exercising and using temporary basal rates during the fasting basal rate testing. The basal rate testing protocol was as follows:
Day 1 Omit evening meal with self-monitoring at 18:00, 20:00, 22:00.
Day 2 Omit lunch with self-monitoring at 12:00, 14:00, 16:00, 18:00
Day 3 Omit breakfast with self-monitoring at 06:00, 08:00, 10:00: 12:00
Day 4 Overnight testing with self-monitoring at 22:00, 00:00, 02:00, 04:00 and 06:00
At study visits on day 11, 18, and 32 of pump therapy, insulin pump and CGM data were downloaded, and insulin basal rates were adjusted based on the fasting basal rate testing. For a glucose increase more than 1.7 mM during sleep or over a 4-hour period when fasting, the basal rate was increased by 10% for 2 hours at the start of period in which the blood glucose rose. If glucose fell more than 1.7 mM during sleep or over a 4-hour period when fasting, the basal rate was reduced by 10% for 2 hours at the start of the period in which the blood glucose fell. Outside of this regimen, insulin basal rates were only adjusted in the case of severe hypoglycemia, recurrent hypoglycemia (<4 mmol/L) at the same time on 3 consecutive days, or recurrent hyperglycemia (>12 mmol/L) at the same time on 3 consecutive days.
On the final study visit (day 32) participants were asked to repeat the questionnaires to measure their hypoglycemia awareness (Gold), fear of hypoglycemia (HFS), general quality of life (WHOQoLBref), diabetes distress (DDS-2) and diabetes quality of life (DQOL). Blood samples to repeat HbA1c were taken.
The primary outcome was absolute change in insulin basal rate over 24 hours after 3 rounds of basal rate testing (calculated by the sum of absolute changes for each 1-hour block compared with baseline). Secondary outcomes included the number of insulin time blocks changed, the number of insulin basal rate changes, the change to total daily insulin dose, CGM outcomes of times spent in ranges and glucose variability and severe hypoglycemia.
Data were entered onto a database with restricted fields by the research team and, following data checking and cleaning, statistical analysis was performed using Stata 14.2 statistical software (StataCorp, College Station, Texas). The primary and secondary outcomes were analyzed by intention to treat analysis. The difference in basal rate change between the two groups at the endpoint was assessed using a Friedman test. Secondary outcomes were assessed using a Mann–Whitney test. Missing data were minimized by reducing manual data entry, using technology to collect glucose data and by carefully supporting participants. Where present, missing data was handled by full analysis of the available data only.
Results
Seventeen participants (mean age 33.3 (SD 8.6) years, mean weight 76.6 (SD 11.2) kg, duration of diabetes 17.2 (SD 9.5) years, Gold score 2.1 (SD 0.9), HbA1c 59.7 (SD 14.4) mmol/mol were recruited. Nine participants were randomized to the circadian group, and eight to the flat group. Three participants did not meet the inclusion criteria at screening, and one participant withdrew before commencement on CSII. All participants who commenced CSII completed the study.
There was no significant difference in the primary outcome of absolute change in insulin basal rates between the two groups (P = .85) from baseline to the end of the study (circadian group median 1.8 (interquartile range (IQR) 0.63-2.99) units of insulin and flat basal group median 1.49 (IQR 0.83–1.94) units of insulin. In the circadian basal group, there was significant variation in the number of changes made across the 24 one-hour blocks with the most changes early in the morning (P = .005), as shown in Table 1. In the flat basal group, there were no significant variation in the number of changes made across the time points (P = .09).
Median (IQR) Number of Changes Made to Hourly Basal Rate Settings after Three Sets of Fasting Basal Rate Tests in the Circadian and Flat Basal Rate Groups.
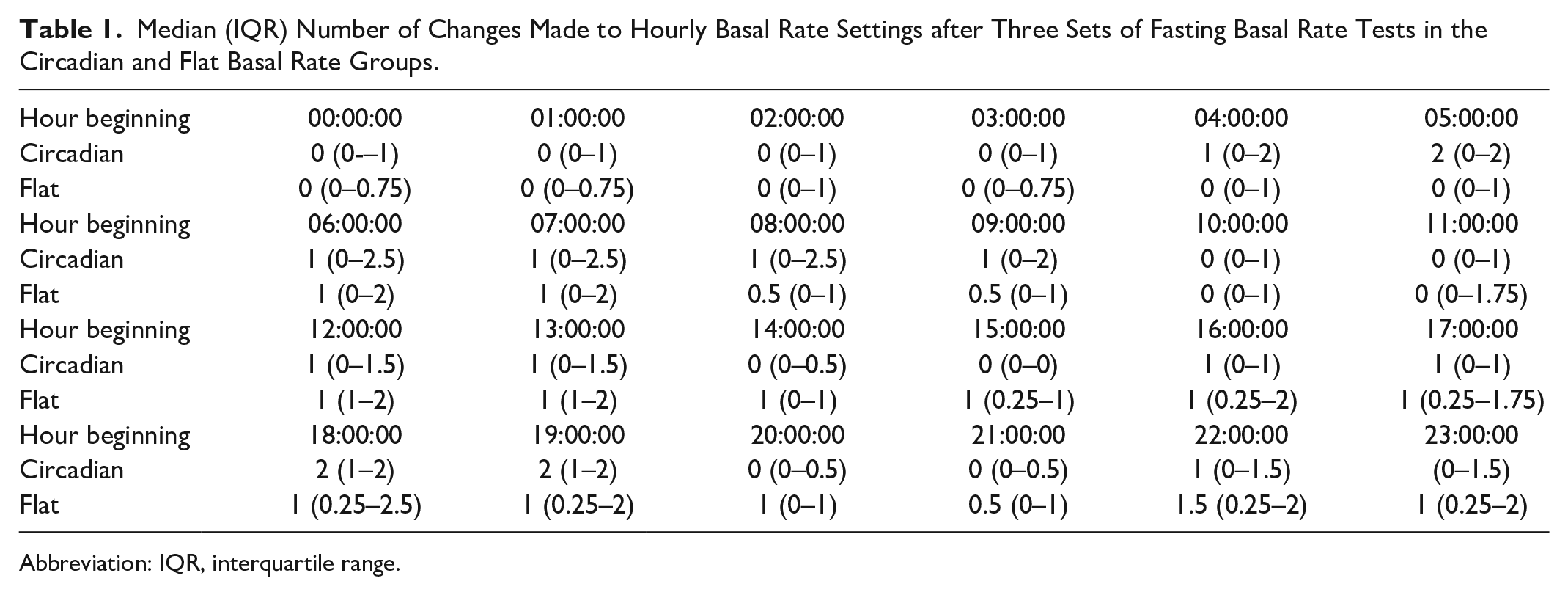
Abbreviation: IQR, interquartile range.
Change in total daily insulin dose in the flat (−7.48 (IQR −11.90-1.23) units and circadian (−14.1 (IQR −22.5-12.95) units) were significantly different (P = .021).
Deviation from the protocol happened only when there was clinical concern about hypoglycemia, in which case basal rates were reduced by 20% instead of 10%. A total of 28 instances of protocol deviation occurred over the three rounds of basal rate testing in the two groups, of which 17 were in the flat and 11 were in the circadian group. The most prevalent times for protocol deviation in the circadian group was 20.00 and 21.00 hours, and in the flat group was 08.00, 09.00, 14.00, 15.00, 21.00, and 22.00 hours.
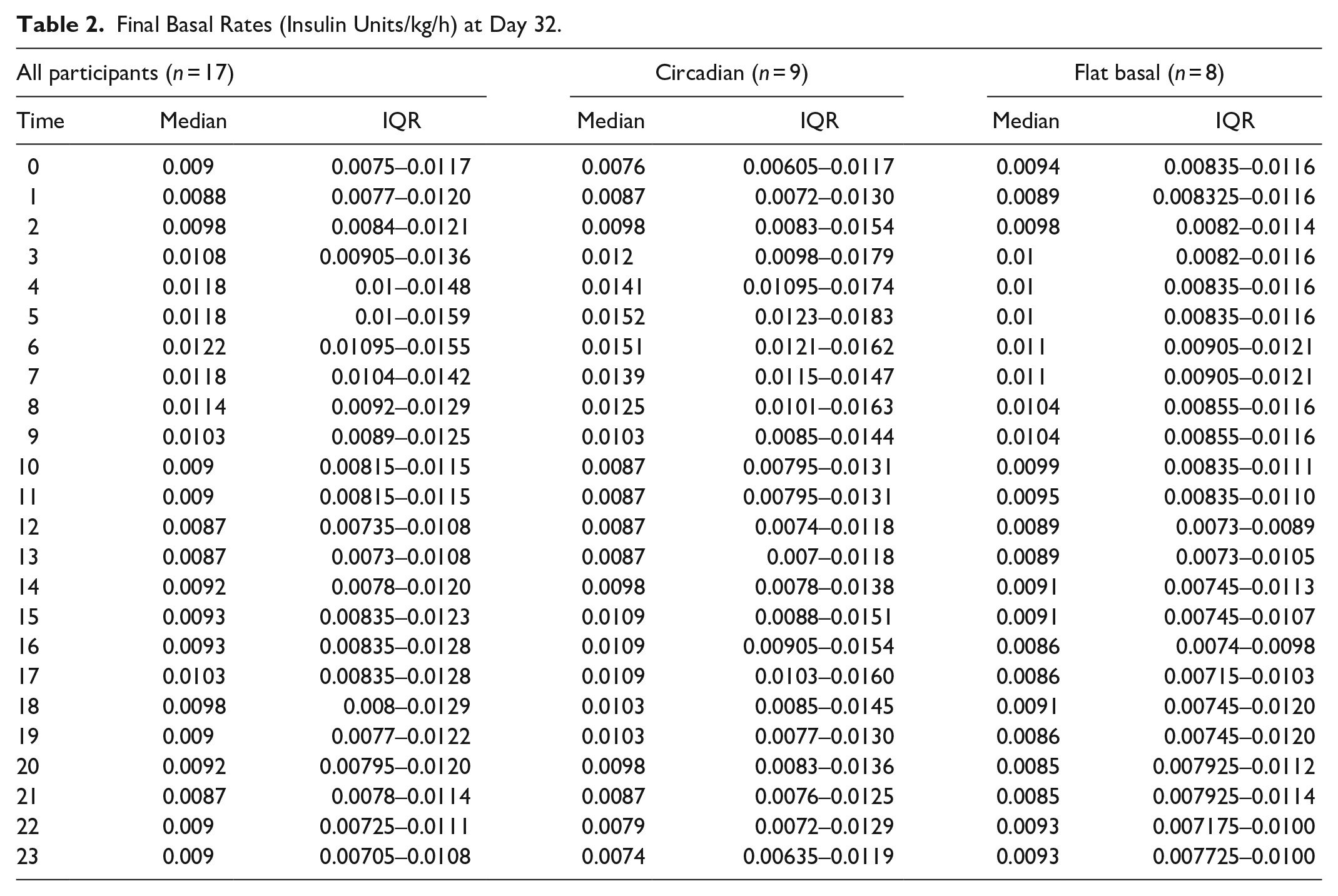
The basal per kilogram bodyweight per hour at the end of the study was calculated for all participants, as well as for the two different groups and is displayed in Table 2, and pictorially in Figure 2.
Final Basal Rates (Insulin Units/kg/h) at Day 32.
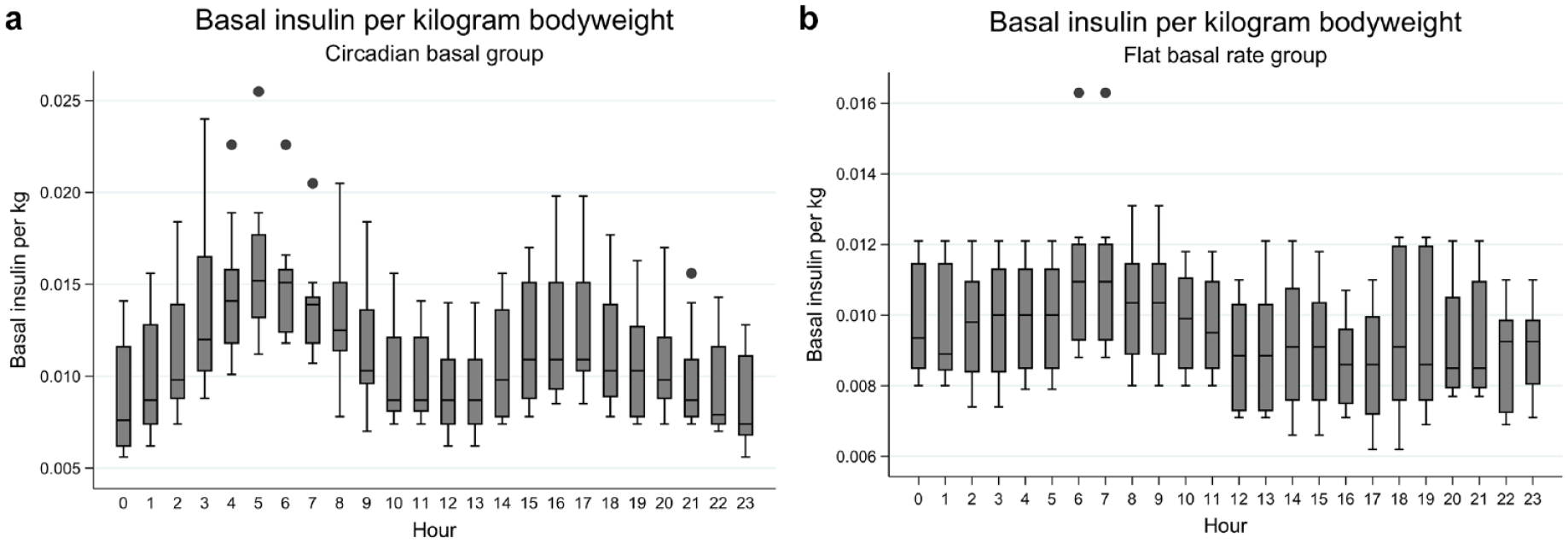
(a) and (b) Basal insulin per kg bodyweight in the circadian group and the flat basal group.
There was no significant difference in the change in Gold, WHOQOL, HFS-ii, DDS or DQOL questionnaire scores between the two groups.
There were no significant differences between the two groups on analysis of the CGM data (Table S1), including %time in hypoglycemia (<2.8 mmol/L), %time in hypoglycemia (<3.3 mmol/L), %time in hypoglycemia (<3.9 mmol/L), %time in target (3.9-10 mmol/L), %time spent in hyperglycemia (>10 mmol/L), mean absolute glucose, mean amplitude glucose excursion and low blood glucose index. There were no incidences of severe hypoglycemia or diabetic ketoacidosis.
Discussion
In this study of 17 people with T1D commencing insulin pump therapy, we showed that there was no significant difference in absolute change in insulin basal rates between the two groups from baseline to the end of the study, suggesting there may be no benefit in selecting a circadian profile. However, there was a significant variation in the number of changes made across the 24 one-hour blocks in the circadian group but not the flat basal group. These changes were observed around 06:00 to 07:00 hours and 18:00 to 19:00 hours, when the circadian basal rates are most different to the baseline. The circadian group had a significantly greater reduction in total daily insulin dose than the flat group with fewer protocol deviations to address hypoglycemia. The absence of variation in basal rate adaptation across the 24 hour period suggests that the flat basal rate may be suboptimal at all times through the day while the circadian basal profile needed most adaptation in the early morning and evening, with protocol deviations occurring around the evening, suggesting that the other times of day may be close to optimal at initiation.
Taken together these data suggest a circadian approach has the potential to be the optimal insulin pump initiation strategy, but that the present profile requires improvement to reduce overall insulin exposure, particularly at the times of peak infusion rates. Analysis of the hourly basal rates per kilogram body weight recorded at the end of the study following three rounds of structured basal testing could inform evidence-based changes to the circadian profile, and further work to assess this in a larger group of people with T1D, over a longer period, would be of interest. It is particularly striking that the flat and circadian profile groups converged between 11:00 and 14:00 hours, a longer study may have shown a similar impact throughout the 24-hour period.
No significant difference was observed in the quality of life measures between the two groups, which may be due to the small numbers and short time period, contained within the study. There was also no difference in change in times in ranges (TIR), or glucose variability measure calculated from CGM data between the two groups. This could be due to the small numbers included in this pilot study or could suggest that basal rates were not optimized by the end of the study. A study of longer duration or with more aggressive changes to the basal rates may have a greater effect on TIR.
Limitations of this study include the sample size. Additionally, commencing insulin pump therapy based on total daily dose (TDD) of multiple dose injection (MDI) in a group of participants who meet the NICE criteria for pump therapy may mean that participants are selected where MDI is not adequate to achieve target, resulting in some TDD estimates that may be incorrect. Using basal insulin per kilogram bodyweight is an alternative way to commence CSII but is limited by varying insulin sensitivities between individuals.
A further limitation is that the data are based on three reviews of the basal rate settings. In clinical practice, it may take longer than this to be established on insulin pump therapy, and further review, analysis and changes to the basal rate may lead to a more informed conclusion of what the ideal basal rate should look like. Biological variability in basal insulin requirement, especially overnight, may also mean that basal rate testing results are not applicable to future requirements. 10
More advanced algorithmic approaches to basal rate and bolus settings optimization are being developed and may enable more frequent and complex adaptation using artificial intelligence approaches. 11 The aim of our study was to identify the optimal strategy for insulin pump initiation to minimize the immediate healthcare resource required to optimize and does not remove the need for regular review and adjustment of insulin pump settings, underpinned by education and support, to achieve ideal times in ranges.
The data presented do not definitively guide initial insulin pump basal rates but suggest an optimized circadian profile may be identified, requiring minimal further optimization with benefits to insulin pump users and healthcare multidisciplinary teams.
Supplemental Material
RilstoneTable_S1 – Supplemental material for A Pilot Study of Flat and Circadian Insulin Infusion Rates in Continuous Subcutaneous Insulin Infusion (CSII) in Adults with Type 1 Diabetes (FIRST1D)
Supplemental material, RilstoneTable_S1 for A Pilot Study of Flat and Circadian Insulin Infusion Rates in Continuous Subcutaneous Insulin Infusion (CSII) in Adults with Type 1 Diabetes (FIRST1D) by Siân Rilstone, Monika Reddy and Nick Oliver in Journal of Diabetes Science and Technology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This investigator-initiated study was funded by Roche Diabetes. Infrastructure support was provided by the NIHR Imperial Biomedical Research Centre. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
