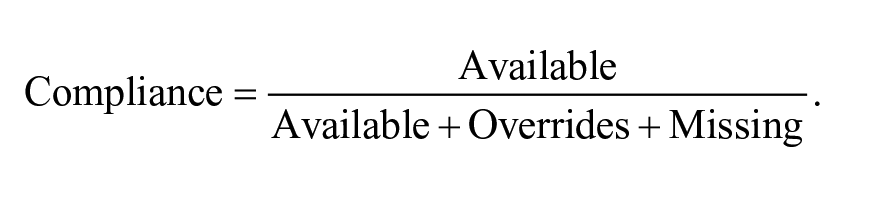Abstract
Introduction:
Glycemic control in patients with type 1 diabetes can be difficult to achieve. One critical aspect of insulin delivery is site rotation, which is necessary to reduce dermatologic complications of repeated insulin infusion. No current application is designed to help patients track sites and instruct on overused sites.
Objective:
The objectives of this study were to (1) design a smartphone app, Insulin Site Guide, to gather real-time information on continuous subcutaneous insulin infusion (CSII) and continuous glucose monitor (CGM) site location and rotation compliance and instruct subjects on the use of an overused site; (2) conduct a usability study to measure site rotation compliance; and (3) report subject satisfaction with the app.
Design:
The app is installed on the subject’s smartphone. Subjects use the app to record CSII and CGM placement in real-time. Data are sent to the study team at the end of the study. Subjects complete a questionnaire concerning the app.
Results:
We report site rotation compliance data for eight subjects and survey responses for 10 subjects. Initial data from eight subjects indicate a high site rotation compliance of 84% for insulin pumps. In general, the majority of users indicate high satisfaction with the app.
Conclusions:
Insulin Site Guide is a mobile app that uses a novel algorithm to better guide site rotation. Use of the app has the potential to improve site rotation and decrease dermatologic complications of diabetes with long-term use.
Keywords
Background and Significance
Type 1 diabetes (T1D) is a chronic autoimmune disease that affects 1.84 million people in the United States. Effective treatment for type 1 diabetes requires administration of insulin through multiple daily injections (MDI) or through use of an insulin pump or continuous subcutaneous insulin infusion (CSII). In adults with T1D, there is increasing evidence that use of technology such as CSII, continuous glucose monitors (CGMs), and hybrid closed-loop systems (HCL) can improve glycemic control, as evidenced by improved glycated hemoglobin (HbA1c), time in range (TIR), as well as reduce the risk of hypoglycemia. Use of CSII in the outpatient setting has increased from 57% in 2010-2012 to 63% in 2016-2018, and it also has been shown to be decrease the burden of managing T1D and improve quality of life as compared with MDI.1,2
Despite these technological advancements, blood glucose control remains difficult to achieve. One critical aspect of insulin delivery is the strategy of site rotation, which is essential to reduce the side effects of repeated placement of CSII infusion sets in the same anatomical site. Lipohypertrophy (LH) is a common complication of repeated insulin injection in the subcutaneous tissue of patients with T1D, appearing as soft nodules on the skin surface that are fibrous and poorly vascularized lesions in the adipose tissue.3,4 It results from the lipogenic action of insulin and repeated trauma at the site of insulin infusion. Although the exact prevalence of LH is unknown, Gentile et al 5 reported LH prevalence ranging from 3.6% to 64% in people with T1D. Data vary depending on the type of insulin therapy (MDI or CSII) and the method used to diagnose LH. Methods used to detect LH include palpation, visual inspection, and ultrasonography. In a larger study of 13 289 patients from 42 countries, LH prevalence was reported to be around 30% based on an injection technique questionnaire, visual inspection, and manual palpation of injection areas, whereas a meta-analysis of data from 26 865 participants found the overall prevalence of LH to be 41.8%.6,7 In a recent study of 79 CSII-treated patients with T1D, Ucieklak et al 8 report LH detected by ultrasound in 94.9% of patients as compared with 49.4% diagnosed by visual inspection and 74.7% by palpation in the same subset of patients.
Development of LH is strongly correlated with nonrotation of sites. Poor rotation of sites and development of LH can lead to poor glycemic control, a higher frequency of unexplained hypoglycemic episodes, and increased glucose variability. In addition, use of a site affected by LH can lead to increases in insulin dosing and therefore a higher cost of insulin therapy. 7 Studies have shown improved glucose control and reductions in insulin dosage after instructing patients on proper site rotation techniques and avoidance of LH lesions. Despite the importance of site rotation, current studies indicate that patients with T1D rotate sites consistently only 40% to 60% of the time.9 -14 There is no current application designed to help patients track use of CSII sites and ensure awareness of overused sites. We have developed an innovative smartphone application (app), Insulin Site Guide, that uses a novel algorithm to better guide site rotation. 15 The hypothesis is that use of the app can improve CSII site rotation, improve insulin site sensitivity, and reduce site rotation injuries. In this article, we describe the app and report data from a small pilot study on subject-site rotation compliance and satisfaction with the app.
Materials and Methods
The objectives of this study were to (1) design a smartphone app, Insulin Site Guide, to gather real-time information on CSII and CGM site location and rotation compliance and inform subjects about overused sites; (2) test out the app in a small usability study to measure site rotation compliance; and (3) report subject satisfaction with the app. Institutional review board approval from Mayo Clinic (19-012737) was granted for the usability study.
Design of the App
A literature search was conducted to gather information on the prevalence of LH with use of CSII therapy in patients with T1D and gather information on current devices and software commercially available in the United States to aid patients in site rotation. Discussions with endocrinologists and patients with Type 1 diabetes confirmed the need for an application to aid patients in site rotation. Based on what was learned from these searches, Insulin Site Guide was created to achieve the following goals: (1) gather real-time information on CSII and CGM site placement, (2) inform subjects on overused sites, and (3) allow providers to remotely access these data.
The Insulin Site Guide app runs on Apple iPhones and iPads and is designed to collect information from subjects on placement of insulin injections, CSII, and CGM sites. The app does not provide medical advice; rather, it records the site history of users to allow for informed decision-making on device placement to encourage site rotation. The app is installed with Apple’s TestFlight application. At startup, the app prompts the user on the proper setup.
Upon opening the app, the user sees available preset anatomical sites for site placement (Figure 1). At the time of initial app setup, the user has the option to accept the preset default locations or customize locations based on user preference. If the user desires, he or she may customize available locations and add or delete sites at any time. In addition, the template images in the app can be used as is, or the user can insert actual personal photos of the sites. Each anatomical location has several spots to choose from. Each spot is color coded to indicate status of that site based on the history of usage. The photo overlays and grid arrangement of spots help with the orientation and positioning of spots.
Home screen of the app. Available spots in blue and unavailable spots otherwise.
Along the bottom is the tab menu to navigate to the various screens. Just above that is the device menu showing the three device types: pump, syringe, and CGM. In the upper-right corner is a lock icon. Locking the screen in between uses prevents accidental changes.
The spots are color coded to indicate status. In Figure 1, the blue spots indicate recommended sites for placement of a device; these are termed “available spots.” This is determined by the amount of time that has passed since last use of each particular site. The red (and pink) spots are in use or have been used recently and are not recommended for current device placement. These are termed “unavailable spots.” When the user places a pump on a spot, the app logs the time and colors the spot pink. When the rest period for the spot has expired, the spot color reverts to blue indicating availability for reuse. Resting periods are customizable in the Settings screen. Default values are: pump 15 days, and CGM 60 days. These values were chosen to represent five cycles since the last placement of the device. The color coding is customizable in the Colors screen. The app will allow for the user to override a spot if they try to place a device on a nonrecommended site but will alert the user that the site has been used recently. The device menu with pumps, syringes, and glucose monitors allows simultaneous management of each device type.
Choosing a Spot
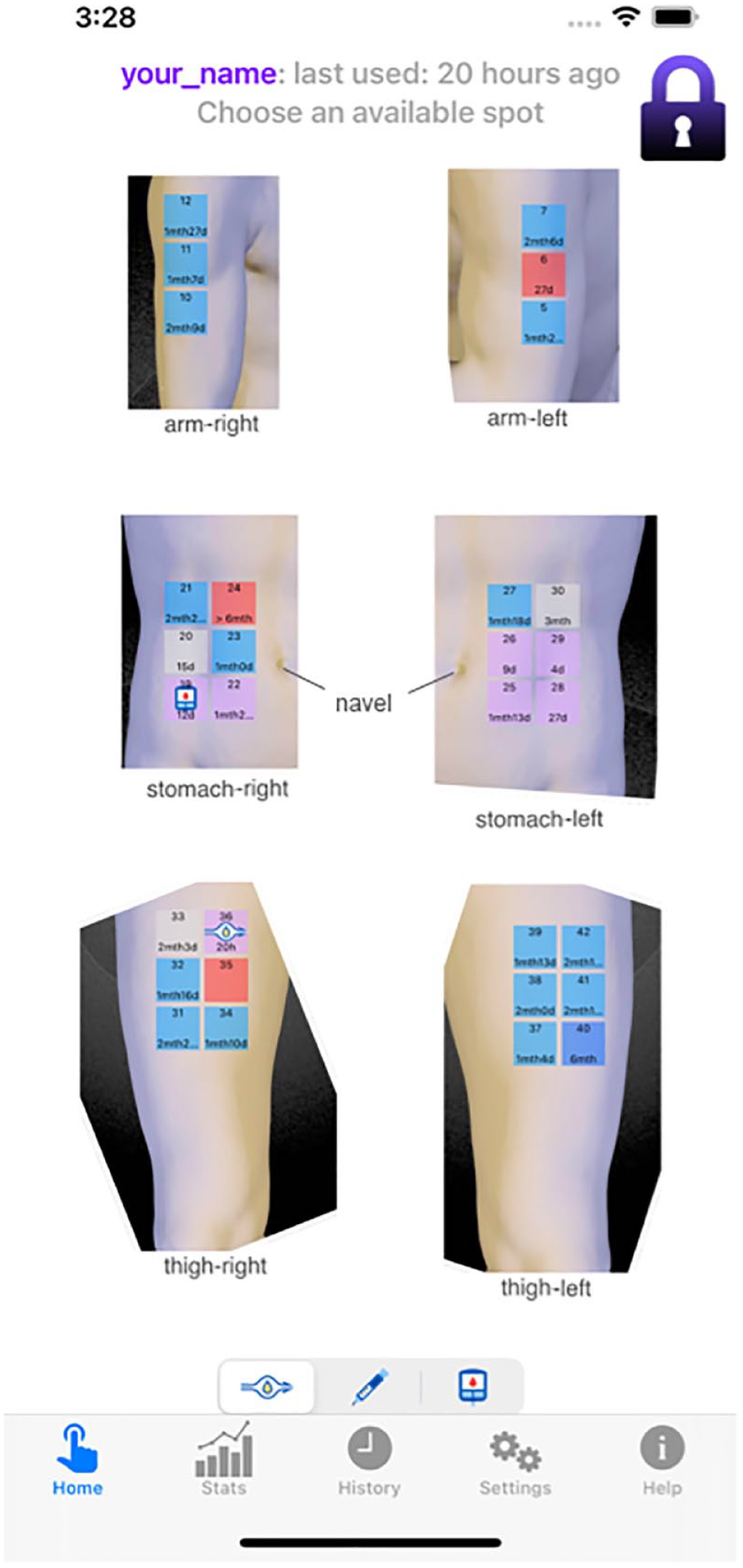
When an available spot is tapped, it changes status and shows the device type chosen from the device menu. The color changes reflecting the new status according to the color scheme and gives visual feedback confirming the selection. In Figure 2-Left, the subject has selected a pump placement site on the top of the thigh. If an unavailable spot is selected for use, the app will allow the user to override the spot. Tapping on an unavailable spot brings up the Override alert box (Figure 3-Right). The alert warns the user the spot is not available and asks the user to record a reason for use of the spot.
Left: a pump has been placed on the top thigh. The spot shows the pump icon and is temporarily green. Right: the override alert box appears when the user selects an unavailable spot.
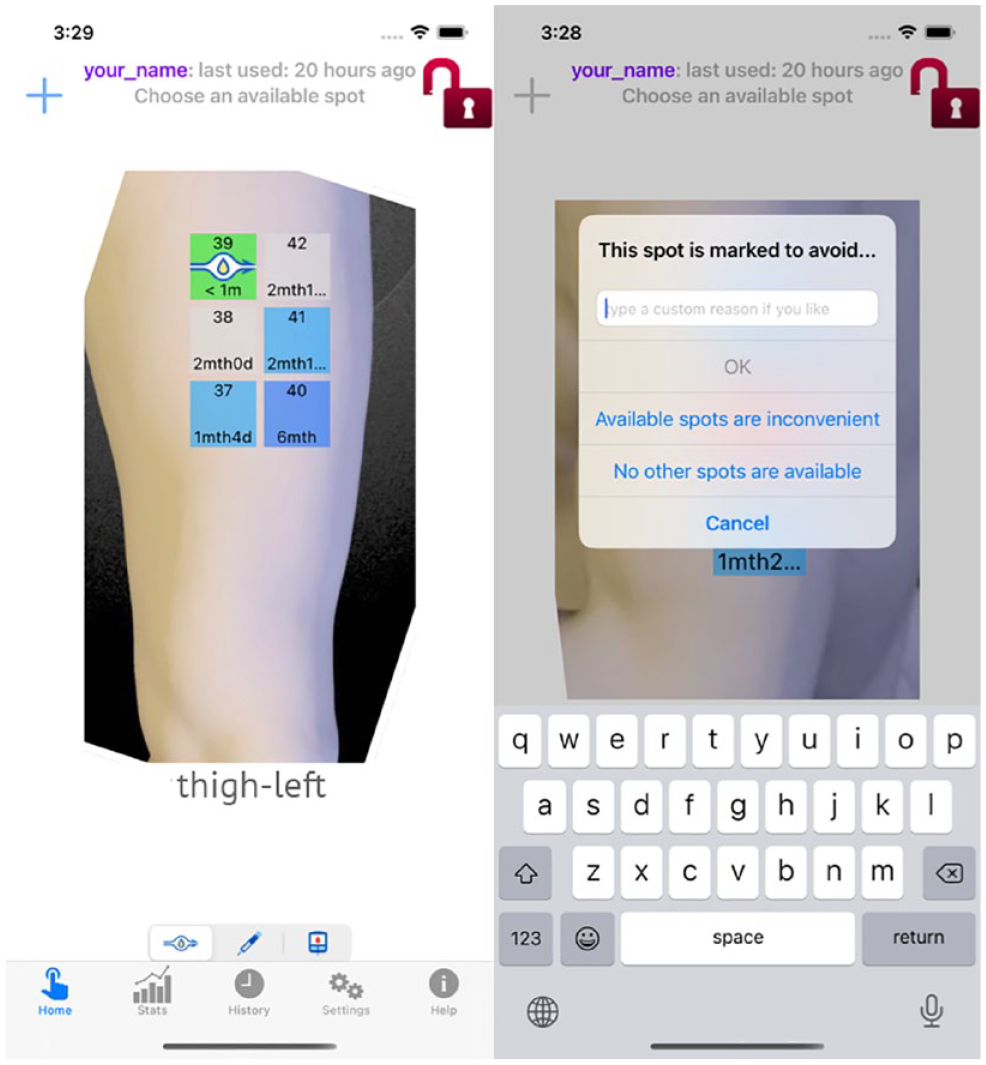
Left: the statistics screen showing the site rotation compliance for insulin pumps over the last 90 days. Right: the count screen shows potentially overused sites.
Site Rotation Compliance and Spot Usage
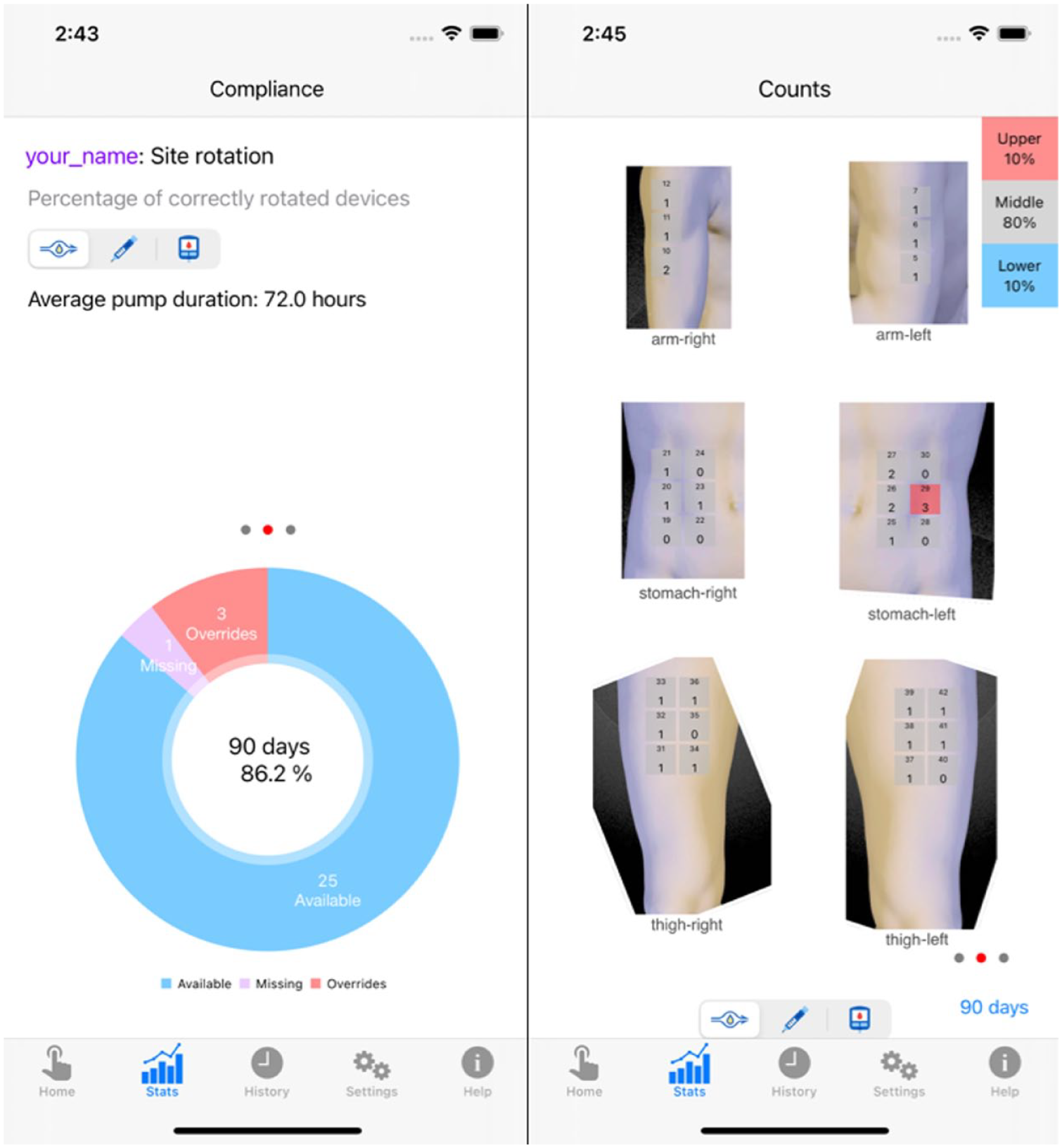
The Statistics screen displays site rotation compliance and spot usage patterns. Compliance is defined as the percent number of times proper site rotation has been achieved. Proper site rotation is defined as use of a spot that is not marked as unavailable by the app based on recent usage history. This is calculated for each device type. Compliance is calculated as the available spots divided by the total spots chosen (which includes available, override, and missing spots). Missing data occur when the user forgets to record a choice of spot. In this case, the app notices a gap in the data and infers a missing history item:
Figure 3-Left shows the pump site rotation compliance for the prior 90 days. In this example, the user has an overall compliance with site rotation 86.2% of the time. Figure 3-Right shows the spot usage counts for the prior 90 days. Overused spots are red. Figure 4-Left shows the spot override counts for the prior 90 days. This displays how often each spot has been overridden. Tapping a spot brings up a list of reasons given for the overrides (Figure 4-Right).
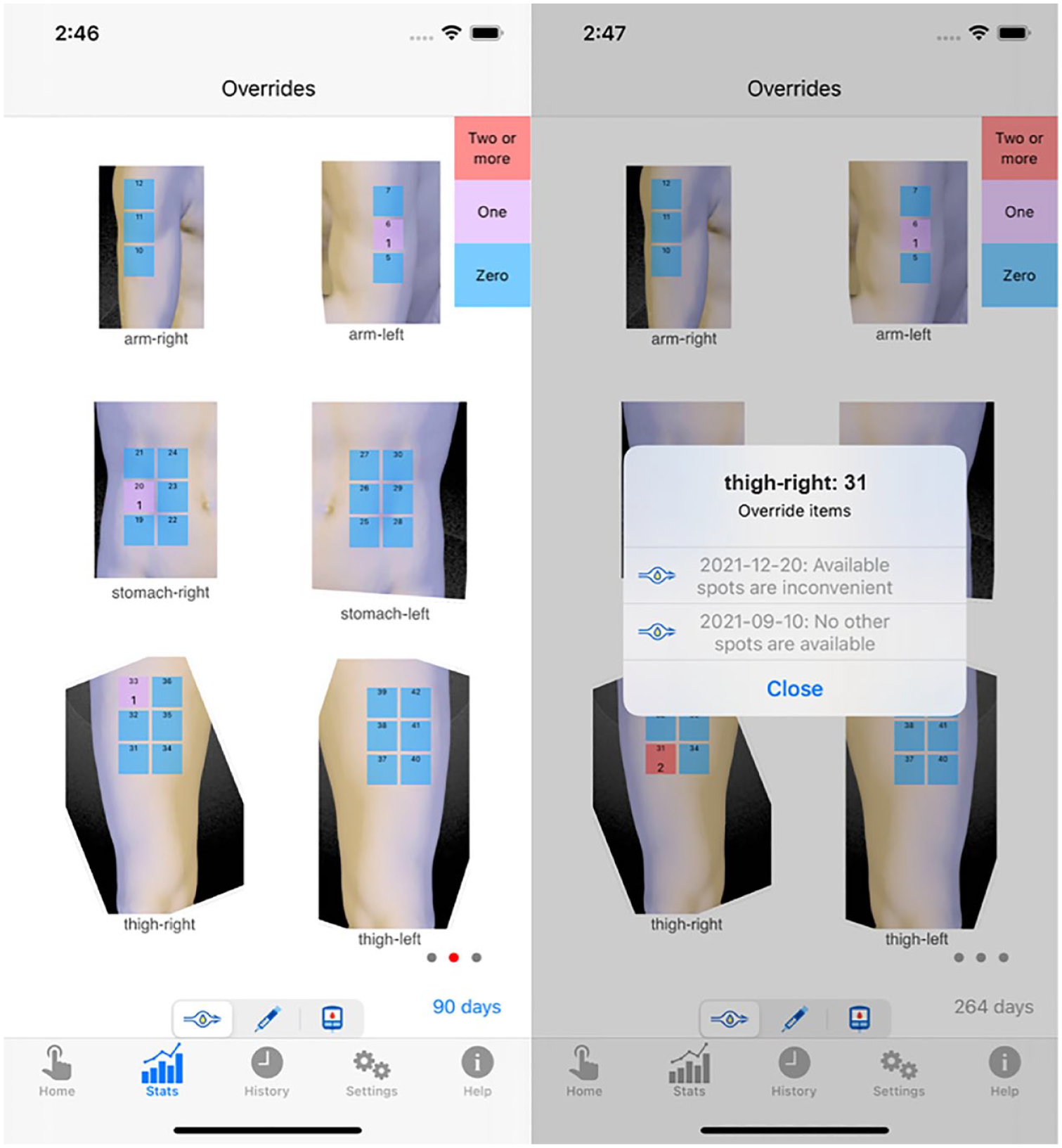
Left: the override screen shows where overrides have occurred. Right: tapping a spot shows the override history.
Results
Usability Study and Demographics
We conducted a pilot study to study the Insulin Site Guide app. Institutional review board approval from Mayo Clinic was granted for the pilot study. Subjects with T1D using a Tandem pump and Dexcom sensor between the ages of 18 and 80 who were actively being followed in the outpatient clinic were identified for participation during their routine clinic visits. Only patients using the Tandem pump and Dexcom system in Control-IQ mode were recruited to allow for consistency in data collection and eliminate any differences between pump systems. Interested subjects were contacted by the study coordinator and enrolled in the study. Instructions were provided for installation of the app and its basic use. After five weeks of continuous app usage, app compliance data were sent remotely from the subject to the study coordinator. Insulin pump and CGM data were downloaded for this time period. Subjects were also asked to complete an online survey regarding use of the app.
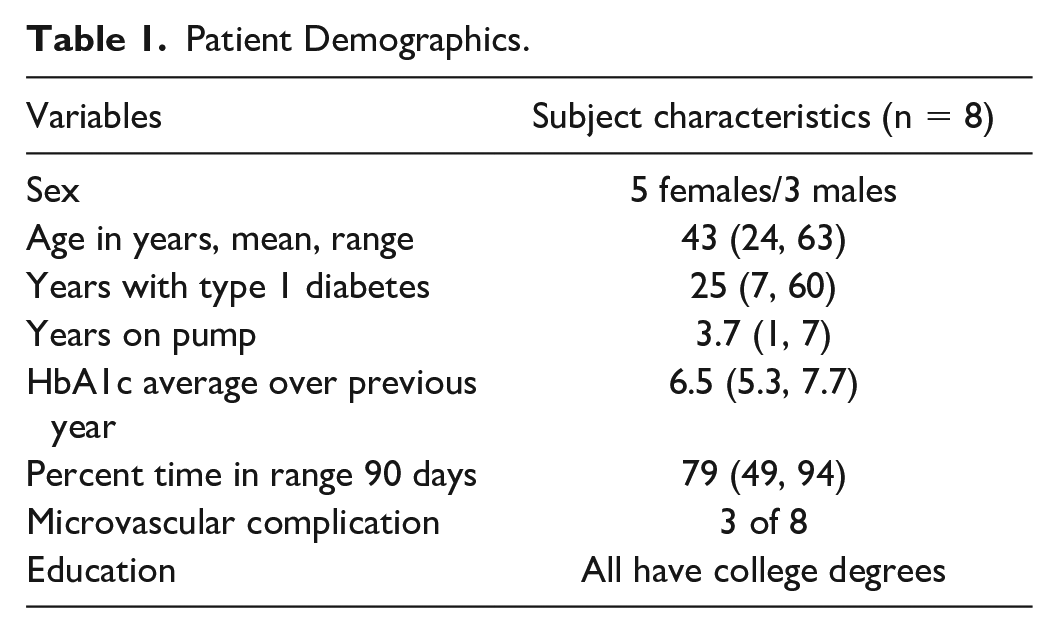
In total, 10 subjects used the app and completed the study, but we received compliance data from only eight subjects. Here, we describe rotation compliance results from eight subjects and survey results on use of the app from 10 subjects. Of the eight subjects, male:female was 3:5, with an average age of 43 years (Table 1). On average, subjects had a diagnosis of T1D for 25 years and had been using the Tandem pump for 3.7 years. Average HbA1c was 6.5% with an average three-month TIR of 79%. Microvascular complications, reported as either evidence of retinopathy or microalbuminuria, were present in three of eight patients. All subjects had completed at minimum a college education.
Patient Demographics.
Site Rotation Compliance
Site rotation compliance is calculated using the app. Initial data from eight subjects indicate a site rotation compliance of 84% for insulin pumps (Figure 5). In the six subjects who tracked CGM rotation, the compliance rate was lower at 62%.
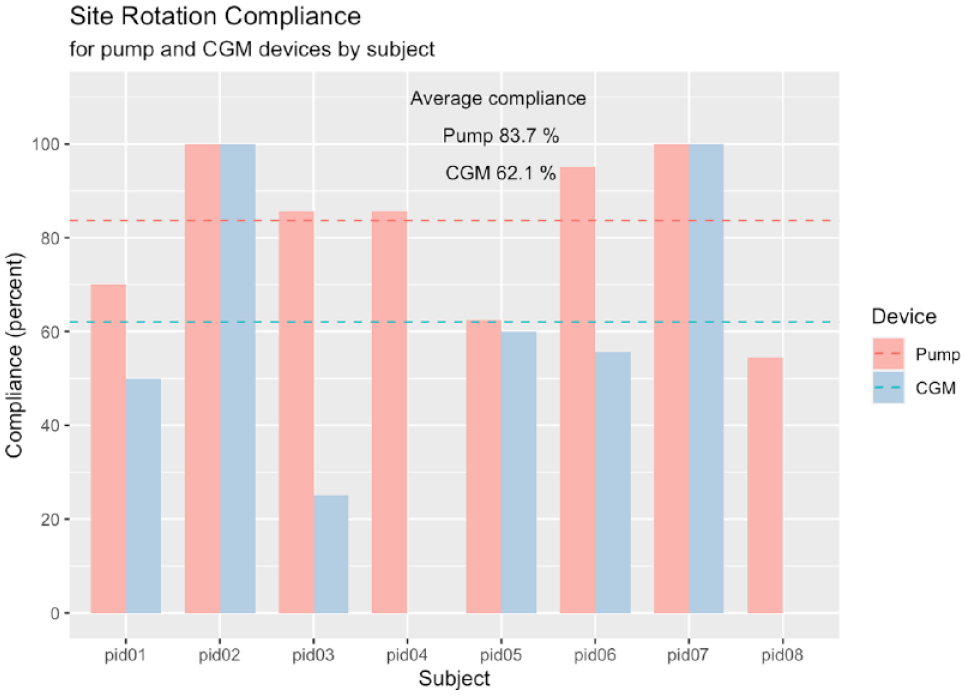
Subjects show 84% pump site rotation compliance. Abbreviation: CGM, continuous glucose monitor.
Figure 6 shows the counts of compliance, overrides, and missing for pump site rotation. In the preliminary usability study of eight subjects, pump rotation compliance is 84%, overrides are 6.6%, and missing are 9.8%. Figure 7 shows a subject with good site rotation and shows spot preferences as measured with the app.
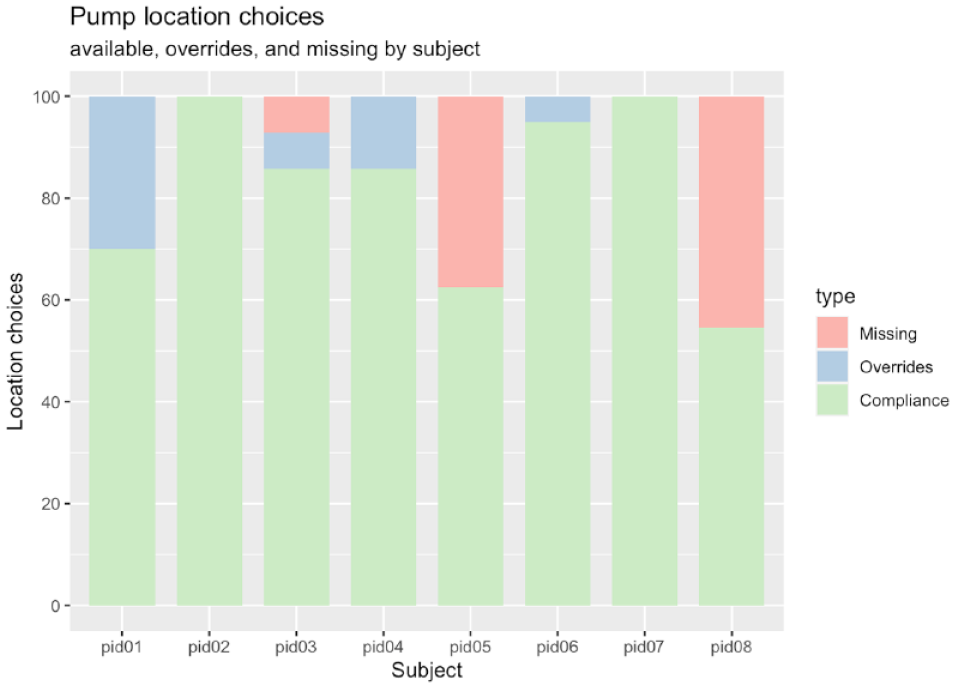
Proportions of pump location choices.
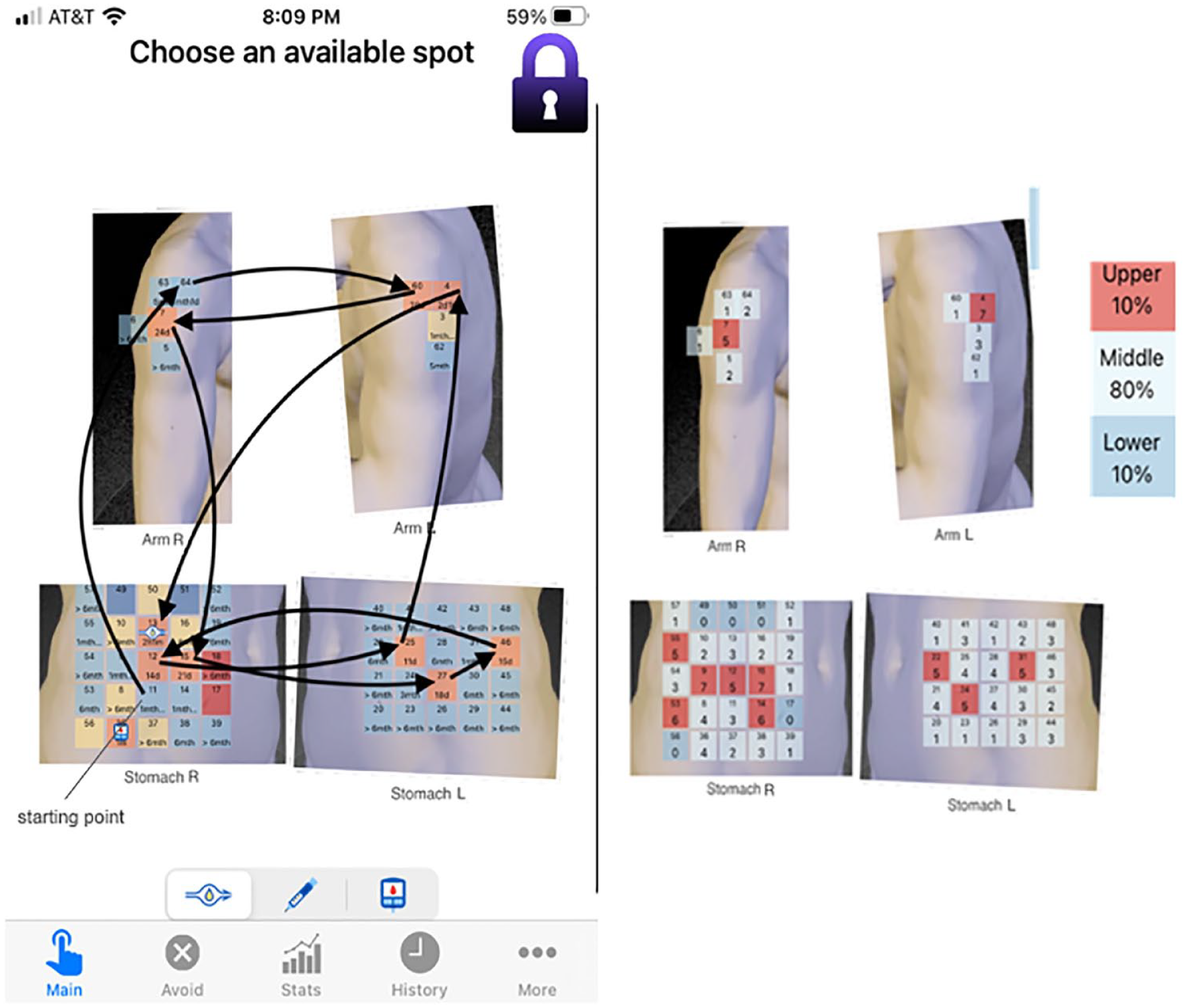
Mapping of insulin pump infusion sites with the Insulin Site Guide App. Arrows indicate the insulin infusion site change for the last 10 events. Left: subject demonstrated good site rotation. Right: subject shows spot preferences within the upper arm and abdomen region. Red color indicates top 10% of sites that are frequently used.
App Survey
At the end of the study, subjects were asked to complete a survey on use of the app to gain insights into user experience, behavior, and experience with general healthcare apps. Ten subjects completed the online survey. All 10 of 10 subjects reported prior use of a health-related app to track weight, exercise, or nutrition. Four of 10 subjects had used a diabetes self-management app (outside of pump and CGM apps) previously. Four of 10 subjects reported a behavior of periodically losing motivation to continue self-tracking with healthcare apps.
The survey next asked questions on the subject’s experience with the Insulin Site Guide app. Eight of 10 users considered the app to have a clear and uncluttered design, easy to comprehend, and easy to perform the fundamental task of choosing an available site. Six of 10 users were able to easily correct mistakes in the History screen. Six of 10 users found the color coding clear from the start and three of 10 users reported successfully adjusting the color scheme. Only two of 10 users found it difficult to customize the site locations. Six of 10 users sometimes desired more options for site placement choices on the app as a result of not customizing locations. Four of 10 users found it difficult to match the current location of a device with the location indicated on the app. In addition, one of 10 users found the left/right orientation of the app confusing.
Discussion
Rotation of CSII sites is critical for prevention of LH and other dermatological complications of continuous insulin infusion. The exact prevalence of LH in patients with T1D using CSII is unknown due to the difficulty of and varied methods in diagnosing LH. Depending on the technique used, prevalence has been reported to be between 49.4% and 94.9% in patients on insulin pump therapy. 8 However, it is well known that rotation of CSII sites is integral for prevention of LH. Despite the importance of site rotation, current studies indicate that patients with T1D rotate sites consistently only 40% to 60% of the time.9 -14 Available studies on site rotation, although informative, rely on patient recall and are subject to recall error (ie, inaccurately remembering and reporting behaviors) and social desirability (overreporting of desirable and underreporting of undesirable behaviors). Here, we describe the development and use of a smartphone app to track placement of CSII and CGM sites allowing for real-time tracking of site placement and position to accurately record and aid the user in making better decisions regarding site rotation. To our knowledge, there is no current application designed to help patients track use of CSII sites and instruct them on use of an overused site. In this article, we report data from a small feasibility trial of an innovative smart device application, Insulin Site Guide, that uses a novel algorithm to better guide site rotation.
Digital diabetes technology, including mobile health applications, has been shown to improve diabetes outcomes in patients with diabetes. Research has shown that people do not adhere to mobile health applications long term with reports of decreasing participant engagement over time with long-term use. 16 Some of the reported barriers to long-term app use include technical barriers, lack of recommendations to use apps from healthcare providers, lack of motivation, poor design, or cost.17,18 We hypothesized that use of the app could improve CSII site rotation in patients with T1D. Our preliminary site rotation compliance data from eight subjects indicates a high rotation compliance percentage of 84% that is significantly higher than currently reported values in the literature.9 -14 However, CGM site rotation compliance was significantly lower at 62%. It is unclear why CGM rotation was less than pump site rotation. It could be that patients are not counseled on rotation of CGM sites as much or are unaware of the need to rotate these sites. Regarding our app, there is likely immediate benefit in terms of providing patients insight on locations for site placement. This may serve as motivation for continuing use of the app. In addition, as these data are available to healthcare providers remotely and can be accessed easily, we are hopeful that these features will help combat user fatigue.
Of the 1000+ diabetes apps available for download, it is unknown how many have been tested clinically.19 -22 Usability testing in a clinical population of patients allows for real-time testing of the Insulin Site Guide app in the intended set of users. User feedback can be used to improve the functionality and design of the app. Survey results from the preliminary pilot of 10 users were generally positive. Subjects reported the app was easy to use, although some found it difficult to match the correct site placement site on the app with the body site. Some users desired more options for site placement, while a minority felt the app configuration was confusing. Many of these functionalities can and are being improved in future versions of the app. In addition, some of these issues may improve with better training on the app. Currently, the app is available only on the iPhone. Further iterations of the app will focus on making it compatible with android phones as well.
There are several limitations to the study. Patients in the study, by virtue of using the app, are being reminded to rotate sites that may be responsible for this higher-than-average rate of site rotation. Also, subjects in our study were very well controlled (HbA1c on average of 6.5% with TIR of 79%) and in addition were well educated. Site rotation compliance in this well-controlled and well-educated population may not be representative of the overall population of patients with T1D. In addition, this pilot study encompasses only five weeks of data and there is a high possibility that over time use of the app could decrease due to patient app fatigue. Future studies using the app should include more subjects, be longer in length, and include a more diverse patient population with regard to diabetes control and education status to allow for more accurate compliance data over time. Future directions with the app include integration of the app with insulin pumps to allow users to simultaneously track site location when placing an infusion site. In addition, future studies should expand use of the app into those patients on injection therapy (both type 1 and 2 diabetes) to allow for assessment of rotation compliance in these larger subsets of patients with diabetes using insulin.
Conclusions
Insulin Site Guide is a smartphone app designed to aid subjects in maintaining good site rotation compliance and avoiding the use of overusage of sites. Initial data indicate a high site rotation compliance rate and good satisfaction with the functionality of the app. Improvement in site rotation can potentially reduce dermatologic complications of diabetes and improve insulin absorption and glycemic control. Further controlled studies with increased participant numbers are needed to test the app.
Footnotes
Acknowledgements
The authors would like to thank Dr Timothy Whitsett for scientific editing assistance.
Abbreviations
CGM, continuous glucose monitors; CSII, continuous subcutaneous insulin infusion; HbA1c, glycated hemoglobin; HCL, hybrid closed-loop systems; LH, lipohypertrophy; MDI, multiple daily injections; T1D, type 1 diabetes; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We have received funding from the Flinn Foundation (#2258) for the pilot study to develop and study the Insulin Site Guide app to address the unmet need of site rotation for IP therapy.