Abstract
Background:
Continuous glucose monitor (CGM) systems were originally intended only for people with diabetes. Recently, there has been interest in monitoring glucose concentrations in a variety of other situations. As data accumulate to support the use of CGM systems in additional states unrelated to diabetes, the use of CGM systems is likely to increase accordingly.
Methods:
PubMed and Google Scholar were searched for articles about the use of CGM in individuals without diabetes. Relevant articles that included sufficient details were queried to identify what cohorts of individuals were adopting CGM use and to define trends of use.
Results:
Four clinical user cases were identified: (1) metabolic diseases related to diabetes with a primary dysregulation of the insulin-glucose axis, (2) metabolic diseases without a primary pathophysiologic derangement of the insulin-glucose axis, (3) health and wellness, and (4) elite athletics. Seven trends in the use of CGM systems in people without diabetes were idenfitied which pertained to both FDA-cleared medical grade products as well as anticipated future products, which may be regulated differently based on intended populations and indications for use.
Conclusions:
Wearing a CGM has been used not only for diabetes, but with a goal of improving glucose patterns to avoid diabetes, improving mental or physical performance, and promoting motivate healthy behavioral changes. We expect that clinicians will become increasingly aware of (1) glycemic patterns from CGM tracings that predict an increased risk of diabetes, (2) specific metabolic glucotypes from CGM tracings that predict an increased risk of diabetes, and (3) new genetic and genomic biomarkers in the future.
Introduction
Continuous glucose monitoring provides detailed information about glucose fluctuations. This information is very useful for people with diabetes who have better outcomes when they bring their abnormal glucose concentrations into the target range. Recently, a trend has emerged for people without diabetes to use continuous glucose monitors (CGMs) as a personal health device to track their glucose concentrations and promote healthy habits such as increased activity, changes in nutrition, and sleep habits. The motivation behind people without diabetes wearing CGMs is to (1) improve their glucose patterns and avoid diabetes, prediabetes, or other states of glycemic fluctuations that are outside of the typical normal range; (2) optimize their blood glucose concentrations to achieve peak mental or physical performance; and (3) utilize personal data to promote/motivate sustained healthy behavioral changes.
Various CGMs are approved by the United States Food and Drug Administration (FDA) for use in adults and children with diabetes. However, little is known about the role and utility of CGMs for improving health outcomes in individuals without diabetes. Twenty-four-hour CGM profiles may detect a more detailed glycemic profile than self-monitored blood glucose and are less burdensome than checking glucose by fingerstick or by an oral glucose tolerance test. There is a growing interest in utilizing CGM technology for clinical and research purposes to augment lifestyle modification techniques and promote health outcomes across all age groups.1 -3 Continuous glucose monitoring provides an opportunity to support lifestyle modification by providing individualized biofeedback on how behavior change strategies impact biological responses and thus promote health. 4
Methods
PubMed and Google Scholar were searched for articles about the use of CGM in individuals without diabetes. Relevant articles that included sufficient details were queried to identify what cohorts of individuals were adopting CGM use and to define trends of use.
Normative Data
Studies of glucose concentrations using CGMs in populations of people with normal baseline fasting and glucose tolerance tests have demonstrated glucose concentrations almost completely in the tight range of 70 to 140 mg/dl.5-12 Over the past fifteen years, eight such studies from Germany in 2007, 5 the United States (US) in 2010, 6 Japan in 2018, 7 Spain in 2018, 8 Sweden in 2018, 9 the US in 2019 10 and 2021, 11 and Sweden in 2022 12 are summarized in Figure 1.

Results of studies reported since 2007 using CGMs to determine times spent in various ranges of glucose concentrations in populations of people with normal baseline fasting and glucose tolerance tests. Under “reported time in ranges,” the gray bars represent the range of glucoses reported in each study. If the original study reported exact minimum or maximum values, the gray bar has hard edges; otherwise it has faded edges if only “less than” or “greater than” were used in the data reporting. The dotted lines at 70mg/dL and 140 mg/dL are not shown for studies that used different cut-off values. Established target times in ranges for patients with type 1 diabetes per the International Consensus on Time in Range. 13 Abbreviations: CGM, continuous glucose monitor; T1D, type 1 diabetes; TAR, time above range; TBR, time below range; TIR, time in range.
Effects of Diet on Glycemia Learned From Using CGM Data
It is well described that blood glucose levels change after a meal and that postprandial glucose responses (PPGR) depend on the macronutrients consumed, the time of day, and the individuals underlying beta-cell function.14,15 Postprandial glucose responses are increasingly considered a major determinant of glycemic control, yet methods for predicting PPGR to food remain elusive and of limited efficacy. 16 CGMs can be utilized to evaluate the frequency with which individuals demonstrate elevations in postprandial glucose, the types of patterns, and how patterns vary between individuals given an identical nutrient challenge.17,18 There are a growing number of scientists and physicians who are developing analytical frameworks that can group individuals according to specific patterns of glycemic responses called glucotypes.19,20 Despite the association of certain glucotypes or PPGR with increased risk of conversion to diabetes and diabetes-related complications, predicting the impact of specific foods on PPGR has been challenging because of the high variability in different individual responses to the same food. 21 Some attempts have been made to estimate PPGR using the carbohydrate content of meals or using glycemic indices. Few studies have attempted to build or test predictive models of PPGR based on specific foods eaten in individual meals. In a seminal study on personalized nutrition, Zeevi et al 16 used CGM to track the glucose response of 800 adults for one week. The study team then developed a machine learning model that could predict the glucose response of a meal for each participant based on individual factors, such as anthropometric variables, PPGR, blood panels, and gut microbiome. Subsequently, this model has been tested on several other independent cohorts and was able to generate personalized nutrition plans that resulted in improved glycemic variability. 22 Since these findings were published, several commercial companies have been working to integrate CGM technology for personalized nutrition and health.
Effects of Exercise on Glycemia Learned From Using CGM Data
Few studies have generated normative data about the effects of exercise on glycemic profiles solely on people with normal glucose tolerance. DuBose et al 23 studied 153 adults with normal glucose tolerance who performed either aerobic or resistance exercise. The mean glucose concentration during nights following exercise days was 82 mg/dl (distributed as 83 mg/dl for aerobic exercise and 76 mg/dl for resistance exercise) compared with 85 mg/dl during nights following non-exercise days (P = 0.05). 23 As measured by CGMs in adults with normal glucose tolerance and body mass index, exercise was shown to impact the metabolic effects of a high calorie diet. Greater activity resulted in lower glucose concentrations, as well as decreased visceral fat and increased cognition. 24 We expect that CGMs will be used in the future to assess the effects of exercise on glycemia in people with normal glucose tolerance. The use of CGM to identify glycemic profiles which might reflect the rate of glucose clearance has been proposed as a surrogate for evaluating resting energy expenditure, 25 although variations in the release of insulin and catecholamines can mask differences in glucose clearance. 26 Using CGM in a cohort of ten sub-elite athletes (6 hours per week), Thomas and colleagues 27 found that 4 out of 10 spent more than 70% of the total monitoring time above 108 mg/dl (even after excluding the 2-hour period after meals) and 3 out of 10 had fasting blood glucose concentrations in the prediabetes range. Although some athletes have expressed concern about hypoglycemia during exercise, 28 the small number of athletes in this study did not experience any hypoglycemia.
Effects of Stress on Glycemia Learned From Using CGM Data
The incidence of diabetes is lower among most populations living at higher altitudes than at lower altitudes. 29 However, among hikers with normal glucose tolerance wearing a CGM in Nepal, Hill et al 30 observed a significant increase in nocturnal glucose at 3600 meters and higher compared with a baseline altitude of 1100 meters. It is possible that the stress response to exercise during high altitude dominates exercise-enhanced insulin sensitivity, resulting in relative hyperglycemia. 30 Furushima et al 31 found that in a series of 40 patients in an intensive care unit with sepsis, the mean amplitude of glycemic excursions, measured by CGM, during the first 48 hours of treatment were associated with increased all-cause mortality and the concentration of urinary 8-iso-prostaglandin-F2α, which was assumed to be a surrogate of oxidative stress. The effect of the stress of an infection on glycemia was also demonstrated with CGM data in a comparison of times within target range, above target range, and below target range in a study of 31 participants with normal glucose tolerance. In this study that spanned a median time of six days per participant, 13 participants had mild COVID-19 and 18 participants did not have COVID-19. The participants with COVID-19 compared with the healthy controls experienced, respectively, significantly greater time above target ranges of both 140 mg/dl and 180 mg/dl (13.9% vs 2.3% and 1.9% vs 0%, respectively) and significantly less time within one of two target ranges (80.1% vs 93.1% for glucose 70-140 mg/dl), but not significantly less time for glucose 70-180 mg/dl (92.1% and 100%). 32 Given the evidence for an increased incidence of diabetes following a COVID-19 infection, 33 such data might be useful to identify patients with COVID-19 without diabetes who are at increased risk of developing diabetes.
Results: Potential Uses of CGM by People Without Diabetes
Researchers and clinicians have been interested in the monitoring capabilities of CGMs beyond diabetes since the technology became available. Broadly speaking, current and potential CGM applications can be organized into four domains (Figure 2):
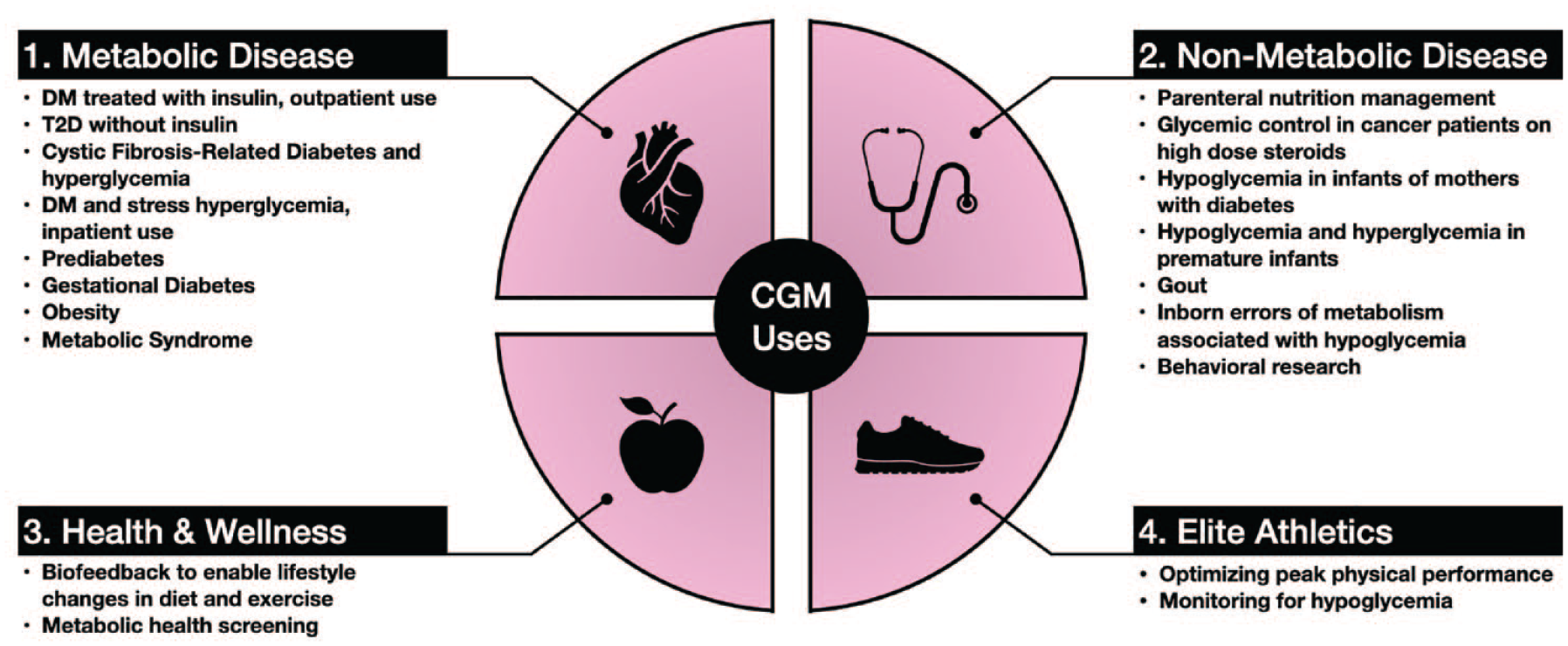
Clinical use case domains for continuous glucose monitors. Abbreviations: CGM, continuous glucose monitor; DM, diabetes mellitus; T2D, type 2 diabetes.
Metabolic Disease
The first CGM, a professional model, was cleared by the FDA in 1999 for monitoring interstitial glucose levels in persons with diabetes mellitus.34,35 CGMs have evolved over the past two decades, but the vast majority of research (and the only FDA-cleared indication) continues to be focused on diabetes. CGMs are indicated for outpatients with diabetes who are using insulin. 36 CGMs are also being used in selected outpatients with diabetes who are not using insulin 4 as well as people with cystic fibrosis-related diabetes 37 and cystic fibrosis-related hypoglycemia. 38
CGMs have been used to achieve tight glycemic control in critically ill hospitalized patients with secondary diabetes39,40 or stress hyperglycemia in adults 41 and children. 42 This type of practice is currently investigational and requires protocols specifying sensor glucose and point of care glucose measurements for sensor validation as well as highly trained staff members and appropriate hospital policies. 43 The authors of this article believe that patients with related metabolic diseases characterized by inadequate insulin production or activity, like prediabetes, 44 gestational diabetes, 45 obesity, 46 and metabolic syndrome 47 may also benefit from CGM use, but more research is required to support and clarify these indications.
On March 1, 2022, the FDA granted breakthrough device designation to the Dexcom Hospital CGM System (Dexcom Inc., San Diego, CA). This designation will provide prioritized pre-market review and increased opportunities to interact with FDA’s experts, ultimately leading to authorization and timely access to this technology for hospitalized patients and healthcare professionals. 48
Prediabetes
For diagnosing diabetes, Hemoglobin A1c does not necessarily correlate with a fasting plasma glucose or a two-hour glucose concentration in a glucose tolerance test, and these two glucose concentrations do not necessarily correlate with each other. Hemoglobin A1c, as well as fasting, and postprandial glucose concentrations do not always cleanly differentiate between diabetes, prediabetes, and a normal metabolic state. CGMs can provide additional diagnostic information. Thus, CGM data might facilitate earlier diagnosis and treatment for people with prediabetes or at increased risk of developing type 2 diabetes (T2D). 44 A standardized method and standardized diagnostic result will be needed before CGMs will be widely accepted as a tool to diagnose diabetes, but a CGM result might provide a signal that a traditional diagnostic test for diabetes is needed. It should be noted that youth onset T2D progresses much more aggressively than adult-onset T2D and results in earlier time to onset of macro and micro vascular complications. 49 This means that early detection and treatment is key to recognition in order to prevent the early onset of these life limiting complications for youth with T2D.
Gestational diabetes
CGMs may provide information that can help predict gestational diabetes. 45 Use of CGMs for monitoring glucose concentrations during pregnancy has been beneficial in type 1 diabetes, T2D, and gestational diabetes. 50
Obesity
Obesity is the greatest risk factor for the development of diabetes. 51 CGM data can be used as biofeedback to help support behavioral and lifestyle interventions, including portion control and both timing and quality of food consumed.1,52 Obesity interventions can prevent or delay onset of T2D as well as other obesity-related diseases (e.g. cardiovascular disease, osteoarthritis, and worse outcomes with COVID-19) in those at risk. 53 Current interventions for weight management are limited, labor and time intensive, and costly. Also, they often suffer from attrition and lack of durability. New tools that leverage technology to support behavior modification could be helpful in preventing the morbidity and mortality associated with obesity. 54
Non-Metabolic Disease
The literature contains a very limited number of case reports of using CGM in a variety of primarily non-metabolic diagnoses. These uses include to manage glycemia during parenteral nutrition for inpatients, 55 monitor cancer patients with hyperglycemia due to steroid therapy,56,57 detect neonatal hypoglycemia in infants of mothers with diabetes, 58 prevent hyperglycemia and hypoglycemia in premature infants, 59 assess the relationship between glycemic fluctuations and serum uric acid concentrations in gout patients, 60 assess glucose management and optimize treatment in patients with inborn errors of metabolism associated with hypoglycemia, 61 and facilitate behavioral research. 62
Health and Wellness
Healthy behavior (e.g. portion control, increased activity and sleep, and avoidance of processed foods and emotional eating) can delay or prevent onset of T2D. If people without diabetes can see their glucose concentrations in real time and respond with healthy behavior when the concentrations are above a physician-determined target or above some concentration that is currently not standardized, then a feedback loop can be developed. Therefore, CGM information may promote healthier behaviors and prevent onset of T2D. 63 A small amount of evidence in the literature supports the idea that knowing one’s data from a CGM can lead to sustained behavior change across all activities of daily living. 44
CGMs have the potential to be used to promote behavior modifications for health benefits. 64 Liao and Schembre 65 studied a cohort of 30 people with normal glucose tolerance who wore a CGM and used a mobile app to track exercise and diet. The people found the measurement tools were easy to use and able to provide relevant information of interest to them. 65 Liao et al 66 then used CGMs along with counseling and an exercise tracker to motivate physical activity in 19 obese individuals who did not have diabetes. Participants gave high satisfaction ratings to the intervention. 66 See Table 1 for results of the acceptability survey by Liao et al.
Mean of Acceptability Score for Each Data Collection Tool and Comparisons of Their Mean Scores.
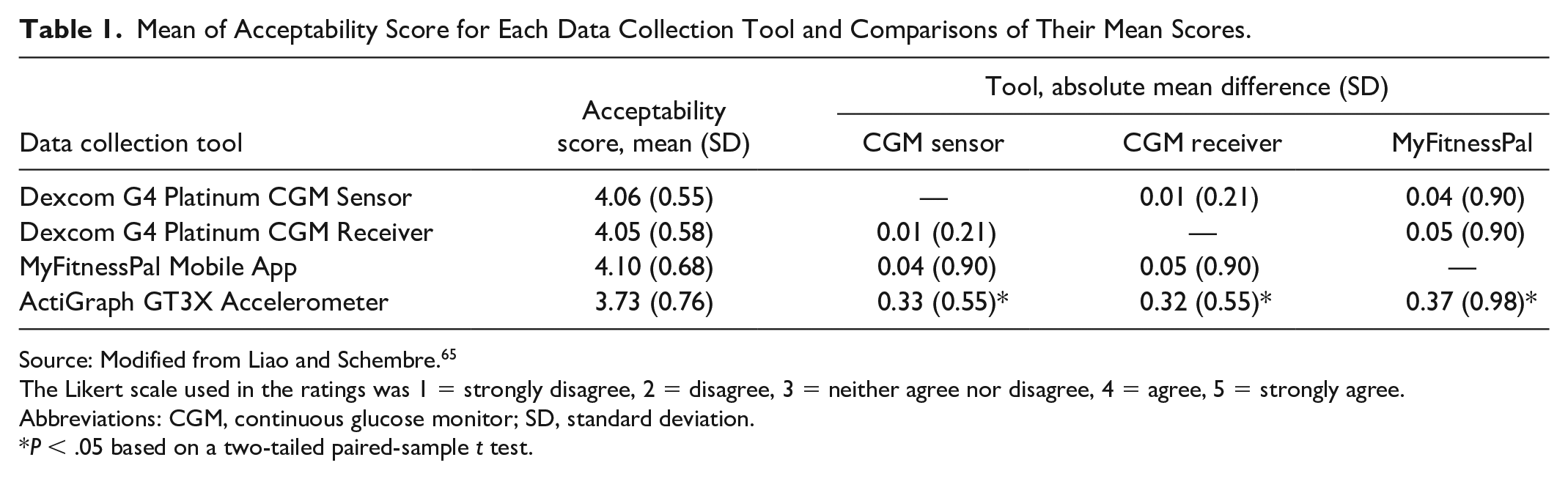
Source: Modified from Liao and Schembre. 65
The Likert scale used in the ratings was 1 = strongly disagree, 2 = disagree, 3 = neither agree nor disagree, 4 = agree, 5 = strongly agree.
Abbreviations: CGM, continuous glucose monitor; SD, standard deviation.
P < .05 based on a two-tailed paired-sample t test.
Whelan et al 67 conducted a qualitative study exploring intuitive engagement with real-time glucose from CGM data and physical activity feedback and found that accessing behavioral and physiological feedback increased self-awareness of how lifestyle impacts short-term health in adults with obesity at risk for T2D. Participants in this study reported changing what food types they selected at meals based on seeing a prominent spike on their CGM from prior consumption. In addition, they reported that motivation to avoid food that resulted in large glycemic excursions encouraged them to make healthier eating choices. 67 Other studies have utilized CGMs as behavioral modification tools both to provide real-time feedback and for hunger cue training and impulse control response. 1
The team at January AI was the first to investigate the potential benefit of using a CGM with a mobile app in people without diabetes in the Sugar Challenge study. They tested a regimen consisting of a ten-day CGM combined with a mobile app that links an individual’s glucose tracing to meal composition, heart rate, and physical activity and provides feedback in a cohort of 473 participants, who self-declared as being healthy (n = 448) or having prediabetes (n = 25). The result was a significant improvement in the time in range (defined as 54-140 mg/dl) from the first two days until the final two days of the wear period. 68 A study sponsored by Signos, Inc, is currently recruiting 20,000 people without diabetes to participate in a study that will use a CGM and a mobile health app to optimize general wellness and body weight. 69
Elite Athletics
Some clinicians have touted the idea that there is a glucose concentration which is a subset of glucose levels that are not widely considered pathological where the body functions at peak performance. These clinicians believe that maintaining normal baseline glucose levels in a narrow peak performance range will improve body function by avoiding occasional hyperglycemia that is inherently unhealthy and providing particularly protection from developing T2D. 70 There is virtually no evidence for this belief, but there has been little research in this area because CGM data is so new and almost all the CGM data in the literature are in people with diabetes. In testing the value of CGM use by athletes without diabetes, it is helpful to determine sensor accuracy both at rest and during exercise. 71 Ishihara and colleagues 72 studied CGM data on seven ultramarathon (160 km) runners and found that their glucose concentrations ranged from 61.9 to 252.0 mg/dl. Average glucose concentrations over the entire race in the runners ranged from 104 to 164 mg/dl. Running speeds correlated significantly with glucose concentrations and with energy/carbohydrate intake. The article did not specify whether the athletes were aware of their glucose concentrations. 72 In a study of sub-elite athletes wearing CGMs by Thomas and colleagues, 27 one of ten participants spent a significant amount of time (between 10% and 20%) with glucose concentrations below 72 mg/dl. Olsson reported in an unpublished white paper that among four Swedish national elite swimmers monitored with a CGM for six days, three had occurrences of hypoglycemia (below 70 mg/dl). These three swimmers were hypoglycemic for an average of 224 minutes per week, which represented 2.59% of the entire week. 73 No study has been reported in the medical literature as to (1) whether an otherwise healthy person with normal glucose tolerance or an elite athlete can use CGM to attain a narrow glucose range generally accepted as peak performance level of glycemia and (2) whether increased time living at such a level indeed improves performance at any task. Are the potential benefits of CGM real for people without diabetes, including elite athletes? At this time, the answer is unproven.
Discussion: Summary of Benefits
The evidence for CGM use is scant beyond the metabolic and non-metabolic disease domains, and consensus is virtually non-existent outside of diabetes. Since using CGMs is not without cost, most endocrinologists are awaiting data before recommending CGM to people without diabetes. On the other hand, some people are choosing to self-pay for CGM equipment because of the appeal of knowing their glucose concentrations under various circumstances, including following ingestion of specific foods and performance of specific activities. Some endocrinologists are advocating use of CGM to prevent T2D without evidence in the medical literature based on the idea that absence of proof is not proof of absence. 74 Many CGM users without diabetes are advocates of the quantified self-movement, which advocates self-monitoring and self-management of multiple types of physiological data streams. 75
Potential Drawbacks of CGM Use by People Without Diabetes
There is virtually no risk to using a CGM other than (1) cost, which is currently not paid by insurance companies outside of an evidence-based covered wellness program, (2) tape allergy, which is infrequent, (3) skin trauma, which is rare, and (4) stress or distress, which could be associated with having access to the data. Two drawbacks to the use of CGMs by people without diabetes are not specifically risks but rather problems with using the data and these include (1) an absence of consensus standards for defining abnormal values and (2) an absence of consensus standards for how to respond to abnormal values. Finally, regardless of the use case, a CGM system may provide inaccurate readings if it is applied incorrectly or malfunctions, which could potentially lead to inappropriate or dangerous corrective action. It is important for a patient to understand the product’s indications and limitations. At the high end of the target range for most people (70-180 mg/dl) measured by CGMs, most of these products on the market function at approximately their published levels of accuracy, but at the low end of the target range, they are usually less accurate. 76 Several questions are currently unanswered (Table 2). With further research, it is hoped that the indications, benefits, and drawbacks of CGM for people without diabetes will become clearer.
Questions About CGM in People Without Diabetes.
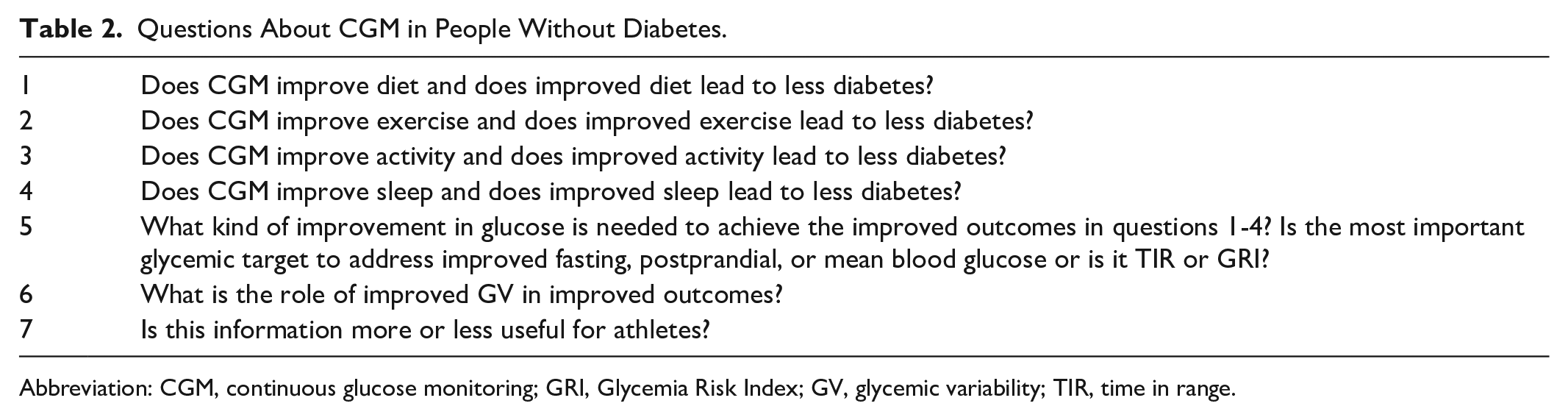
Abbreviation: CGM, continuous glucose monitoring; GRI, Glycemia Risk Index; GV, glycemic variability; TIR, time in range.
Regulation of CGM Systems Used for People Without Diabetes
Four manufacturers produce all the FDA authorized CGM systems in the United States. They include Abbott Diabetes Care (Alameda, CA), Dexcom Inc. (San Diego, CA), Medtronic Diabetes (Northridge, CA), and Senseonics (Germantown, MD). All of those systems have either been cleared or approved as prescription use only devices for the management of patients with diabetes. Currently, therefore, manufacturers are restricted in making any claims for the use of their product on people without diabetes, although clinicians are able to prescribe them for such use.
Will the FDA regulate the use of CGM systems in use for people without diabetes? The FDA has broad authority to regulate diagnostic devices, yet it has recognized that the use of such devices to promote healthy living or for sport performance is low risk, so it has concluded that is not worth spending its limited resources regulating such devices. On September 27, 2019, FDA issued a final guidance on “General Wellness: Policy for Low Risk Devices.” 77 Two of the possible uses mentioned, which are to inspire healthy behavior or to function at peak performance, fit squarely within the FDA stated policy. A claim for detecting prediabetes does not. Besides making general wellness claims, a device must be non-invasive. The guidance defines invasive as: “penetrates or pierces the skin or mucous membranes of the body.” As such, CGM devices that are non-invasive will likely not be regulated by the FDA, but devices that are implanted, as most of currently approved and cleared devices are, will have to make a filing with the Agency before such devices can be placed in the market. Even for non-invasive CGMs, the FDA will be concerned with the risks associated with an over-the-counter device that has wellness claims. Chief among those will be the use of such devices by people with diabetes to manage their condition. Given that the FDA still has jurisdiction over such devices, it can issue safety alerts or develop labeling or other controls to mitigate the risks those devices may pose, or change the policy for those devices that pose an increased risk to health.
Commercial Health and Wellness CGM Programs
In the US and Canada, CGMs require a medical prescription and are only approved for use in patients with diabetes. In recent years, several digital health companies (Table 3) have started offering factory calibrated CGMs from Abbott Diabetes Care or Dexcom to consumers paired with a mobile app and often coaching or dietician services to “improve general health.” Most of these companies do not detail on their website how they provide CGMs to people without diabetes and they do not claim to be recruiting for clinical trials of CGM. However, the fact that these digital health companies do not accept insurance and yet they offer to provide prescriptions suggests that the companies employ or contract with physicians that write off-label prescriptions for CGMs that the patients pay for out of pocket. The basic premise of these services is not dissimilar to the role of CGMs in more traditional clinical and research settings: to provide real-time biofeedback to help individuals make lifestyle changes. However, whether CGMs are effective behavior change tools in people without diabetes is not known. It is also not known who is most likely to benefit from using a CGM and how often diabetes can be prevented or delayed. Finally, it is unclear what, if any, is the impact of glycemic variability in individuals with normal glucose homeostasis. Research is needed to define the best role for CGMs in populations without diabetes. Several consumer health product companies are developing noninvasive glucose monitors (mostly with a wrist watch form factor) for people without diabetes to measure glucose optically from the skin in the wrist.78-81 It remains to be determined how accurate these new devices will be and how they will be regulated.
Companies That Market CGMs for Health and Wellness Applications.
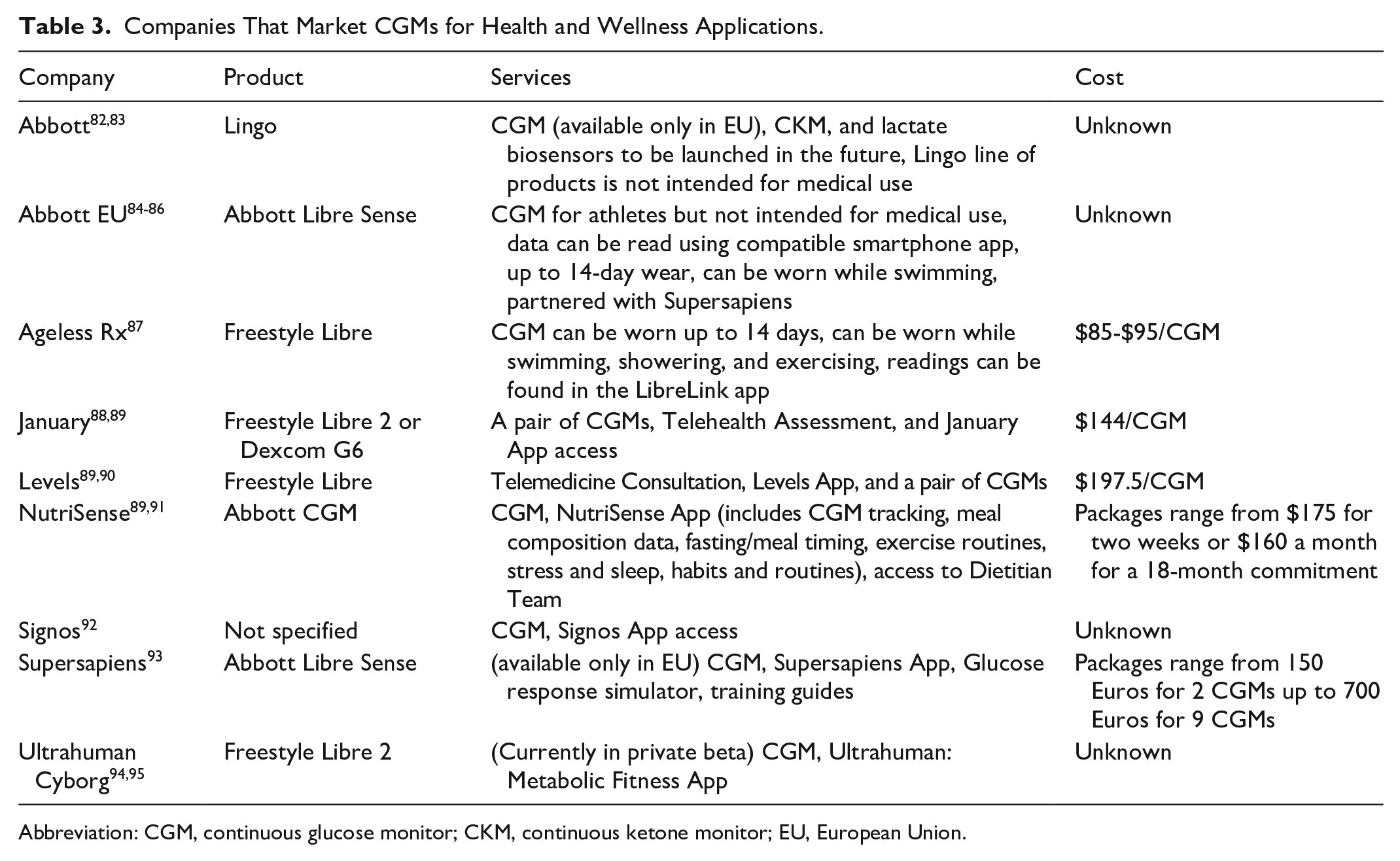
Abbreviation: CGM, continuous glucose monitor; CKM, continuous ketone monitor; EU, European Union.
Conclusions: The Future of CGM Use by People Without Diabetes
We expect increasing adoption of CGM use in four areas: metabolic diseases other than diabetes, non-metabolic diseases, health and wellness, and elite athletics. We see seven trends in the use of CGM systems in people without diabetes (Table 4). These trends can pertain to both FDA-cleared medical grade products as well as anticipated future products, which may be regulated differently based on intended populations and indications for use. 64 We expect that clinicians will become increasingly aware of (1) glycemic patterns from CGM tracings that predict an increased risk of diabetes, (2) specific metabolic glucotypes from CGM tracings that predict an increased risk of diabetes, and (3) new genetic and genomic biomarkers in the future that are linked to an increased risk of developing diabetes in the presence of any atypical CGM readings. It is likely that a role for CGMs will be discovered for people without diabetes who are (1) at increased risk of developing diabetes based on emerging biomarkers and glucotype classifications, (2) in an overweight or obese state, or (3) elite athletes at risk of hypoglycemia. Although little data has been reported to date on the benefits of CGM for people without diabetes, a groundswell of interest in defining the role of this type of sensor is now beginning. We expect that well designed clinical trials will demonstrate a specific role for CGM use in people without diabetes, and that consensus guidelines will establish how to interpret and respond to this type of data. As our understanding of this technology and the data it generates improves through rigorous science, more and more people without diabetes will be able to benefit from CGMs.
Future Uses of CGM Systems in People Without Diabetes.

Abbreviation: CGM, continuous glucose monitor.
Footnotes
Acknowledgements
The authors thank Lynn Kysh for her help in designing and conducting the background literature search, and Annamarie Sucher-Jones for her expert editorial assistance.
Abbreviations
CGM, continuous glucose monitor; CKM, continuous ketone monitor; COVID-19, coronavirus disease 2019; DM, diabetes mellitus; EU, European Union; FDA, United States Food and Drug Administration; GRI, Glycemia Risk Index; GV, glycemic variability; PPGR, postprandial glucose responses; TAR, time above range; TBR, time below range; TIR, time in range; T1D, type 1 diabetes; T2D, type 2 diabetes; US, United States.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DCK is a consultant for Eoflow, Eli Lilly, Lifecare, Integrity, Medtronic, Roche Diagnostics, Rockley Photonics, and Thirdwayv. KTN and NYX are consultants for Abbott Diabetes Care. AG is a contract consultant for the ProPharma Group and helps medical device companies with regulatory issues. JCE is a paid consultant for AI Health. JCE’s time is supported in part by the Food and Drug Administration under award number P50FD006425 for The West Coast Consortium for Technology & Innovation in Pediatrics (PI: Espinoza). APV receives CGM product in kind from Dexcom for research conduct. The content is solely the responsibility of the authors and does not necessarily represent the official views of the FDA.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
