Abstract
Insulin pumps are used by many patients with diabetes to manage their diabetes therapy. Adequate handling of the systems is important to avoid errors. In this study, one aspect of device handling—the number of steps required to operate the system—was evaluated for different insulin pump systems. Specific tasks that are usually performed by insulin pump users were simulated and all necessary actions were documented. Differences between the required numbers of steps strongly depended on the specific task. So did the level of guidance for these tasks provided by the systems. Results of this study provide an overview of this particular aspect of insulin pump handling rather than a general advice.
Continuous subcutaneous insulin infusion (CSII) has become a therapeutic option for many people with type 1 diabetes. Today’s insulin pump systems offer multiple functions to optimize insulin delivery to individual patients’ needs and increase adherence to CSII. 1 In turn, an increasing number of functions and options may be associated with increased handling effort as well as with susceptibility to errors, especially in the initial pump set-up, but also during daily life use. To investigate the number of steps required for specific tasks, a study was performed comparing one given pump (Accu-Chek® Insight system; Roche Diabetes Care GmbH, Mannheim, Germany) with other currently available insulin pumps.
Methods
In this study, eight insulin pump systems (Table 1) were evaluated regarding the number of steps that are required to perform different routine tasks associated with pump usage (if applicable, with the corresponding blood glucose meter [BGM] or remote control):
Programming a bolus (standard bolus via menu and as quick bolus on pump)
Editing a basal rate profile
Choosing a different basal rate profile
Programming a temporary basal rate
Changing the cartridge, infusion set and cannula
Filling the cartridge
Connecting a new infusion set to the pump
Replacing or charging the battery
Pairing the pump with the BGM/remote control
Evaluated Insulin Pump Systems.
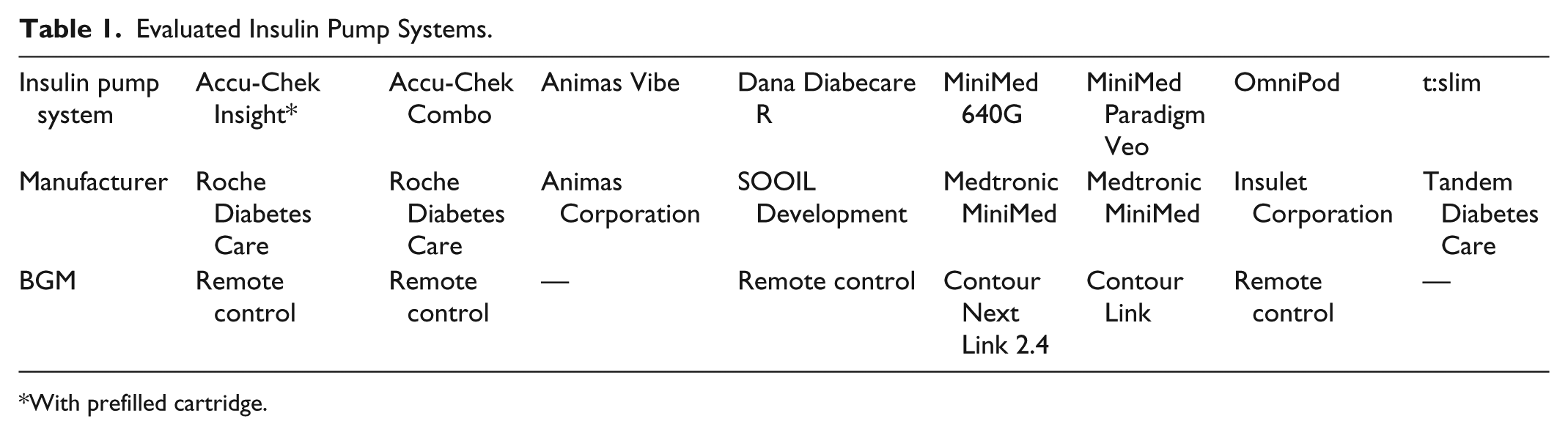
With prefilled cartridge.
For the tasks concerning bolus and basal rate, test settings reflecting clinical practice were used (Table 2); other standard settings were maintained.
Test Settings.
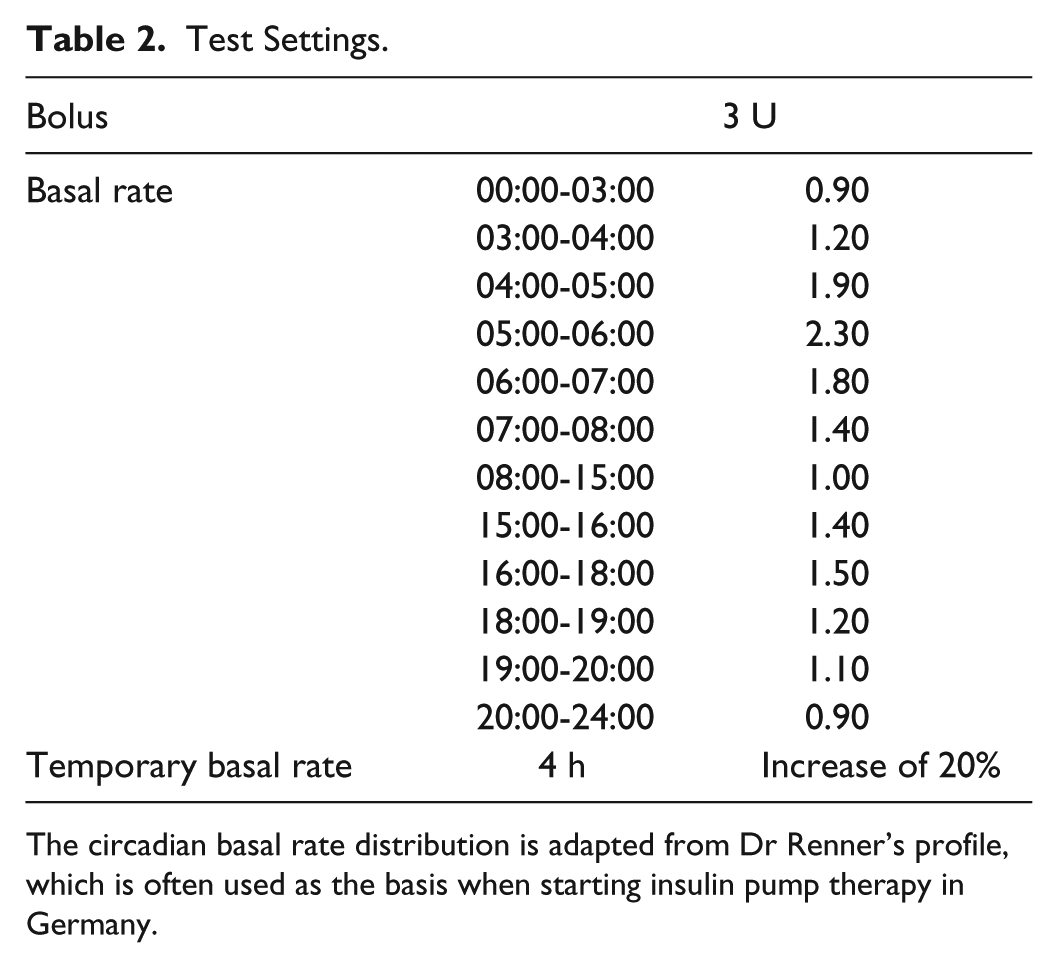
The circadian basal rate distribution is adapted from Dr Renner’s profile, which is often used as the basis when starting insulin pump therapy in Germany.
Two different experienced and trained investigators independently performed all tasks according to the respective instructions for use. The following actions were documented and counted (Figure 1):
Click step: step requiring single or repeated pressing of the same button or tapping on the same spot of a touchscreen
Handling step: step not requiring pressing of a button or tapping on a touchscreen, eg, removing the insulin cartridge
Steps: sum of handling steps and click steps
Clicks: the number of button presses or touchscreen taps required for click steps
Guided step: steps were documented as “guided” if the display indicated how to perform the specific step or announced the next step
Steps that were independent from the systems (like washing hands, warm up insulin) were not counted
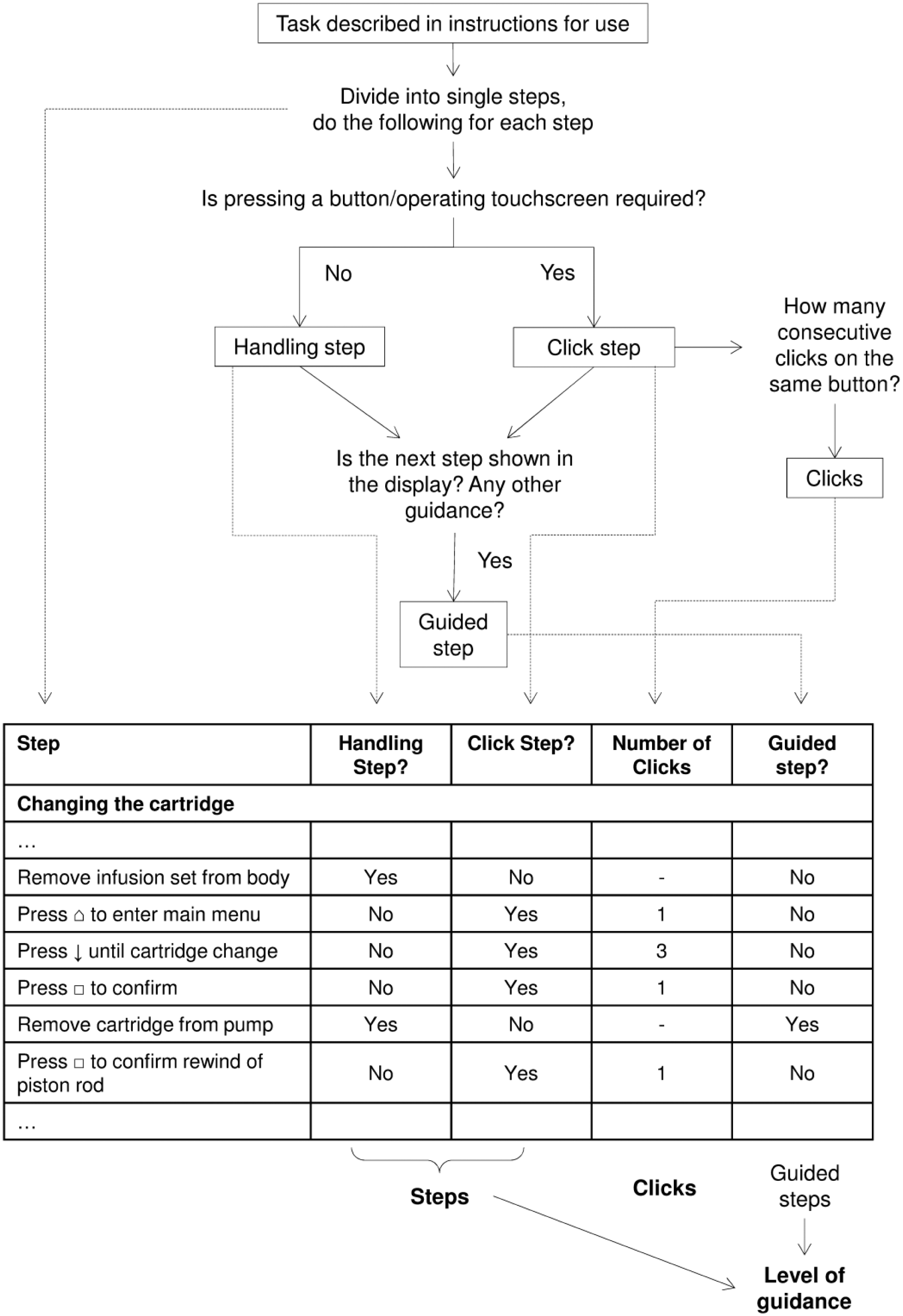
Scheme of step classification and documentation. The inlaid table shows an exemplary excerpt.
For each insulin pump system, the number of steps (handling steps + click steps) and the corresponding number of clicks were evaluated. In addition, the level of guidance was calculated by dividing the guided steps by the total number of steps. It indicates how much support is provided by the pump during the task.
Subsequently, for each task the relative number of steps and clicks compared to one pump (the Accu-Chek Insight system; 100%) was calculated.
The study was performed in two parts: in 2013, seven of the eight pumps were evaluated: Accu-Chek Insight system, Accu-Chek Combo system (Roche Diabetes Care GmbH), Animas® Vibe® insulin pump (Animas Corporation, West Chester, PA), Dana Diabecare® R system (SOOIL Development Co Ltd, Seoul, Korea), MiniMed® Paradigm® Veo™ system (Medtronic MiniMed, Northridge, CA), OmniPod® system (Insulet Corporation, Billerica, MA), t:slim® insulin pump (Tandem Diabetes Care, Inc, San Diego, CA). In 2016, another pump (MiniMed® 640G system; Medtronic MiniMed) that was launched in the meantime was evaluated using the same methods as in the first evaluation. In addition, the first evaluation was repeated for one pump (Accu-Chek Insight system), because a prefilled cartridge became available since then.
Since 2014, this insulin pump system is available in certain European countries. It is intended to be user-friendly and easy to use, but nevertheless offers options to suit individual needs. The system has two main components: the insulin pump and the diabetes manager, a handheld for remote control and other functions. The insulin pump is an external, microprocessor-controlled durable pump equipped with color display screen. In 2014, a prefilled cartridge (NovoRapid® PumpCart®; Novo Nordisk, Bagsværd, Denmark) that contains 160 units of insulin aspart became available for this pump. With its “pause” mode, the pump automatically compensates for insulin not delivered while replacing the cartridge, the infusion set, or the battery, without the need to reprogram ongoing boluses or temporary basal rates as required by some other pumps. The diabetes manager with color touchscreen communicates wirelessly with this pump and can remotely control most of its functions. In addition, it includes a BGM that has an integrated bolus advisor and data management capabilities like display of graphs and reports.
Results
Regarding the evaluated tasks with the selected sample settings, differences between the numbers of steps required with the different pumps were strongly dependent on the specific task. While for some tasks, like programming a standard bolus or temporary basal rate, numbers of steps were similar among the pumps, larger differences between the evaluated pumps were observed especially for editing a basal profile, filling the cartridge and pairing the pump with the corresponding BGM/remote control (Table 3). Similarly, numbers of clicks associated with these tasks varied.
Number of Steps (Handling Steps + Click Steps) and Defined Clicks Required to Perform Different Tasks.
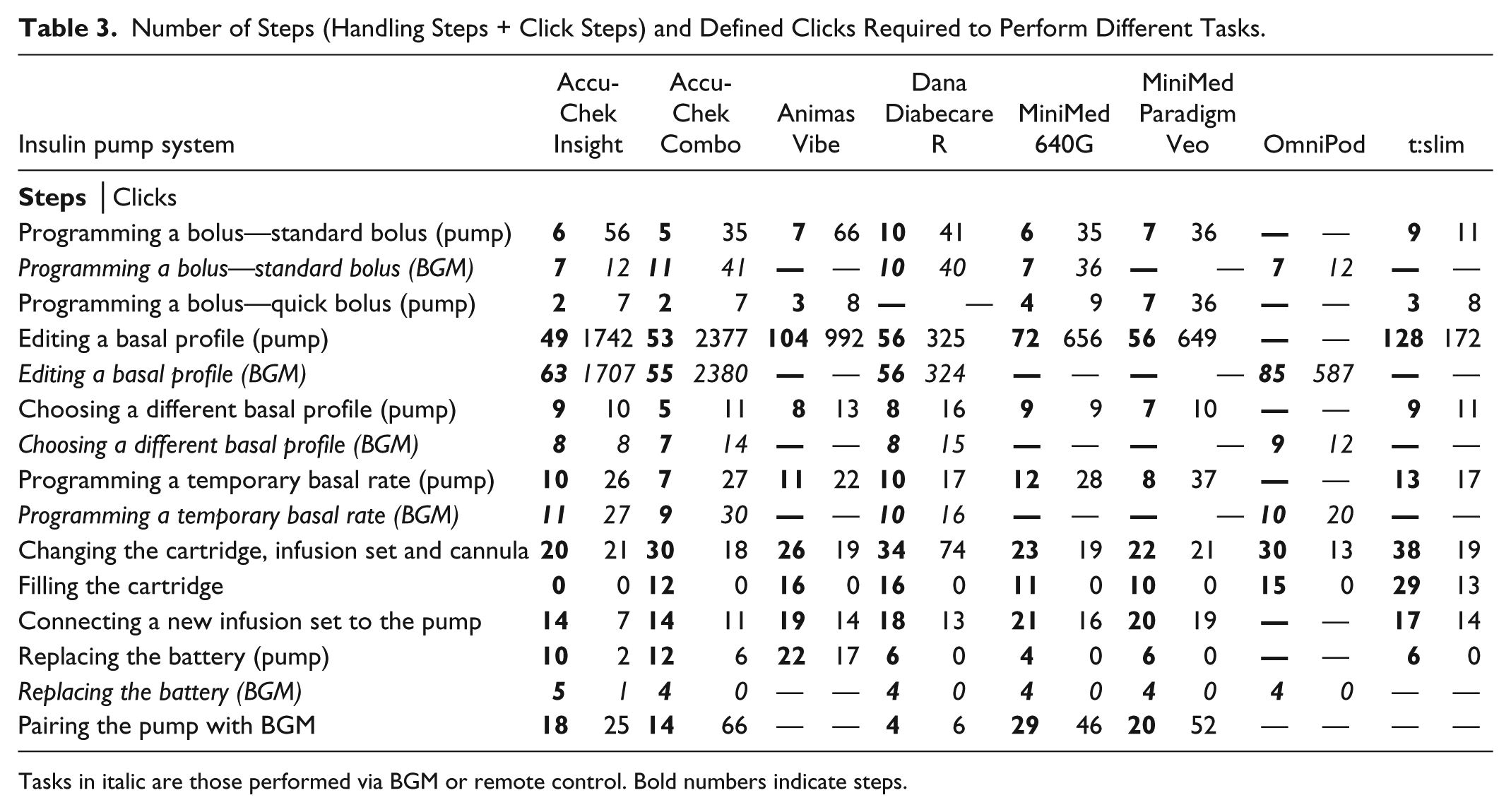
Tasks in italic are those performed via BGM or remote control. Bold numbers indicate steps.
When comparing the number of steps associated with specific tasks, one pump (Accu-Chek Insight system) showed advantages in the following tasks: programming a bolus via remote control, quick bolus delivery, editing a basal rate profile via the pump, changing cartridge, infusion set and cannula, filling the cartridge, and connecting a new infusion set to the pump (Figure 2). Due to the availability of prefilled cartridges, no steps are required for the task filling the cartridge, for which other pumps required at least 10 steps. When comparing the number of clicks required to perform specific tasks, these advantages were observed less often (Figure 3).
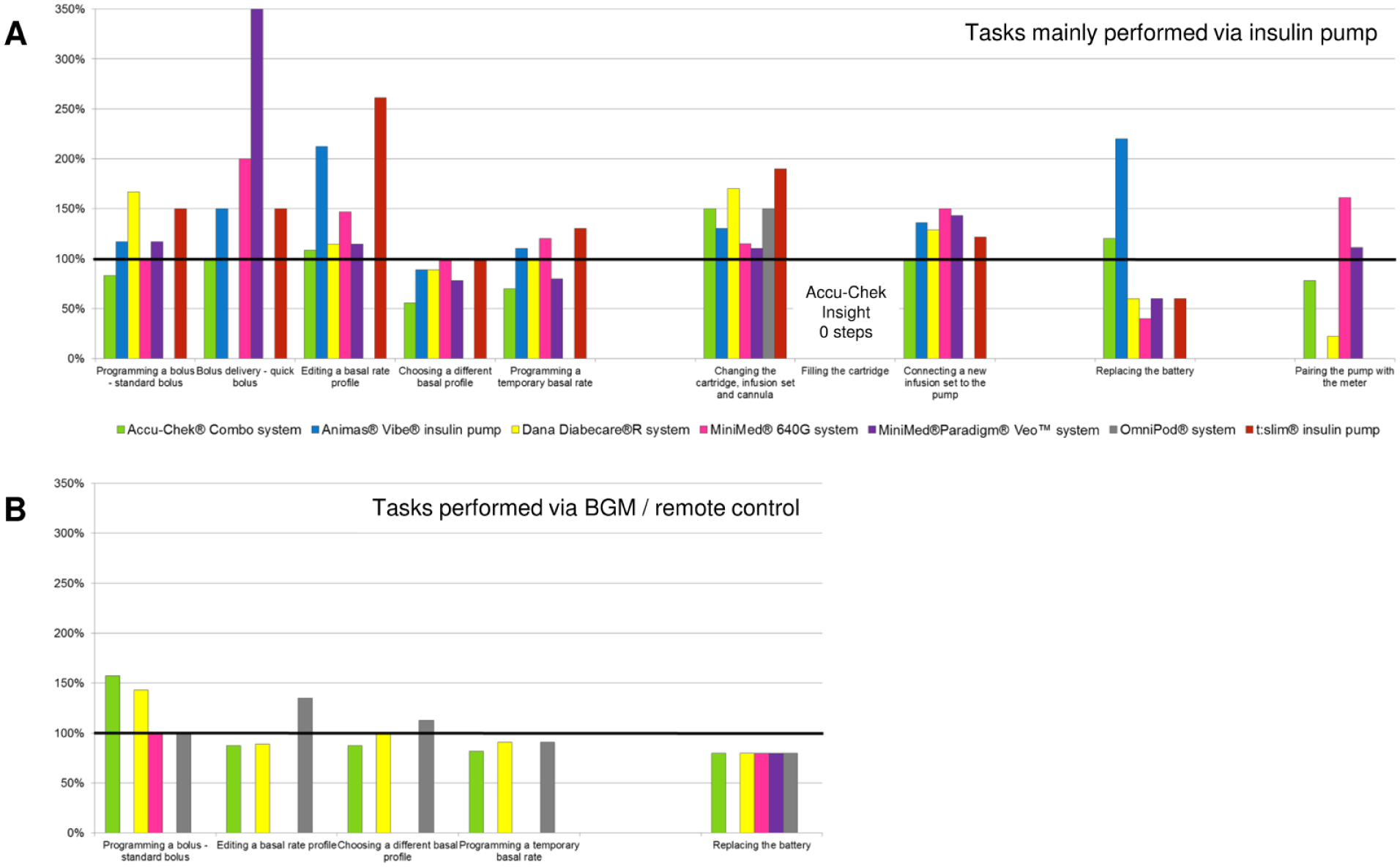
Comparison of steps of seven insulin pump systems to one system (=100%, horizontal black bar). (A) Tasks mainly performed via insulin pump. (B) Tasks performed via remote control or corresponding BGM.
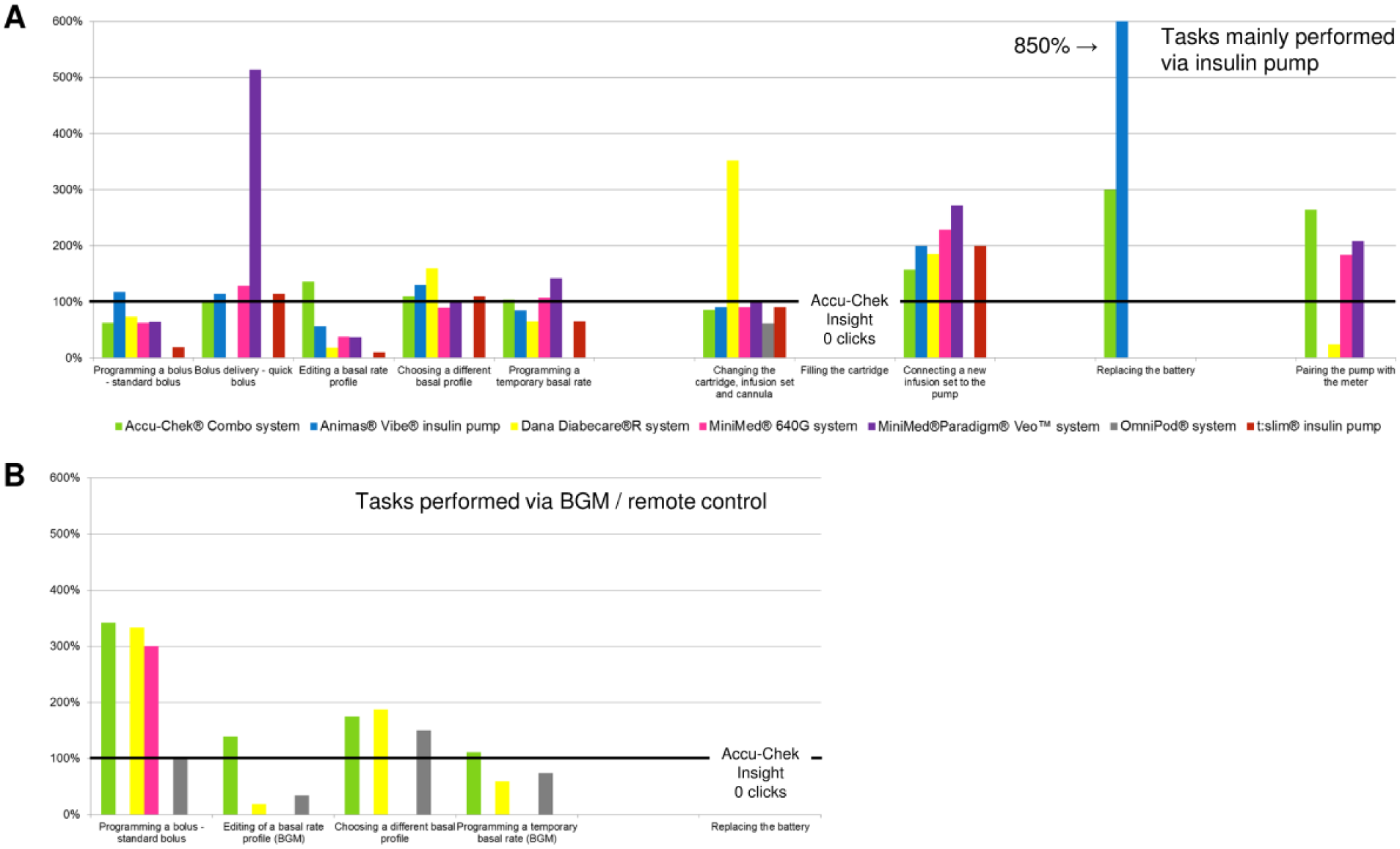
Comparison of clicks of seven insulin pump systems to one system (=100%, horizontal black bar). (A) Tasks mainly performed via insulin pump. (B) Tasks performed via remote control or corresponding BGM.
The level of guidance ranged from 0% up to 98% between all tasks and pumps (Table 4). The largest differences between the pumps in the level of guidance were observed in changing cartridge, infusion set and cannula, and connection of a new infusion set.
Level of Guidance in Different Tasks: Guided Steps Divided by the Total Number of Steps.
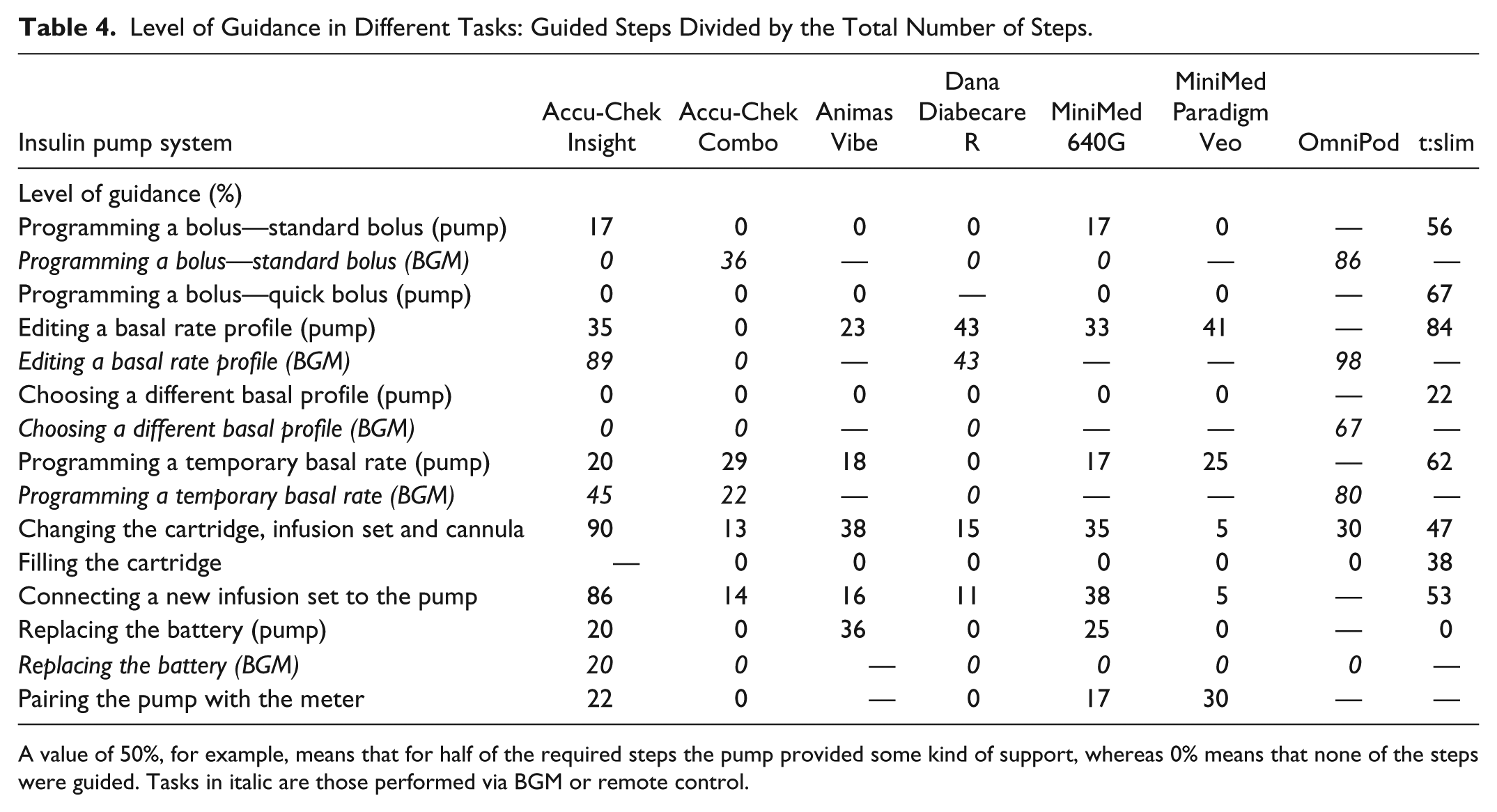
A value of 50%, for example, means that for half of the required steps the pump provided some kind of support, whereas 0% means that none of the steps were guided. Tasks in italic are those performed via BGM or remote control.
Discussion
Besides reliability and functionality, easy handling of an insulin pump system contributes to patient acceptance and last to therapy adherence in CSII, 2 further to diabetes education and health literacy. Using a pump should therefore be neither an additional burden to the patients, nor an unnecessary source for errors. Evaluating handling of a pump is complex as there are a lot of individual preferences. To minimize the influence of such individual preferences, this study aimed at providing an objective assessment by counting steps that are required for tasks commonly performed by patients on CSII. This does not include an evaluation of the ease of use or fine motor skill requirements of any particular step, but rather provides an overview of common steps. Because of the different frequencies with which the evaluated tasks are performed by individual patients, individual needs would have to be translated into a specific weighting of the different tasks. The required level of guidance also is patient-specific, as some may want to have a high level of support whereas others may even perceive any guidance as disturbing. Therefore, the present study cannot give general advice on which insulin pump system is the best for which patient in terms of handling, but it can serve as a guide. In addition, all the numbers shown only apply to the exact test settings that were used in this study and will differ if, for example, other basal rates are programmed.
Not all tasks could be performed with all pumps. Only 10 of the 16 tasks could be performed for those pump systems that had no integrated remote control or BGM. Similarly, the patch pump evaluated can only be operated via the remote control and does not have a separate infusion set, which makes a comparison difficult or at least incomplete. Having paired BGM or remote control has some advantages, like discreetness or a better display, but may require carrying an additional device. The focus of this evaluation was the comparison between one pump system and other currently available pumps. This system showed certain advantages in many tasks regarding the number of steps, and in some tasks regarding the number of clicks, and still provided an appreciable level of guidance. Assuming that more handling steps also mean more potential error sources (especially in tasks like filling cartridge or changing infusion set), fewer handling steps may contribute to patient safety. However, in some cases, more handling steps might contribute to improved safety even at the expense of decreased convenience. The main advantage of the insulin pump in focus in this analysis was the prefilled cartridge, which reduces effort for the patients and, in addition, excludes errors like air bubbles within the cartridge or contamination during self-filling of cartridges.
Conclusion
In general, handling and ease of use of insulin pump systems should not be underestimated as factors that may impact compliance with and efficacy of CSII. There are considerable differences between available insulin pump systems.
Footnotes
Acknowledgements
The authors would like to thank Stefan Pleus (IDT) for assistance in data management and for reviewing the manuscript and Ralph Ziegler (Diabetes Clinic for Children and Adolescents Münster) for reviewing the manuscript.
Abbreviations
BGM, blood glucose meter; CSII, continuous subcutaneous insulin infusion.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DW, KS, and AW are employees of the Institut für Diabetes-Technologie Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany (IDT). GF is general manager of the IDT, which carries out clinical studies on the evaluation of BG meters and medical devices for diabetes therapy on its own initiative and on behalf of various companies. GF/IDT have received speakers’ honoraria or consulting fees from Abbott, Ascensia, Bayer, Berlin-Chemie, Becton-Dickinson, Dexcom, LifeScan, Menarini Diagnostics, Novo Nordisk, Roche, Sanofi, Sensile, and Ypsomed. LH holds share in the Profil Institute for Metabolic Research, Neuss, Germany, and the Profil Institute for Clinical Research, San Diego, USA. He is consultant for a range of companies that develop new diagnostic and therapeutic options for the treatment of diabetes.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Performance of the study and writing of the manuscript were supported by Roche Diabetes Care, Germany.
