Abstract

A Performance-Enhancing Drug
During the recent Winter Olympics, a Russian figure skater tested positive for the heart medication, trimetazidine (TMZ). In February 2022, the Court of Arbitration for Sport ruled that the athlete could nevertheless continue to compete in the 2022 Beijing Olympic Games. The court accepted her claim that her grandfather uses TMZ, and that she inadvertently ingested this drug. 1 Previously, a Chinese swimmer served a three-month ban in 2014 and a Russian bobsledder was disqualified from the 2018 Olympics and received an eight-month ban after each tested positive for TMZ. 2
TMZ was placed on the World Anti-Doping Agency (WADA) banned list in 2014. 3 However, a 2018 review of WADA-prohibited substances identified no well-designed studies of performance enhancement with athletes using TMZ.4,5 The only three studies in the literature related to performance with the use of TMZ (compared with placebo) include two studies demonstrating increasing exercise tolerance in a setting of peripheral artery disease6,7 and one study demonstrating improved psychomotor performance in healthy subjects. 8 Although TMZ has been reported to decrease lipid oxidation and to increase glucose oxidation, this mechanism has not been shown to enhance striated (cardiac and skeletal) muscle performance in athletes during exercise. 9
TMZ is used worldwide for the treatment of angina and congestive heart failure and is manufactured in 53 countries (Table 1). However, the drug has not been approved by the US Food and Drug Administration (FDA). Meanwhile, in the United States, the safety and efficacy of sodium-glucose cotransporter-2 (SGLT2) inhibitors have also not been established in patients with type 1 diabetes (T1D), and the FDA has not approved them for use in these patients. With the increased awareness of the benefits of SGLT2 inhibitors in type 2 diabetes (T2D), especially in patients with established cardiovascular and renal disease, there has been growing interest in their use in patients with T1D. To date, use of SGLT2 inhibitors in T1D patients has been reported to be associated with increased risk of euglycemic diabetic ketoacidosis (DKA). 10 In this article, we explore the question of whether TMZ, which affects both fat and glucose metabolism, and which has already been shown to improve glycemia in T2D patients in some11-13 but not all14,15 trials, may have a role to play to prevent euglycemic ketoacidosis in T1D patients treated with SGLT2 inhibitors.
Countries Where Trimetazidine is Manufactured.

Cardiovascular Properties of TMZ
During ischemia, limitations in blood flow and oxygen delivery result in myocardial acidosis that adversely affects contractility. 16 TMZ acts as an exercise mimetic through inhibition of cardiac long-chain 3-ketoacyl coenzyme A (CoA) thiolase (LC 3-KAT) and stimulation of pyruvate dehydrogenase, an enzyme involved in regulation of glucose oxidation, that together increase glucose and glycogen oxidation and decrease fatty acid oxidation. 17 These two effects together appear to improve angina symptoms 18 (Figure 1).
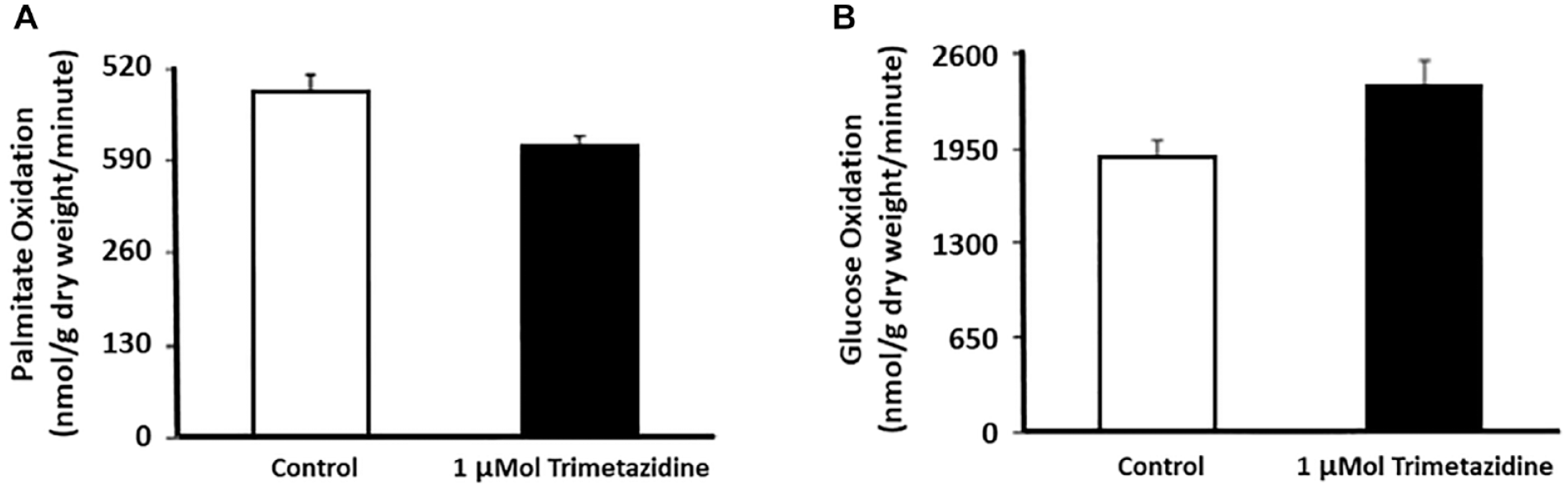
Effects of trimetazidine in perfused rat hearts on metabolic flux through (a) fatty acid oxidation and (b) glucose oxidation. In both cases P < .05.
Compared with glucose oxidation, the oxidation of fatty acids produces more adenosine triphosphate (ATP) per gram; however, in terms of ATP per oxygen molecule, glucose and lactate yield more energy than ketone bodies or fatty acids. For example, the oxidation of fatty acids, compared with glucose, requires 11% to 12% more oxygen for a given amount of ATP produced. 19 In a state of oxygen deficiency, such as myocardial ischemia or perhaps in a state of extreme athletic activity, a switch from metabolism of fatty acids to glucose and lactate 20 is more oxygen-effective and can balance the mismatch between oxygen requirements and oxygen supply. The switch in substrate preference can theoretically protect an ischemic heart.
The effect on cardiac function is different for TMZ compared with other antianginal drugs, which reduce demand for oxygen and also have a cardiodepressive effect. TMZ has an anti-ischemic effect without associated depression of heart rate, blood pressure, coronary artery blood flow, or left ventricular contractility. This effect occurs without increased cardiac perfusion (Figure 2).
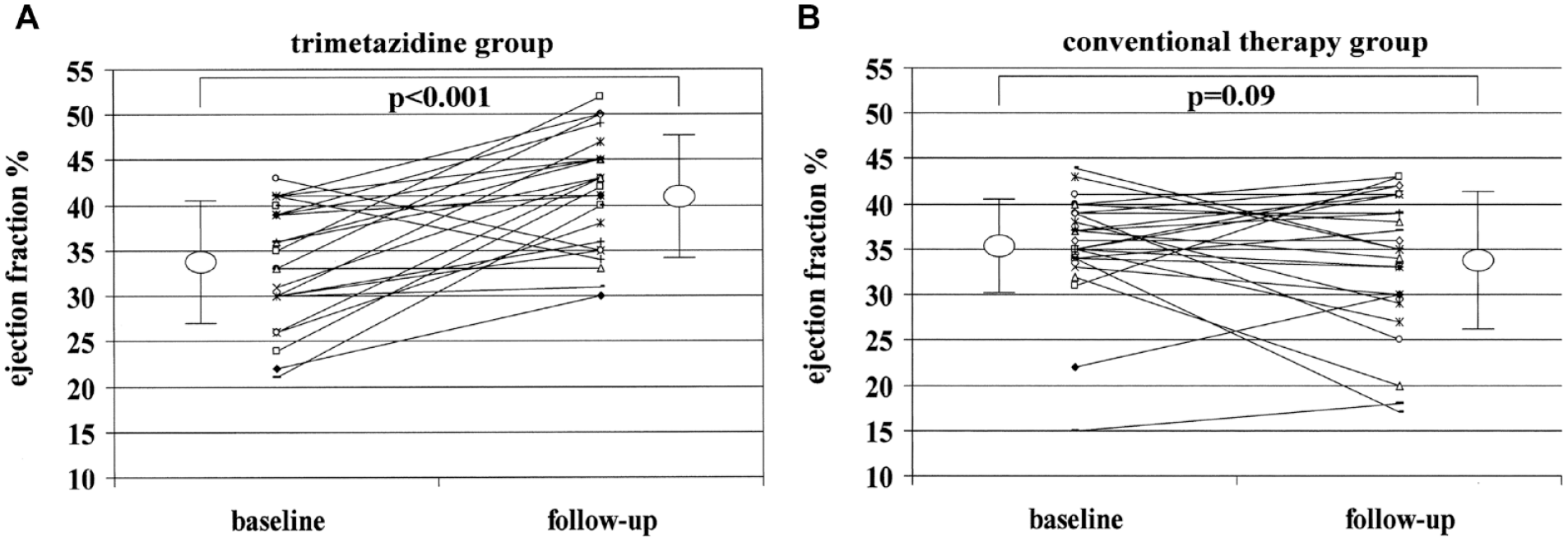
Effects of trimetazidine added to conventional therapy (a) compared with conventional therapy alone; (b) on left ventricular ejection fraction (LVEF), at baseline, and at follow-up. Trimetazidine significantly improved LVEF, whereas a trend toward a decline in left ventricular function was observed in patients treated with conventional therapy alone.
Use of SGLT2 Inhibitors in Addition to Insulin Therapy in Type 1 Diabetes
SGLT2 inhibitors are approved by the FDA to improve glycemic control in adults with T2D 22 (Table 2). These drugs may also be useful for heart failure 23 and chronic kidney disease. 24 SGLT2 inhibitors act by blocking SGLT2 transporters in the proximal renal tubules, which are involved in the reabsorption of filtered glucose and sodium. 25 This results in glycosuria and lower blood glucose concentrations. Three mechanisms have been proposed to account for increased ketone levels in the circulation with the use of SGLT2 inhibitors. 26 First, the metabolic response to the decreased availability of circulating glucose due to glycosuria can be similar to that of starvation where there is a shift from carbohydrate (glucose, glycogen, and lactate) metabolism to fat metabolism associated with decreased insulin activity, which leads to increased lipolysis and then increased ketone production. 27 In a similar sense, patients with T1D 28 and T2D 29 who use insulin usually will decrease their dose when they use an SGLT2 inhibitor because glucose is being disposed of via renal excretion, so less exogenous insulin is needed to transfer glucose into cells. Second, along with decreased insulin activity, SGLT2 inhibitor therapy results in increased glucagon activity (although the exact mechanism is not understood), which also stimulates lipolysis with increased formation of ketone bodies. 30 The third is a potential small decrease in ketone excretion, although this has only been reported in an animal model. 31 The first mechanism is probably dominant, resulting in a decreased insulin concentration with increased lipolysis and shifted substrate utilization from glucose to free fatty acids (FFAs), in turn resulting in release of FFAs followed by ketogenesis.27,32
Single-Ingredient SGLT2 Inhibitors Approved by the FDA.

Abbreviation: FDA, Food and Drug Administration.
SGLT2 inhibitor therapy has been proposed as adjuvant therapy in addition to insulin in T1D patients. This combination of two drugs has been associated with improved glycemic control, increased time in range, improved quality of life measures, and weight loss. 33 However, this combination is associated with an increased risk of DKA. For this reason the addition of an SGLT2 inhibitor to insulin in T1D patients has not been approved by the FDA, although this combination of drugs has been approved in Europe for sotagliflozin, a dual sodium-glucose cotransporter-1 (SGLT1) and SGLT2 inhibitor, 34 and dapagliflozin, a SGLT2 inhibitor. 35 The manufacturer of the latter drug withdrew the drug for the indication of T1D from the European market in 2021. 36 The FDA has required Warnings and Precautions to be added to the labels of all SGLT2 inhibitors to describe the risk of DKA. 37 TMZ for heart disease has also been associated with drug-induced parkinsonism. 38
Metabolic Properties of TMZ
In perfused rat hearts, TMZ has been shown to be an inhibitor of FFA oxidation that increases the use of glucose as a fuel 17 (Figure 1). Therefore, one might hypothesize that a blocker of fatty acid oxidation might lead to a decrease in the amount of ketoacids produced through lipid oxidation. In that case, TMZ might be a protective agent from ketoacidosis, where an absolute insulin deficiency is not the main problem, such as euglycemic ketoacidosis in patients with T1D who are using an SGLT2 inhibitor. Could TMZ’s effect of decreased lipid oxidation protect from formation of ketoacids?
A drug that blocks FFA oxidation could have diverse effects on ketoacid formation. Unless lipolysis is blocked so that fatty acid production is limited, suppression of lipid oxidation could result in backup of fatty acid carbon flux and cause ketone formation through a mass action effect. 39 However, most of the research on the effects of TMZ has been on cardiac muscle rather than skeletal muscle. TMZ’s effect on skeletal muscle fatty acid oxidation appears to be extremely mild. 40 If TMZ significantly prevented lipolysis in white adipose tissue and skeletal muscle (which has not been demonstrated), or significantly prevented lipid oxidation in skeletal muscle (which has also not been shown), then there would be a reasonable basis for optimism that TMZ could block formation of ketoacids, which are derived from nonoxidative disposal of fatty acids. However, the effect of TMZ on cardiac muscle is probably not sufficient to block systemic formation of ketoacids during states of insulin deficiency. Likewise, it has not been shown that TMZ therapy will prevent a clinically significant amount of ketone formation. It is probable that without a significant inhibition of skeletal muscle FFA oxidation, TMZ’s intended effect (of decreased FFA oxidation and associated decreased ketone formation) would not substantially decrease total ketone production in humans.
TMZ in Type 1 Diabetes Patients Using Insulin Plus an SGLT2 Inhibitor
If TMZ would decrease FFA oxidation and the development of ketoacidosis, then one could hypothesize that this drug might be an effective treatment to be used by T1D patients who are also receiving an SGLT2 inhibitor off label. In that case, the increased risk of ketoacidosis when SGLT2 inhibitors are added to insulin therapy could perhaps be reduced by adding this metabolic modulator, which would unlock the possibility of safer SGLT2 inhibitor therapy for people with T1D. However, in T1D patients receiving insulin and SGLT2 inhibitor therapy experiencing increased lipolysis and impending ketoacidosis, there is insufficient clinical or basic science evidence to expect that repurposing TMZ would likely prevent ketoacidosis. In conclusion, the mechanism of TMZ is unfavorable for preventing euglycemic ketoacidosis in T1D patients using insulin plus an SGLT2 inhibitor.
Footnotes
Acknowledgements
The authors would like to thank Annamarie Sucher-Jones for her editorial expertise.
Abbreviations
ATP, adenosine triphosphate; CoA, coenzyme A; DKA, diabetic ketoacidosis; FDA, Food and Drug Administration; FFAs, free fatty acids; LVEF, left ventricular ejection fraction; LC 3-KAT, long-chain 3-ketoacyl coenzyme A thiolase; SGLT2, sodium-glucose cotransporter-2; T1D, type 1 diabetes; T2D, type 2 diabetes; TMZ, trimetazidine; US, United States; WADA, World Anti-Doping Agency.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DCK is a consultant to AI Health, Dexcom, Eli Lilly, EOFlow, Integrity, Lifecare, Medtronic, Novo, Roche Diagnostics, Rockley Photonics, and Thirdwayv. NYX is a consultant to Abbott Diabetes Care. KTN is a consultant to Abbott Diabetes Care. DK has received remuneration for participation in Advisory Boards from Sanofi, Novo Nordisk, and Abbott Diabetes Care. CM has nothing relevant to disclose. GEU reports research funds to Emory University from Astra Zeneca and Dexcom. GAB is supported by NIA R01 AG059715.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
