Abstract
Background:
Testing the potential influence of interfering substances on the measurement performance of needle sensors for continuous glucose monitoring (CGM) is a challenging task. For proper function, the sensors need an almost stable fluidic environment. Previously published in vitro interference experiments were measuring under static concentration conditons. Our experimental setup allows for interference testing with dynamic changes of the interferent concentrations.
Methods:
We designed a macrofluidic test stand that is fueled by several high-pressure liquid chromatography (HPLC) pumps generating programmable glucose and/or interferent gradients in phosphate-buffered saline (PBS). After optimizing experimental parameters (channel dimensions, temperature, flow rates, gradient slopes, buffer, pH etc.), we validated the setup using Dexcom G6 (G6) and Freestyle Libre 2 (L2) sensors with/without interferents, and using YSI 2300 Stat plus as the reference glucose device at room temperature.
Results:
Both sensors tracked the programmed glucose changes. After calibration, G6 results closely matched glucose reference readings, while L2 routinely showed ~50% to 60% lower readings, most likely because of the factory-based calibration and temperature compensation. Gradients of maltose, acetaminophen, and xylose were employed to further validate the setup. As expected, both sensors were not affected by maltose. We confirmed previous findings regarding susceptibility of G6 readings to acetaminophen and L2 readings to xylose. Signals from both sensors are influenced by temperature in a linear fashion.
Conclusions:
Our experimental in vitro setup and protocol may provide a useful method to dynamically test CGM sensors for interfering substances. This may help to improve the accuracy of future CGM sensor generations.
Keywords
Introduction
Sensors for continuous glucose monitoring (CGM) in the interstitial fluid have improved over the past decades CGM devices have reached a glucose reading quality leading to regulatory approval for the determination of insulin doses based on their results. However, no CGM device fulfills the ISO standards for blood glucose measurement yet.1,2 Still clinically unexplainable deviations between sensor readings and blood glucose concentrations have been observed on a regular basis, where both factory-calibrated and user-calibrated sensor technologies show high and sometimes clinically unacceptable differences to the blood glucose concentrations, which cannot be explained by the well-characterized glucose dynamics between the interstitial fluid and the blood.3-7 One explanation are concentration changes of substances in the interstitial fluid, which interfere with the glucose measurement technology of the CGM
Similar to blood glucose, test strips using the same measurement technology, sensors for CGM can be subject to interference by other molecules. Well-known examples are the impact of vitamin C and the influence of the frequently used pain drug acetaminophen (paracetamol) on some of the existing and commercially available glucose oxidase-based needle sensors.8-15
At this stage, the only effective way to investigate the relevance of such interference on a CGM sensor are clinical studies in animals or humans, where a potentially interfering substance is ingested in clinically approved amounts or doses. Consecutive comparison of blood glucose and sensor glucose readings under consideration of the pharmacokinetic profile of the ingested substance are required to understand the nature and size of the interference. However, only few clinical studies about CGM interference have been reported in the literature so far.8-11 At the same time, the manufacturers and the users need to understand the relevance of a potential interferent to draw correct conclusions about clinical or regulatory consequences (eg, warnings in the instructions for use). 16
In addition, there is no information existing in the literature at all about a potential interaction of dynamic changes of several potentially interfering substances when taken in parallel on CGM performance. However, this is clinical reality, for example, when patients take multiple drugs in the morning prior to breakfast and sometimes in conjunction with multiple nutritional supplements. In Germany, the Freestyle Libre sensor is very popular and frequently prescribed for patients with type-2 diabetes suffering also from coronary artery disease, atrial fibrillation, hypertension, dyslipidemia, heart failure, and many other diseases in parallel. A significant proportion of these patients take up to 10 and more drugs, for example, prior to breakfast. According to our knowledge, nothing is known about potential potentiating, additive, or subtractive effects of individual components of such drug and nutritional supplement “cocktails” on the measurement performance of CGM sensors.
It is clearly impossible to run clinical trials in people with diabetes to test for each and every possible interferent and cocktail composition. This would be extremely time and resource consuming. Having this as a regulatory requirement would even prevent companies to pursue or enter into future continuous glucose sensor developments. At the same time, long-lasting and economically affordable CGM systems still represent a major unmet medical need for people with diabetes.
In this project, we have tried to design and realize an in- vitro test setup and suitable protocol to be able to address the interference issues raised above under consideration of the specific conditions required for dynamic interference testing of CGM needle sensors in the laboratory. Our plan was to realize a test bench setup, where (1) several CGM sensors can be tested in parallel, (2) different glucose concentrations and gradients can be investigated, (3) dynamic concentration changes of individual substances can be superimposed on a selected glucose concentration, and (4) different substances can be combined for dynamic “cocktail” testing.
Materials and Methods
Setup of the In Vitro Bench Test
After multiple experimental attempts, we managed to design a sensor test bench, consisting of a 3D-printed solid PPE block (15 cm × 15 cm × 4 cm) as the sensor housing component. On one side, a macrofluidic channel was left out from the solid material as shown in Figure 1 (2 mm × 10 mm × 500 mm). The CGM sensors were located on top of the channel and the inner spaces between the sensor needles were carefully filled with chemically inert cotton wool to block any fluidic turbulences around the sensor tips during the measurements. Tubing connections were also 3D-printed on both sides of the channel to allow for fixation of high-pressure liquid chromatography (HPLC) pump tube connectors on one side, and tubing leading to a waste container on the other side. High-pressure liquid chromatography pumps were used to supply the test bench channel with buffer, variable glucose concentrations and different concentrations of candidate substances for interference (Waters 2695, Waters, Eschborn, Germany). A design schematic is provided in Figure 1.
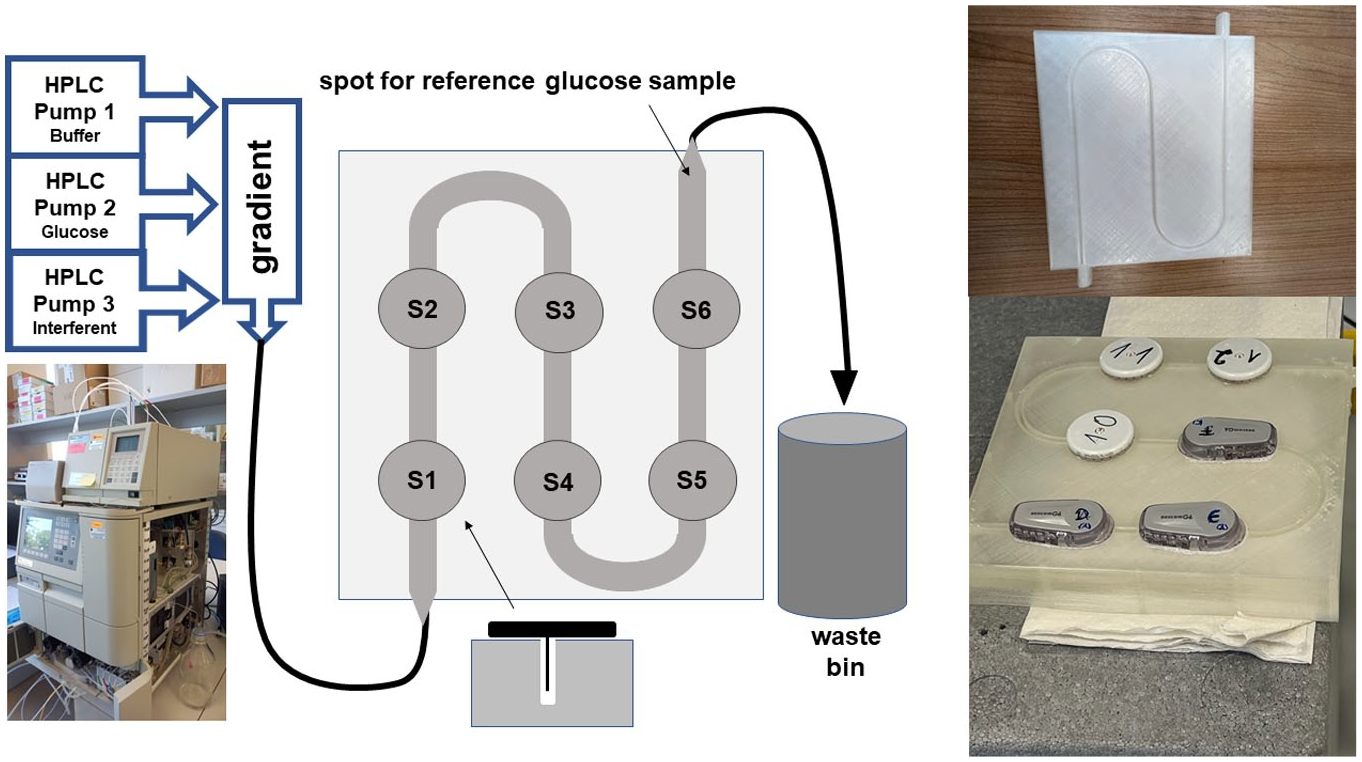
Sketch of the design of the laboratory test stand for dynamic interference testing of continuous glucose monitoring needle sensors.
General Test Protocol
We tested several buffers and finally decided to use a phosphate-buffered saline (PBS) buffer (Carl Roth, Karlsruhe, Germany) in our general experimental protocol (18 g NaCl, 0.20 g KCl, 0.20 g KH2PO4, 1.15 g Na2HPO4×2 H2O in 1000 ml H2O, pH 7.2). The sensors proved to be sensitive to the overall speed of the pumping operations and also to the slope of the glucose or interferent gradients. Finally, we identified an overall pumping speed of maximally 1 ml/min and substance increases of less than 4%/min of the planned maximal concentration as suitable flow conditions. Glucose, xylose and maltose were obtained from Carl Roth Sigma Aldrich (Hamburg, Germany) and acetaminophen from Sigma Aldrich, Darmstadt, Germany).
In this setup, we tested the Dexcom G6 CGM sensor (G6; Dexcom, Mainz, Germany) and the Freestyle Libre 2 sensor (L2; Abbott, Wiesbaden, Germany). Three sensors from each CGM system were placed on the test bench as shown in Figure 1 and were tested in parallel. Initiation of device operation was performed in-line with manufacturer’s instructions, and each was connected to an individual read-out device. The G6 sensors were calibrated as provided in the instructions for use at an initially stable glucose level of 120 mg/dL against the reference method Yellow Springs Instruments (YSI Stat 2300 plus, Yellow Springs Inc., Silver Springs City, US), while the results from the factory-calibrated L2 sensors were recorded as displayed on the Libre readers, and G6 on the respective Dexcom readers. Each experiment was done with 3 sensors per device type in parallel. During all experiments, samples were taken at the outflow tube of the channel at least every 10 min for a YSI reference reading.
Substance Interference Test Protocol
Each individual experiment was conducted at least twice and the final individual experimental settings are shown together with the results in the result section. We first ran a glucose gradient experiment, starting with a glucose level of 100 mg/dL for 30 min, increasing to 300 mg/dL over 100 min (2 mg/dL/min), maintained at 300 mg/dL for 30 min, prior to returning to 100 mg over 100 min (−2 mg/dL/min), then maintaining at 100 mg/dL for another 30 min. In a second experiment, we chose a stable glucose value of 200 mg/dL (to ensure suitable L2 signals in case an interferent would lead to a reduced signal strength). We superimposed an acetaminophen gradient from 0 to 20 mg/dL within 30 min, and maintained this level for 30 min. Thereafter, the levels were reduced back to 0 mg/dL over the next 30 min and were finally maintained at 0 mg/dL for final 30 min. Similar experiments were run with xylose (up to 200 mg/dL) and maltose (up to 600 mg/dL). The supraphysiological interferent concentrations were chosen in these proof-of-concept experiments to surely induce signal interference if it existed.
Temperature Interference Test Protocol
For this experiment, a glucose concentration of 200 mg/dL was programmed, and the system ran for 1 h at room temperature (20°C). The test platform was placed into a warming chamber and the temperature was increased to 30°C within 30 min, kept stable at 30°C for 30 min, increased to 37°C within 30 min, kept stable at 37°C for 30 min, increased to 40°C within 30 min and kept stable for 30 min at 40°C. Thereafter, temperature was dropped again to room temperature within 30 min and kept there for another 60 min.
Statistical Analysis
A proper calibration of G6 allows to define substance-specific interference, if differences of >20% occur between the reference glucose readings and the sensor readings at the maximal interferent concentration. The same is not possible for the factory.calibrated L2 sensor as the signal deviates from the reference readings most likely due to the in vitro nature of our experiments. We therefore determined the mean absolute relative deviation from the glucose signal measured at baseline with no present interferent at the timepoint of maximal interferent concentration as a benchmark parameter (BOB = bias over baseline). Interference was asumed if BOB was >20%.
Results
The results of testing dynamic glucose gradients with our bench test are provided in Figure 2. The flow rate of 1 mL/min was determined to be a suitable experimental flowrate. Faster flow results in frequent signal errors from the sensors. Slower flow leads to unpractical signal delays between sensors sitting at the later positions in the test channel and makes comparisons more difficult.
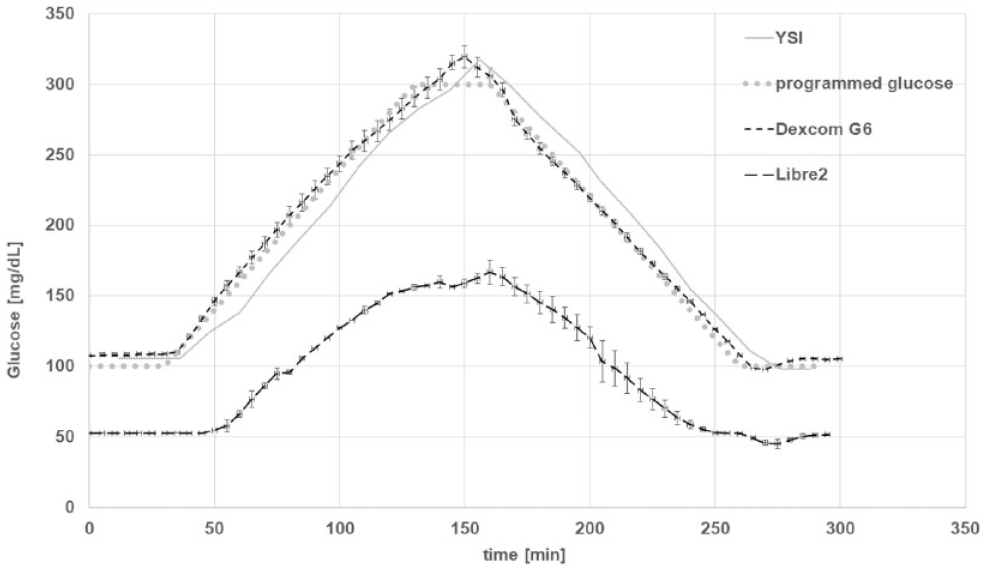
Glucose gradient experiment (YSI: Yellow Springs Instruments Stat 2300 plus, glucose reference method, n = 3 for each sensor type).
It can be seen that the calibrated G6 sensors closely followed the programmed glucose gradients and the reference glucose readings. The factory calibrated L2 sensors had a corresponding and parallel signal pattern, but the displayed results were consistently about 50% to 60% lower than the reference readings. This difference could not be resolved, for example, by an individual sensor calibration of the L2 sensors, and it was hence observed throughout all performed experiments.
When running interference experiments at stable glucose levels and at room temperature, the L2 sensor signals showed minor signal variations when substance concentrations dynamically increased and decreased. This repetitive signal pattern was observed for all substances irrespective of their chemical nature and was interpreted as an experimental setup-related sensor-specific measurement artifact. These signal variations were considered to be either a temperature artifact, or induced by local fluidic disturbances at the sensor tip or a result of corrective actions of the sensor software algorithm to compensate for local signal disturbances at the sensor tip, or as a combined effect of all of the above.
Testing Interefering Substances
The results of the maltose interference experiment with maltose concentrations between 0 mg/dL and 480 mg/dL are shown in Figure 3.
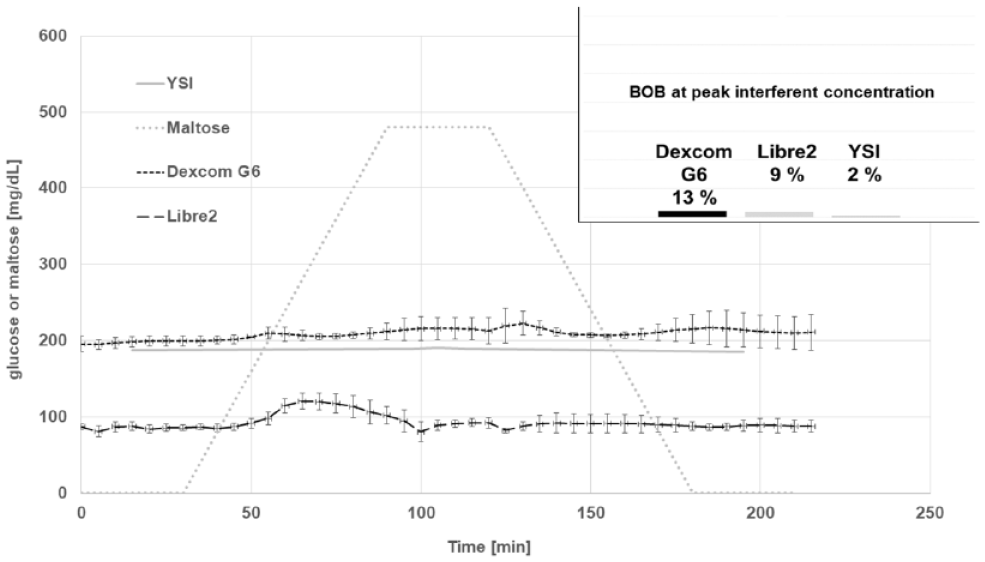
Sensor response to a maltose gradient (0-480 mg/dL) at stable glucose concentrations of 200 mg/dL at room temperature. The Dexcom G6 sensor was calibrated to the baseline glucose level prior to the experiment, while the factory calibrated Libre 2 sensor does not provide this option and displayed 50% to 60% lower results (BOB = bias over baseline, n = 3 for each sensor type).
It can be seen that with exception of minor L2 signal differences induced by increasing interferent concentrations both sensors showed a stable signal, which was not affected by the maltose gradient.
The results of the interference experiments with xylose (0 mg/dL to 600 mg/dL) are provided in Figure 4. There was no impact of increasing or decreasing xylose concentrations up to 600 mg/dL on the G6 sensor signals (BOB: 13%). The L2 sensor was substantially impacted by increasing xylose concentrations in an additive manner. Bias over baseline at peak xylose concentrations was 308%.
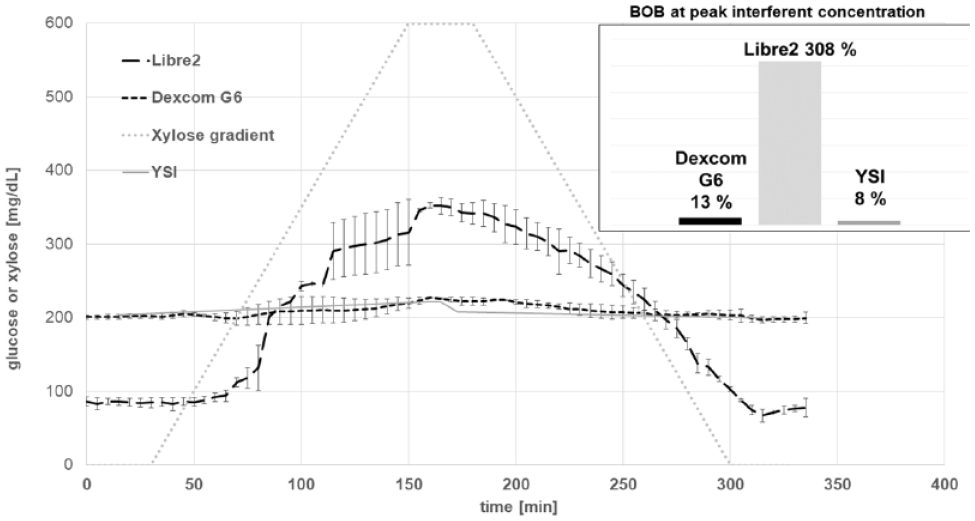
Sensor response to a xylose gradient (0-600 mg/dL) at stable glucose concentrations of 200 mg/dL at room temperature. The Dexcom G6 sensor was calibrated to the baseline glucose level prior to the experiment, while the factory calibrated Libre 2 sensor does not provide this option and displayed 50% to 60 % lower results (BOB = bias over baseline, n = 3 for each sensor type).
The results of the interference experiments with acetaminophen (0 mg/dL to 20 mg/dL) are provided in Figure 5. While L2 showed no interference (BOB: 0%), G6 showed significant interference starting directly with increasing acetaminophen concentrations. Bias over baseline at peak acetaminophen concentration was 104%.
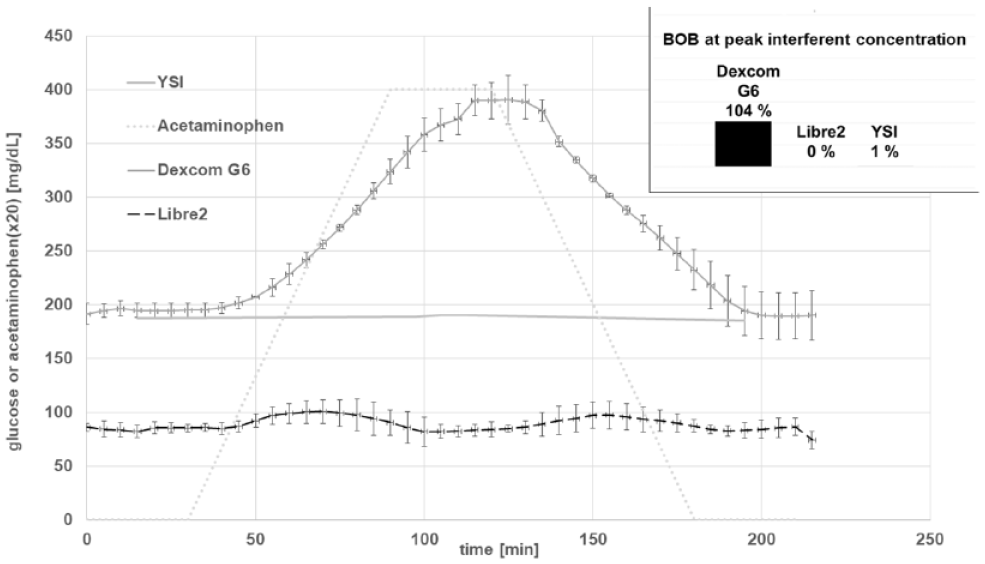
Sensor response to an acetaminophen gradient (0-20 mg/dL) at stable glucose concentrations of 200 mg/dL at room temperature. The Dexcom G6 sensor was calibrated to the baseline glucose level prior to the experiment, while the factory calibrated Libre 2 sensor does not provide this option and displayed 50-60 % lower results (BOB = bias over baseline, n = 3 for each sensor type).
Except for the observed minor variations with increasing and decreasing acetaminophen levels, L2 was unaffected by this molecule, and there was no measurable difference at peak acetaminophen level (BOB: 0%). The reference readings with the YSI 2300 Stat plus device closely followed the programmed glucose concentrations in all experiments (BOB: 1%).
The temperature experiment revealed a major influence of the ambient temperature as shown in Figure 6. From the results, it was calculated that the mean signal increase per 1°C was similar for both devices (2.3%/°C for L 2 and 2.4 %/°C for G6).
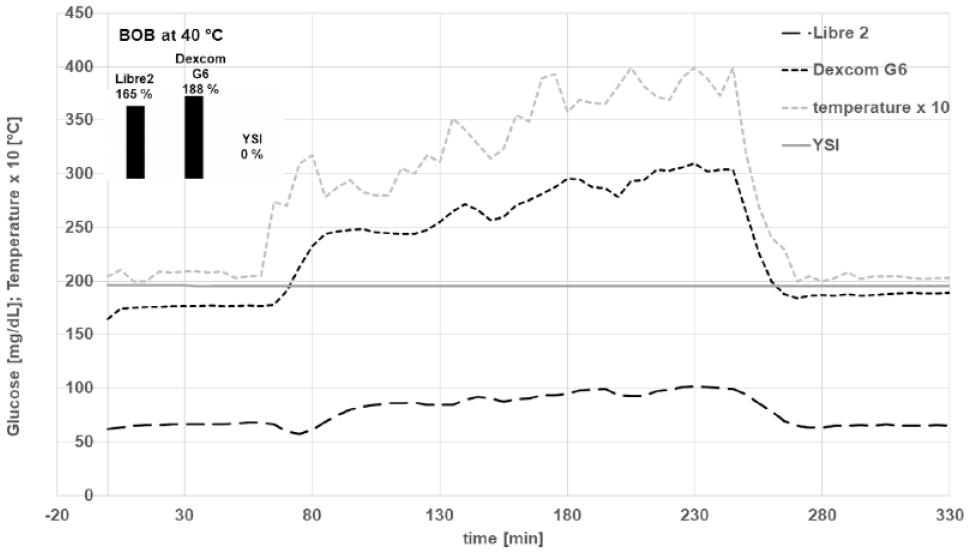
Impact of ambient temperature on the sensor signals in our in vitro test setup (n = 3 for each sensor type).
In additional experiments conducted with acetaminophen, maltose, and xylose at 37°C, it was observed that the influence of temperature on the sensor signals is linear. A G6 sensor properly calibrated at 37°C showed the same curves as compared to calibration and running the experiments at 23°C. As a result, BOB was similar for the same substances at the different temperature levels (data not shown).
Discussion
The susceptibility of CGM devices to erroneous readings in the presence of interferences by common pharmaceuticals or nutritional supplements is an open and at least partially unclear issue in diabetes technology. 16 Only few reports exist in the current literature about such issues. We believe that it is fair to state that there is a general lack of experimental know-how and understanding in the scientific community about the required testing conditions, which as of today prevents comprehensive standardized testing and data generation to fully explore the CGM interference situation.
The most valid but also most resource-consuming approach is to assess CGM error to pharmacologic interferences by means of clinical trials with oral administration of the candidate interference substance. Basu et al reported a clinical trial protocol in 2017, with the aim to examine the responses of several different Food and Drug Administration–approved and commercially available CGM systems (Dexcom Seven Plus, Medtronic Guardian, Dexcom G4 Platinum) to oral acetaminophen administration in healthy volunteers without diabetes. 9 Blood and microdialysate ISF samples were collected periodically and analyzed for glucose and acetaminophen concentrations before and after oral ingestion of 1g of acetaminophen. They observed interference with glucose measurements in the tested CGM devices that coincided temporally with appearance of acetaminophen in the ISF.
In a follow-up report, the same group used a similar protocol with 19 healthy drug-naïve subjects without diabetes to compare CGM glucose patterns to actual plasma glucose concentrations after uptake of several drugs. They showed that lisinopril, albuterol, and acetaminophen, and also red wine appear to interfere with commonly used CGM devices. 10
At the same time, the manufacturers tried to solve known interference issues and in 2018, Calhoun and coworkers reported a next generation CGM device (G6), where acetaminophen interference appeared to be solved by use of a novel permselective membrane coating for the needle sensor. In their study with 66 subjects with type 1 or type 2 diabetes, the observed mean difference of glucose levels induced by a single dose of 1 g of acetaminophen was found to be only 3.1 ± 4.8 mg/dL (with an impact of 10 mg/dL defined as clinically relevant interference). 17
In a complex experimental in vitro approach, 7 sugars, 7 sugar alcohols, and 3 artificial sweeteners were in vitro screened for interference with amperometric glucose oxidase (GOx) sensors at concentrations greater than physiologic concentrations, by Boehm and coworkers in 2019. 18 Fourteen of the 17 tested substances did not exhibit any interference. Galactose, xylose and mannose, affected sensor signals with MARDs > 20% but in concentrations significantly higher than normal physiologic concentrations. Galactose exhibited a concentration-dependent MARD of 47-72% and was subjected to further testing. In this setup, the highest recorded mean relative difference (MRD) was 6.9 ± 1.3% when testing physiologically relevant galactose concentrations (0.1-10 mg/dL). Enzyme kinetic analysis conducted with galactose supported the notion that the reactivity of glucose-oxidase toward nonglucose sugars and the presence of enzymatic impurities (such as galactose oxidase) are 2 potential sources for sugar interference with glucose oxidase-based CGM- sensors.
The laboratory in vitro setup chosen by Boehm et al required a static stable interferent concentration prior to obtaining a read-out from the investigated needle sensors. Unfortunately, the authors did not disclose the names of the commercially available sensors investigated in their study. In addition, their experimental setup did not work with dynamic interferent concentration changes. Hence their results are of limited value for a real-life dynamic in-vivo situation. Still, it provides valuable information about a potential interference impact of galactose on glucose oxidase-based CGM devices. 18
Dynamic measurement of interferences provides an additional degree of interference information in comparison to static measurements. A single static measurement only provides qualitative interference information and only for the interferent concentration tested. Dynamic testing allows for determination of the necessary interferent concentration to meet a predefined interference criteria (eg, a 20% difference) independent from the highest interferent concentration tested. In addition, in future experiments, we will be able to determine if (and to what extent) prior presence of an interfering substance may continue to influence sensor performance even when the interferent has disappeared from the testing site.
In our in vitro setup, the maximal acetaminophen concentration tested was 10 to 20 times higher than the maximal ISF concentration measured by Basu et al after application of 1 g of the drug. 9 This may explain the difference between our findings and the work of Calhoun et al. 17 Still, Denham D reported increasing MARD values for the G6 sensors with multiple consecutive uptakes of doses of 1 g of acetaminophen every 4 hours. 11 At this initial protocol development stage, our approach was to explore the overall suitability of the designed in vitro setup to provide valid interference results rather than simulating daily clinical circumstances. This will be the topic of future studies.
One limitation of our methodology is a lack of valid information regarding the normal ranges and expected concentrations of potential interferents in the ISF after oral, subcutaneous or intravenous administration. Further basic research work will be necessary to close this information gap and to allow for determination of realistic maximal testing concentrations for interference assessment of CGM sensors. Until this information becomes available, we suggest to employing the corresponding plasma concentrations or interferent levels suggested by the FDA guidance to industry for blood glucose meters for patient self-testing. 19 Whenever possible, the physiological and transfer factors that may arise due to differences between the blood and ISF compartments should be considered.
Another—experimental—limitation of our test bench is the fact that the sensors are connected in series. This results in a time offset in the change of the concentrations, which can be seen very clearly in the figures. In the chosen series, the 3 G6 sensors react first, followed by the 3 L2 sensors, and the reference measurement at the very end. The time delay depends on the volume of the flow channel and the flow rate. This limitation can be easily overcome in future experiments, if the sensors were arranged in parallel or in randomized order. However, this limitation does not affect the suitability of the system to detect interferences.
We were able to show a direct linear influence of ambient temperature on sensor signals. We had initially conducted our presented proof-of-concept dynamic interference experiments at room temperature, and repetition of the experiments at 37°C resulted in similar signal patterns (and even similar curves for G6 after calibration at 37°C). While we consider it feasible to investigate substance interference at both temperatures, we have decided to choose 37°C for our standard protocol for future work to stay closer to the physiological operational sensor conditions in the body.
By adding more HPLC infusion pumps to our setup, we expect to be able to simulate the physiologic situation of parallel increases of multiple substances with interference potential, as can be frequently observed in daily practice in multimorbid patients, for example, at breakfast and dinner. No information exists in the literature regarding additive, subtractive, or potentiating effects of several interferents occuring in the ISF at the same time as it may occur after uptake of several drugs and nutritional supplements in the context of food uptake. Such “cocktail” assessment options represent a new opportunity for understanding interference of CGM sensors (and also for disposable single-use blood glucose test strips), which has not been explored so far.
At this stage, our experimental in vitro setup for interference testing of CGM sensors was able to confirm the known impact of acetaminophen at higher concentrations on the Dexcom G6 sensor signal and of xylose on the Libre 2 sensor signal. We also confirmed that maltose does not exhibit interference on the signals of both sensors. We therefore believe that our experimental protocol may be a suitable and low resource-consuming method to further investigate the impact of potentially interfering substances on CGM sensor device performance in an in vitro setup. It may provide an option for large scale in vitro substance screening to identify potential clinically relevant interfering substances (single or cocktails) and in particular also to rule out non-interfering molecules. This will allow focus on suspect interference candidates, when assessing the clinical relevance of observed in vitro interferents in subsequent resource-consuming clinical trials.
Footnotes
Abbreviations
BOB, %bias over baseline; CGM, continuous glucose monitoring; FDA, Food and Drug Agency; GOx, glucose oxidase; HPLC, high pressure liquid chromatography; ISF, interstitial fluid; ISO, International Standardization Organization; MARD, mean absolute relative difference; MRD, mean relative difference; PBS, phosphate buffered saline, PPE, Polyphenylene Ether; YSI, Yellow Springs Instruments.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: In the context of this manuscript, APF declares that he is a consultant or has receive travel support and/or speaker fees from Sanofi/Germany, Lifescan/USA, Lifecare/Norway, and Abbott/Wiesbaden. The other authors have no conflict of interest to report.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has received funding from the European Union’s Horizon 2020 research and innovation program under grant agreement No 951933 (ForgetDiabetes) and also from Lifescan Global Corporation, Malvern, PA, USA.
