Abstract
Background:
While continuous glucose monitoring (CGM) systems allow precise and real-time blood glucose control, current electrochemicalbased CGM technologies inherently harbor enzyme instability issues. The direct electron transfer (DET) type open circuit potential (OCP) based enzyme sensing principle can minimize the catalytic turnover of the enzyme reaction, thereby providing longer-term operational stability in future CGM glucose sensors.
Method:
DET-type OCP based glucose sensors were constructed using gold disk electrodes with glucose dehydrogenase capable of DET which was immobilized using a self-assembled monolayer (SAM). The single enzyme layer prepared on the gold electrode was operated in the presence of glucose, using in vitro buffer solution, continuously for over 3 months with the OCP sensor signal monitored every 10 seconds at 25°C.
Results:
The DET-type OCP glucose sensor was continuously operated for more than 3 months without a significant decrease of the sensor signal and sensitivity (slope). These results suggest that the DET-type OCP glucose sensor is far more stable than the sensor constructed based on the amperometric principle. The long-term stability of DET-type OCP glucose sensor is attributed to the enzyme’s minimized catalytic reaction during the operation, thereby extending the lifetime of enzyme.
Conclusion:
The DET-type OCP glucose sensor can be continuously operated for more than 3 months at 25 °C, in vitro without significant decreases in sensor signal and sensitivity. While the further investigation will be required for in vivo validation, the DET-type OCP glucose sensor is ideal for next generation CGM’s, especially in long duration implantable use cases.
Keywords
Introduction
The continuous glucose monitoring system (CGM) is the gold standard technology for precise and real-time blood glucose control. The continuous monitoring of patient glucose levels with CGM sensors in real-time, promotes euglycemia, reducing chronic and acute diabetic complications including death. Although CGMs have been researched and developed for more than 20 years, they still have inherent issues regarding the accuracy and stability of the sensor, especially for long term use. In fact, current electrochemical glucose sensor based CGM users are still required to replace the implanted sensor every 1 to 2 weeks due to the degradation of the sensor’s accuracy and stability. These limitations make patients less motivated toward diabetes management, increasing the risk of poor outcomes. Fortunately, the optical based CGM sensor with abiotic glucose sensing polymer became available and is approved for use of up to 180 days in the United States and in Europe. However, current CGM technologies still have challenging technical limitations to improve the accuracy and stability of their sensors. 1 The electrochemical based CGM’s inherently harbor stability issues relating to the nature of enzymes used. In addition to this, sensors employing an amperometric measurement principle with glucose oxidase (GOx) as the glucose sensing principle, produces H2O2 continuously, which will inactivate the enzyme itself. The more catalytic reactions proceeded, the more oxygen radical specifies produced which can inactivate the enzyme or synthetic electron acceptors (mediators). Furthermore, using amperometric detection requires a potential application (usually between 0.2 and 0.5 V) which may cause oxidation of interfering substances directly on the electrode surface and can cause signal bias. To overcome these limitations of CGM, additional modifications have been made in the sensor layer; for example, using a glucose limiting membrane to create diffusion limited regime and/or with the exceeded amount of glucose sensing enzyme. 1 However, technology improvements are still needed to overcome inherent limitations seen in all the sensors currently on the market.
We have reported a third generation glucose sensor employing direct electron transfer (DET) type FAD dependent glucose dehydrogenase (FADGDH).2-6 This enzyme consists of three subunits; a catalytic subunit, an electron transfer subunit and a small subunit which is a hitchhiker protein. The generated electron by oxidizing glucose is transferred from the catalytic subunit to the electron transfer subunit through intramolecular electron transfer.7,8 Consequently, the electron transfer subunit allows the transfer of electrons generated from glucose oxidation directly to the electrode via the electron transfer subunit without requiring oxygen or a redox mediator to facilitate electron transfer to the electrode. A CGM sensor employing DET-type FADGDH has various advantages compared to the predecessors. First, sufficient electrochemical signals can be generated by the enzyme itself without the use of any artificial redox mediator or oxygen, reducing the number of essential reactions for glucose detection. Second, electrochemical signals can be generated at a lower bias potential. The potential applied over the sensor depends on the reduction/oxidation potential of either hydrogen peroxide, or the artificial electron mediator. Traditional methods using hydrogen peroxide, or an artificial redox mediator require elevated potentials leading to electroactive interference. On the other hand, the redox potential of DET-type FADGDH is far lower than the oxidation potential of an artificial mediator or hydrogen peroxide. The lower redox potential will reduce the oxidation of interfering substances at the electrode, and thus simplify electrode surface treatments required to reduce interference. Since the sensor fabrication steps and surface chemistry can be greatly simplified by employing DET-type FADGDH, the accuracy and stability of the CGM sensor employing DET-type FADGDH are also expected to be improved, as oxygen fluctuation and high bias potential no longer interact with glucose detection. However, there remains the issue of the continuous bias potential which may inactivate the sensor.
To overcome the technological limitations of current CGM systems and achieve more sustainable diabetes management, we recently reported an open circuit potential (OCP) based third generation glucose sensor employing DET-type FADGDH. 9 While CGM detection principles require continuous potential application, leading to degradation of sensor accuracy and stability, OCP measures the potential change and thereby does not require continuous bias potential application during its operation. During OCP operation, negligible current flows across the anode and cathode, minimizing the enzyme turnover and reducing the probability of enzyme degradation and extending the functional lifetime. In other words, the issues arising from continuous bias potential application can be solved with the OCP principle and ultimately improves the accuracy and stability of CGM sensors. In our previous work, combining DET-type FADGDH with OCP principle, glucose could be detected without any artificial mediator or oxygen. Furthermore, the sensor signal could be obtained without being affected by ascorbic acid and acetaminophen, which are representative electroactive interferences of the glucose sensor. Also, this sensor could be operated stably for 7 days continuously without any significant change in the sensor signal.
Therefore, in this study, we investigated the long-term continuous operation stability of DET-type glucose sensor. The enzyme was immobilized using self-assembled monolayer (SAM); therefore, a single monolayer of the enzyme was utilized on the surface of the electrode. 10 The DET-type OCP glucose sensor can be continuously operated for more than 3 months without any significant decrease of the sensor signal and sensitivity and will be the ideal next generation sensor for CGM system, especially suitable for the future chronic implantable CGM systems, heralding the next CGM sensor.
Method
Chemicals and Materials
In this study, the bacterial FAD-dependent glucose dehydrogenase complex (DET-type FADGDH) was used. The DET-type FADGDH comprises 3 subunits: the catalytic subunit, heme c containing electron transfer subunit, and small subunit. A recombinant DET-type FADGDH was prepared using the expression vectors pTrc99A containing the structural gene for the DET-type FADGDH and pAYCYC184 containing the structural genes for heme c maturation (pEC86). The vectors were transformed into Escherichia coli strain BL21 (DE3) and cultivated as previously described. 6 Dithiobis(succinimidyl hexanoate) (DSH) was purchased from Dojindo Molecular Technologies, Inc. (Kumamoto, Japan). Gold disk electrode, platinum (Pt) wire and a silver/silver chloride (Ag/AgCl) reference electrode were purchased from BAS Inc. (Tokyo, Japan). Acetone and D (+)-Glucose were purchased from Sigma-Aldrich (St. Louis, MO, USA). Micro cloth polishing pads (brown in color) and 0.05-micron micro polish powder were purchased from CH Instruments Inc. (Austin, TX, USA)
Preparation of DET-Type FADGDH Immobilized Glucose Sensor With Self-Assemble Monolayer (SAM)
Gold disk electrode (electrode surface area: 7 mm2) was polished using a 0.05-micron micro polish power on microcloth polishing pads, and then the polished electrode was sonicated in ultra-pure water for 5 minutes to remove alumina particles from electrode surface. Next, the electrode was soaked in 50 mM KOH and 25% H2O2 solution for 10 minutes and the electrode washed with ultra-pure water. After that, the electrode, Ag/AgCl and Pt wire were placed in 50 mM KOH solution as a working electrode, reference electrode and counter electrode, respectively, and the potential was swept for 1 cycle, Scan range: 0.2 V ~ -1.2 V (vs. Ag/AgCl), Scan rate: 50 mV/s. 11 The cleaned electrode was washed with acetone and incubated in 200 μl of 10 μM DSH solution overnight at 25°C, as was described previously 10 to modify SAM on the gold surface. The SAM modified electrode was washed with acetone again, and then the electrode was incubated in 200 μl of enzyme solution, including 0.014 mg/ml of DET-type FADGDH solution overnight at 25°C. The prepared DET-type FADGDH immobilized glucose sensor with SAM was stored in 100 mM potassium phosphate buffer (pH 7.0) at 4°C until use.
The Sensor Performance Evaluation of DET-Type FADGDH Immobilized Glucose Sensor With SAM
The prepared DET-type FADGDH immobilized glucose sensor with SAM was placed in 20 ml volume of water jacket cell including 10 ml of 100 mM potassium phosphate buffer (pH 7.0) as a working electrode, with the Ag/AgCl and Pt wire as a reference and counter electrode, respectively. A potentiostat measured and recorded OCP change continuously under several different glucose concentrations (0.1, 1, 3, 5, 10, 15, and 20 mM) at 25°C. To discharge and reset the electrical status of electrode surface before every measurement, 9 the other potentiostat applied +0.1 V (vs. Ag/AgCl) for 10 seconds before OCP data sampling.
The Continuous Operation of DET-Type FADGDH Immobilized Glucose Sensor With SAM
The prepared DET-type FADGDH immobilized glucose sensor with SAM was placed in 10 ml of 100 mM potassium phosphate buffer (pH 7.0) including 20 mM glucose as a working electrode, with the Ag/AgCl and Pt wire as a reference and counter electrode. A potentiostat measured and recorded OCP change continuously in the presence of 20 mM glucose at 25°C and the other potentiostat was programmed to apply +0.1 V (vs. Ag/AgCl) potential to the glucose sensor (working electrode) for 10 seconds with 5 minutes interval, repeatedly. The OCP calibration curve of glucose sensor was investigated under the several different glucose concentrations (0.1, 1, 3, 5, 10, 15, and 20 mM) before starting the continuous operation (t = 0 day), 4, 6, 10, 14, 20, 38, 48, 55, 60, 69, 78, 87, 99, and 108 days after starting the continuous operation at 25°C.
Results
The Sensor Performance Evaluation of DET-Type OCP Glucose Sensor
Figure 1a shows the response of DET-type OCP glucose sensor. The OCP rapidly decreased after glucose addition, and it is revealed the DET-type FADGDH can oxidize glucose and accept an electron changing the surface potential of the electrode. After +0.1 V (vs. Ag/AgCl) potential application, the OCP immediately returned to the OCP values before potential application, demonstrating that the electrical state of DET-type FADGDH electrode can be reset to the same potential by the potential application with the electrons generated by the enzyme reaction changing the surface potential of the electrode again (Figure 1a). Figure 1b shows the calibration curve of ΔOCP, which was calculated as the difference between the OCP value at each glucose concentration and the OCP value in the presence of 0.1 mM glucose, and the curve showed linearity within the range of glucose concentration from 0.1 mM to 20 mM, confirming that the DET-type OCP glucose sensor can detect 0.1 mM to 20 mM glucose concentration. The reproducibility and detailed characterizations of this sensor, such as the interference effect, was previously reported. 9
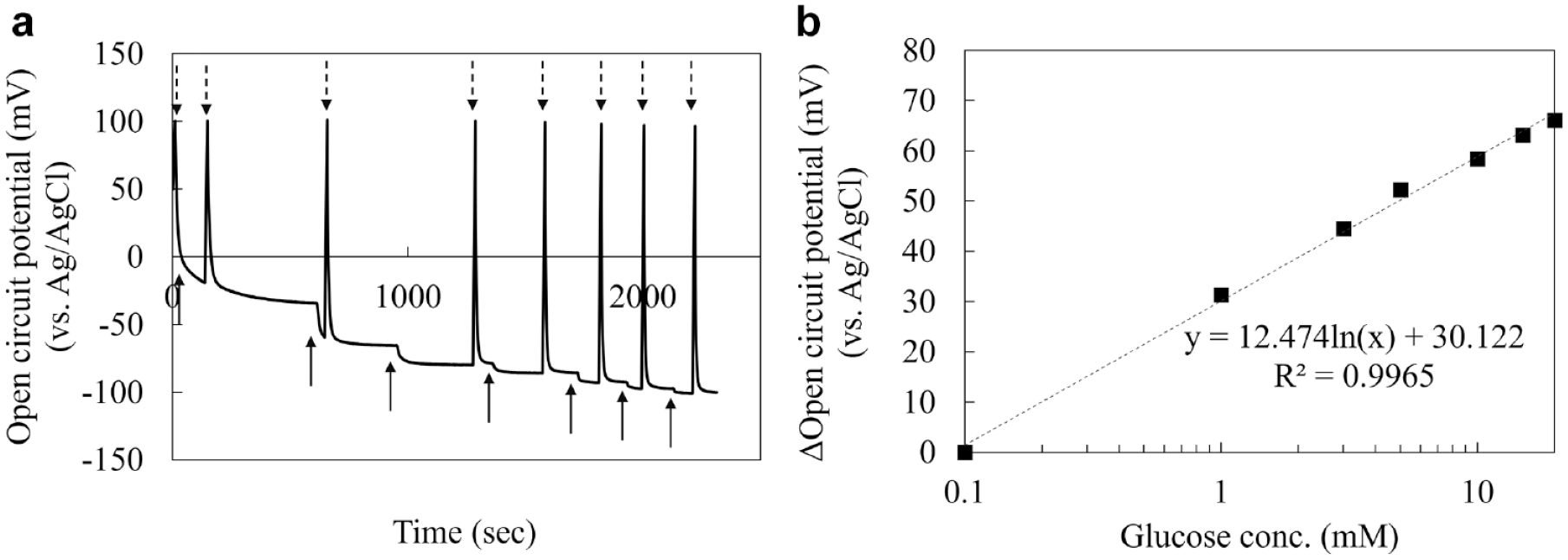
The electrochemical evaluation of DET-type OCP glucose sensor: (a) OCP evaluation of the direct electron transfer FADGDH immobilized electrode with SAM in 100 mM potassium phosphate buffer (pH 7.0), followed by the addition of 0.1, 1, 3, 5, 10, 15, and 20 mM glucose at the solid arrows and + 100 mV (vs. Ag/AgCl) potential application at the dashed arrows and (b) a representative calibration curve of direct electron transfer FADGDH immobilized electrode in 100 mM potassium phosphate buffer (pH 7.0). Before each OCP data sampling, + 100 mV (vs. Ag/AgCl) was applied to the OCP-based glucose sensor for 10 seconds. Abbreviations: DET, direct electron transfer; OCP, open circuit potential; FADGDH, FAD dependent glucose dehydrogenase.
The Continuous Operation of DET-Type OCP Glucose Sensor
We then investigated the in vitro continuous operation of the DET-type OCP sensor for 108 days, with DET-type FADGDH immobilized gold electrode by SAM with single enzyme monolayer. The sensor was placed in the 100 mM potassium phosphate buffer (pH 7.0) including 20 mM glucose at 25℃, and the OCP change was continuously measured and recorded with 5 minutes interval of +0.1 V (vs. Ag/AgCl) potential application (10 seconds) for 108 days. Figure 2 shows the entire time course of OCP which was chose the stabilized OCP signal until right before potential application for 108 days continuous OCP measurement. The OCP increased over the first several days from starting continuous operation (Figure 2), and the calibration curve of the sensor also varied for the first 10 days of continuous operation (Figure 3a). Considering that under the current immobilization status, the sensor required at least 10 days for conditioning, we used the OCP value after 10 days continuous operation as the initial sensor signal value (100%), and thereby normalized OCP sensor signals afterward relative to this (Figure 2). During the 108 days continuous operation, the OCP was able to keep a relatively stable value which is within a range from 85% to 110% of the initial OCP value. The calibration curves also showed very stable slope and great linearity from 14 days to 48 days continuous operation. Between 48 days and 55 days, a relatively large slope change was observed, however the slope became stable and maintained stability until 69 days of continuous operation. Between 69 days and 78 days, although a slightly large slope change was observed again, the slope was stable thereafter, and the sensor still showed glucose response even after 108 days continuous operation without any decrease of the slope. Based on the change of curve slope, we divided entire calibration curves to 4 different groups, and the calibration curves were clearly overlapped in the same group (Figure 3b-3d) except for the electrode conditioning period (Figure 3a). Furthermore, to investigate the change rate of curve slope in these 4 different groups, we calculated the slope on the first day in each group (0 day, 14 days, 55 days, and 78 days) as 100% and normalized all slope values to the percentage. As a result, the rate of change for each slope was within ±10% from the first day in each group except for the electrode conditioning period (Figure 4), and it is indicated that the sensor requires only 3 calibrations at the time points which showed large slope change (14 days, 55 days, and 78 days) during continuous 3 months operation once after they completed the electrode conditioning.
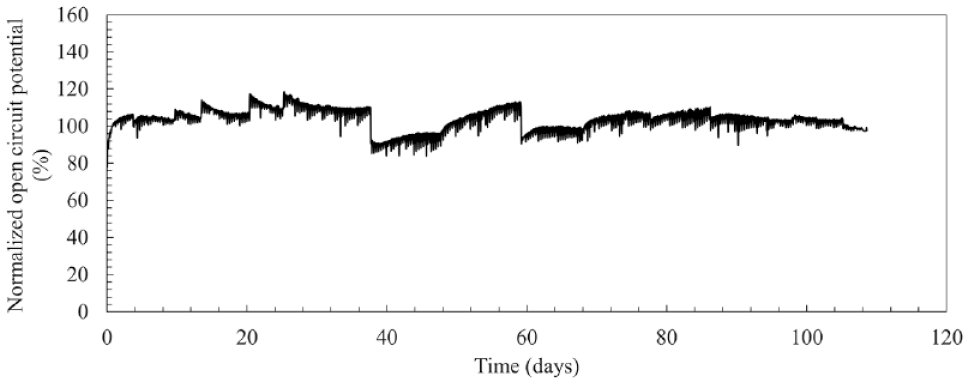
Continuous operation of the DET-type OCP glucose sensor for 108 days. OCP of direct electron transfer FADGDH immobilized electrode was continuously measured in 100 mM potassium phosphate buffer (pH 7.0) including 20 mM glucose. Before each OCP data sampling, + 100 mV (vs. Ag/AgCl) was applied to the OCP-based glucose sensor for 10 seconds. We recalculated the OCP value after 10 days continuous operation as 100 and normalized all OCP to the percentage. Abbreviations: DET, direct electron transfer; OCP, open circuit potential; FADGDH, FAD dependent glucose dehydrogenase.
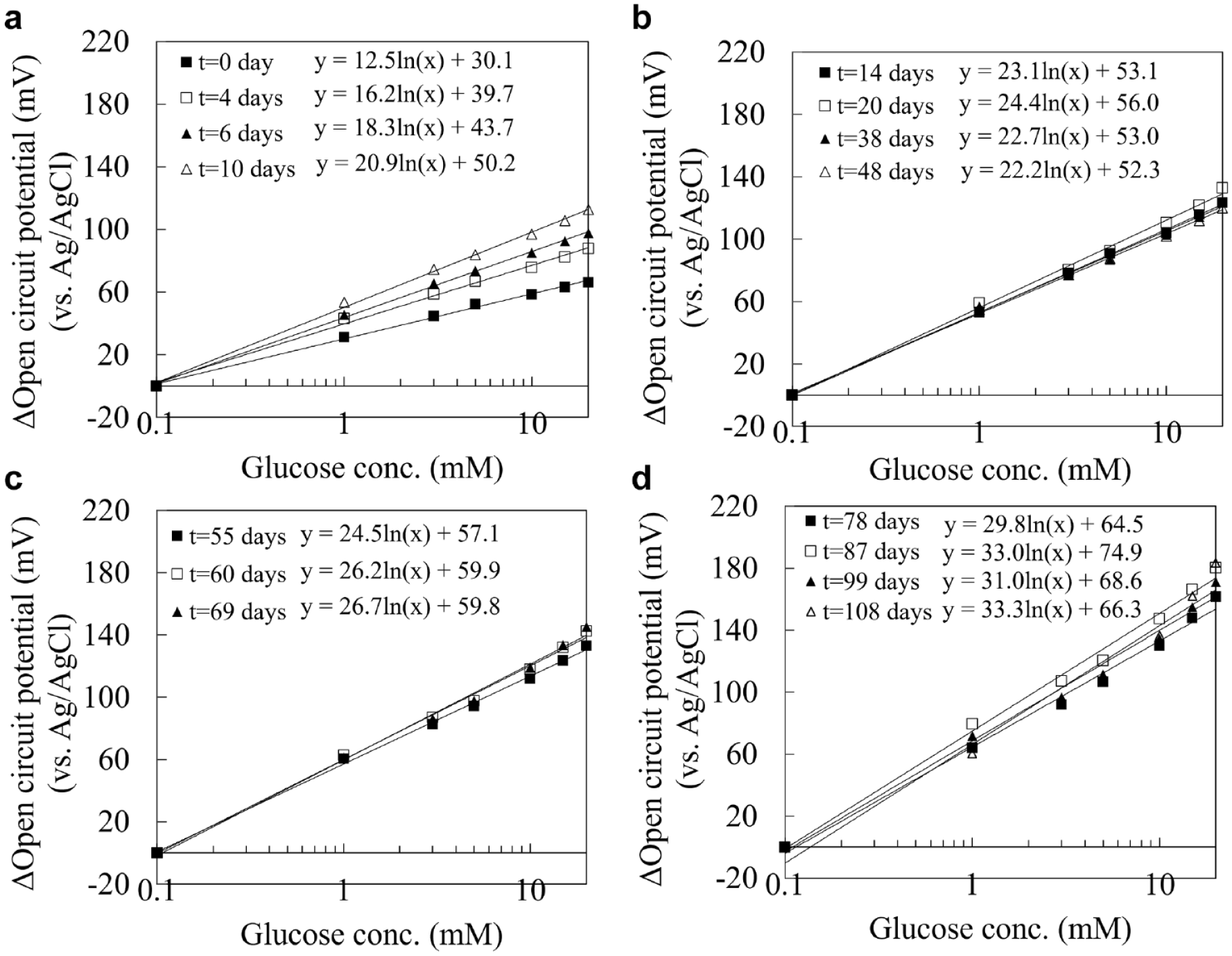
Representative calibration curves of DET-type OCP based glucose sensor with direct electron transfer FADGDH immobilized electrode in 100 mM potassium phosphate buffer (pH 7.0) for 108 days continuous operation. Before each OCP data sampling, + 100 mV (vs. Ag/AgCl) was applied to the OCP-based glucose sensor for 10 seconds: (a) Filled squares showed the calibration curve before starting continuous operation, opened squares showed the calibration curve 4 days after, filled triangles showed the calibration curve 6 days after and opened triangles showed the calibration curve 10 days after starting continuous operation, (b) Filled squares showed the calibration curve 14 days after, opened squares showed the calibration curve 20 days after, filled triangles showed the calibration curve 38 days after and opened triangles showed the calibration curve 48 days after starting continuous operation, (c) Filled squares showed the calibration curve 55 days after, opened squares showed the calibration curve 69 days after and filled triangles showed the calibration curve 38 days after starting continuous operation, and (d) Filled squares showed the calibration curve 78 days after, opened squares showed the calibration curve 87 days after, filled triangles showed the calibration curve 99 days after and opened triangles showed the calibration curve 108 days after starting continuous operation. Abbreviations: DET, direct electron transfer; OCP, open circuit potential; FADGDH, FAD dependent glucose dehydrogenase.
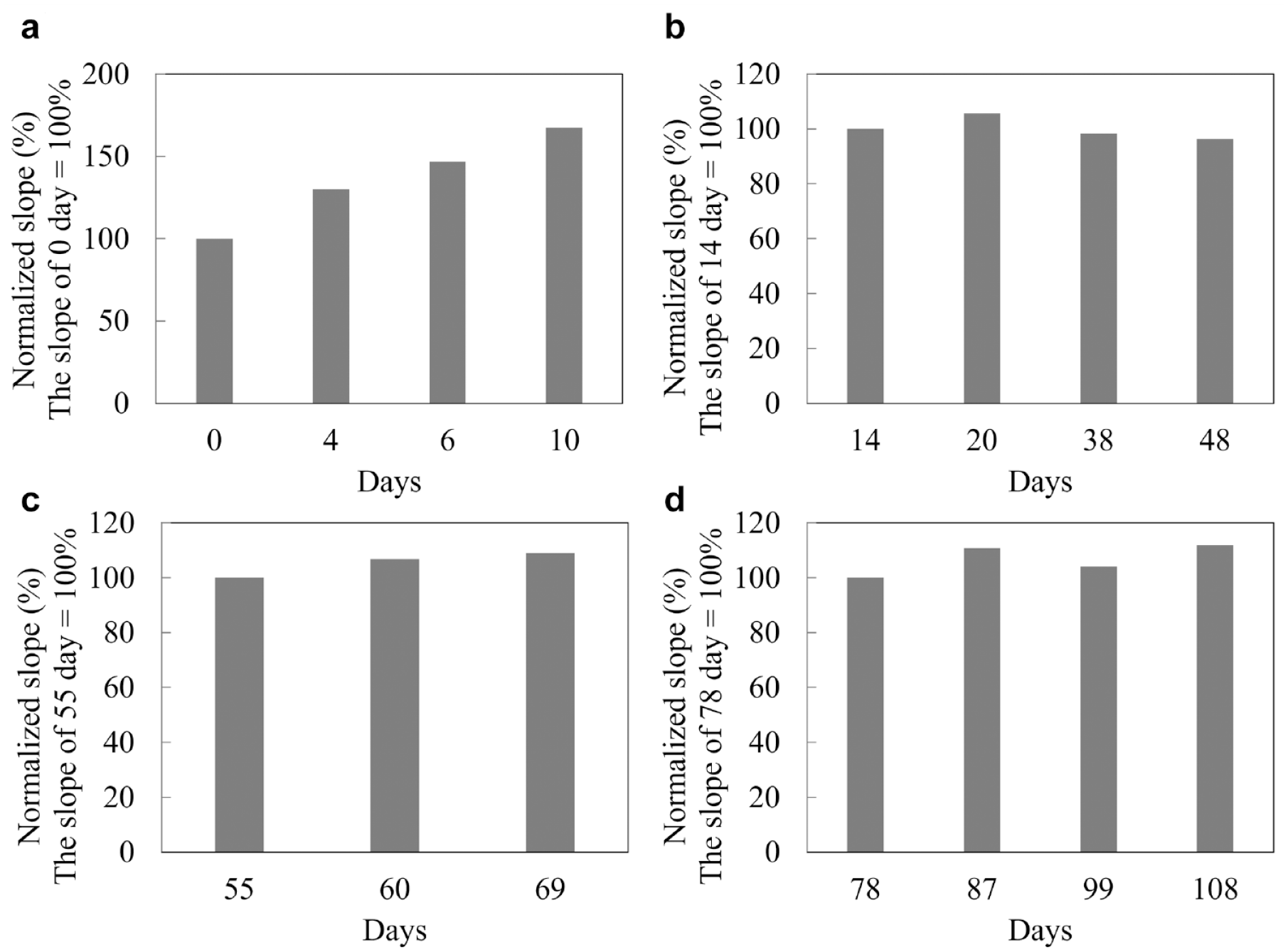
The change rate of curve slope between 0 day to 10 days, 14 days to 48 days, 55 days to 69 days and 78 days to 108 days: The change rate of curve slope was calculated the slope at the first day in each group (a: 0 day, b: 14 days, c: 55 days, and d: 78 days) as 100 % and normalized all slope value to the percentage.
Discussion
In this study, we demonstrated 108 days of in vitro continuous operation using a DET-type OCP sensor. Since DET-type FADGDH was immobilized by SAM, a single molecule enzyme layer was formed on gold electrode. Even with the minimum amount of enzyme, the sensor showed continuous operation for 108 days without any significant signal change. We previously reported the CGM sensor based on the chronoamperometric principle with glutaraldehyde crosslinking immobilized DET-type FADGDH, and the sensor showed a diminished signal after 36 hours of continuous operation. 12 We also reported on amperometric DET-type glucose sensor, with SAM immobilized DET-type FADGDH prepared by the same protocol of this study, the sensor also showed diminishment of the signal after 12 hours of continuous operation. 10 Even compared with recently reported continuous operation of enzymatic glucose sensors,13-15 this sensor is stable, indicative of the development of continuous longer-term operation of the enzymatic glucose sensor. The long-term stability of the current sensor may be attributed to the use of the OCP principle which can greatly reduce the number of catalytic reactions during the sensor operation. As our goal of this research is the practical and long-term use of OCP based glucose sensor, several investigations are still required to use this sensor as the future CGM application. For example, the 10 days of electrode conditioning period is too long for actual use and the sensor performance also should be investigated using in vivo conditions. In addition, the sensor performance and stability should be investigated at 37 °C, using the simulated interstitial fluid to validate the sensor performance, before its in vivo application. Based on the data presented here, we believe that the DET-type OCP glucose sensor is an ideal technology improvement for next generation CGM’s.
Conclusions
The DET-type OCP glucose sensor can be continuously operated in vitro at 25°C for more than 3 months without significant decreases in sensor signal and sensitivity. While the further investigation will be required in vitro and in vivo, the DET-type OCP glucose sensor should be the ideal next generation sensor for CGM, especially in long duration implantable CGM systems.
Footnotes
Acknowledgements
Joint Department of Biomedical Engineering, University of North Carolina at Chapel Hill and North Carolina State University; Arkray Inc.; Japan Society for the Promotion of Science. The authors would like to thank Mr. David Probst, PhD student at the Joint Department of Biomedical Engineering, UNC Chapel Hill and North Carolina State University, for English proofreading of this article.
Abbreviations
CGM; Continuous Glucose Monitoring, DET; direct electron transfer, FADGDH; flavin adenine dinucleotide-dependent glucose dehydrogenase, GOx; glucose oxidase, OCP; open circuit potential, DSH; Dithiobis(succinimidyl hexanoate), SAM; self-assembled monolayer, Pt; Platinum.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Inyoung Lee and Koji Sode are full time employees of the University of North Carolina at Chapel Hill. Sode’s research project relating to this contribution at UNC was financially supported by ARKRAY Inc. Tsugawa Wakako and Kazunori Ikebukuro are full time employees of Tokyo University of Agriculture and Technology.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Joint Department of Biomedical Engineering, University of North Carolina at Chapel Hill and North Carolina State University; Arkray Inc.; Japan Society for the Promotion of Science
