Abstract
Background:
The bacterial derived flavin adenine dinucleotide (FAD)-dependent glucose dehydrogenase (FADGDH) is the most promising enzyme for the third-generation principle-based enzyme sensor for continuous glucose monitoring (CGM). Due to the ability of the enzyme to transfer electrons directly to the electrode, recognized as direct electron transfer (DET)-type FADGDH, although no investigation has been reported about DET-type FADGDH employed on a miniaturized integrated electrode.
Methods:
The miniaturized integrated electrode was formed by sputtering gold (Au) onto a flexible film with 0.1 mm in thickness and divided into 3 parts. After an insulation layer was laminated, 3 openings for a working electrode, a counter electrode and a reference electrode were formed by dry etching. A reagent mix containing 1.2 × 10−4 Unit of DET-type FADGDH and carbon particles was deposited. The long-term stability of sensor was evaluated by continuous operation, and its performance was also evaluated in the presence of acetaminophen and the change in oxygen partial pressure (pO2) level.
Results:
The amperometric response of the sensor showed a linear response to glucose concentration up to 500 mg/dL without significant change of the response over an 11-day continuous measurement. Moreover, the effect of acetaminophen and pO2 on the response were negligible.
Conclusions:
These results indicate the superb potential of the DET-type FADGDH-based sensor with the combination of a miniaturized integrated electrode. Thus, the described miniaturized DET-type glucose sensor for CGM will be a promising tool for effective glycemic control. This will be further investigated using an in vivo study.
Keywords
Introduction
Continuous glucose monitoring (CGM) systems have been recognized as the ideal monitoring systems to ensure tight glycemic control of patients with diabetes,1-6 which is key to reducing the risk of various complications. Current commercially available CGM systems generally use a small sensor inserted or implanted under the skin for the measurement of glucose concentration in interstitial fluid. The principle of sensing glucose for CGM systems are mainly electrochemical based on enzymatic reactions. One exception is a system with fluorescent detection principle by employing an abiotic synthetic receptor.7-10 Current enzyme-based electrochemical CGM systems only use glucose oxidase, the gold standard enzyme, with the combination of the monitoring hydrogen peroxide or reduced synthetic electron acceptor (mediator).11-13 Among the various enzyme electrochemical glucose-sensing methods, direct electron transfer (DET), has shown several advantages compared to traditional methods. These including not requiring a soluble mediator or oxygen and uses a low redox potential. This helps mitigate several error states caused by variation to oxygen concentration around the electrode, or electroactive interference. In addition, thanks to the low redox potential of detection, the elimination of the interference caused by electrochemical active ingredients in the biological sample is expected. Unlike the commonly used fungi-derived enzymes used in self-monitoring glucose sensors, bacteria-derived flavin adenine dinucleotide (FAD)-dependent glucose dehydrogenase (FADGDH) is capable of DET with an electrode, and it is expected to realize DET-type, or third-generation electrochemical glucose sensors for CGM system.14-16 Recognizing the benefit of DET-type enzyme reaction with electrode, Sode and his group have reported various glucose-sensing principles, which could be used for the future CGM systems.17-21
In vitro characterization of a miniaturized electrode capable of being used in subcutaneous area is essential to realize a functional CGM system based on DET-type FADGDH. In this study, we report the in vitro evaluation of a miniaturized glucose sensor employing DET-type FADGDH, for the future use for an in vivo CGM system.
Materials and Methods
Chemicals and Enzyme
Phosphate-buffered saline (PBS) was purchased from Takara Bio Inc. (Kusatsu, Japan), D-glucose was purchased from Nacalai Tesque Inc. (Kyoto, Japan). Acetaminophen was purchased from Sigma-Aldrich, Inc. (St. Louis, MO, USA). DET-type FADGDH was recombinantly prepared according to the previous report. 22
Experimental Setup for the Evaluation of Miniaturized DET-Type Glucose Sensor
Figure 1 shows the experimental setup for the evaluation of miniaturized DET-type glucose sensor. The setup comprises 3 flow cells; each of them equipped with miniaturized DET-type glucose sensor connected with a custom-made potentiostat, and a pump/mixer system (Chromaster 5110 pump, Hitachi, Tokyo, Japan) with reservoirs containing 100 mM PBS solution pH 7.4, 1000 mg/dL glucose solution in PBS, and 100 µg/mL acetaminophen solution in PBS. The mixing ratio of these solution is controlled by chromaster system manager (Hitachi, Tokyo, Japan). The system is operated at 37°C.
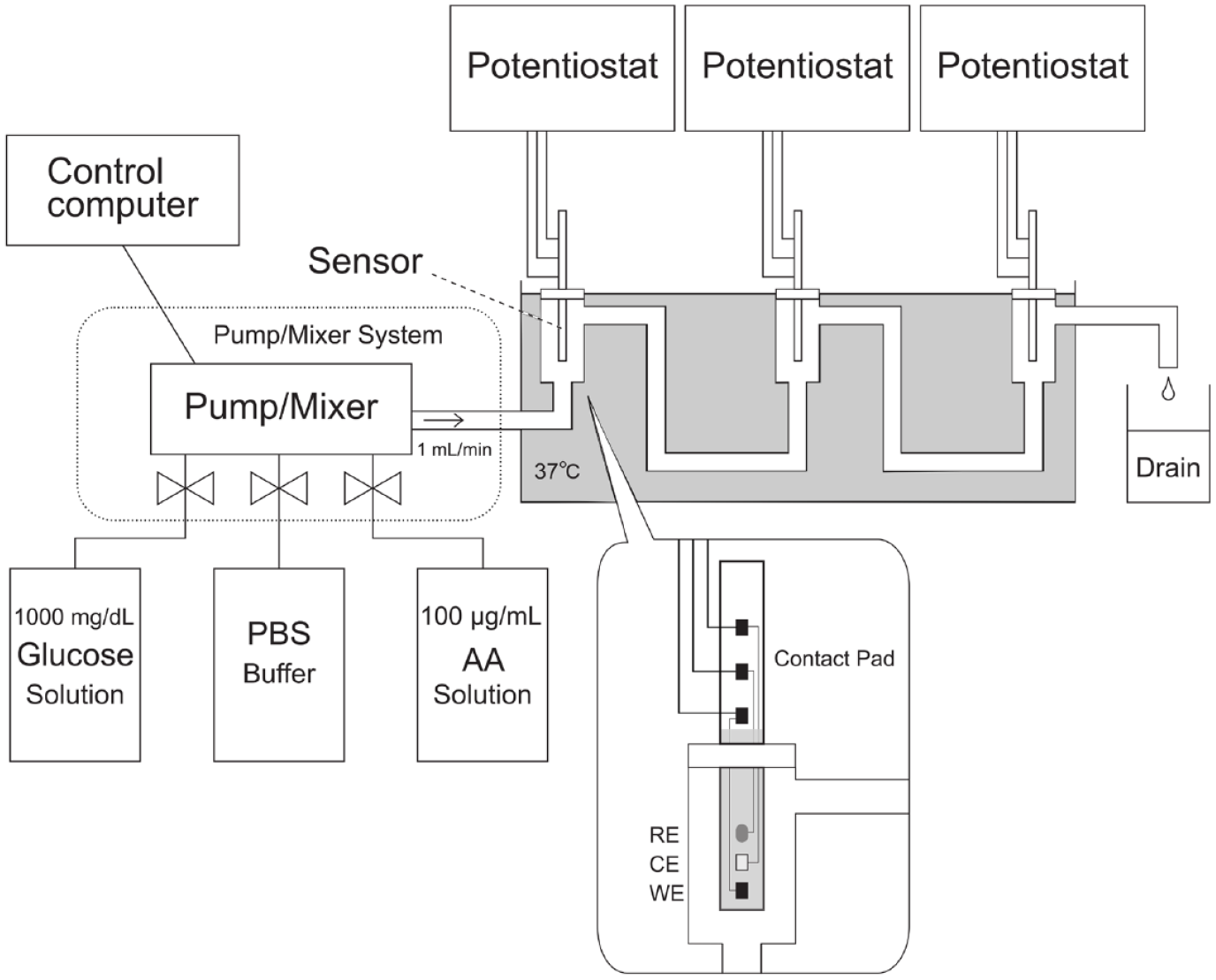
Experimental setup to investigate miniaturized DET-type glucose sensor. In this setup, a pump/mixer system was connected to 3 reservoirs, containing 1000 mg/dL glucose in PBS, 100-mM phosphate buffer saline solution pH 7.4 (PBS), and 100 µg/mL acetaminophen (AA) in PBS. The system mixed and pumped the solutions to a drain via 3 flow cells, each of them equipped with the miniaturized DET-type glucose sensor. A working electrode (WE), a counter electrode (CE), and a reference electrode (RE) of the sensor were connected to a potentiostat. The volume of each flow cell was 1 mL and the solutions flowed to the cells at a flow rate 1 mL/min. Abbreviation: DET, direct electron transfer.
Operation and Characterization of Miniaturized DET-Type Glucose Sensor
Using the experimental setup shown in Figure 1, 500 mg/dL glucose solution was continuously supplied to each flow cell equipped with miniaturized DET-type glucose sensor. Each miniaturized DET-type glucose sensor was operated by applying 0.15 V versus Ag/AgCl, and the current was recorded continuously. After the sensor signals provided steady-state current, then the solution with different glucose concentration (40, 70, 100, 180, 300, and 500 mg/dL) was provided by changing mixing ratio of the flow from the reservoirs for 1000 mg/dL glucose in PBS.
Investigation of the Effect of Acetaminophen on the Sensor Signal
The effect of acetaminophen on the sensor signal was investigated using same experimental set up, under glucose concentration at 100 mg/dL. Using a flow system, the miniaturized DET-type glucose sensors were exposed to 4 concentrations of acetaminophen (5, 10, 15, 20 μg/mL) for each 60 min, and the currents were measured continuously under an applied voltage of 0.15 V versus Ag/AgCl.
Investigation of the Effect of Oxygen Partial Pressure on the Sensor Signal
The effect of oxygen partial pressure (pO2) on the sensor signal was investigated using a miniaturized DET-type glucose sensor immersed in a reaction vial filled with 12 mL solution of 500 mg/dL glucose in PBS at 37°C. The sensor response was measured by applying 0.15 V versus Ag/AgCl using a potentiostat (PG580RM, Uniscan Instrument, Buxton, UK). After stable response of the sensor was obtained, nitrogen gas was purged into the glucose solution at a flow rate of 1 L/min to decrease pO2. pO2 was measured using a blood gas analyzer, RAPIDLab 348Ex (Siemens Healthcare Diagnostics, NY, USA), every 5 to 15 min.
Results and Discussion
Sensor Configuration
Figure 2 shows the configuration of a miniaturized electrode used for this study. The electrode was formed by sputtering gold (Au) onto a flexible film, 0.1mm in thickness and divided into 3 parts by wet etching. After an insulation was laminated, 3 openings for a working electrode, a counter electrode and a reference electrode were formed by dry etching. A reagent mix containing 1.2 × 10−4 Unit of DET-type FADGDH, and carbon particles was deposited. The reactive area was covered with an outer membrane, which is composed of the mixture of cellulose acetate (CA) and polyvinyl pyrrolidone (PVP) by dip coating. CA has been generally used as a biocompatible membrane covering sensor for in vivo use and limiting diffusion of substrate (glucose), 23 while PVP was employed to improve the mechanical stability of membrane.
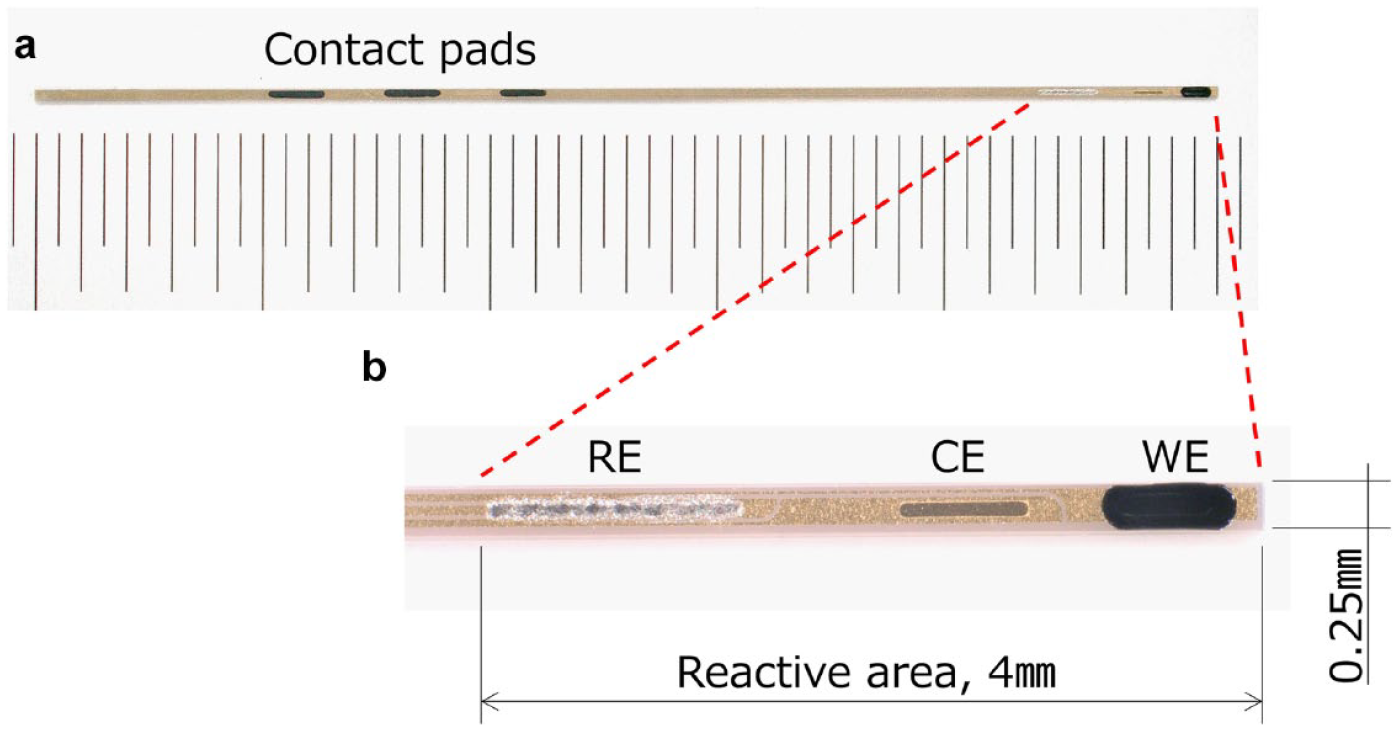
The miniaturized DET-type glucose sensor used in this study. (a) Outline view of a DET-type glucose sensor with the reactive area at one end and 3 contact pads to make electrical connection to a potentiostat at the other end. (b) A closeup of the DET-type glucose sensor on the reactive area including a working electrode (WE), a counter electrode (CE) and a reference electrode (RE) which consists of Ag/AgCl. The areas of WE, CE and RE were approximately 0.18, 0.05 and 0.17 mm2 respectively. Abbreviation: DET, direct electron transfer.
Continuous Operation of Miniaturized Glucose Sensor Employing DET-Type FADGDH
Figure 3a shows the representative sensor response toward consecutive change of glucose concentration. The sensor response increased in a stepwise manner according dependent on glucose concentration. The sensors (n = 3) showed a good linear correlation between glucose concentration and sensor response within the range from 40 to 500 mg/dL (Figure 3b). Figure 3c shows the result of the continuous operation of the miniaturized DET-type glucose sensor for 11 days. Identical sensor responses were repeatedly observed during the 11 days operation. This was confirmed by comparing the linear correlations between sensor signal and glucose concentration observed at day 1, day 6, and day 11, which are overlapped within ±9.5%. These results reveal that the miniaturized DET-type glucose sensor is stably operated for 11 days without any change of the sensor responses.
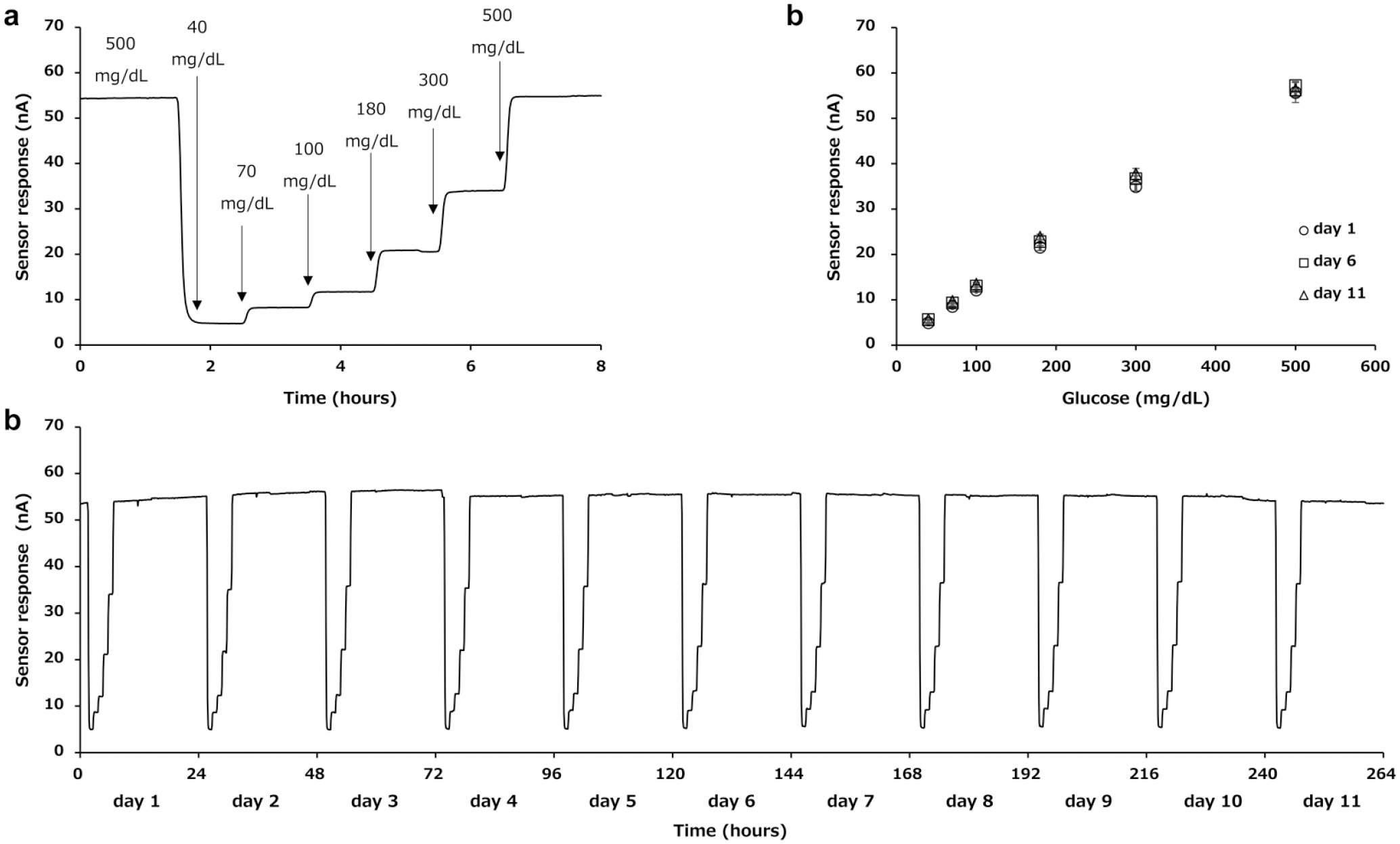
In vitro continuous monitoring of glucose using DET-type glucose sensor. The sensors (n = 3) were operated by applying 0.15V versus Ag/AgCl at 37°C. Glucose monitoring was carried out under consecutive change of glucose concentration in the carrier solution (40, 70, 100, 180, 300, and 500 mg/dL) following 18 h continuous operation with glucose concentration at 500 mg/dL. (a) Representative time course of the sensor response (day 1). (b) Calibration curves on day 1, day 6, and day 11. (c) Continuous operation of miniaturized DET-type glucose sensor for 11 days. Abbreviation: DET, direct electron transfer.
Effect of Acetaminophen
Table 1 shows the results of investigation about the effect of acetaminophen on the sensor response to glucose. In the presence of 20 μg/mL acetaminophen in the measurement solution, the relative sensor response was 99.8% compared to the response in the absence of acetaminophen, which we recognized as a not significant impact. The concentration of acetaminophen in human interstitial fluid after clinical dose administration is less than 20 μg /mL. 24 Thus, we concluded that daily use of acetaminophen does not affect the sensor response. This prevention of the interference signal from acetaminophen was achieved by applying relatively low potential on working electrode versus reference electrode, which is one of the biggest advantages of employing DET-type FADGDH. The redox potential of acetaminophen was reported around 0.2 V (vs. Ag/AgCl); 25 therefore, the impact of acetaminophen was minimized because the sensor was able to generate sufficient sensor signal with 0.15 V (vs. Ag/AgCl) which is lower than the redox potential of acetaminophen. In addition, it is also anticipated that the impact of salicylate and hydroxyurea would be minimized or negligible, considering the fact their redox potentials are similar or higher than acetaminophen.26-29
Acetaminophen Effect on Sensor Response.
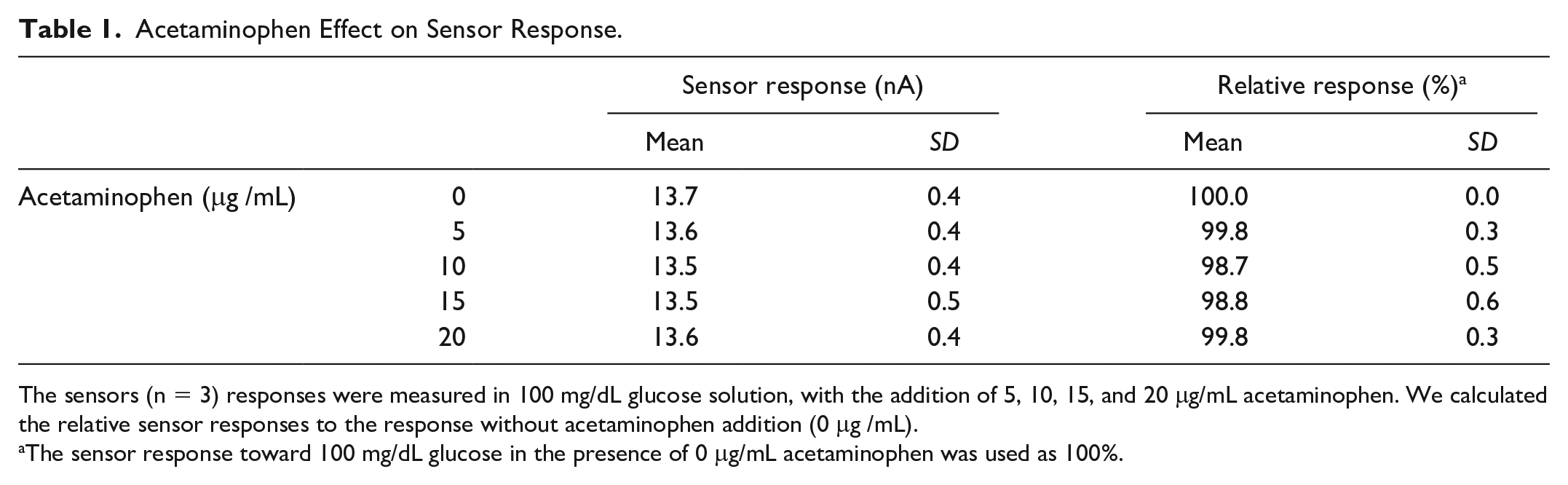
The sensors (n = 3) responses were measured in 100 mg/dL glucose solution, with the addition of 5, 10, 15, and 20 μg/mL acetaminophen. We calculated the relative sensor responses to the response without acetaminophen addition (0 μg /mL).
The sensor response toward 100 mg/dL glucose in the presence of 0 μg/mL acetaminophen was used as 100%.
Effect of Partial Pressure of Oxygen on the DET-Type Glucose Sensor Response
Figure 4 shows the sensor response under atmospheric condition, where pO2 is 150 mmHg, and under N2 bubbling condition, where pO2 is 40 mmHg. As shown in Figure 4, the sensor response slightly reduced during low (40 mmHg) pO2 condition, though which was negligible impact. Direct electron transfer-type FADGDH does not use oxygen as the electron acceptor, so theoretically the sensor employing this enzyme is not affected by the change of pO2. Therefore, the reduced signal might be caused by another physical condition, such as by the micro-bubble during the N2 bubbling condition. From these results, unlike the sensors employing glucose oxidase, 30 the effect of pO2 level on the DET-type glucose sensor response is negligible. As a future research direction, the sensor performance will be investigated in the solution mimicking interstitial fluid, 31 as well as in vivo, to demonstrate its feasibility of using this sensor for the future CGM system.
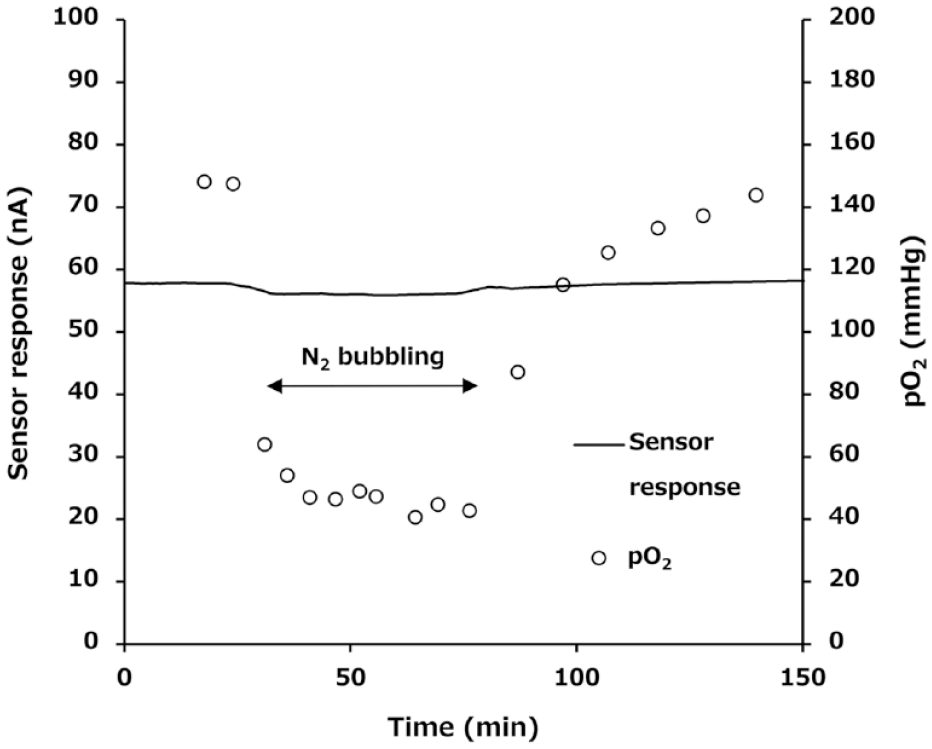
The impact of the partial pressure of oxygen change on DET-type glucose sensor response. The sensor response was monitored using 500 mg/dL glucose solution with or without N2 bubbling. The oxygen partial pressure (pO2) level was monitored using a blood gas analyzer. Abbreviation: DET, direct electron transfer.
Conclusion
The in vitro evaluation of miniaturized glucose sensor employing DET-type FADGDH, DET-type glucose sensor was investigated. The miniaturized DET-type glucose sensor showed stable response for more than 10 days without significant drift of calibration curve, which contributes to good accuracy during continuous measurement. In addition, the presence of acetaminophen and the change in pO2 level showed no significant effect on the sensor response. We believe that our miniaturized DET-type glucose sensor will be expected as a promising future CGM sensor which is an ideal tool for glycemic control in patients with diabetes.
Footnotes
Acknowledgements
The authors would like to thank Mr. David Probst, PhD student at the Joint Department of Biomedical Engineering, UNC Chapel Hill and North Carolina State University, for English proofreading of this article.
Abbreviations
FAD, flavin adenine dinucleotide; GDH, Glucose dehydrogenase; FADGDH, flavin adenine dinucleotide-dependent glucose dehydrogenase; CGM, Continuous Glucose Monitoring; DET, Direct Electron Transfer; WE, Wworking Electrode; CE, Counter Electrode; RE, Reference Electrode; AA, Aacetaminophen; pO2, Oxygen partial pressure.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Y.I., Y.K., and K.S. are full-time employees of ARKRAY, Inc. I.L. and K.S. are full-time employees of the University of North Carolina at Chapel Hill. K.S.’s research projects at UNC are partially financially supported by ARKRAY, Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by ARKRAY, Inc.
