Abstract
Background:
Continuous glucose monitoring (CGM) systems are increasingly being adopted as an alternative or adjunct to self-monitoring of blood glucose (SMBG) by patients receiving insulin therapy. However, the available evidence on the role of intermittently scanned CGM or flash CGM (isCGM) remains limited. This consensus aims to evaluate the degree of agreement among Spanish experts on the role of isCGM in the evaluation of glycemic variability, reduction of glycosylated hemoglobin (HbA1c) levels, and selection and adjustment of insulin therapy.
Methods:
Delphi methodology was used to achieve consensus in two survey rounds. A total of 431 Spanish endocrinologists participated in the first round of a 34-item questionnaire survey on isCGM and 427 participated in the second round. Any disagreement was resolved in round 2.
Results:
Consensus was reached for 32 statements, and four items were ultimately agreed upon SMBG after round 2. There was a high degree of consensus that isCGM helps to evaluate glycemic variability, improves HbA1c levels, and can guide therapeutic changes in type 1 diabetes patients. However, there was no consensus on the routine use of the interquartile range to evaluate glycemic variability or the selection of HbA1c as the main parameter for monitoring glycemic control.
Conclusions:
Most Spanish experts believe that the isCGM system is appropriate for: (1) identifying glycemic variability and facilitating its management, (2) evaluating hyperglycemia as a complement of HbA1c levels, and (3) guiding therapeutic decisions on insulin selection and dosing. The isCGM system is a useful tool for patients and health care professionals to improve glycemic control in insulin-dependent diabetes.
Keywords
Introduction
Self-monitoring of blood glucose (SMBG) is a well-established tool for daily optimization of glycemic control in people with diabetes mellitus. 1 However, this method requires several finger pricks, which many patients find uncomfortable and painful. 2 Furthermore, significant fluctuations in blood glucose may be missed with SMBG, hampering daily glycemic management. 3
Continuous glucose monitoring (CGM) technology provides near-continuous data by showing the glucose concentration in the body’s interstitial fluid every five minutes. 4 Glucose levels in interstitial fluid correlate well with plasma glucose concentrations and lag time is generally less than ten minutes. 5 This information can help identify, analyze, and prevent undesirable episodes of hypoglycemia and hyperglycemia, improving overall glycemic control.6,7
Two types of CGM systems are currently available: (1) real-time CGM (rtCGM) devices that continuously provide the real glucose concentration on a display; and (2) devices that perform intermittent scanning, also called Flash CGM (isCGM). Real-time CGM devices like Medtronic Guardian 4 Sensor® (Medtronic Inc, Northridge, Calif.), Dexcom G5® and G6® (Dexcom Inc, San Diego, Calif.), Eversense® (Senseonics Inc, Germantown, Maryland), and more recently, GlucoMen® Day CGM (Menarini Diagnostics, Firenze, Italy) and TouchCare® Nano CGM (Medtrum Technologies Inc, Shanghai, China) automatically measure glucose values and transmit them every five minutes to a monitoring device that provides alerts for predicted high and low glucose levels. 4 Available evidence indicates that rtCGM, compared with SMBG, improves glycemic management in adult and pediatric diabetes populations.6,8-15 New Guardian 4 system only requires a one-time calibration at the beginning to switch to Smart Guard mode. However, rtCGM devices (except for DexCom G6®) still need finger-stick glucose determinations for calibration once or twice daily. 16
Current isCGM systems, in contrast, require intermittent scanning of the sensor to obtain glucose values, and this should be done at least every eight hours to avoid data loss. The FreeStyle Libre™ (Abbott Diabetes Care, Witney) is currently the only isCGM system available. This device consists of a combined glucose sensor/transmitter and a separate touch-screen reader device, although most users download a specific application to view data and trends onto their smartphone. The sensor measures glucose levels in the interstitial fluid every minute and stores glucose data automatically every 15 minutes, using wired enzyme glucose-sensing technology. 17
isCGM is associated with a series of advantages: daily calibration with finger-stick glucose determinations is unnecessary, it is easy to use, and it costs less than rtCGM systems. CGM technology is reimbursed in many countries, including Spain, although under some prespecified conditions. Two large randomized controlled trials have demonstrated significant improvements in nocturnal and daytime hypoglycemia, associated with lower SMBG measurements in people with type 1 diabetes (T1D) 18 and type 2 diabetes receiving insulin therapy. 19 Although reductions in glycosylated hemoglobin (HbA1c) were not seen in either of these two studies, smaller observational studies have shown notable improvements in both HbA1c and hypoglycemia.20-27
In the absence of new randomized controlled studies with isCGM, there is currently no definitive evidence on the effects of these systems in reducing HbA1c levels. Furthermore, some experts believe that HbA1c is no longer the only parameter of interest in the follow-up of individuals with diabetes given the new metrics derived from CGM/isCGM data analysis. This study aims to evaluate the degree of consensus among Spanish adult and pediatric endocrinologists on the role of isCGM in identifying glycemic variability, achieving HbA1c reduction, mitigating the risk of hypoglycemia, and managing insulin selection, injection timing, and dose adjustments in adult and pediatric patients receiving insulin therapy, especially those with T1D.
Methods
This qualitative, multicenter study was conducted between September 2020 and January 2021. The consensus was developed in two stages: panel selection and a two-round modified Delphi process. The study protocol was approved by the Research Ethics Committee of the San Carlos Clinical Hospital in Madrid.
Panel Selection
Potential panelists were invited by e-mail to participate, and the inclusion criteria were confirmed: participants had to be (1) health specialists in endocrinology and nutrition or pediatric endocrinology within Spain and (2) experienced in the use of isCGM and experts in diabetic clinical practice. The invitation e-mail included a web link to access the study protocol and the Delphi questionnaire. When respondents accessed the web link, they were asked for their consent to participate before registering.
Questionnaire Development
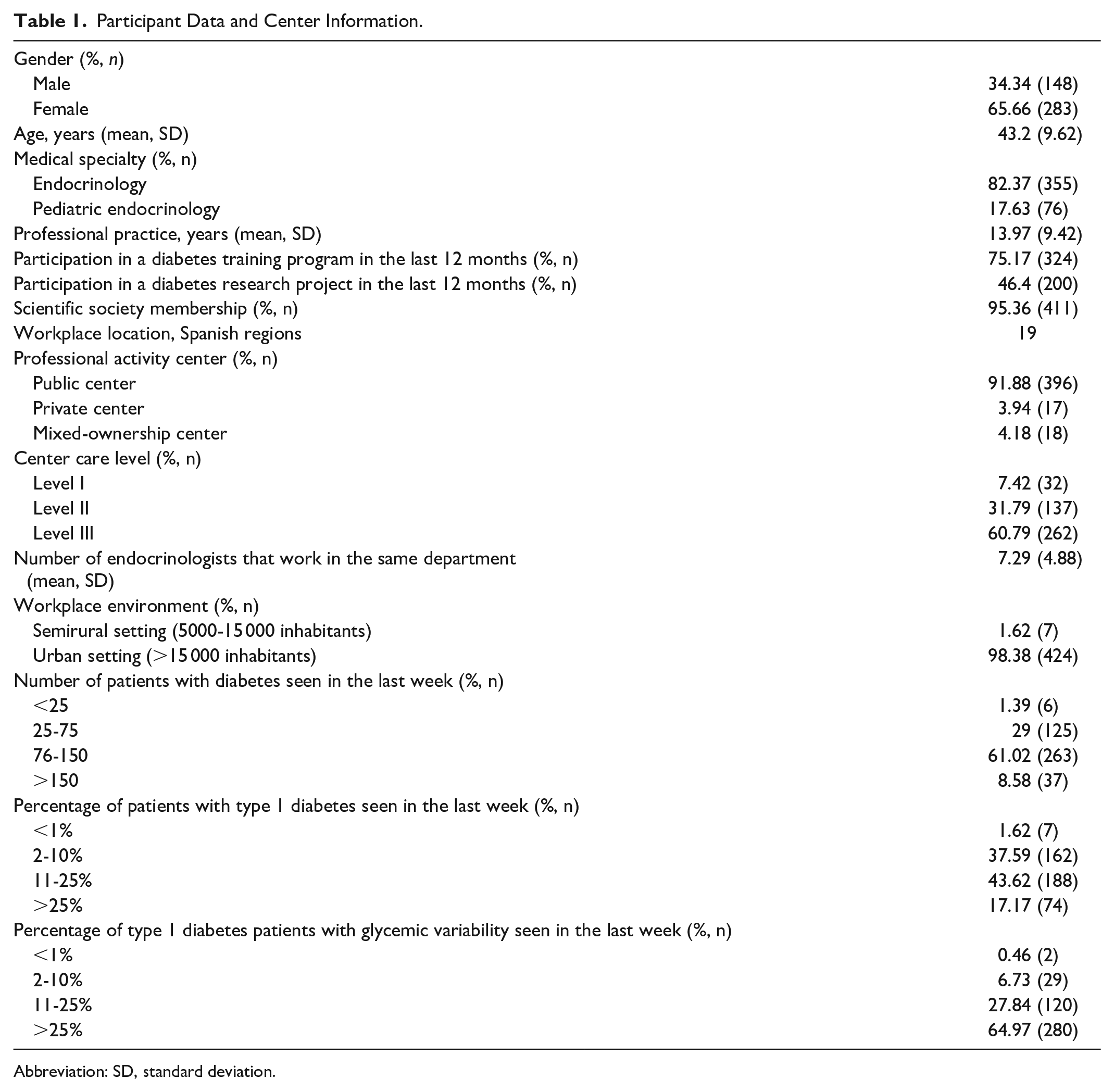
The steering committee defined a consensus framework based on a combination of publication review and clinical practice. Five key topics were initially identified (Supplementary Material), and the questionnaire was structured in 34 items grouped in three dimensions: (1) glycemic variability (14 items); (2) isCGM and HbA1c reduction (10 items); and (3) the role of isCGM in guiding insulin selection and therapeutic adjustments to minimize glycemic variability, the risk of hypoglycemia, and exaggerated postprandial glucose excursions (10 items). Some researcher data (six items) and center data (eight items) were also collected (Table 1).
Participant Data and Center Information.
Abbreviation: SD, standard deviation.
Delphi Method
Two rounds of questionnaires were delivered to a selected expert panel to reach a final consensus.
Round 1
Panelists signed into the microsite where they were asked to rank on a 5-point Likert scale their level of agreement with each item on the use of isCGM in glycemic variability management, HbA1c reduction, and the selection of and switching between insulin products (1 = strongly disagree; 2 = disagree; 3 = undecided; 4 = agree; 5 = strongly agree). Consensus for each item was considered if more than 75% of the experts rated it with a score of 4 to 5 out of 5 (agree or strongly agree) or 1 to 2 (disagree or strongly disagree). 28 Consensus was defined as “high” at 75% to 90% and “very high” at ≥90%. Completed questionnaires were automatically registered in a standardized, secure microsite database. The answers for each item retrieved in round 1 were statistically analyzed with SAS Version 9.1.3 Service pack 3 and organized according to whether consensus was achieved or not. For the comparison between the degree of agreement between groups, due to the ordinal nature of this parameter, the Mann-Whitney U test was used in the case of two comparison groups, and the Kruskal-Wallis test was used for three or more groups. In the case of comparison of two categorical variables, Fisher’s exact test was used. All statistical tests were performed with a significance level of .05.
Round 2
In the second round, the experts had to assess items that did not reach consensus in the first round. Panelists were informed of aggregate-level summary statistics of the individual responses and consensus. Similarly, to round 1, the responses for each item were analyzed and grouped by specialty to identify differences in the respondents’ agreement.
Results were expressed as a percentage for categorical variables and mean and standard deviation (SD) for continuous variables.
Results
Characteristics of Participants
A total of 431 Spanish panelists took part in round 1, and 427 in round 2. Most panelists (82.3%) were adult endocrinologists distributed within Spain, while only 17.6% were pediatric endocrinologists. Panelists’ mean age was 43.2 years (±9.6), and more panelists were female (65.6%). Table 1 lists panelists’ data and center information.
Questionnaire Results
Of the 34 questions on isCGM domains, 32 (94.1%) achieved a consensus score exceeding the 75% threshold, and 20 of these (58.8%) exceeded 90% consensus. Only two questions (5.8%) did not achieve consensus, scoring less than 75%.
Domain 1: Glycemic variability
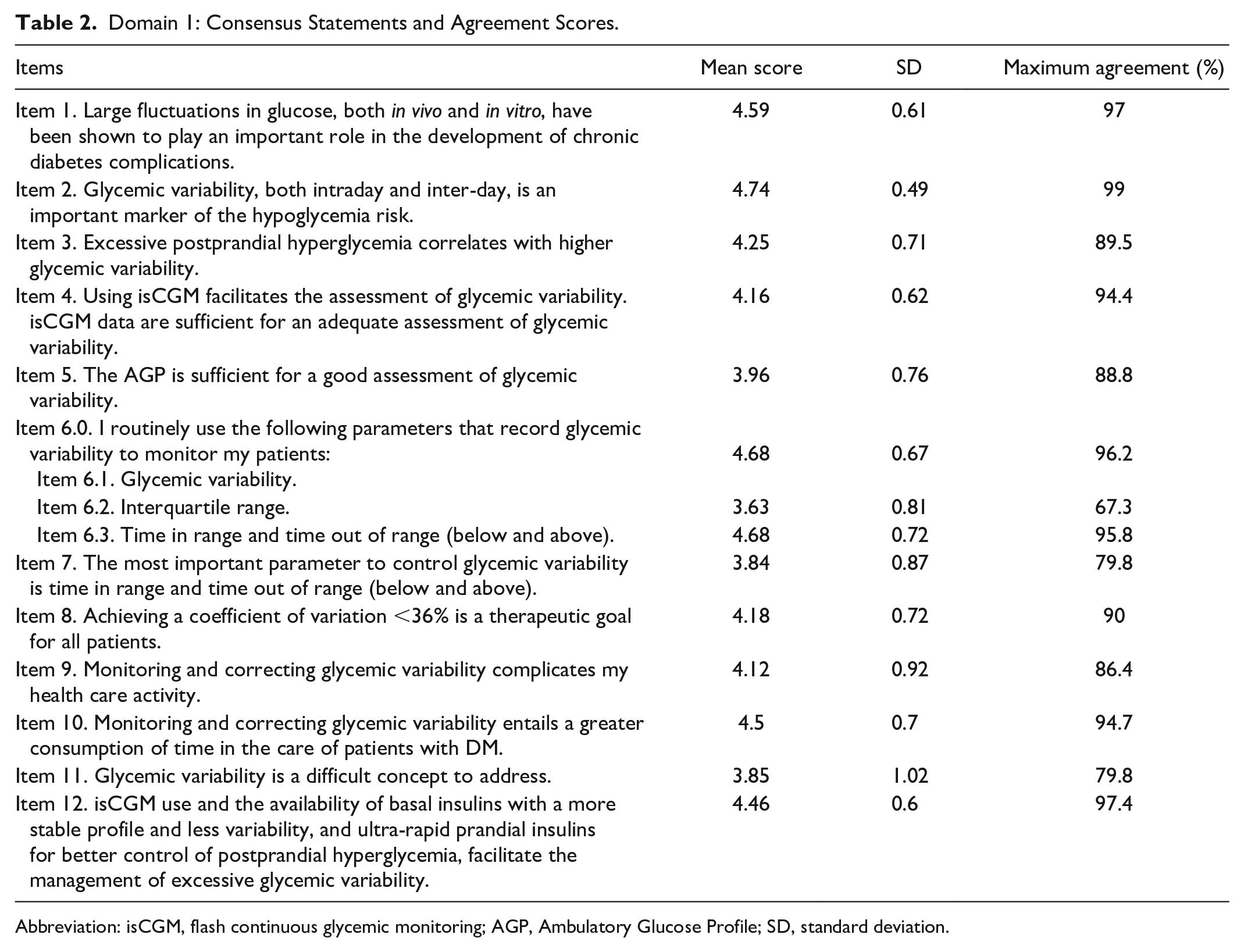
Ninety-seven percent of respondents agreed (mean ± SD: 4.59 ± 0.61) that large fluctuations in glucose, both in vivo and in vitro, play an important role in the development of chronic complications of diabetes (Table 2). Furthermore, there was a very high degree of consensus (99% of respondents) that glycemic variability, both intraday and inter-day, is an important marker for hypoglycemia risk (4.74 ± 0.49). Among the factors involved in glycemic variability, 89.5% of participants agreed (4.25 ± 0.71) that excessive postprandial hyperglycemia correlates with higher glycemic variability. Experts also agreed strongly (94.4%) that isCGM facilitates the assessment of glycemic variability, and data obtained are sufficient for adequate monitoring of glycemic variability (4.16 ± 0.62). However, 88.8% of panelists agreed that the Ambulatory Glucose Profile (AGP) was appropriate for assessing glycemic variability (3.96 ± 0.76). On the other hand, a very high consensus was achieved on the routine use of glycemic variability (96.2% of participants, 4.68 ± 0.67) and time out of range as metrics to monitor people with diabetes (95.8% of respondents, 4.68 ± 0.72). Despite this, no consensus was reached on the use of the interquartile range as a metric for recording glycemic variability (agreement of 67.3% of panelists). Indeed, in the first round, there was no consensus as to whether the most important parameter for controlling glycemic variability was time in range (TIR) and time out of range (below and above). In the second round, 79.8% of the experts reached agreement (3.84 ± 0.87). A clear consensus (90%) was reached on achieving a coefficient of variation <36% as a therapeutic goal for all people with diabetes (4.18 ± 0.72)
Domain 1: Consensus Statements and Agreement Scores.
Abbreviation: isCGM, flash continuous glycemic monitoring; AGP, Ambulatory Glucose Profile; SD, standard deviation.
Integrating glycemic variability monitoring into clinical practice can be challenging for several reasons. Common issues reported include data overload, increased clinical visit time, and the need for training on data interpretation. In this line, 94.7% of respondents agreed (4.5 ± 0.7) that monitoring and correcting glycemic variability complicates their health care activity, while in the second round 79.8% of the experts agreed (3.85 ± 1.02) that glycemic variability is a difficult concept to address. In addition, 86.4% of the experts admitted that monitoring and correcting glycemic variability complicates their care activity (4.12 ± 0.92). A very high consensus was achieved on the role of isCGM in this respect, and 97.4% of respondents agreed (4.46 ± 0.6) that isCGM use along with the availability of basal insulins with a more stable profile and ultra-rapid prandial insulins facilitates the management of excessive glycemic variability.
Domain 2: isCGM and HbA1C reduction
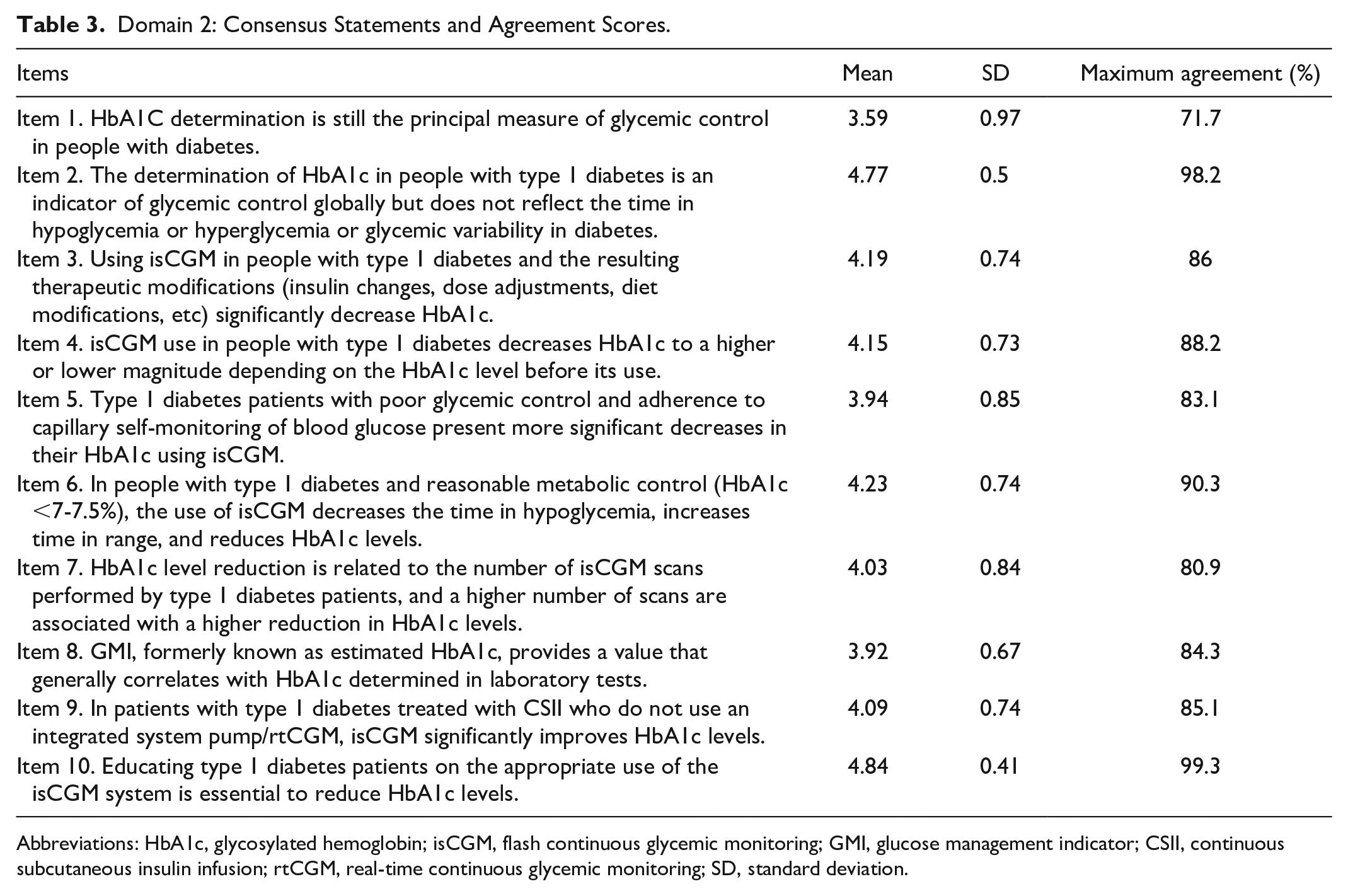
There was a very high consensus (98.2%) that the determination of HbA1c in people with T1D is a global indicator of glycemic control but that it does not reflect the time in hypoglycemia or hyperglycemia or glycemic variability (4.77 ± 0.5, Table 3). However, no consensus was reached among the expert group in favor of the statement that HbA1c determination is still the gold standard for monitoring glycemic control in people with diabetes (71.7% as maximum agreement).
Domain 2: Consensus Statements and Agreement Scores.
Abbreviations: HbA1c, glycosylated hemoglobin; isCGM, flash continuous glycemic monitoring; GMI, glucose management indicator; CSII, continuous subcutaneous insulin infusion; rtCGM, real-time continuous glycemic monitoring; SD, standard deviation.
There was agreement (>85%) that using isCGM in people with T1D and the resulting therapeutic modifications significantly decrease HbA1c (4.19 ± 0.74), to a higher or lower magnitude depending on the starting HbA1c level before its use (4.15 ± 0.73). In fact, 90.3% of panelists agreed that in people with T1D and reasonable metabolic control (HbA1c <7-7.5%), the use of isCGM decreases time in hypoglycemia, increases TIR, and reduces HbA1c levels (4.23 ± 0.74). Furthermore, there was good consensus (80.9%) that reducing HbA1c levels is related to the number of isCGM scans performed by T1D patients. However, a second round was needed to reach an 83.1% consensus among panelists on whether T1D patients with poor glycemic control and adherence to SMBG present more significant decreases in HbA1c levels.
Only 84.3% of respondents ultimately agreed after round 2 (3.9 ± 0.6) that the glucose management indicator provides a value that generally correlates with HbA1c determined in laboratory tests. There was also agreement (85.1%) among experts that isCGM significantly improves HbA1c levels in patients with T1D treated with continuous subcutaneous insulin infusion who do not use an integrated system pump/rtCGM (4.09 ± 0.74). Finally, consensus that educating T1D patients on the appropriate use of the isCGM system is essential to reduce HbA1c levels was very high (99.3%, 4.84 ± 0.41).
Domain 3: Role of isCGM in the selection, dosing, and switching between insulin formulations
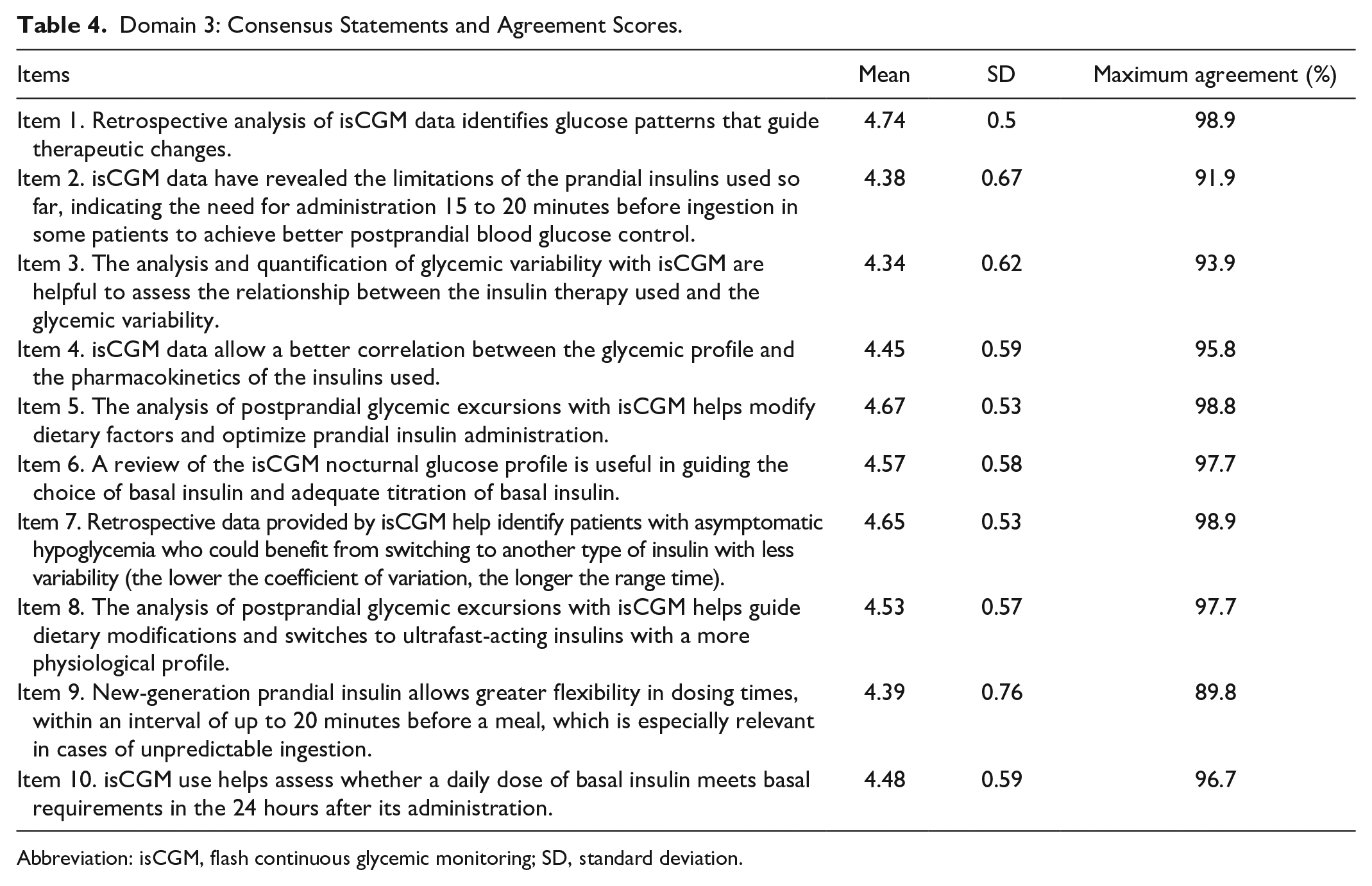
In the first round of the questionnaire, a consensus of 75% was obtained for all statements for this domain (Table 4). Therefore, the steering committee decided to avoid a further round. A very high consensus was found among the experts (>90%) who agreed that isCGM is useful for: (1) guiding therapeutic changes by identifying glucose patterns retrospectively (4.74 ± 0.5); (2) assessing the relationship between an insulin formulation and glycemic variability (4.34 ± 0.6); (3) finding a better correlation between glycemic profile and insulin pharmacokinetics (4.45 ± 0.59); and (4) identifying patients with asymptomatic hypoglycemia who could benefit from switching to an insulin with less variability (4.65 ± 0.53).
Domain 3: Consensus Statements and Agreement Scores.
Abbreviation: isCGM, flash continuous glycemic monitoring; SD, standard deviation.
Panelists agreed (91.9%) that isCGM data have revealed the limitations of first-generation prandial insulins, indicating the need in some patients for administration 15 to 20 minutes before ingestion to achieve better postprandial blood glucose control. Furthermore, experts also agree (>97%) that the analysis of postprandial glycemic excursions with isCGM helps to: (1) modify dietary factors and optimize prandial insulin administration (4.67 ± 0.53) and (2) switch to ultrafast acting insulins with a more physiological profile (4.53 ± 0.57). Finally, with regard to other types of insulin, 89.8% of experts agreed (4.39 ± 0.76) that new-generation prandial insulin allows greater flexibility in dosing times, within an interval of up to 20 minutes before a meal, a factor that is especially relevant in cases of unpredictable ingestion.
Among other benefits of using isCGM, the panelists (96.7%) agreed that this system helps assess whether a daily dose of basal insulin meets the basal requirements in the 24 hours after its administration (4.48 ± 0.59). Overall, 97.7% of respondents agreed that a review of the isCGM nocturnal glucose profile is useful in guiding the choice and adequate titration of basal insulin (4.57 ± 0.58).
Subgroup Analysis According to Professional Specialty
Statistically significant differences of opinion were found between professional specialties, that is adult endocrinologists versus pediatric endocrinologists. Spanish adult endocrinologists agreed that glycemic variability is a difficult concept (82% vs 72%), and HbA1c remains the principal method of monitoring glycemic control (75.43% vs 54.66%). Conversely, Spanish pediatric endocrinologists believed that (1) large glucose fluctuations play an important role in the development of chronic complications of diabetes (96.61% vs 98.68%); (2) excessive postprandial hyperglycemia correlates with higher glycemic variability (87.89% vs 97.37%); and (3) routine use of the interquartile range as well as TIR and time below and above range are recommendable (78.87% vs 84.21%) for monitoring glycemic control.
Discussion
This article describes a Delphi consensus on the benefits of isCGM in people with diabetes receiving insulin therapy, based on expert opinions from a large group of Spanish adult and pediatric endocrinologists. High levels of agreement were achieved in 32 out of a total of 34 statements related to the benefits of isCGM in glycemic variability, reduction in HbA1c levels, and the selection and adjustment of insulin therapy, demonstrating very strong comparable views. Furthermore, this level of consensus was achieved for most of the items after a single round of polling. A second round to reach an agreement was needed for only six items.
Most of the statements relating to glycemic variability were strongly supported by the respondents. The main limitations of HbA1c are the lack of information on daily glycemic excursions, acute diabetes complications, and the magnitude and frequency of intraday and inter-day glucose variability.29,30 Indeed, no consensus supporting HbA1c determination as the principal method of monitoring glycemic control in people with diabetes was reached among the experts. The lack of consensus in this respect may be partly explained by an increasing number of experts supporting the assertion that CGM/isCGM systems should be used to deliver valuable additional information on patients with T1D. Although studies that support the association between time in/out of range outcomes and long-term morbidity and mortality are lacking,31-33 CGM/isCGM parameters will play an increasingly important role in monitoring overall glycemic control.
Other consensus documents34-38 have been published with the aim of clarifying the opinion of clinicians on the appropriate use of CGM in the management of people with diabetes. Specifically, in an Italian consensus published in 2019, 35 a panel of 74 experts stated agreement or disagreement with a selection of 59 Likert statements using both rtCGM and isCGM systems. However, our study includes a larger number of participants (431 panelists) than the Italian consensus, and we achieved a higher level of agreement on clinical issues (66% vs 75%, respectively). Indeed, expert opinions on the relevance of identifying glycemic variability and making therapeutic modifications to improve the glycemic profile are of particular importance.39,40 Furthermore, we found a high level of clinical consensus in areas of uncertainty surrounding isCGM, despite participants working in different regional health systems.
Nevertheless, our study has several limitations. First, we describe the consensus views of a large group of clinicians working in different regions of Spain. As such, the perspective presented here may not reflect the situation in other health care systems. Second, our study is a consensus including the opinions of adult and pediatric endocrinologists, so it may underrepresent the point of view of non-endocrinologist experts. This would be an important area for further investigation. Last, we did not achieve consensus on two statements: (1) the routine use of the interquartile range to record glycemic variability and (2) HbA1c determination as the principal method of monitoring glycemic control. Nonetheless, this lack of consensus only represents around 5% of all items and might be explained by potential arguments. For example, in clinical practice, the interquartile range may possibly be assessed visually in the AGP without being quantified or recorded. Furthermore, the disagreement among the experts on HbA1c determination as the principal method of monitoring glycemic control supports the need for complementary methods to control glycemic variability.
Glycemic variability is associated with increased risk of hypoglycemia. Identifying increased glycemic variability is easy but correcting it is not a simple task. Therefore, the study reveals that most experts recognize that monitoring and correcting glycemic variability is difficult. New long-acting insulin analogues (degludec, glargine U300), with a more protracted and stable profile, and new ultra-rapid prandial insulins (faster aspart, URLi) may reduce glycemic variability, but their correct combination in an individual patient may be challenging for some health care providers.
Conclusion
This consensus evaluates the degree of consensus among Spanish adult and pediatric endocrinologists on the role of isCGM in insulin-dependent diabetes, especially people with T1D. The expert panel concludes that the isCGM system: (1) complements information based on HbA1c to identify and facilitate the management of excessive glycemic variability; (2) reduces HbA1c levels; and (3) may guide changes between insulin formulations when necessary. These results suggest that the isCGM system should be considered an important tool to help health care professionals and patients for improving diabetes management.
Supplemental Material
sj-docx-1-dst-10.1177_19322968221087270 – Supplemental material for Spanish Consensus on the Use of Intermittently Scanned Continuous Glucose Monitoring in the Management of Patients With Insulin Therapy: The MONITOR Project
Supplemental material, sj-docx-1-dst-10.1177_19322968221087270 for Spanish Consensus on the Use of Intermittently Scanned Continuous Glucose Monitoring in the Management of Patients With Insulin Therapy: The MONITOR Project by Francisco Javier Ampudia-Blasco, Estibaliz Ugarte-Abasolo, Ana Chico, Jorge García-Alemán and Manuel Galan-Barroso in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors thank Grupo Saned, S.L. for logistic and statistical support and editorial assistance.
Abbreviations
AGP, Ambulatory Glucose Profile; CGM, continuous glucose monitoring; isCGM, intermittently scanned continuous glucose monitoring; HbA1c, glycosylated hemoglobin; MS, mean score; rtCGM, real-time continuous glucose monitoring; SD, standard deviation; T1D, type 1 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that they have participated as speakers and in meetings supported by Novo Nordisk, and in advisory boards with Novo Nordisk.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Novo Nordisk Pharma, S.A., Madrid, Spain.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
