Abstract
Diabetes polyneuropathy is an important complication of diabetes polyneuropathy, and its notable sequelae of foot ulceration, autonomic dysfunction, and neuropathic pain are associated with significant morbidity and mortality. Despite the major impact on quality of life and health economic costs, it remains underdiagnosed until late in its natural history, and there is lack of any intervention that can reverse its clinical progress. Assessment of small fiber neuropathy (SFN) in diabetes offers an opportunity to detect abnormalities at an early stage so that both interventional studies and preventative measures can be enacted to prevent progression to the devastating complications of foot ulceration and cardiac dysautonomic death. Over the last two decades, significant advances have been made in understanding the pathophysiology of diabetes neuropathy and its assessment. In this review, we discuss limitations of the screening methods recommended in current clinical guidelines which are based on large nerve fiber assessments. Thereafter, we discuss in detail the various methods currently available to assess small fiber structure and function and examine their individual strength and limitations. Finally, we discuss the reasons why despite the considerable body of evidence available, legislators and global experts have yet to incorporate the assessment of SFN as routine clinical surveillance in diabetes management. We hope that these insights will stimulate further discussion and be instrumental in the early adoption of these methods so as to reduce the burden of complications arising due to diabetes polyneuropathy.
Introduction
The term diabetes polyneuropathy (DPN) refers to a heterogeneous group of neurological disorders—either clinically evident or subclinical—that occur in the setting of diabetes mellitus after exclusion of other etiologies of peripheral neuropathy. A recent position statement by the American Diabetes Association (ADA) proposed a clinical classification of DPN into typical/diffuse or atypical neuropathy with the former further categorized based on organ-systems affected. 1 Distal symmetrical polyneuropathy (DSPN) is considered the most common form of DPN with estimated prevalence rates reaching up to 75% in some studies. 2 Case definition of DSPN has historically been based either on patient-reporting of neurological symptoms or insensitivity to a 10-gm Semmes-Weinstein mono-filament (SWMF), abnormal vibration sensation using tuning forks, or abnormal ankle reflex during diabetes annual reviews. Electro diagnostic tests like nerve conduction studies (NCS) have been traditionally reserved for atypical presentations such as amyotrophy or entrapment neuropathy3,4 and are not part of routine DPN assessment. All these methodologies assess large fiber neural health. There is a significant body of evidence to suggest that the myelinated Aα and Aβ nerve fibers (Figure 1)—which serve touch, proprioception, position sense, vibration and muscle control and put patients at high risk of foot ulceration and its consequences—are affected late in the natural history of diabetes. 5 However, it is increasingly recognized that small nerve fibers are involved early in the course of DPN, often before large fiber involvement. Therefore, early detection of small fiber neuropathy (SFN) may offer hope for early intervention using disease-modifying strategies such as lifestyle modification, glycemic control, or novel therapeutics, in the same vein as retinal assessments for retinopathy and microalbuminuria for nephropathy have transformed their outcomes.
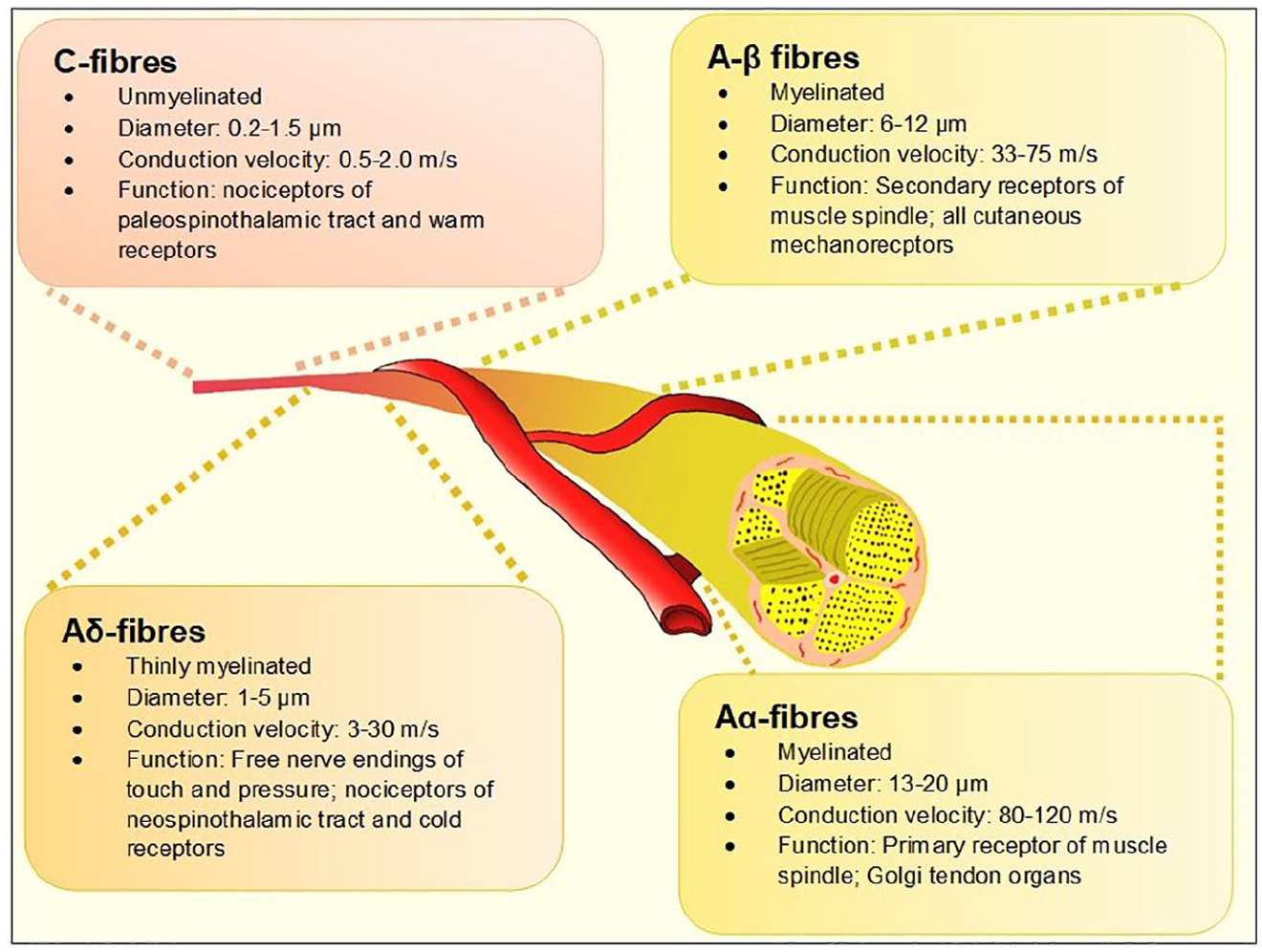
The different types of peripheral nerves based on the Erlanger-Gasser classification. 6
Definition of SFN
SFN is a subtype of peripheral neuropathy affecting the thinly myelinated Aδ or the non-myelinated C fibers (Figure 1). These fibers constitute 79.6% to 91.4%7,8 of peripheral nerve fibers and mediate several key functions including temperature and pain perception, sweating, and tissue blood flow, all of which, when impaired, are pathophysiologically related to adverse outcomes associated with foot ulcerations in people with diabetes. 9 Another important function of small fibers is regulation of the autonomic system and when affected, the term “autonomic neuropathy” is often used.10,11
In 2010, the Diabetic Neuropathy Study Group of the European Association for the Study of Diabetes (NEURODIAB) agreed on the diagnostic criteria for SFN in diabetes 12 (Table 1). It is interesting to note that despite significant developments in the detection of SFN in all forms of diabetes including pre-diabetes, type-1 diabetes (T1DM), and type-2 diabetes (T2DM), this definition has yet to be updated and remains based on length-dependent symptoms, intraepidermal nerve fiber density (IEFND), and quantitative sensory testing (QST). Furthermore, the consensus document posited that “at present, it is not possible to suggest criteria to define the severity of SFN in DPN.” This statement is perhaps in need of reappraisal in view of the advancements in tests for SFN, including the establishment of normative values for these tests.
Diagnostic Criteria for SFN in Diabetes Proposed by the Diabetic Neuropathy Study Group of the European Association for the Study of Diabetes (NEURODIAB)—2010. 12
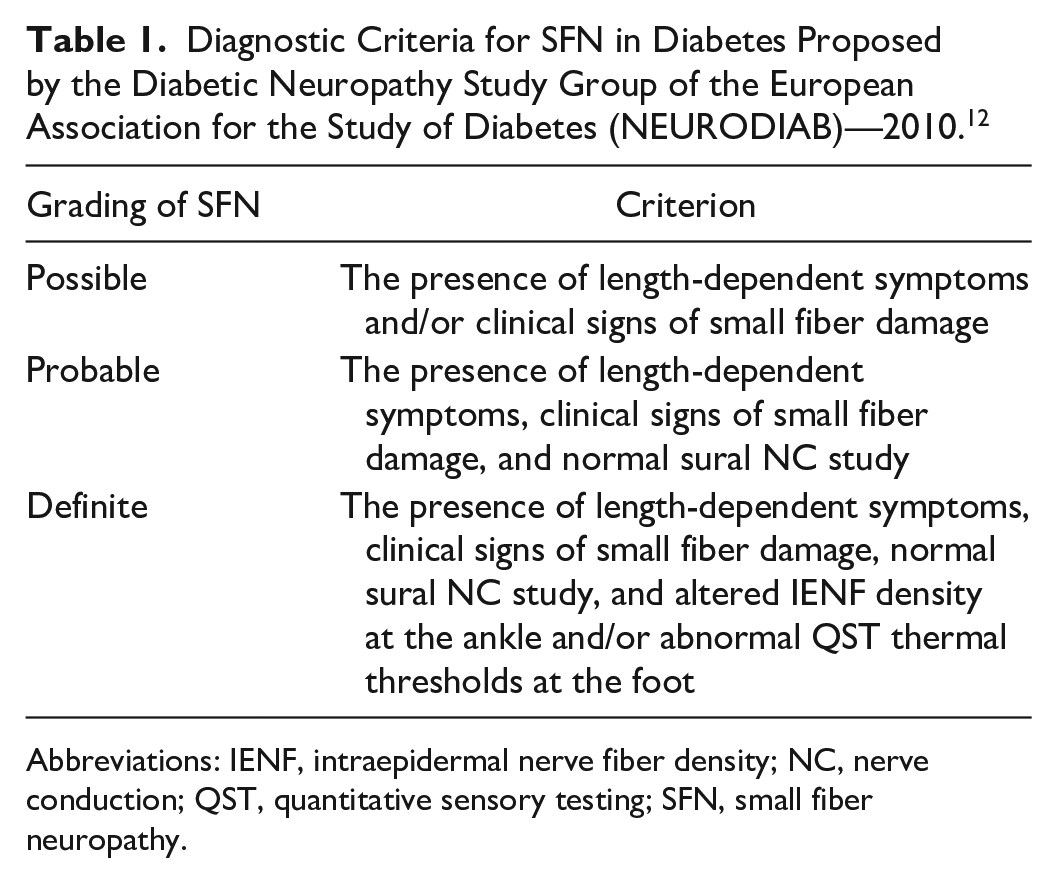
Abbreviations: IENF, intraepidermal nerve fiber density; NC, nerve conduction; QST, quantitative sensory testing; SFN, small fiber neuropathy.
Methods
Methods for evaluating SFN can be broadly categorized into two groups; first, techniques assessing small fiber function (SFF) mediated by the Aδ or the non-myelinated C fibers; and second, those quantitating small fiber structure (SFS). Table 2 provides a summary of the methods currently available.
A Summary of the Various Methods of Assessing SFF and SFS.
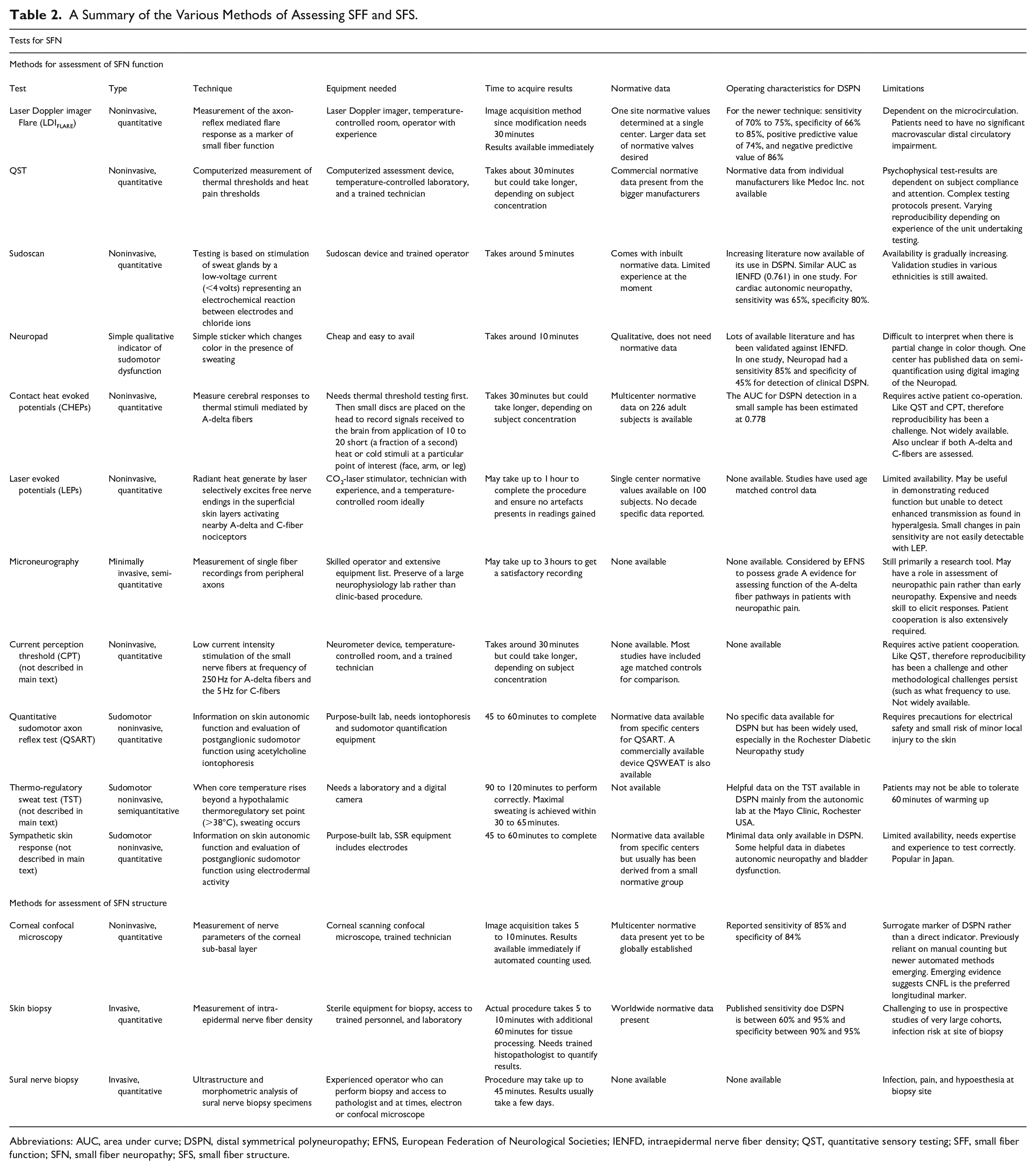
Abbreviations: AUC, area under curve; DSPN, distal symmetrical polyneuropathy; EFNS, European Federation of Neurological Societies; IENFD, intraepidermal nerve fiber density; QST, quantitative sensory testing; SFF, small fiber function; SFN, small fiber neuropathy; SFS, small fiber structure.
Methods of Assessing SFF
Laser Doppler imaging flare (LDIFLARE)
The LDIFLARE is a noninvasive technique of measuring C fiber-mediated cutaneous vasodilatation in the foot skin in response to thermal heating. The heating is done by a 1 cm2 probe and the area of induced hyperemia measured by a 610 nm laser probe. Validation studies have suggested a moderate to strong correlation with IEFND) intraepidermal nerve fiber density, 13 and in-vivo corneal confocal microscopy (CCM) 14 for the detection of DPN. The LDIFLARE is progressively reduced with increasing severity of DPN 15 and has also been noted to be impaired in subjects with impaired glucose tolerance (IGT) with normal thermal thresholds and large fiber assessments. 16 In a cohort of newly diagnosed T1DM subjects, following optimization of glycemic control, a significant increase in the LDIFLARE response was noted, suggesting that it is not only a sensitive method of assessing SFN, but importantly also a versatile measure of short-term fluctuation in SFF affected by metabolic milieu, for example, glycemic improvement or deterioration. 17 In a study of young subjects aged 12 to 16, the LDIFLARE method showed significant SFF reduction as compared to healthy and correlation with HbA1c change. 18 However, it takes around 30 minutes per patient for evaluation, which could be a limiting factor in its applicability in the busy clinical setting. 19 More longitudinal data are required to support its use as a routine tool for the assessment of diabetes-related SFN. It has been shown in other non-diabetes studies to be a sensitive investigation to measure SFN in hypothyroidism, 20 chemotherapy-induced peripheral neuropathy, 21 and hypertriglyceridemia. 22
QST for thermal and pain thresholds
Use of QST is a valuable method for assessing SFF and has been extensively used in both research and clinical settings, including assessment of peripheral neuropathy, acute and chronic pain. The various available devices of QST fundamentally determine the sensory and pain thresholds for cold and warm temperatures (thermal thresholds), and some devices also quantitate vibration perception a large fiber function. Examples include the Computer Aided Sensory Evaluator-IV (CASE-IV) and Medoc TSA-II NeuroSensory Analyzer. In a study of 498 T2DM and 434 healthy subjects, Chao et al 23 showed that an elevated warm threshold was the most frequent abnormality in the T2DM group (60.2%), compared with an abnormal cold threshold (39.6%) and an abnormal sural nerve conduction velocity (12.9%); it was also shown to be related to both symptoms and glycemic control. However, other studies have shown that unlike cold perception thresholds and IENFD, warm perception thresholds did not differentiate people with diabetes with and without symptoms. 24 Similarly, in a study of 191 people with diabetes, there was no difference in heat-pain. In a recent prospective T2DM study of 725 patients, 25 Sheen et al 26 showed there was a high prevalence (74.3%) of subclinical abnormal thermal thresholds (both warm and cold detection thresholds) measured by the Q-Sense Thermal Sensory Analyzer. These were significantly associated with peripheral arterial disease and nephropathy in their cohort population.
However, methods of QST have two important limiting factors. First, they need patient cooperation and can be adversely affected by patient-related altered motivation, inattention, and cognitive issues; second, if subjects have neurological abnormalities such as allodynia/hyperpathia, aberrations of temporal latency, or spatial localization, then QST outcomes can be significantly affected. This may explain why QST techniques demonstrate a modest correlation with IEFND. 27 Newer updates like the Medoc TSA-II have been able to mitigate some of these limitations; however, the psychophysical aspect of the tests is a non-modifiable limitation. Furthermore, there are very limited data on whether QSTs are able to detect small to modest degrees of improvement in SFF, a crucial aspect for suitability of technique for use in neuropathy intervention trials.
Assessment of sudomotor function
The assessment of sudomotor function as a marker for SFN is based on the principle that the degeneration of small nerve fibers reduces sweat gland innervation leading to an impairment in sweating. Based on this, several portable devices are now available, which allow rapid evaluation of sudomotor function. The Neuropad, a plaster comprising of an anhydrous cobalt II compound applied on the plantar forefoot, is centered on the principle of color change from blue to pink when in contact with sweat. 28 In a study of 127 subjects of both T1DM and T2DM subjects, Ponirakis et al 29 have shown that the Neuropad has good sensitivity and specificity for the detection of DPN in those with established clinical neuropathy comparing favorably with established modalities including CCM, NCV, and autonomic tests. The test can currently provide qualitative data only. The Sudoscan provides a quantitative measure of chloride conductance (measured as electrochemical skin conductance [ESC]) serving as a biomarker to assess sweat gland function in relation to sweat gland innervation.30,31 Studies have reported that the device has good correlations with DPN clinical scoring systems, large fiber methods like NCS, VPT, and tests for cardiac autonomic nervous system.30,32,33 However, in a study of 63 patients, Duchesne et al 34 showed that ESC measurement has a weak correlation with sweat gland density and IENFD in skin biopsies, prompting the speculation that mechanisms other than loss of innervating fibers may be responsible for sudomotor dysfunction in DPN.
An older method of assessing sudomotor function is the qualitative sudomotor axon reflex testing (QSART), which evaluated sudomotor function by assessing the local sweat response to iontophoresis of acetylcholine 35 and has been shown to be highly sensitive in the detection of distal SFN. 36 It is however time consuming and requires specialist equipment and laboratory access.
Evoked potentials
Nociceptive laser-evoked potentials (LEP) and contact heat-evoked potentials (CHEPs) are electrophysiological methods to detect functional impairment of small fibers in DPN. 37 However, LEP and CHEPs have drawbacks in clinical routine such as the need for expensive equipment and complex procedural algorithms. 38 Furthermore, reproducibility of these techniques is not fully established, 39 and it remains to be determined which of the variables measured—amplitudes, latencies, or areas—robustly differentiate and follow the course of SFN.39,40 The recording of electrically elicited pain-related evoked potentials (PREP) via concentric electrodes is a useful, noninvasive method to detect early SFN. PREP involves delivering a nociceptive electrical stimulus to the skin using a concentric surface electrode, which imparts a high current density with relatively low intensity, limiting the depolarization only to the nociceptive fibers, which is then detected via standard recording electrodes placed at C2. 41 Mueller et al, in a study of 35 patients with and 22 without painful neuropathy, showed that despite normal electrophysiology, PREP could detect increased latency and reduced amplitude in patients with painful neuropathy. 42 In a recent study by La Cesa et al, 43 LEP demonstrated excellent sensitivity and specificity with abnormal pinprick sensitivity and capsaicin-induced epidermal denervation prompting the authors to speculate that unlike LEPs and CHEPs, PREPs are not selectively mediated by nociceptive system. We believe that these techniques are still in their development phase, and further studies are required to outline their use in the assessment of SFN.
Microneurography
Microneurography is a minimally invasive neurophysiological technique allowing direct recording of unmyelinated postganglionic sympathetic or afferent nociceptive fibers function by inserting tungsten needles into a peripheral nerve fascicle.44,45 Orstavik et al 46 proposed that based on microneurographic studies, change in the distribution of C-nociceptive fibers in the skin as shown in the patients with diabetic neuropathy may help to reveal mechanisms responsible for small-fiber dysfunction. This technique has failed to gain traction beyond research uses since it is very time-consuming as well as requires specialist skill. Its strength is in its unique power for exploration of normal neural mechanisms as well as pathophysiological conditions of various neurological disorders. There have also been reports of nerve injury during this procedure. 47
Methods of Assessing SFS
Corneal confocal microscopy (CCM)
In the last two decades, CCM has emerged as the SFN assessment technique with the largest body of evidence to support its use in DPN. The procedure is noninvasive and relatively quick (taking three to five minutes in experienced hands), well tolerated by patients and provides in-vivo quantification of the sub-basal corneal nerve plexus (in-vivo). 48 Its portability makes it an attractive “add-on option” for neuropathy screening when diabetes subjects have their annual retinal surveillance. 49
CCM will be discussed in further detail in another section of this special supplement. It has been studied in most forms of diabetes in both cross-sectional and a few longitudinal studies. CCM has been shown to be predictive of development of future DPN, 50 correlates with severity of DPN, 51 and with autonomic diabetes neuropathy. 52 Finally, in a smaller study of 38 T1DM subjects, Edwards et al 53 showed that CCM demonstrated the greatest, and most sustained degeneration over a four-year period as compared to NCS, hot and cold thermal thresholds, VPT, 10 gm MF and neuropathy scores.
Skin biopsy
Skin biopsy is considered as the “gold-standard” for the diagnosis of SFN, and most methods already mentioned have to be validated against it for accuracy. It is a minimally invasive procedure using a disposable 3-5 mm punch biopsy device to sample the skin and the pan axonal marker PGP-9.5 to identify the small nerve fibers, which can then be quantified and expressed as the number of IENF per length of section (IENF/mm).54,55 It demonstrates good intra and inter-observer validity, 56 but like many SFN measures reduces with age. 57 This led to an international consortium of investigators to compile an age-related normative values for IENFD. 58 The technique has now been further adapted to specifically quantify dermal nerve fibers, autonomic innervation of the sweat glands, and pilomotor muscles. 59 One limitation is that it needs a specialized laboratory with staff trained to use specific neuronal markers. Repeated measures in study subjects may also be challenging, and certainly this aspect makes it less practical for deployment as a routine clinical test for annual DPN assessments. However, it is likely to remain the “gold-standard” for SFN assessment in research practice. 60
Nerve biopsy
This is a highly specialized invasive procedure traditionally used to quantify myelinated nerve fiber density, which is reduced in DPN and correlates with NCS.61,62 It has also been shown to be beneficial for early detection of DPN. 8 However, due to its invasive nature, requirement of neurosurgical expertise especially when fascicular biopsy is necessary, and electron microscopic interpretation, it cannot be advocated to be useful in the routine assessment of SFN in diabetes. Its mention in this section is for completion of the list of methodologies available.
SFF or SFS or Both?
In the past decade, there have been significant refinements of the methodologies for assessing SFN as described. However, there are several key questions that need to be answered by each of these methodologies before their acceptance into a wider clinical practice:
Adoption of a test to be used in a busy clinical setting requires it to be simple and quick to perform with good reproducibility. In this context, point of care devices for sudomotor function like the Neuropad and SUDOSCAN fit the above attributes. Whilst the Neuropad shows good correlation with CCM, NCV, and autonomic tests, it can only provide binary data, 29 and it is unclear how patchy/limited change on the pad should be interpreted. ESC measured by Sudoscan has a weak correlation with sweat gland density and IENFD in skin biopsies. 34 CCM appears to fulfil most criteria, in particular its noninvasive nature, rapid image acquisition, and potential scalability into large screening/assessment programs but costs around $30 000 per instrument, and there is the recurring cost of consumables including $5 per tomocap per patient and other topical medicines. The LDIFLARE and QST have robust clinical evidence to support their use but take longer to perform, require access to a laser doppler imager, and experienced operators, and hence, probably unsuitable for outpatient use. As it stands, there is a lack of an inexpensive, quantitative method to detect SFN in the current clinical settings.
This is a question of fundamental importance, both for research and clinical settings. The two methodologies that are sensitive to detecting changes in small fiber responses to changing metabolic milieu are the LDIFLARE and CCM. Sharma et al 17 have shown that in newly diagnosed people with LDIFLARE T1DM, SFF assessed by the LDIFLARE, and CCM techniques at diagnosis and then at six months after the commencement of insulin and stable glycemic control, there was a significant improvement but nonsignificant change in CCM indices at six months. This study suggests that in people with T1DM, SFF assessed by the LDIFLARE is more sensitive to short-term changes in glycemia than SFS assessed by CCM. 17 Hassler-Hurst et al 18 similarly showed that a change of HbA1c as measure of glycemic control significantly inversely correlated with LDIFLARE size in young diabetes subjects aged 12 to 16. Azmi et al 63 reported that CCM detects SFN in subjects with prediabetes who go on to develop T2DM and found a dynamic worsening or improvement in corneal indices in relation to change in glucose tolerance status. Corneal regeneration evidenced by CCM has also been demonstrated by Tavakoli et al 64 in patients with T1DM undergoing simultaneous pancreas and kidney transplantation corneal nerve regeneration within six months, whereas other parameters including IENFD, QST, and NCS did not improve. There is also good evidence in support of IENFD in tracking relatively short-term changes in small fiber structure; however, the invasive nature of the technique limits its adoption into routine clinical use where repetitive monitoring would be required. Most of the other techniques do not have studies exploring their sensitivity and accuracy in response to diabetes related fluctuations; therefore, it is difficult to comment on their ability to detect meaningful improvements or decrements.
Is There a Strong Argument Now for Routine Use of SFN Techniques in the Early Diagnosis of DPN and Its Follow-Up?
The current ADA position statement on DPN advocates the following: Assessment for distal symmetric polyneuropathy should include a careful history and assessment of either temperature or pinprick sensation (small-fiber function) and vibration sensation using a 128-Hz tuning fork (for large-fiber function). All patients should have annual 10-g monofilament testing to identify feet at risk for ulceration and amputation.1,65
It is noteworthy that despite the growing body of evidence of early involvement of small fibers in diabetes, the ADA remains reserved on use of modern small fiber techniques to diagnose early neuropathy. The same approach is endorsed by National Institute for Health and Care Excellence (NICE-UK) and European Association for the Study of Diabetes (EASD), which focus on the assessment of loss of protective sensation. 66 In the initial stages, the majority will have normal temperature or pinprick sensation, vibration sensation using a 128-Hz tuning fork, and 10-g monofilament testing. In such an instance, it would seem reasonable to consider the use of tests that will detect neuropathy at its earliest stages. Non-invasive tests like CCM and LDIFLARE have shown this potential but need to be more practical and less expensive for use in clinical envirorment where the focus is on early detection of complications. Annual screening for diabetic retinopathy with digital retinal photography and measurement of urinary microalbuminuria for detection of those at risk of progressing to significant diabetes nephropathy is now an established practice with proven clinical benefit.64,67 Furthermore, the data from such screening have proven important in facilitating the development of therapies that can reduce the decline renal function (eg, Ramipril 68 or more recently the use of sodium-glucose co-transporter-2 [SGLT2] inhibitors).69,70 In contrast, to date, none of the methods for assessing SFS or SFF have been introduced into clinical practice or employed into research for the development of new therapies.
What Are the Challenges That Have Precluded the Adoption of SFN Methods for the Routine Surveillance of DPN?
There may be several reasons why the expert group has not included the assessment of SFN into routine guidelines for the assessment of DPN. First, they have suggested that these methods need further clinical validation before their incorporation.71,72 Second, there are only a few longitudinal studies of the natural history of SFN. A MEDLINE search of this area found 784 citations in the last 10 years of which the majority were cross-sectional. Only six were longitudinal with the longest study duration being 5.3 years. The latter is a multicenter study of 399 T1DM and 191 T2DM patients followed up over 4.4 and 5.3 years, respectively. This important study showed that loss of corneal nerve fiber length (CNFL) of 6% per year occurs in 17% of the study population and “may identify patients at the highest risk of development of DPN.” 73
Another important limiting factor appears to the lack of “established” normative ranges, which would otherwise make its interpretation easier in the clinical world. Unlike IEFND and CCM, which have established worldwide normative ranges,57,74 other SFN methodologies still lack globally agreed normative range for comparison. Furthermore, there is disagreement on which of the variables measured during testing are best suited for clinical adoption and if they vary between the types of diabetes. For example, using pooled data of 998 subjects, Perkins et al 75 have suggested that the optimal threshold for automated CNFL was 12.5 mm/mm2 in T1DM and 12.3 mm/mm2 in T2DM, with a lower and upper threshold values between eight and six and 15.3 mm/mm2 rule out DPN with 88% specificity and 88% sensitivity. However, the authors conclude that “further research must determine to what extent CNFL can be deployed in clinical practice and in clinical trials assessing the efficacy of disease-modifying therapies for DSP,” suggesting that such cutoffs need further validation. Since the publication of this study, more evidence has been published, which supports that normative data set should be age-linked due to age-related decline in SFF and SFS in healthy individuals. 14 So, whist the existing methodologies are effective in smaller cross-sectional studies as biomarkers to improve our understanding of DPN, the lack of widely accepted normative data across might be a major drawback against their widespread use in a clinical environment.
Another important confounder is that protocols have changed over time, making comparisons with previous studies difficult. For example with CCM, although the majority of DPN studies are conducted by investigators based on protocols first established by Malik et al 76 and use the same automated software (Accumetrics) for image interpretation,77-79 these protocols continue to be updated and Petropoulos et al 80 commented that in addition to corneal confocal nerve fiber density (CNFD), nerve branch density (CNBD) and length (CNFL), addition of the inferior whorl length (IWL) may further improve the diagnostic performance of the method. With other investigators, there is considerable heterogeneity on both instruments and protocols involved. 81
Conclusions
The current approach to the assessment of DPN needs urgent reconsideration. Within the minds of many health care practitioners, including most diabetes specialists, determining whether an individual in their clinic has neuropathy involves screening using 10-gm SWMF, or a tuning fork, or even a patellar hammer. These are important as they help the practitioner determine whether the person is “at-risk” of foot ulceration and then to put in place strategies to prevent ulceration and amputation. However, we should be striving to detect the earliest stages of neuropathy during annual screening in line with what is routine for nephropathy and retinopathy. Given that we now have accurate tools to detect the earliest stages of this potentially devastating condition, well before the patient has lost protective sensation, these methods should be refined so that they can be incorporated as part of the annual screening procedure. Adoption of these SFN methodologies might be instrumental to design interventions like aggressive blood pressure or lipid management or even newer drug treatments, which could delay onset or progression of DPN.
We hope that this review will prompt further discussion by global experts on the incorporation of SFN assessment as routine care of DPN.
Footnotes
Acknowledgements
The authors would like to thank the colleagues at the Diabetes Research Unit and staff colleagues at the Diabetes Center, Ipswich Hospital for their continued support and facilitation of research endeavors.
Abbreviations
ADA, American Diabetes Association; CCM, corneal confocal microscopy; CNBD, corneal nerve fibre density; CNFD, corneal nerve fibre density: CNFL, corneal nerve fibre length; DPN, diabetic polyneuropathy; DSP; diabetes symmetrical neuropathy; EFNS, European Federation of the Neurological Societies; HbA1c, glycated hemoglobin A1c; HC, healthy controls; IENFD, intraepidermal nerve fiber density; IGT, impaired glucose tolerance; LDIFLARE, laser doppler imaging flare; NCS, nerve conduction studies; QST, quantitative sensory testing; SFN, small fibre function; SFN, small fibre neuropathy; SFS, small fibre structure; SMWF, Semmes-Weinstein monofilament; T1DM, type-1 diabetes mellitus; T2DM, type-2 diabetes mellitus; VPT, vibration perception threshold.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
