Abstract
Diabetes distal symmetrical peripheral neuropathy (DSPN) is the most prevalent form of neuropathy in industrialized countries, substantially increasing risk for morbidity and pre-mature mortality. DSPN may manifest with small-fiber disease, large-fiber disease, or a combination of both. This review summarizes: (1) DSPN subtypes (small- and large-fiber disease) with attention to clinical signs and patient symptoms; and (2) technological diagnosis and screening for large- and small-fiber disease with inclusion of a comprehensive literature review of published studies from 2015-present (N = 66). Review findings, informed by the most up-to-date research, advance critical understanding of DSPN large- and small-fiber screening technologies, including those designed for point-of-care use in primary care and endocrinology practices.
Keywords
Introduction
Distal symmetric peripheral neuropathy (DSPN) is the most common form of diabetes neuropathy, affecting 50% of adults with diabetes during their lifetime. DSPN afflicts the feet, and less frequently, the hands.1-4 In addition to severe pain, imbalance, and associated risk for fall, a highly concerning sequela of DSPN is foot ulceration.5,6 Foot ulceration confers heightened risk for gangrene and lower extremity amputation. Lower extremity amputations increase risk for mortality with a 5-year survival rate of 40%–48% post-amputation. 7 The economic cost of foot ulcerations and amputations is exceptional, with the cost of foot ulcerations due to diabetes alone estimated at $9-$13 billion annually in the United States. 8 Hence, development of reliable, accurate technological methods to detect DSPN, particularly in the early course of its development (even in asymptomatic stages), is critically needed to slow DSPN progression with prompt interventions to prevent or decrease adverse outcomes and contain healthcare costs.9-16
The pathogenesis of diabetes DSPN is not fully understood but is believed to involve multiple mechanisms.4,17,18 There is strong evidence suggesting that diabetes DSPN is caused by hyperglycemia-induced nerve fiber injury and microvascular ischemia with ensuing nerve fiber degeneration and loss. Hyperglycemia causes direct nerve toxicity through several pathways, such as increased oxidative stress, advanced glycation end-product accumulation, and impaired axonal transport, thereby precipitating nerve degeneration.4,19 At the same time, long-standing hyperglycemia effects the microvasculature, causing nerve ischemia & degeneration. 20 Hypertension, dyslipidemia, elevated body mass index, and smoking may also contribute to DSPN incidence.21-26
DSPN is a progressive condition with no cure. Hence, timely diagnosis of DSPN is essential to prevent or mitigate DSPN-related morbidity and mortality. Prompt diagnosis of DSPN is indicated in prediabetes, new-onset T2D, and well-established T2D and T1D. Among those with prediabetes, DSPN may afflict 10%-30% or more, indicating neuropathic damage may occur at glucose levels not meeting the threshold for diagnosis of T2D.4,27 DSPN prevalence, among patients with newly diagnosed T2D, is estimated at 21%. 28 After 10 years of T2D duration, DSPN rates climb exponentially to 50%. 4 Among patients with T1D, DSPN rates are or exceed 20% after 20 years of disease duration.15,29 Since DSPN rates increase dramatically with longer diabetes duration, early and ongoing surveillance is warranted. Risk reduction strategies for DSPN and its sequelae are optimization of blood glucose, blood pressure, and lipid levels in addition to healthy lifestyle behaviors (daily foot care, weight loss, increased physical activity, smoking cessation).4,21-26,30-33
On the frontline diagnosing and managing prediabetes and diabetes, primary care providers play a critical role in early detection of DSPN. Yet, in primary care, research indicates DSPN may be underdiagnosed or not diagnosed promptly with underutilization of diagnostic tests and sometimes misperceptions about causes or management.34-36
Hence, this study examines: (1) DSPN subtypes (small- and large-fiber disease); (2) DSPN screening with attention to guidelines for clinical diagnosis; and (3) well-established diagnostic and more recent, innovative advances in technological screening for DSPN subtypes with attention to performance, reproducibility, and longitudinal outcomes data. Currently, comprehensive, up-to-date reviews on technologies and related advancements for DSPN screening and diagnosis are lacking.
Study inclusion criteria were: (1) examination of technological diagnosis or screening (specifically, nerve conduction study, quantitative sensory testing, contact heat evoked potentials, corneal confocal microscopy, quantitative sudomotor axon reflex testing, and electrochemical skin conductance) approaches for DSPN; (2) related investigation of diagnostic or screening performance, reliability or validity testing, automation procedure, and/or longitudinal outcomes in detecting DSPN; (3) inclusion of adult participants with T1D, T2D, impaired glucose tolerance (IGT), or impaired fasting glucose (IFG); (4) published in English; and (5) published between 2015-April of 2021. Study exclusion criteria were: (1) exclusive use of a descriptive and/or correlational design; (2) investigation of technology in performing the routine foot exam; (3) exclusive examination of DSPN predictors (eg, demographic and clinical factors); (4) intervention studies; (5) conference abstracts; (6) dissertations; and (7) review articles.
We conducted a literature search using the following search engines: PubMed, Scopus, and MEDLINE. The search strategy was conducted by separately entering key search terms (nerve conduction study, quantitative sensory testing, contact heat evoked potentials, corneal confocal microscopy, quantitative sudomotor axon reflex testing, and electrochemical skin conductance) with each term, respectively, accompanied (one at a time) by AND diabetes, AND type 1 diabetes, AND type 2 diabetes, AND prediabetes, AND impaired glucose tolerance, or AND impaired fasting glucose. Then, the key search terms, respectively, were accompanied, individually, by AND peripheral neuropathy AND diabetes; AND peripheral neuropathy AND type 1 diabetes; AND peripheral neuropathy AND type 2 diabetes; AND peripheral neuropathy AND prediabetes; AND peripheral neuropathy AND impaired glucose tolerance; or AND peripheral neuropathy AND impaired fasting glucose. The literature search yielded 66 studies for analysis (see Figure 1).
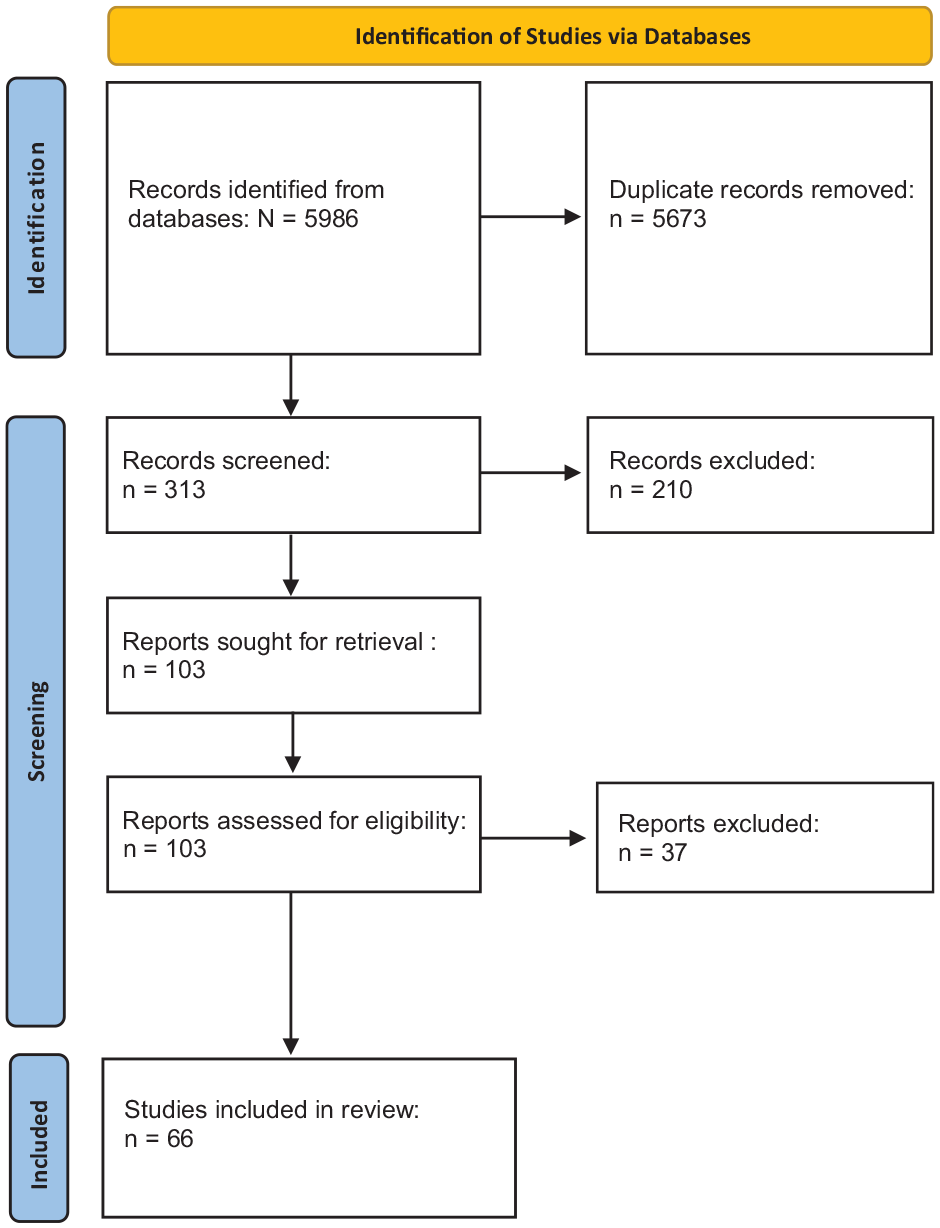
PRISMA Flow Diagram.
DSPN Subtypes
The subtypes of DSPN are small- and large-nerve fiber disease. Small- and large-nerve fiber DSPN may present exclusively or together while each subtype may increase risk for foot ulceration due to reduced sensory function, and thereby heightened risk for lower extremity amputations. 4 Small-fiber DSPN typically precedes large-fiber neuropathy. Small-fiber DSPN impairs functional integrity of the small thinly myelinated Aδ and unmyelinated C fibers. These small, peripheral nerve fibers prominently convey pain to the central nervous system. In DSPN, they may stimulate profound pain. 3 Small-fiber DSPN may also adversely affect local autonomic (eg, decreased sweating, dry skin, impaired vasomotion) and thermoreceptor (cold, warm sensations) functions.3,37,38
Often, pain and other symptoms and signs (see Table 1) first manifest in the feet and progress proximally to the lower extremities and, in some cases, to the hands with a stocking and glove pattern. However, some with small-fiber DSPN may not experience pain.39,40 A proportion of patients with small-fiber neuropathy may present with little evidence of the disease, which may delay DSPN diagnosis.41-42
Symptoms & signs of small & large fiber DSPN. 3

Large-fiber disease refers to impairment of Aα and/or Aα/β fibers. These fibers are large myelinated fibers. Aα fibers control motor functions and muscle control while Aα/β fibers are related to sensory functions. 3 In DSPN, damage to Aα fibers may manifest with muscle weakness, painful cramps, among other symptoms. 40 Aα/β fibers are implicated in reduced perception to touch, vibration, balance, and position as well as pain (see Table 1).3,42 Damaged large Aα/β fibers may increase risk for falls and fractures with reduced or absent sensory input for the control of movement.3,43
DSPN Screening and Diagnosis
The American Diabetes Association (ADA) recommends screening for DSPN to promote early interventions (eg, glycemic control and lifestyle modifications), particularly given treatments targeting the underlying nerve damage are lacking. Screening should commence upon the diagnosis of T2D and at least annually thereafter. In the setting of prediabetes, screening may be considered although research suggests 11%-25% of this population may exhibit DSPN and 13%-26% may present with neuropathic pain.4,44 The ADA suggests, after exclusion of other causes, a diagnosis of DSPN may be based on a supportive patient history plus assessment of either gross temperature or pinprick sensation (small-fiber function) and vibration sensation using a 128-Hz tuning fork (large-fiber function). An annual 10-g monofilament test is recommended to assess for risk of foot ulceration and amputation. According to the ADA, the clinical history and physical examination often are sufficient for diagnosis of DSPN. Yet, up to 50% of individuals with DSPN may be asymptomatic. 4 Also, screening methods may not yield valid data to detect early or subclinical stages of DSPN and/or diagnose DSPN in diverse populations.42,45-48 Therefore, additional DSPN testing may be warranted.
Large-Fiber DSPN Diagnostic Testing and Screening
Nerve Conduction Study (NCS)
NCS is considered the gold standard for diagnosing large-fiber neuropathy. NCS has documented sensitivity and specificity, ranging, for example, from 40%-81% and 91%-95%, respectively, for DSPN.49-50 NCS measures the rate at which an electrical impulse travels through nerves, such as the sural sensory, peroneal motor, and tibial nerves. Two electrode patches are placed on the nerve being testing with one electrode stimulating the nerve and the other measuring the electrical impulse. Given NCS findings are complex, data are interpreted by health professionals with requisite advanced training. 3
Conducted from 2015-present, the literature search on NCS yielded 10 studies that revealed technological advances (see Table 2).51-60 In particular, a point-of-care device (POCD), DPN-Check (Neurometrix, Waltham, MA, USA), was identified, which rapidly reproduces part of the NCS (sural nerve conduction).61 Overall, studies reveal DPN-Check may be a highly promising automated technology while allowing increased access to DSPN testing and limited clinician technological expertise in its application.51-60 DPN-Check is a handheld device placed on the lateral aspect of the lower extremity (posterior to the lateral malleolus), following the same measurement principles as standard NCS. 61 Sampling T1D and T2D populations, studies tend to report good to excellent sensitivities (80%-96%) and specificities (80%-97%) for DPN-Check in detecting DSPN and its severity (see Table 2).51,52,55-59 In comparing NCS to DPN-Check, data reveal intraclass correlation coefficients are excellent overall (intrarater reliability: velocity 0.77, amplitude 0.81; interrater reliability: velocity 0.97, amplitude 0.83). 60
Point-of-Care Sural Nerve Conduction.

Abbreviations: Amp, amplitude; AMP, amplitude potential; AUC, area under the curve; AUROC, area under the receiver operator characteristic; CV, conduction velocity; DM, diabetes mellitus; DN, diabetic neuropathy; DPN, diabetic peripheral neuropathy or diabetic polyneuropathy; DSPN, diabetic symmetric sensorimotor polyneuropathy; eMBC, estimated severity in modified Baba classification; EMGS, electromyography study; HC, healthy control; IQR, interquartile range; JRF, Japanese regression formulas; LDIFLARE, laser doppler image flare; M, mean; Med, median, m/s, meter per second; MBC, modified Baba classification; NCA, nerve conduction abnormality; NCS, nerve conduction study; NDS, neuropathy disability score; POCD, point-of-care-device; ROC, receiver operator characteristic; SN, sural nerve; SNAP, sensory nerve conduction potential; SNC, sural nerve conduction; SNCV, sensory nerve conduction velocity; T1D, type 1 diabetes; T2D, type 2 diabetes; USRF, regression formulas of individuals from USA; µV, microvolts; yrs, years
Among Japanese and possibly other East Asian populations, review findings indicate US normative ranges for DPN-Check parameters, particularly sural nerve action potentional amplitude, may not be valid. Hirayasu et al. propose a promising Japanense regression formula to promote accurate DSPN assessment with DPN-Check. 54
Small-Fiber DSPN Diagnostic Testing
Intraepidermal Nerve Fiber Density Testing (IENFD)
IENFD testing is the gold standard for diagnosing small-fiber DSPN. IENFD is an invasive procedure wherein a punch biopsy is obtained from the distal leg for quantification of small-fiber densities. Small-fiber DSPN is identified by reduced intraepidermal nerve fiber density or morphological changes.62,63 IENFD has variable sensitivities and specificities, ranging, for example, from 78%-88% and 64%-90%, respectively. 62 Varied diagnostic accuracy may be partially attributed to IENFD cutoff and reference values used in assessing for small-fiber neuropathy as well as specific location of the biopsy site. Normative values require adjustment for age and gender or results may likely be biased.62,64-65 A number of studies suggest IENFD values are affected by ethnicity, which may also bias results as revealed in Asian populations.66-68
Quantitative Sensory Testing (QST)
QST assesses large- and small-fiber function. With respect to small-fiber function, QST technologies assess the thresholds at which small nerve fibers detect pain and thermal sensations. In suspected DSPN, a thermode is placed on the foot and/or hand areas with pain and/or sensory deficits. Patients are instructed to quickly respond to changes in specified sensations, which yield mean peak values or thresholds for pain and thermal sensations. QST may thus be conceived as a psychophysical test wherein sensation stimuli are controlled and stimuli responses are dependent on the active participation of the patient. QST is thus susceptible to bias related to patient motivation, attention, or cognitive impairment. Interpretation of QST findings requires attention to the clinical context while data is best compared to normative data stratified for age, gender, and body site. QST is not recommended as a stand-alone diagnostic test given variable study results and testing methodologies.69-70 Compared to IENFD and clincial examination, published reports document the sensitivity of thermal testing ranges from 36%-100% for small-fiber neuropathy although diagnostic criteria for the condition were not uniform across studies. 70
Since 2015, the literature search identified thirteen studies comparing, for instance, different QST devices and evaluating new methodologies or technological approaches (see Table 3).71-83 NerveCheck (Phi Med Europe SL, Barcelona, Spain), in particular, reveals technological advancement of traditional QST. NerveCheck is a small, portable device, and yields rapid results. NerveCheck assesses vibration (VPT), cold (CPT) and warm (WPT) perception thresholds, and heat pain threshold. NerveCheck has demonstrated an intraclass agreement for VPT (large-fiber vibration testing), CPT (small-fiber thermal testing) and WPT (small-fiber thermal testing) at 0.79, 0.86, and 0.71, respectively. 82 The diagnostic accuracy of NerveCheck in detecting sensory loss, based on the area under the curve (AUC), is reportedly 0.70 for CPT and 0.69 for WPT with IENFD as the reference. 83
Quantitative Sensory Testing (QST).

Abbreviations: ACVT, autonomic cardiovascular tests; AUC, area under the curve; AUROC, area under receiver operator characteristic; CCM, corneal confocal microscopy; CDT, cold detection threshold; CIP, cold induced pain; CPT, cold pain threshold or cold perception threshold; CST, cold sensation threshold; CT, cold threshold; DM, diabetes mellitus; DNP, diabetic neuropathy; DPN, diabetic peripheral neuropathy; DSP, diabetic sensorimotor polyneuropathy; DSPN, diabetic symmetrical peripheral neuropathy; ESC, electrochemical skin conductance; FU, follow-up; HC, healthy control; HPT, heat pain threshold; HRV, heart rate variability; ICC, intraclass correlation coefficient; IENFD, intraepidermal nerve fiber density; IGT, impaired glucose tolerance; IQR, interquartile range; LDIFLARE, laser doppler imager flare; LEP, laser evoked potentials; M, mean; Med, median; NCS, nerve conduction study; NGT, normal glucose tolerance; QST, quantitative sensory testing; ROC, receiver operator characteristic; SFN, small fiber neuropathy; SNAP, sensory nerve action potential; SNCV, sural nerve conduction velocity; T1D, type 1 diabetes; T2D, type 2 diabetes; TSA, Thermal Sensory Analyzer; TSL, thermal sensory limen; VPT, vibration perception threshold; WDT, warm detection threshold; WIP, warm induced pain; WPT, warm pain or perception threshold; WST, warm sensation threshold; WT, warm threshold; yrs, years.
More well established, the TSA NeuroSensory Analyzer (Medoc, Ramat Yishai, Israel) has advanced over the years with development of a precision enhanced, small portable device for QST; i.e., the TSA-II NeuroSensory Analyzer. Using the TSA-II, Farooqi and colleagues advanced the science of QST by validating cooling detection thresholds (CDT) to detect DSPN in a sample of participants with T2D. While not assessed against IENFD, CDT was found to outperform other measures of DSPN. CDT had acceptable discriminatory ability for detecting clinical DSPN (AUC 0.79) with a sensitivity of 64% and specificity of 83%. Good discriminatory performance for pre-clinical DSPN (AUC 0.80), with a sensitivity of 83% and specificity of 72%, was also observed. 77
Contact Heat Evoked Potentials (CHEPs)
CHEPs examine the integrity of Aδ and C fibers by measuring cerebral responses to thermal pain stimuli. A heat evoked potential stimulator is used with a thermode placed on the lower extremity. A heat pulse is delivered from a baseline to increasing safe, warm temperatures. Heat stimuli are repeatedly applied to the same lower extremity area. Concurrently, CHEPs are recorded with electrodes placed on the vertex of the head. An evoked potential system generates waveform tracings reflecting responses in the cerebral cortex.3,84-86
The literature search identified one study meeting eligibility criteria for inclusion in the review of CHEPs. 87 The study sample was comprised of 255 adults with 188 diagnosed with neuropathy (38.5% had diabetes) and 57 controls. Tests of equality were performed to assess the area under the receiver operator characteristic (AUROC) curves with respect to the diagnostic accuracy of CHEPs (Medoc, Ramat Yishai, Israel) versus QST (TSA NeuroSensory Analyzer, Medoc, Ramat Yishai, Israel). Results revealed that CHEP amplitudes (AUC 0.79) had significantly greater accuracy realtive to the QST warm (AUC 0.71) but not cold (AUC 0.72) threshold for diagnosis of small-fiber neuropathy. Using a cutoff of 29.1 mV, CHEPs had a diagnostic sensitivity of 80.7% and specificity of 68.8% for DSPN. 87
Corneal Confocal Microscopy (CCM)
The cornea is abundantly innervated by small thinly myelinated Aδ and unmyelinated C fibers, and thereby uniquely provides indirect assessment and quantification of small-fiber DSPN. CCM has emerged as a noninvasive, valid, reliable technique for assessing small-fiber DSPN.88-90 CCM involves visualization, via image acquisition, of corneal microstructures. Analysis of corneal images allows for detection of small nerve fiber loss with assessment of corneal nerve fiber density (CNFD), nerve branch density (CNBD), and nerve fiber length (CNFL), among other corneal parameters. 91
Since 2015, the literature search identified 32 studies meeting study criteria, including technological advancements of CCM (see Table 4).72-73,75,78,81,92-118 Advancing CCM technology, automated quantification of corneal images was frequently examined with respect to its accuracy, validity, and reliability (reproducibility and repeatability) with very promising results reported.72,93-98,104,105,107,109-110,113-115,118. Compared to traditional manual methods, automated methods were found to be relatively equivalent with overall high levels of agreement.92-94,96-98,107,110,113-115,118 With manual or automated methods for image quantification, CCM parameters tended to have moderate to excellent accuracy (as assessed by AUROC or AUC values) in detecting DSPN, including its levels of severity. However, sensitivies and specifities for CCM parameters tended to be highly variable and not uniformly adequate across studies.72-78,95-98,103,110-112,116-117 Beginning evidence suggests CCM and IENFD may have roughly comparable performance in detecting DSPN although additional research is warranted.72,96
Corneal Confocal Microscopy (CCM)

Abbreviations: AAC, adjusted area calculation; ACNBD, automated corneal nerve branch density; ACNFD, automated corneal nerve fiber density; ACNFL, automated corneal nerve fiber length; ACNFrD, automated corneal nerve fiber fractal dimension; AMC, age-matched controls; AMHCs, age-matched healthy controls; ANFL, average nerve fiber length; AUC, area under the curve; AUROC, area under the receiver operator characteristic; BD, beading density; CC, central cornea; CCM, corneal confocal microscopy; CG, control group; CN, corneal nerve; CNBD, corneal nerve branch density; CNBDA, corneal nerve branch density with automated analysis; CNBDFA, corneal nerve branch density with fully automated analysis; CNBDM, corneal nerve branch density with manual analysis; CNBDSA, corneal nerve branch density with semi-automated analysis; CNFD, corneal nerve fiber density; CNFDA, corneal nerve fiber density with automated analysis; CNFDFA, corneal nerve fiber density with fully automated analysis; CNFDM, corneal nerve fiber density with manual analysis; CNFDSA, corneal nerve fiber density with semi-automated analysis; CNFL, corneal nerve fiber length; CNFLA, corneal nerve fiber length with automated analysis; CNFLcenter, corneal nerve fiber length at the central cornea; CNFLFA, corneal nerve fiber length with fully automated analysis; CNFLM, corneal nerve fiber length with manual analysis; CNFLSA, corneal nerve fiber length with semi-automated analysis; CNFLwhorl, corneal nerve fiber length at the whorl; CNFrD, corneal nerve fractal dimension; CNFT, corneal nerve fiber tortuosity; CNL, corneal nerve length; CNM, corneal nerve migration; CNN, convolutional neural network; CNT, corneal nerve thickness or corneal nerve tortuosity; CSNP, corneal sub-basal nerve plexus; CTBD, corneal total branch density; DLA, deep learning algorithm; DM, diabetes mellitus; DN, diabetes neuropathy; DPN, diabetes peripheral neuropathy; DSP, diabetic sensorimotor polyneuropathy or distal symmetric polyneuropathy; DSPN, diabetes symmetrical peripheral neuropathy or diabetic sensorimotor polyneuropathy; EEP, equal error point; EER, equal error rate; EERP, equal error rate point; FU, follow-up; HCs, healthy controls; ICC, interclass correlation coefficient; IENFD, intraepidermal nerve fiber density; IGT, impaired glucose tolerance; IVCCM, in vivo corneal confocal microscopy; IWL, inferior whorl length; IWNFrD, inferior whorl nerve fractal dimension; M, mean; MCNBD, manual corneal nerve branch density; MCNFD, manual corneal nerve fiber density; MCNFL, manual corneal nerve fiber length; MDL, mean dendric length; MFU, mean follow-up; mm, millimeter; mm/mm2, millimeter/square millimeter; mm2, millimeters squared; NBD, nerve branch density; NBe, number of beadings; NBi, number of bifurcations; NBr, number of branches; NCs, normal controls; NDS, Neuropathy Disability Score; NFA FIJI, nerve fiber area determined by total number of pixels within the nerve plexus; NFA W×L, nerve fiber area determined by length times average width; NFD, nerve fiber density; NFL, nerve fiber length; NFLD, nerve fiber length density; NFT, nerve fiber tortuosity; NGT, normal glucose tolerance; no., number; no./mm2, number/square millimeter; NSP, Neuropathy Symptom Profile,; NT, number of trunks; PN, peripheral neuropathy; PNCV, peroneal nerve conduction velocity; PPV, positive predictive value; QST, quantitative sensory testing; RCNFL, rapid corneal nerve fiber loss; RNFL, retinal nerve fiber layer; ROC, receiver operator characteristic; S/S, sensitivity/specificity; SBP, sub-basal nerve plexus; SCNP, sub-basal corneal nerve plexus; SD, standard deviation; SD-OCT, spectral domain optical coherence tomography; SNF, small nerve fiber; T1D, type 1 diabetes; T2D, type 2 diabetes; VPT, vibration perception threshold; vs., versus; WST, warm sensation threshold; yr, year; yrs, years; μm2, square micrometer.
Further, the current longitudinal literature suggests CCM may predict incident DSPN, identify patients at high-risk for DSPN, and monitor progressive nerve damage over time.73,75,81,101,106,108 Sampling adults with T1D, Pritchard et al. found, for example, that baseline CNFL predicted incident DSPN at 4-year follow-up. Participants who developed DSPN had significantly lower baseline CNFL (14.0 ± 4.1) relative to participants (16.2 ± 3.5) who did not develop small-fiber neuropathy. 81 Additional longitudinal research, reviewed in this study, support these findings, revealing reduced CNFL may be an important biomarker for future development of DSPN.101,106,108
Sudomotor Function Testing (SFT)
SFT is used to assess small-fiber DSPN. 3 SFT, a measure of autonomic function, may be assessed with quantitative sudomotor axon reflex testing (QSART), and newer approaches, including electrochemical skin conductance (ESC).3,119-121 SFT non-invasively assesses C fiber function, specifically innervation of sweat glands.
QSART
QSART measures C fiber induced sweat production in predetermined sites (forearm, distal and proximal leg, and foot) with application of iontophoresis and measurement of sweat responses with iontophoretic stimulators.119-120 Abnormal QSART test findings reveal reduced sweat volume or latency. 120 The literature search identified 2 studies specific to QSART technology and one related, novel technological advancement (see Table 5)122-124 The Q-Sweat System (WR Medical Electronics, Stillwater, MN, USA) was found to have suboptimal sensitivity (58%), optimal specificity (100%) in one study while another documented lack of discriminatory power in identifying participants with and without DSPN and controls.122-123
Quantitative Sudomotor Axon Reflex Testing (QSART).
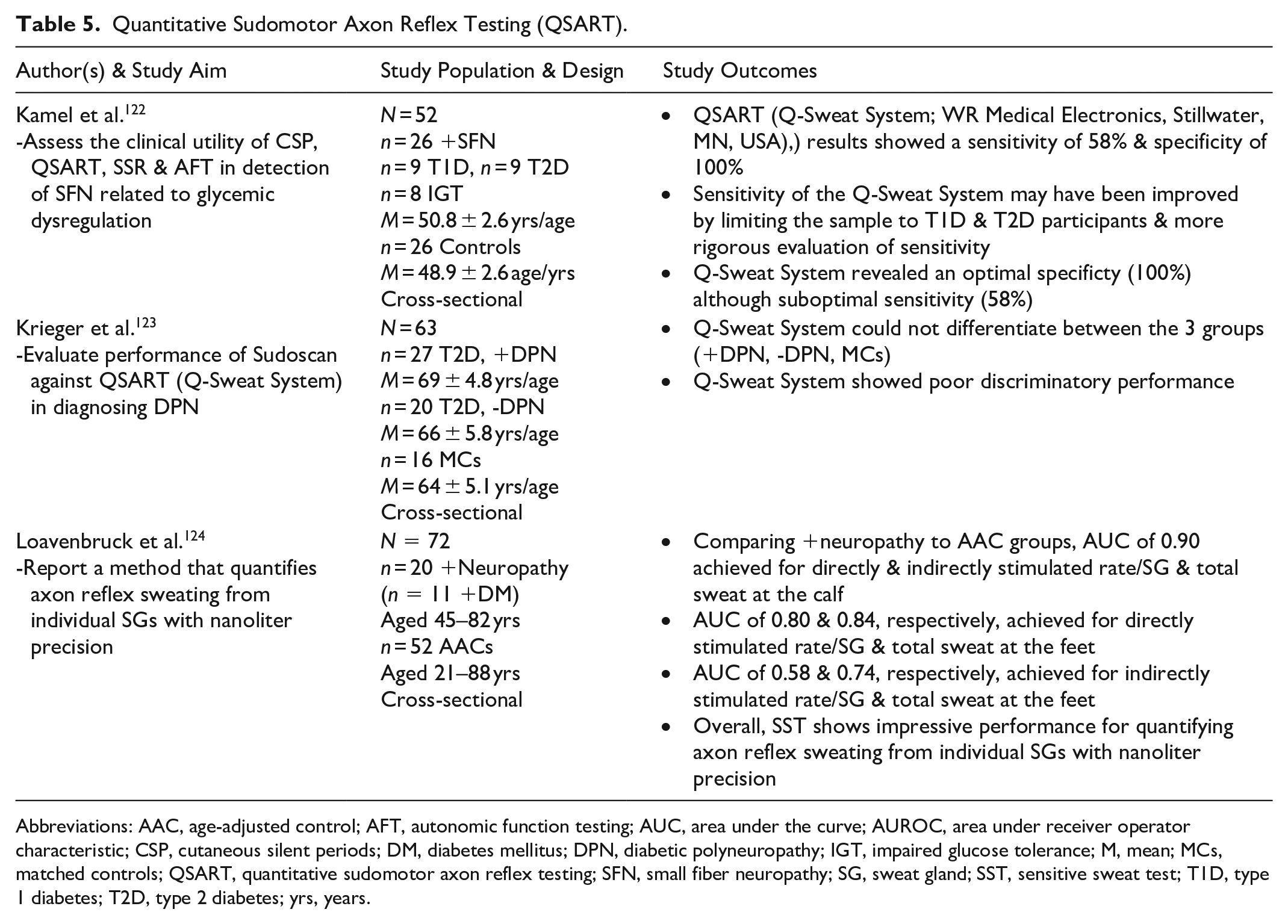
Abbreviations: AAC, age-adjusted control; AFT, autonomic function testing; AUC, area under the curve; AUROC, area under receiver operator characteristic; CSP, cutaneous silent periods; DM, diabetes mellitus; DPN, diabetic polyneuropathy; IGT, impaired glucose tolerance; M, mean; MCs, matched controls; QSART, quantitative sudomotor axon reflex testing; SFN, small fiber neuropathy; SG, sweat gland; SST, sensitive sweat test; T1D, type 1 diabetes; T2D, type 2 diabetes; yrs, years.
Distinct, although related to traditional QSART, Loavenbruck et al. developed a brief, sensitive sweat test (SST) with nanoliter precision, using high-definition videography, to assess C fiber innervation in the foot, calf, thigh, and hand. In SST, the anatomical site being assessed has a small, handheld camera (with starch tape across its lens) pressed firmly, face down. Acetylcholine (ACh) gel, loaded in an anode iontophoresis capsule, is applied directly adjacent and lateral to the camera. Directly stimulated by ACh gel, affected sweat gland (SG) ducts secrete sweat, which is absorbed by the starch tape as small dark spots. Recorded dark spots accurately correspond with sweat volume. In comparing participants with neuropathy to controls, ROC analyses showed the greatest distinction between groups at the calf (directly stimulated rate/SG and total sweat had an AUC of 0.90 and 0.90, respectively) and foot (directly stimulated rate/SG and total sweat had an AUC of 0.80 and 0.84, respectively). 124
ESC
ESC assesses the electrical potential of sweat glands to evaluate sudomotor function. ESC directly evaluates C fiber stimulated sweat gland function through measurement of sweat chloride concentrations using reverse iontophoresis and chronoamperometry.125-127
Since 2015, the literature search identified 14 studies evaluating ESC, namely Sudoscan (Impeto Medical, Paris, France; see Table 6).51,76,123,128-139 Advancing earlier EZSCAN technology, Sudoscan is a POCD wherein testing requires that the patient place his/her hands and feet, respectively, on 2 stainless steal electrodes.125-126 While the patient’s hands and feet remain on the electrodes for 2 minutes, a computer records ESC values. 125
Electrochemical Skin Conductance (ESC).

Abbreviations: AUC, area under the curve; AUROC, area under receiver operator characteristic; CAN, cardiovascular autonomic neuropathy; DM, diabetes mellitus; DNS, Diabetic Neuropathy Symptom Scale; DPN, diabetic peripheral neuropathy or diabetic polyneuropathy; DSPN, diabetic distal symmetric peripheral neuropathy or distal symmetrical polyneuropathy; EMG, electromyography; ESC, electrochemical skin conductance; FU, foot ulceration; HC, healthy control; HVs, healthy volunteers; IENFD, intraepidermal nerve fiber density; IGT, impaired glucose tolerance; IQR, interquartile range; kg, kilogram; LE, lower extremity; LSR, lumbosacral radiculopathy; M, mean; MC, matched controls; MNSI B, Michigan Neuropathy Screening Instrument B; NCS, nerve conduction study; NDS, Neuropathy Disability Score; PN, peripheral neuropathy; PPN, peripheral polyneuropathy; QSART, quantitative sudomotor axon reflex testing; ROC, receiver operator characteristic; SFN, small fiber neuropathy; SFN-AD, small fiber neuropathy classified according to associated disorders; SFN-I, small fiber neuropathy classified as idiopathic; SGNFD, sweat gland nerve fiber density; T1D, type 1 diabetes; T2D, type 2 diabetes; VPT, vibration perception threshold; yrs, years; μS, microSiemens.
Most studies revealed overall adequate to good performance of Sudoscan.51,76,123,130-131,133-134,136,138-139 For example, sampling Mexicans with T2D, one study examined the performance of Sudoscan in predicting DSPN among 2 groups: (1) Mexicans with T2D for greater than 5 years; and (2) Mexicans with T2D for less than 5 years. In the first group, the AUROC curves for hands and feet ESC were 0.84 and 0.78, respectively, with the Michigan Neuropathy Screening Instrument as the reference. The sensitivity of abnormal hands or feet ESC for detection of neuropathy was 97%, while the positive predictive value was 87%. In the second group, AUROC curves of hands and feet ESC were 0.66 and 0.72, respectively. The sensitivity of abnormal hands or feet ESC for detection of neuropathy was 91%, while the positive predictive value was 88%. 130 These findings, coupled with the work of Goel et al, 134 suggest Sudoscan is a technology that may detect early DSPN. Sudoscan has also demonstrated respectable repeatability and reproducibility. Bordier and colleagues reported that, for feet, the ESC mean coefficient of variation for repeatability was 2.8 ± 1.6% in healthy controls and 6.9 ± 6.3% in participants with T2D, while the coefficients of variation for reproducibility were 3.1 ± 1.5 and 6.9 ± 6.3%, respectively. 129
Discussion
The diagnosis of DSPN is typically informed by a complete patient history and clinical exam. This approach is often sufficient to diagnose DSPN but not in its early asymptomatic and subclinical stages. Also, more advanced DSPN small- and large-fiber disease may present exclusively or together and sometimes without signs or symptoms.4,39-42 Given DSPN may not present with clinical signs and symptoms, utilization of highly sensitive screening technologies may be warranted to promote early detection of DSPN. Also, when signs or symptoms are present, but the distinction between small- and large-fiber disease remains unclear, confirmation of the DSPN subtype may require diagnostic testing. NCS is recommended for confirming large-fiber DSPN. 3
Advances in NCS technologies have yielded a very promising POCD, DPN-Check. DPN-Check assesses sural nerve conduction. Research suggests that DPN-Check, overall, has very respectable accuracy in detecting the presence and severity of DSPN.51-52,56-59 Also, DPN-Check has strong reliability and good agreement with standard electromyography. 60 However, a few studies indicate the POCD may have measurement bias compared to standard NCS.56,140 Yet, DPN-Check, approved by the Food and Drug Administration (FDA), is sufficiently accurate and reliable for use as a screening technology. The POCD is inexpensive ($500), provides rapid results, and does not require highly specialized training. DPN-Check has the potential to widen access to nerve conduction testing in primary care, internal medicine, and endocrinology practices although patients with abnormal or borderline results should undergo standard NCS. 56
Typically preceding large-fiber DSPN, small-fiber DSPN has negative findings upon NCS testing. 3 The gold standard for small-fiber diagnostic testing is IENFD. IENFD may identify early DSPN.141-142 Yet, this diagnostic technology is invasive and may be painful for patients. Serial skin biopsies for detecting early changes in IENFD may not be practical. More recent technological advances for small-fiber DSPN screening provide alternative, more rapid, non-invasive approaches – some of which show promise for early or asymptomatic, subclinical detection of small-fiber disease.
NerveCheck, a POCD, is an inexpensive ($500) QST device. It has demonstrated adequate to good reproducibility but mixed results with respect to diagnostic accuracy.82-83 The TSA-II NeuroSensory Analyzer, while also having varying results with respect to performance, may discriminate between clinical and subclinical DSPN in line with prior QST research.77,143-146 CHEPs has not advanced technologically and lacks practicality given its complex testing approach.
CCM technology has advanced, including automated image assessment. Prior and more current CCM research reveals its high promise to detect incident and early risk for DSPN 73,75,81,101,106,108,147-151 In the present study, CCM, compared to IENFD, was found to have fairly comparable performance.72,96 A limitation of CCM is that it is currently considered investigational for the purposes of detecting DSPN, and thus, is an out-of-pocket patient expense although smartphone CCM may be on the horizon, which may reduce costs in tandem with automated image evaluation.
In terms of sudomotor function testing, technological advancements are also impressive. A novel POCD, using high-definition videography, assesses C fiber innervation in the foot, calf, thigh, and hand with a brief SST. Beginning data indicate that the POCD has respectable accuracy in detecting small-fiber DSPN. 124 Yet, evidence is lacking in SST’s ability to detect asymptomatic or subclinical DSPN. Further rigorous research is needed prior to clinical adoption.
When considered with respect to past and more recent research, Sudoscan has a substantial evidence revealing it may potentially identify early DSPN (although this not its main use) and monitor DSPN progression over time.51,76,123,130-131,133-134,136,138-139,152-154 Sudoscan also has adequate reproducibility and repeatability.129,155-156 Sudoscan is approved by the FDA and may be reimbursed through proper billing. Sudoscan is thus a PCOD worthy of clinical adoption to detect and monitor DSPN in clinical settings, which may be accompanied by initial, confirmatory IENFD results.
While established and more recent technological advances may detect DSPN, it must be underscored that early detection of the disease is critical. At this time, IENFD (gold standard) and CCM have the strongest evidence for early detection of small-fiber disease, a precursor to large-fiber disease. The small-fiber technological screening devices reviewed may build on their respective research bases to further examine performance measures in detecting small-fiber DSPN in asymptomatic, subclinical populations. Furthermore, it appears that additional examination of normative values may be required, as indicated, for ethnic/racial, gender, and age groups to minimize the risk for bias in DSPN device outcomes data.
Conclusion
There is no cure for DSPN. Hence, its early detection is critical. Early, regular screening for DSPN is essential, particularly for small-fiber disease. Although IENFD is the gold standard for assessing small-fiber disease, innovative, non-invasive technologies, such as Sudoscan and particularly CCM, for early disease detection may be useful alternatives. Regardless of whether screening reveals small-fiber disease or not, healthy lifestyle interventions are warranted, if not ideally implemented earlier. Healthy lifestyle behaviors (weight loss, increased physical activity, and smoking cessation), may prevent, modify the progression, or mitigate symptoms of DSPN.9,21,33,157-161 In T1D populations, tight glycemic control tends to delay or prevent DSPN whereas, in T2D populations, tight control, for some, may slow DSPN progression.15,30,162-164 Optimization of blood pressure and lipid levels is also warranted.22-25
Footnotes
Abbreviations
ADA, American Diabetes Association; CCM, corneal confocal microscopy; CHEPs, contact heat evoked potentials; DSPN, distal symmetrical peripheral neuropathy; DTRs, deep tendon reflexes; ESC, electrochemical skin conductance; IENF, intra-epidermal nerve fiber density testing; NCS, nerve conduction study; QST, quantitative sensory testing; QSART, quantitative sudomotor axon reflex testing; SFT, sudomotor function testing; T1D, type 1 diabetes; T2D, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
