Abstract
Background:
Diabetic foot ulceration (DFU) affects only a subgroup of patients with diabetic neuropathy, that is, those with pain-insensitivity due to end-stage sensory failure. Pain perception failure develops insidiously and remains asymptomatic until first DFU. As loss of pain perception is clinically significant, timely detection is mandatory.
Objectives:
A novel suprathreshold pinprick pain stimulus of 512 mN force made from optical glass-fiber was explored in a prospective cross-sectional diagnostic accuracy study to detect DFU-related end-stage sensory failure.
Methods:
A total of 116 participants were studied (25 healthy people, 22 patients with diabetes without relevant complications, 19 patients with previous painful foot or leg injuries, and 50 patients with previous or active painless DFU [reference standard]). Palmar and plantar surfaces were stimulated in a standardized fashion. At the feet, the second and third toe skinfolds and the middle of the plantar arch were stimulated. Participants scored stimulated pricking discomfort or pain intensity 0 to 10 on a numerical rating scale.
Results:
At hands, intensity was rated on average 5 (1-10) [median (range)] by 114/116 participants. Per foot, participants without DFU scored 5 (1-10), while those with DFU scored 0 (0-3) (P < .0001). At plantar toe skinfolds, the absence of pinprick pain perception detected DFU-associated sensory failure with an accuracy of 99.5% (sensitivity 99.5%, specificity 99.4%, positive likelihood ratio 248, and negative likelihood ratio 0.005).
Conclusion:
In this pilot study, nociceptive stimulation of a plantar toe skinfold with a 512 mN optical glass-fiber pinprick accurately identified DFU-associated end-stage sensory failure.
Keywords
Introduction
Painless diabetic foot ulceration (DFU) and Charcot osteoarthropathy (COA), affecting less than 10% of patients with diabetic neuropathy according to established diagnostic criteria, is associated with severe, end-stage diabetic somatosensory neuropathy.1,2 This stage of diabetic neuropathy is characterized by failure of the small A-delta and C-fiber afferents of the lower limbs, due to axonal degeneration. Subsequently, their abundantly arborizing intraepidermal endings (>1000/mm²) are dying-back, which function as receptors for noxious impacts (nociceptors). The gradual degeneration process remains asymptomatic, even with >90% of the intraepidermal nociceptors lost.3-6 It becomes symptomatic only with the advent of painless foot injuries from which DFU and COA arise. 7 Accordingly, pain perception studies, eg, by pinprick stimulation, have demonstrated perception thresholds above the upper limit of measurement8-10 in feet with DFU or COA.
To date, the conventional plastic 10 g Semmes-Weinstein monofilament (SWM) is recommended to diagnose and screen for diabetic neuropathy, 11 assessing innocuous punctate touch sensitivity, that is the function of large myelinated A-ß fibers serving subepidermal Meissner corpuscles and Merkel disks. 12 While healthy persons’ SWM touch perception thresholds range between 0.28 mN (equivalent to 0.028 g) and 40 mN (4 g), 13 most feet with DFU-associated sensory failure display SWM perception threshold above 75 g, as Holewski et al 14 have shown. However, the lack of 10 g SWM touch perception to diagnose DFU-associated end-stage sensory failure has a questionable reproducibility, and sensitivity or specificity as low as 51% to 63% in some studies. 15 As cutaneous pain perception rather than touch perception is causative for painless DFU, it seems reasonable to assess pain perception for diagnosing DFU-associated end-stage sensory failure. The following study tests a novel diagnostic approach using a force-controlled suprathreshold glass-fiber pinprick-pain stimulus, which combines several important features: it targets nociception (and thereby the dominant ulceration risk factor loss of nociception), its force is constant and of a critical magnitude (since greater than threshold pain intensity is necessary to trigger physiologic avoidance reaction), 16 its detectability in the general population is particularly high, and the stimulus response can be determined by the robust yes/no method.
Study Protocol
Study Design
Cross-sectional proof-of-principle study of a diagnostic test for end-stage sensory failure; validation against clinical judgment (confirmed history of painless foot ulceration as reference standard). This comparator was chosen because objective measurements of nociception failure (like laser-evoked potentials or histopathology to determine intraepidermal nerve fiber endings)4-6 were not available. The test employs punctate cutaneous pain stimulation by a special glass-fiber pinprick with a force of 512 mN. In healthy people, the detection probability of such a pain stimulus is >98%, 17 and the pain response is mild (on average 1.5 on numerical rating scale 0-10 in perception threshold studies).18,19
Objective
The objective of this study is to assess the test performance and its potential for diagnosing end-stage sensory failure among people with or without diabetes, with or without a history of painless or painful foot lesions.
Outcome
The outcome of this study is to identify diseased/not diseased according to the absence/presence of pricking pain sensation.
Setting
The setting includes private practices of general medicine and diabetology in Düsseldorf/Germany.
Study Sample
A convenience sample of 116 German speaking volunteers recruited from practice patients, staff member, relatives, and friends. Sample size was not based on power calculation, but on the available literature.
Materials and Methods
The handheld pinprick-pain stimulating device consists of an optical glass-fiber mounted to a holder.20,21 The glass-fiber is round, has a diameter of 0.34 mm, and a flat tip of 0.091 mm² surface. Its length is 38.5 mm. The cylindrical holder has a length of 110 mm, a diameter of 8 mm, and a blunt, convex ending. The glass-fiber is more rigid, but less thick than a conventional plastic 10 g SWM with a diameter of 0.475 mm and a tip surface of 0.18 mm². 22 It is also much thinner than the tip of an ordinary wooden toothpick commonly recommended for assessing diabetic neuropathy 11 (Figure 1).
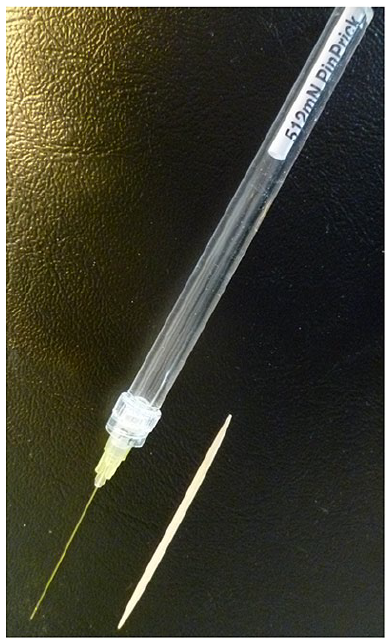
Picture of a 512 mN optical glass-fiber pinprick stimulator and a conventional wooden toothpick.
According to the instructions for use, the glass-fiber is pressed by hand perpendicular to an object until slight buckling. The glass-fiber then will exert a force of 512 mN, consistent with a calculated pressure of 5625 mN/mm² (corresponding to a load of 562.5 g/mm², which is around eightfold the load per mm² exerted by a 10 g SWM). 22 Before use, the bending force of the actual glass-fiber was checked several times on a top-loading electronic laboratory balance, measuring 50 to 53 g (consistent with 512 mN). 22 Applied to the skin, the glass-fiber’s force exceeds the pinprick-pain perception threshold in >98% of healthy persons and evokes a sting-like cutaneous sensation that can be described as “sharp pricking discomfort or pain.”
Mode of Operation
The pricking pain or discomfort in response to punctate noxious stimulation is encoded and transduced mainly by A-delta-fiber nociceptors and to a lesser extent also by C-fiber nociceptors. 23 Mechanically, a pinprick-pain probe operates by funnel-shaped skin indentation, depending on the compliance of the skin and subcutaneous structures.24,25 Indentation by the high-force 512 mN probe in particular activates both high- and low-threshold intraepidermal mechanoreceptors (at variance to SWM, which activates low-threshold mechanoreceptors only). Receptors for pain are high-threshold nociceptive free nerve endings (nociceptors), whereas those for touch and vibration like Meissner and Pacinian corpuscles are low-threshold mechanoreceptors. The deeper the indentation, the more skin becomes involved and the greater is the ensuing afferent signal from adjacent receptors being stimulated simultaneously (the phenomenon of spatial summation).
A considerable variance of the pinprick stimulation of intraepidermal nociceptors is caused by mechanical skin compliance. For instance, with very soft skin in a mouse model, a minimum force of 10 to 25 mN is sufficient to activate nociceptors, 26 while at the human forehead skin, at least 55 mN is required. 27 At the palmar side of the hand, the average pinprick-pain perception threshold is around 250 mN, and at the plantar side of the foot, it is around 120 mN.28,29 Due to acquired individual psycho-physical irritability, 30 the pain response to pinprick stimulation varies little intraindividually, but considerably between people. Hence, in healthy human populations, the range of pinprick-pain perception thresholds is wide, reaching from <10 mN (1 g) to >1000 mN (100 g), with a bell-shaped logarithmic distribution, see Mücke et al. 17
Study Participants
Four categories of German speaking people were studied: (I) 25 healthy control persons (staff members, relatives, and friends), (II) 22 control patients with diabetes, without neuropathy according to the current German disease management regulations (64 Hz Rydel-Seiffer tuning fork vibration sensation at the hallux >4/8 or 10g SWM perception), (III) 19 patients with a history of painful foot or leg lesion (venous ulceration, soft tissue trauma, eg, dog bite, sprain, fracture, pressure sore, and necrobiosis lipoidica), and (IV) 50 patients with diabetes and a history of painless DFU or COA, serving as reference standard. Consecutive patients were retrieved from the practices’ international classification of diseases ICD-10 based data storage systems and were examined during one of their regular practice appointments. Exclusion criteria were age <18 years, inability to comply with the experimental protocol, mental problems (eg, borderline personality disorder or drug addiction), comedication affecting sensory functions (eg, tranquilizers, antiepileptic drugs, and pain-killers), and skin pathology like keratosis palmoplantaris. The medical histories were taken from the patients’ files; the healthy controls were briefly interviewed. Of the healthy control persons and control persons with diabetes, a total of 17 had undergone pinprick-pain threshold assessment already seven years ago, revealing an average pain threshold of 256 (64-512) mN at the hands and 192 (48-512) mN at the feet along with perceived pain intensity (numerical rating scale 0-10) of 2.5 (1.2-9) at the hands and 2.5 (1.2-9) at the feet [median (range)].
Experimental Procedures
Preparation
The whole investigation took about ten minutes to complete. Each study person was placed in supine position and bare-footed in a separate quiet room with ambient temperature around 18°C. After accommodation, the person was familiarized with the test by the examiner. To this end, the device was demonstrated and a sharp (painful) as opposed to nonsharp (touch) stimulus was applied (the tip of the glass-fiber vs the convex ending of the holder), first to the examiner’s and then to the study person’s forearm.
Explanation of the Sensation of Interest
The sensation of interest “sting-like sharp, pricking discomfort, or pain” was explained in plain language. The common German expression is “pieks,” which is compared to the English “sting” or “prick.”
Stimulus Response Ascertainment
The stimulus response was ascertained according to the yes/no signal detection paradigm. Every stimulation was accompanied by the examiner’s question: “Does it sting (prick)—yes or no ?” Whenever “yes” was answered, intensity should be rated and told to the examiner, and the details of which foot had been pricked should be given. When “no” was responded, the person was asked to communicate if anything else was felt, eg, “touch,” “movement,” “pressure,” or “nothing at all.” Occurrence of visible involuntary retraction movements of the limbs (nocifensive somatomotory withdrawal reflexes) was noted, while audible reactions (exclamations indicative of emotional suffering) were disregarded.
Stimulus Intensity Rating
Whenever “sting-like sharp, pricking discomfort, or pain” (pieks) was perceived, the participant should rate the intensity (absolute magnitude estimation) on an 11-point numerical rating scale, ranging from 0 (no “sting-like sharp, pricking discomfort, or pain”) to 10 (maximal imaginable “sting-like sharp, pricking discomfort, or pain”).
Pattern and Procedure of Stimulation
The tip of the glass-fiber was regularly applied and maintained for one to two seconds. The stimulation was repeated twice, with one to two seconds interval. Unlike the previous studies applying unmeasured pinprick stimulation at the eponychium11,31 or the plantar surface 32 of the big toe only, in the present study, three sites on the plantar surface were consecutively stimulated (with 5-10 seconds interval): the plantar arch and the skinfolds of the second and third toe innervated by the terminal branches of the tibial nerve. Areas with thickened skin near the metatarsal heads 33 were avoided; hammertoes were gently stretched by the examiner to expose the plantar skinfold for stimulation. Both feet were tested randomly. The study persons were unaware of the numbers of stimulations and sites of the foot to be stimulated. For control purpose, the middle of the palm of a hand was also stimulated consistent with the previous usage. 28 Catch trials (by applying the blunt ending of the holder) were occasionally interspersed to check the truth of the answers. The stimulation course was run top down from hand to feet and was repeated about three minutes later in reversed order. All stimulations were performed by the same examiner (the author).
The study was carried out in accordance with the World Medical Association’s Declaration of Helsinki and the Ethics Commission of the Heinrich-Heine University medical faculty (Project no. 3718; “Pain perception in diabetic neuropathy”). Written informed consent was provided by all study participants.
Data Processing and Analysis
Data were assessed per foot (unless stated otherwise), considering potential unilateral foot affections, eg, from radiculopathy due to intervertebral disk herniation, or amputation. Primary outcome was the correct “prick” perception per stimulated site, distinguishing between not diseased and diseased (end-stage sensory failure associated with DFU). Secondary analyses comprised pain scores and withdrawal reactions and statistical evaluation of the test. 34 Likelihood ratios were calculated, adding 0.5 to all cells to avoid problems with “zero” ratios, according to Deeks et al.35,36 Mann-Whitney U test, chi-square test, and intraclass correlation coefficients were applied, as appropriate. A P < .05 was considered significant. Data are presented as median (range), unless indicated otherwise.
Results
The experiments were well-tolerated without adverse effects. The clinical characteristics of all 116 participants, the healthy control people, the control persons with uncomplicated diabetes, the patients with painful leg injuries, and the patients with painless DFU are summarized in Table 1.
Clinical Characteristics of the Participants [Median (Range)].
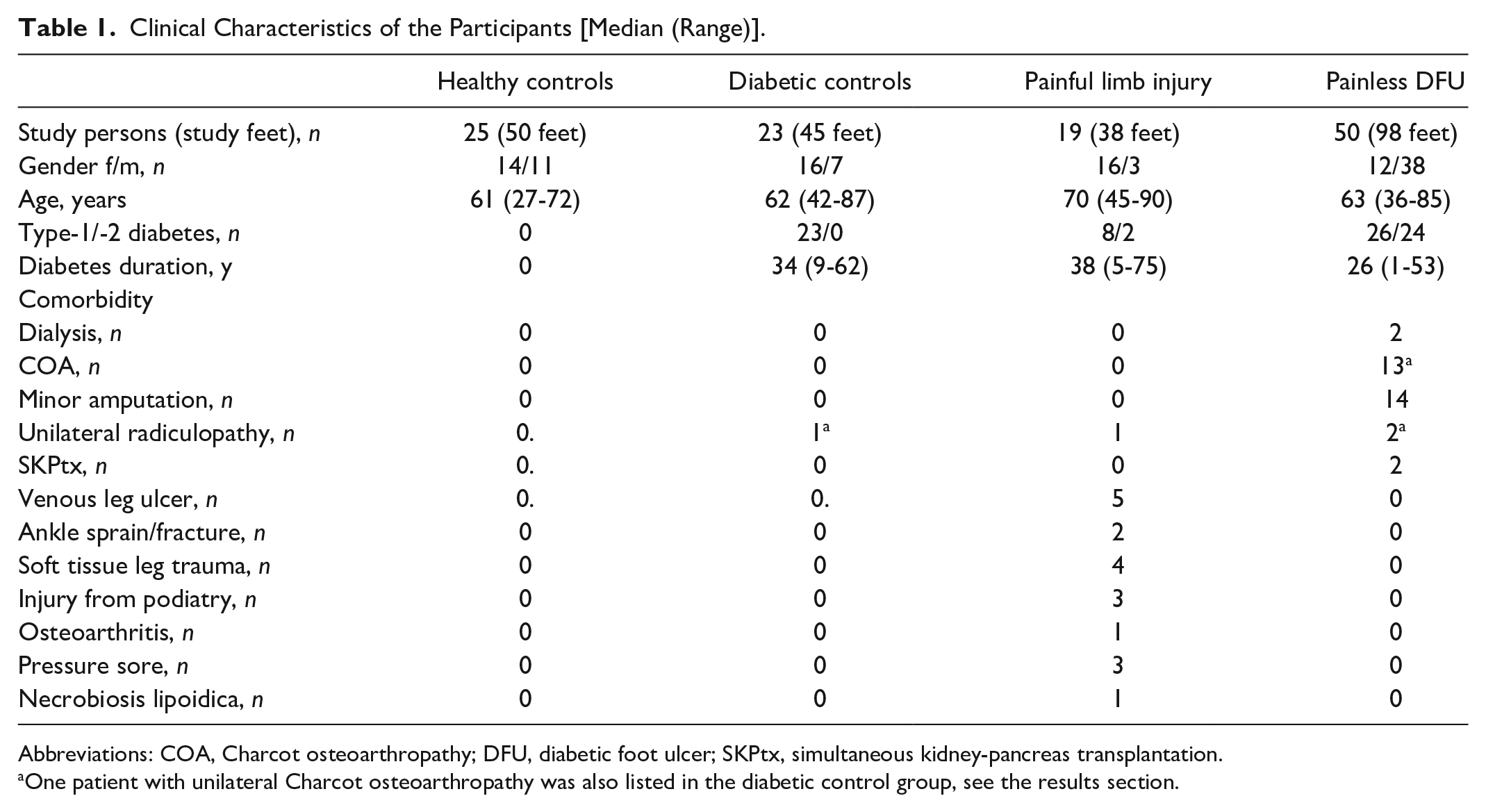
Abbreviations: COA, Charcot osteoarthropathy; DFU, diabetic foot ulcer; SKPtx, simultaneous kidney-pancreas transplantation.
One patient with unilateral Charcot osteoarthropathy was also listed in the diabetic control group, see the results section.
One patient with DFU had a midfoot amputation, and one had unilateral COA and only mild neuropathy on the opposite side (this person was listed twice in the DFU group and also in the diabetic control group). Hence, 50 patients in the reference group provided a total of 98 feet with DFU-associated end-stage sensory failure (42% of the whole sample of 231 feet).
At the feet without DFU-associated sensory failure, pain perception was intact at all stimulated sites, and intensity was scored on average 5 (1-10) per foot. By contrast, pinprick-pain was scored on average 0 (0-3) per foot (P < .0001) by 50 participants with DFU. The stimulus responses were similar on the right and the left foot except for three subjects, who had radiculopathy with different severity of sensory defects on either side. Pinprick-pain was perceived at the hands by 114 participants and was rated on average 5 (1-10). Test-retest reliability was assessed by comparing the pain scores of the first and the second run of stimulations. The findings in each participant group are virtually superimposable, confirmed by intraclass correlation coefficients >0.95 (P < .0005). Details are summarized in Table 2.
Pain Scores per Anatomical Site During First and Second Run of Stimulations [Median (Range)].
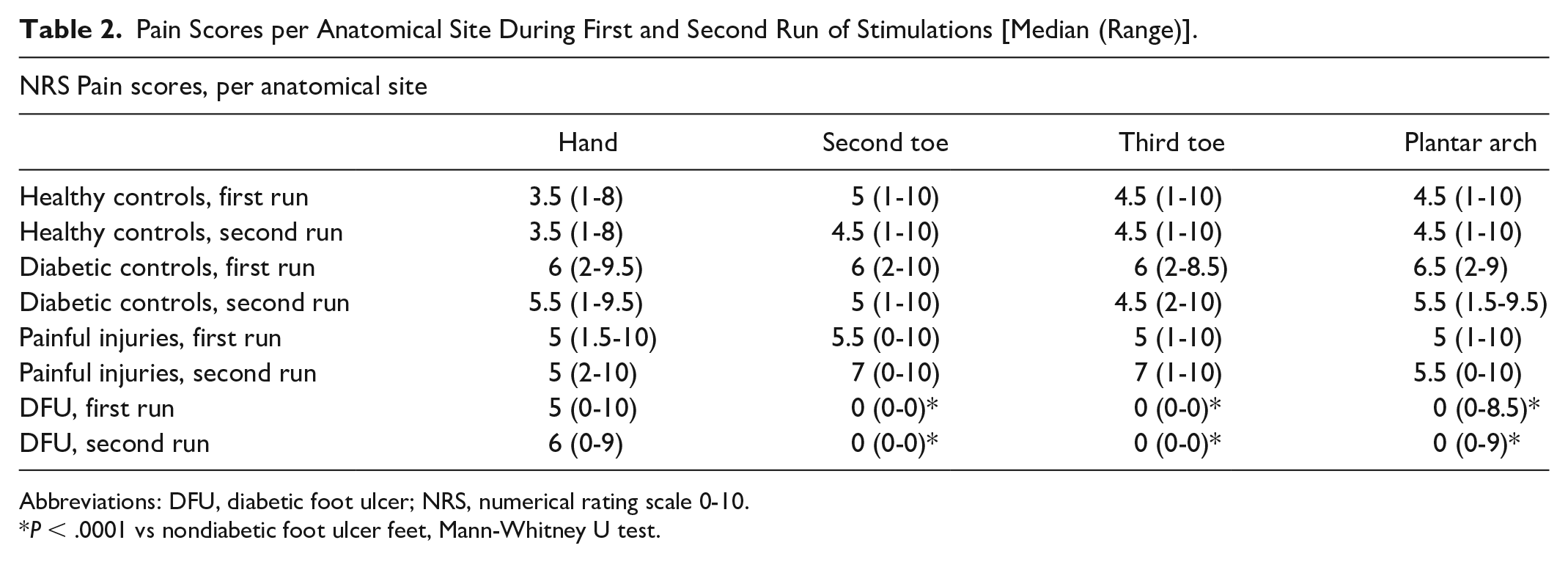
Abbreviations: DFU, diabetic foot ulcer; NRS, numerical rating scale 0-10.
P < .0001 vs nondiabetic foot ulcer feet, Mann-Whitney U test.
Prevalence of pinprick-pain perception per participant group is summarized in Table 3. Perception threshold was above 512 mN at toe skinfolds of all of the reference feet, whereas it was below 512 mN in all of the other feet (healthy and uncomplicated diabetic control feet, and feet with painful leg injuries).
Prevalence of Pinprick-Pain Perception in Participant Groups [Median (Range)].
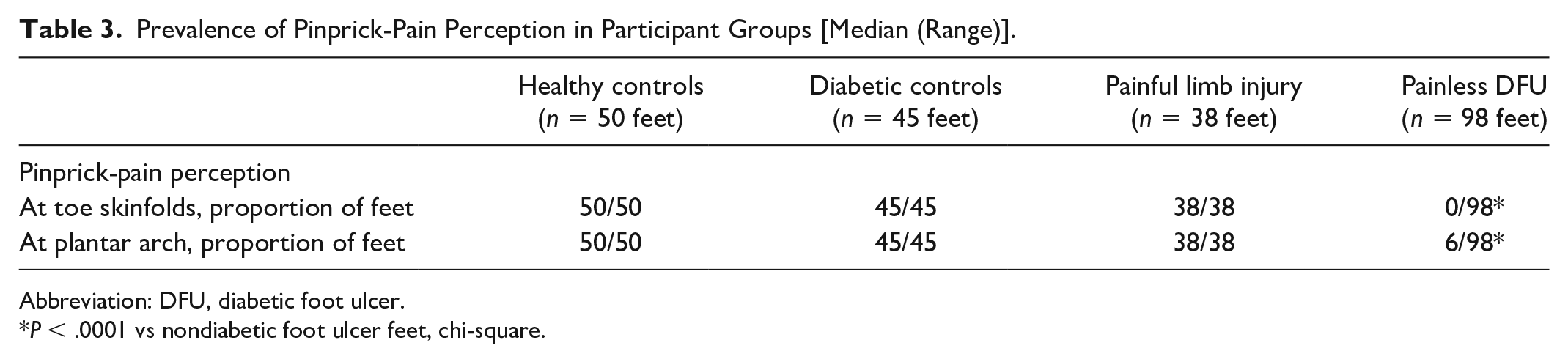
Abbreviation: DFU, diabetic foot ulcer.
P < .0001 vs nondiabetic foot ulcer feet, chi-square.
The validity of a pinprick-pain perception threshold >512 mN at the skinfold of the second or third toe to detect DFU-associated end-stage sensory failure in the sample studied was determined as follows: sensitivity 99.5%, specificity 99.4%, positive likelihood ratio 248, negative likelihood ratio 0.005, positive predictive value 99.5%, negative predictive value 99.6%, and diagnostic accuracy 99.5%. Given a 10% prevalence of DFU-associated end-stage sensory failure, like in the actual German diabetic population (author’s estimate, unpublished; pretest probability), such a test result would increase the chance of a patient with diabetes of having the condition to 96% (post-test probability).
Only three persons with DFU perceived pinprick-pain and only at their longitudinal arches (indicative of local A-delta and C fiber afferent function); one person scored 8.5 and 9, see Table 3. At a further 30 longitudinal arches, patients with DFU reported ill-defined sensations of pressure, touch, or skin movement in response to pinprick stimulation, suggestive of residual A-ß fiber afferent function. There were three wrong responses to catch trials in the DFU group only. Withdrawal reflex movements of the hands were shown by 18% to 56% of participants with and without DFU. Reactive movements of the feet were noticed in 67% to 76% of participants without DFU and in 3% of participants with DFU.
Discussion
Contrary to what the NeuroDiab "diabetic neuropathy community" demands (that is simple tests which identify early subclinical neuropathy and not advanced neuropathy), the present study focuses on severe, advanced small fiber sensory neuropathy which remains asymptomatic, too, until a first-ever DFU has developed. In this respect, diabetic neuropathy is similar to diabetic retinopathy which also develops insidiously, remains asymptomatic for long, and becomes clinically significant and symptomatic only in its advanced sight-threatening stage. The present data show that DFU-associated sensory failure can reliably be identified by demonstrating a pain perception threshold above 512 mN at the plantar skinfold of the second or third toe, using a simple calibrated monofilament pinprick stimulator.
Detection of the principal risk factor, loss of protective pain sensation, is crucial when preventing first-ever DFU or COA, and eventually amputation 37 is considered. The consequences are self-evident, imperative, and comprehensive, the key features being patient education, among others, “on ways to substitute other sensory modalities (palpation or visual inspection using an unbreakable mirror) for surveillance of early foot problems” and on “selection of appropriate footwear and footwear behaviors at home.” 38 There are also consequences for the education of the medical personnel, as previously addressed by Professor Boulton and here quoted in full: “It is pain that leads to many medical consultations; our training in health care is oriented around cause and relief of pain. Thus the care of the patient with no pain sensation is a new challenge for which we have no training. It is difficult for us to understand, for example, that an intelligent patient would buy and wear a pair of shoes three sizes too small, and to come to our clinic with extensive, shoe-induced ulcers. The explanation, however, is simple: with reduced sensation, a very tight fit stimulates the remaining pressure nerve endings, and this is interpreted as normal fit.” 39
For pain stimulation, ordinary toothpicks have been applied, or devices with a spring-driven stimulus of an unspecified force of 400 mN)40-42 or with a disposable 23 G injection needle 43 loaded with various weights. 44 The latter, however, must not be used in cases with suspected elevated pain perception threshold, since injection needles may perforate the skin already at a force of 150 mN (15 g). 45
Expectedly, healthy people in the present study perceived greater pain intensity from suprathreshold stimulation, as compared to threshold-stimulation applied in the previous studies.18,19 Moreover, the present data are consistent with punctate touch perception threshold above 75 g SWM in diabetic or leprotic feet with painless ulcers,13,14,46 which will heal at identical rates when treated identically. 47 Of note, 100 g SWM testing detects painless leprotic ulcers with >95% sensitivity and specificity. 13 In the present study, the lack of 512 mN pinprick-pain perception at the second and third toe skinfold had higher sensitivity and specificity (99.5% and 99.4%) to detect people with DFU as compared to a previous report (97% and 77%). 9 This difference is most likely due to methodology. The previous study has used the method of limits for assessing perception thresholds by serial sub- and suprathreshold stimulations. In the present study, the yes/no method was applied with only one suprathreshold stimulus and two possible results: normal or abnormal. Perception threshold determination by serial stimulations is more intricate and prone to biases, eg, from errors of expectation and habituation, than a simple yes/no response to a single suprathreshold pain stimulation.
The present data must be regarded as preliminary due to certain weaknesses in study design: (a) the subjects were not matched regularly by age and gender; (b) the assessments were neither independent nor blind, because reference standard was clinical judgment implying that foot deformities were visible to the investigator. However, including clinical nerve conduction studies for comparison was deemed unwise, because these objective methods do neither assess small fibers nor A-delta and C-fiber afferent function in particular. 48
Conclusion
Reliable detection of end-stage sensory failure associated with painless DFU was feasible by stimulation of second or third toe skinfold with a glass-fiber pinprick of 512 mN force. Prospective diagnostic and screening trials are required to corroborate these pilot data, and—by comparison with traditional clinical methods, nociceptive-evoked potentials, or skin biopsy (note Costa et al 49 )—to establish the efficiency of this new way of identifying neuropathic diabetic feet particularly prone to DFU.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
