Abstract
Background:
Callus formation in the diabetic foot increases the risk of ulcer onset. It is standard procedure to remove these dead tissue layers to reduce rising pressures. In a surgical procedure known as scalpel debridement, or chiropody the callus tissue is removed up to the epidermal layer. Factors may influence the outcome of this surgical process such as clinician inexperience. In an effort to standardize the debridement process, tissue oxygenation (TO) measurements are obtained before and after to study the effect of debridement on callus tissue.
Methods:
Fifteen debridement cases were analyzed using near infrared (NIR) imaging to study changes in TO. The NIR-based device used in this study estimates effective changes in TO in terms of oxy-, deoxy-, total hemoglobin, and oxygen saturation. Weber contrasts between callus tissue and the surrounding normal tissue were compared following debridement for all TO parameters. In a secondary analysis, callus tissue was segmented into quadrants and a percent of significance (in terms of total TO change) was calculated using a t-test.
Results:
Results show majority of cases displayed greater than 80% as the significant change in TO following debridement, except in cases with the presence of blood clot (a common precursor for ulceration). In cases where incomplete debridement was suspected, a significant change in TO was still observed.
Conclusions:
With extensive systematic studies in the future, NIR imaging technique to measure changes in TO may be implemented as a low-cost hand-held imaging device useful for objectively assessing the effectiveness of the scalpel debridement process.
Keywords
Introduction
Diabetic Foot Ulcers and Calluses
Complications associated with Diabetes Mellitus (DM) include maintenance of the diabetic foot requiring extensive supervision and treatment to prevent Diabetic Foot Ulcers (DFUs) and/or amputation.1-3 Prior to DFU onset the patient often presents with hyper-keratosis or callus formation due to increased pressures in the diabetic foot compromising motor, sensory, and autonomic neurons within soft tissue.4-8 Increased pressures may occur due to stress-based injuries, from (internal) biomechanical deformations or from pressure based (external) deformations.9-11 Neuropathy of: (i) motor cells causes changes in gait (additional/misplaced pressure), (ii) sensory cells causes loss of sensation (abnormal pressures not felt), and (iii) autonomic cells causes reduced tissue repair/viability (normal tissue functions disrupted).12-14 A loss of coordinated arterial functions and ischemia in the extremity is often associated with limb dysfunction, promoting increased pressures and callus formation. 15 As a result of callus deposition, there is a higher risk for DFU onset as excess build-up further increases pressure on surrounding tissue causing more cell deformation and death.16-18 Underlying necrotic tissue can become infected or ulcerative and may be masked by superficial callus layers. Neuropathic ulcers are commonly found beneath plantar calluses. Therefore, it is recommended that calluses are removed to prevent the onset of diabetic foot ulcerations and/or reduce pressure in the diabetic foot.19-21
Scalpel Debridement
Topical treatments have been developed to remove or maintain callus sites such as salicylic acid pads and keratin dissolving agents.22,23 One common method is force removal in a surgical process known as scalpel debridement (SD), or chiropody. 24 During SD, callus skin is removed until the clinician observes a desirable color or texture to the callused area (eg, rose color refers to healthy skin), indicating the callus tissue has been removed. Other approaches to determine the extent of debridement (differentiating callus to healthy tissue) are practiced by clinicians but with no scientific evidence of a standardized approach (limited by the clinician). Figure 1 illustrates SD process in which the callus (see Figure 1a) is removed (see Figure 1b) revealing healthy tissue below (see Figure 1c). Vascular changes following debridement are observed as a pink color in the underlying dermal region compared with the surroundings.
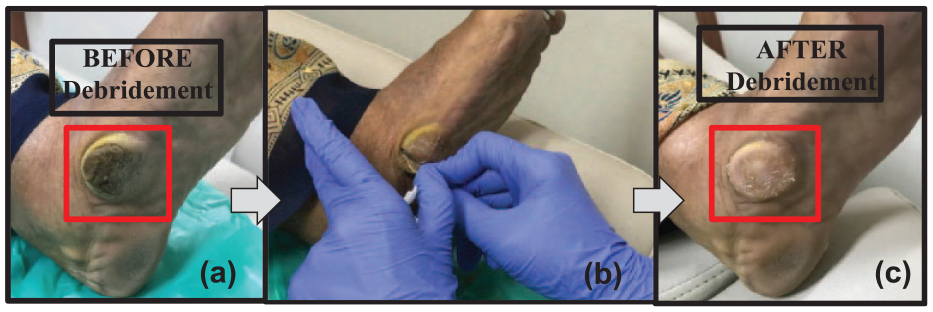
Location of callus is highlighted within the red-boxed region on the inner sole of patient’s foot. (a) Prior to debridement the callus tissue is dark in color. (b) Following removal of the superficial layers, (c) only pink skin remains, satisfying the clinician for completion of the scalpel debridement process (or chiropody).
Assessment of Scalpel Debridement
Other factors could alter the outcome of SD such as patient sensitivity and surgical inexperience. Manual clinician skills are learned with time, which can be labor-consuming and lengthy. In the past, the effect of SD has commonly been determined using pain scales and/or foot pressure measurements but many report conflicting results.25-31 Other measurements to study the effect of debridement include skin surface hydration (capacitance based), skin elasticity (negative pressure), and skin surface texture (high-resolution imaging). 32 Digital images are acquired to analyze skin texture following debridement and on a long-term tracking area reduction compared to baseline (day one of treatment). Color images are useful for surface analysis, but other imaging modalities have been explored in parallel. Fluorescence imaging was used to assess penetration depths of contrast dye comparing the effect of a topical therapeutic agent on callus tissue. 33 Thermography and ultrasound techniques have been explored to assess differences in callus tissue. 34 Optical coherence tomography (OCT) and near-infrared (NIR) imaging were used to measure changes in optical depth and contrast (of diffuse reflectance) in callus tissue following dissolved keratin treatment. 35 In this study, NIR imaging is used to assess tissue oxygenation (TO) changes following SD in pre-ulcerative cases.
Near-Infrared Imaging of Diabetic Foot Ulcers
In wound imaging applications, obtaining multiple NIR diffuse reflectance images are used to estimate oxygen saturation in soft tissues 36 and as a healing indicator in DFUs. 37 A hand-held Near-InfraRed Optical Scanner (or NIROS) was developed in our lab recently.38-42 The device measures effective (or relative) changes in TO in terms of 4 parameters: oxy-hemoglobin (∆HbO), deoxy-hemoglobin (∆HbR), total hemoglobin (∆HbT), and oxygen saturation (∆StO2). NIROS imaging studies were performed on DFU subjects across weeks of treatment to study changes in the four TO parameters. Two dimensional (2D) pseudo-color maps for each parameter showed distinct differences between TO concentrations within the wound (W) compared to the surrounding background, also known as the peri-wound (PW). It was observed that as the wound healed, contrast between the wound and peri-wound (W:PW) approached zero, while in non-healing cases this contrast remained or increased.40-42 Various imaging studies have been conducted with emphasis on changes occurring within the wound and peri-wound (W:PW).43,44 Important features occurring at this W:PW boundary such as presence of cellulitis/erythema, temperature, and oxygen saturation are potential biomarkers to monitor wound healing.40-42,45-47
Near Infrared Imaging of Callus Tissue
It is possible that similar approaches may be employed comparing contrast in TO between callus (C) and peri-callus (PC) tissue in pre-ulcerative cases. Understanding how C:PC contrast changes in response to debridement is of particular importance in this study. Due to the dense morphology and lack of vasculature the hypothesis is that callus tissue (compared to surrounding peri-callus tissue) will be spatially distinct and observable on 2D TO maps. Hence the objective of this study is to obtain TO measurements before and after SD and compare the resulting C:PC contrasts across subjects with diabetes using NIROS. Changes in TO (due to SD procedure) will be assessed and compared with clinical observations in a retro-observational, feasibility study. On a long-term, objective assessment of SD could be beneficial for maintenance of the diabetic foot in pre-ulcerative cases.
Methods
Instrumentation
A continuous wave (CW) NIR based, non-contact optical device (NIROS) was used in this study. A dual wavelength (735/805nm) LED light source is illuminated onto the callus region and surrounding tissue. The LED driver maintains a stable maximum optical power of 7 mW (measured point blank) per LED (and per wavelength). Wavelengths are multiplexed to acquire NIR diffuse reflectance images using CMOS camera after passing through a 645 nm long pass filter. A uniformly diffusing calibration sheet is centered over the callus region to obtain the reference intensity distribution. NIROS is mounted on an articulating arm to stabilize the entire device during imaging. The source and detector are in-sync via a custom-developed (using Matlab) graphical user interface (GUI) controlling image acquisition and storage for further processing. The clinical setup is shown in Figure 2.
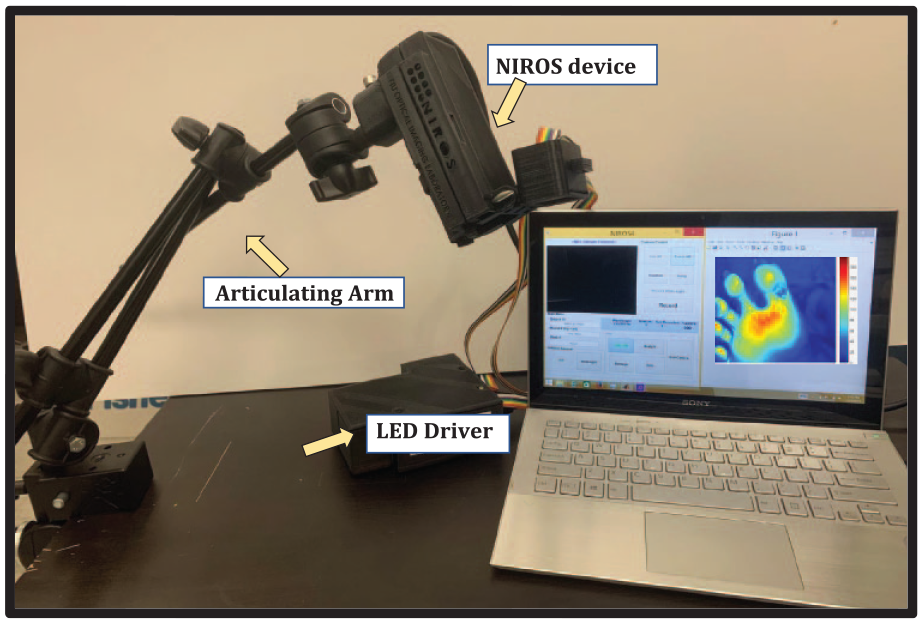
NIROS device using CMOS detector and multi-wavelength NIR LEDs, with laptop for data acquisition and processing. The articulating arm allows for easy and stable positioning during imaging.
Subject Recruitment
Subjects with diabetes and callus(es) requiring SD were recruited for this study (45 to 90+ years old) during their periodic visit to Dr. Mohan’s Diabetes Specialties Center (Chennai, India) to avoid the risk of DFU onset. In this IRB (Institutional Review Board) approved study, between Florida International University (FIU) and Madras Diabetes Research Foundation (to which Dr. Mohan’s Diabetes Specialties Center is affiliated to), all participants agreed and signed a written consent in English or Tamil (the local language). Ten subjects were recruited in this study with single or multiple calluses (on one or both feet), for a total of 15 callus regions that underwent debridement. The data was collected at Dr. Mohan’s Center and was analyzed in a retro-observational study at FIU.
Data Acquisition
NIROS device was always positioned to encompass the entire callus and surrounding regions. Diffuse reflectance and respective calibration images were acquired (at 735 and 805 nm illumination) immediately before and after SD. Figure 3 displays the raw diffuse reflectance data at one NIR wavelength (735nm) on the respective foot for 15 cases, the callus region is highlighted using a pseudo-color map. The higher intensity regions appear as red (max) while lower intensity regions appear as blue (min). Data collected at both wavelengths (before and after SD) are used to estimate oxygenation maps in the following section.
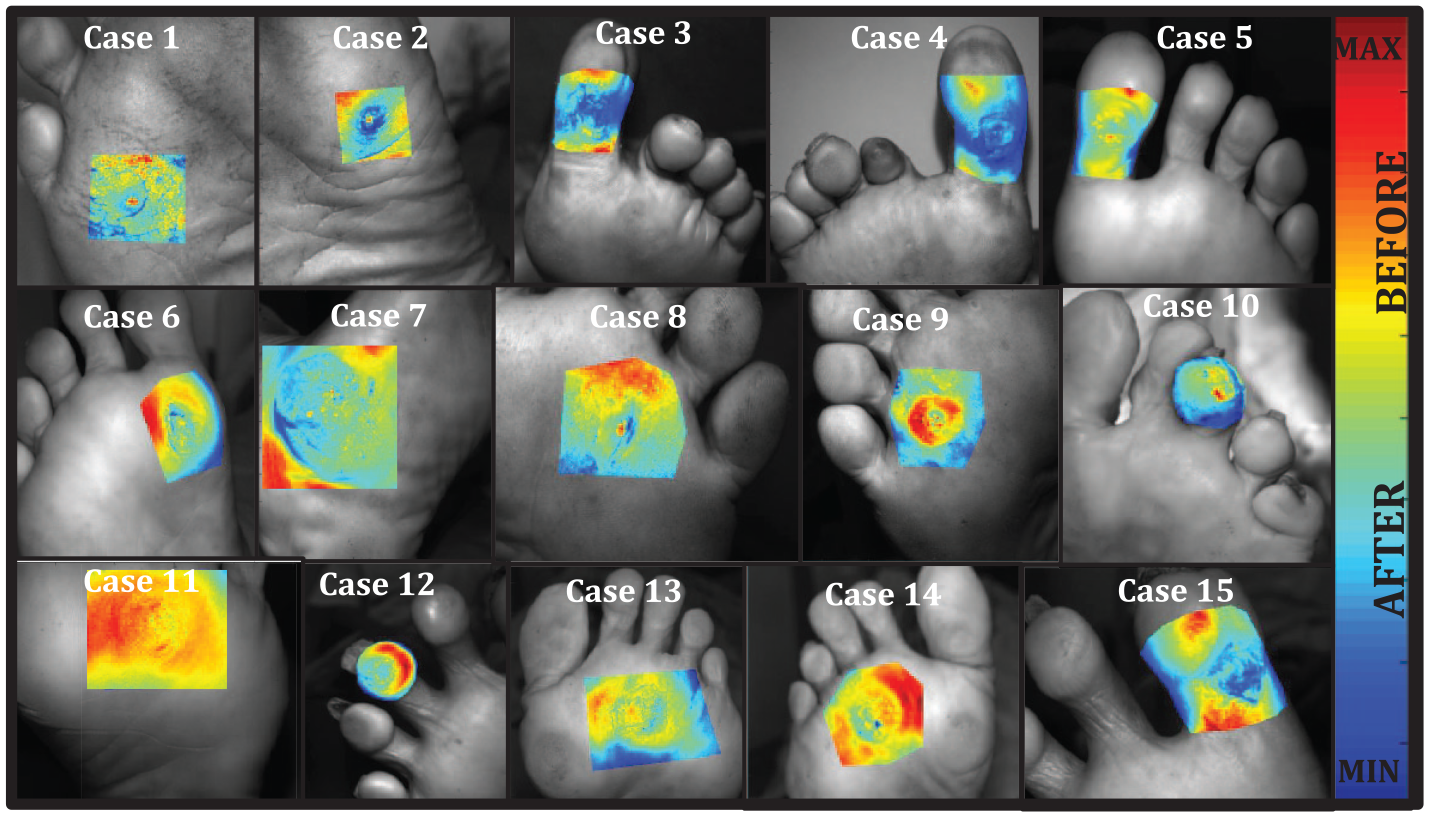
Fifteen callus sites are shown before debridement as diffuse reflectance NIR signal (at 735nm) demarcating the overall field of view imaging area (gray) and the region of interest (pseudo-color area) where imaging analysis was performed. Here, red corresponds to higher intensities and blue corresponds to lower intensities of the measured diffuse reflectance signals.
Data Processing
Tissue oxygenation maps
Images were analyzed as 2D arrays in a pixel-by-pixel approach using Modified Beer Lambert’s Law (MBLL) in which the diffuse reflectance signal collected at dual wavelengths is proportional to the effective (or relative) concentrations of ∆HbO and ∆HbR according to a previously described approach for NIR imaging. 46 ∆HbT and ∆StO2 measurements are given by ∆HbO+∆HbR and ∆HbO/∆HbT, respectively. Figure 4 depicts the resulting 2D spatial maps of the TO parameters in terms of ∆HbR, ∆HbO, ∆HbT, and ∆StO2 obtained before and after SD (shown for one example case #14). Images were normalized across ∆HbR, ∆HbO (since they are related parameters), and independently for ∆HbT and ∆StO2. Qualitative differences in TO can be seen in individual plots within the callus and surrounding regions on each pseudo-color map. Regions of red indicate higher effective concentrations (values closer to one) and regions of blue indicate lower effective concentrations (values closer to zero) of TO. However, a qualitative comparison is not always sufficient for identifying significant changes in TO concentration in response to SD, and possibly introduces variability across clinicians’ assessment. With a focus to avoid ambiguity in qualitative assessments of images, pseudo-color maps for each parameter are compared further in a quantitative analysis following SD.”
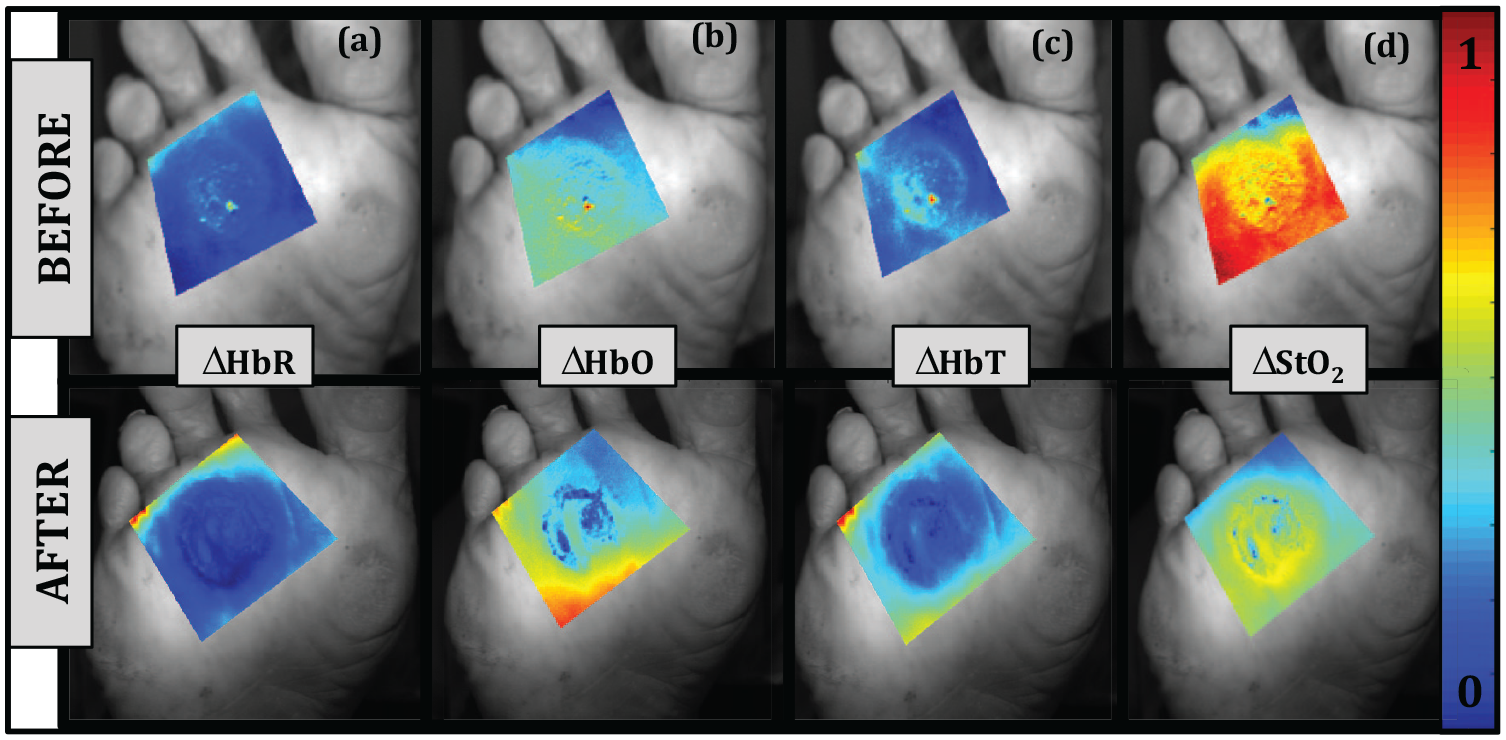
Tissue oxygenation maps in terms of (a) ∆HbR (b) ∆HbO (c) ∆HbT and (d) ∆StO2 before (top) and after (bottom) scalpel debridement for case #14. Here, red corresponds to higher concentrations and blue corresponds to lower concentrations of the given parameter.
Callus: Peri-callus contrast ratios
The C:PC based contrast was determined for each TO parameter (∆HbO, ∆HbR, ∆HbT, ∆StO2) obtained before and after SD. Callus formation generally proliferates from a localized central point and the approximate area of each callus can be calculated as a circular or oval region. 32 Hence, the callus was approximated as a circle in this contrast analysis and was demarcated by selecting the callus center and greatest edge. To capture changes occurring at the C:PC boundary, the region of analysis will extend slightly further by 10 pixels. A second circle is automatically generated with a radius of 10 pixels larger than the callus (as shown in Figure 5). The PC tissue is the area between these two circles, positioned directly next to callus but presenting a normal morphology. The C:PC contrast ratio is estimated for each TO parameter (before/after) across all cases. A secondary Weber contrast or relative contrast with respect to a chosen baseline (here it is TO measured prior to SD) was calculated to quantify how much each TO parameter increased or decreased in response to treatment (ie, before vs after). An example analysis is shown in Figure 5 for case 9. This Weber contrast analysis was performed three times (circle selected three times each, during data analysis) for each case.
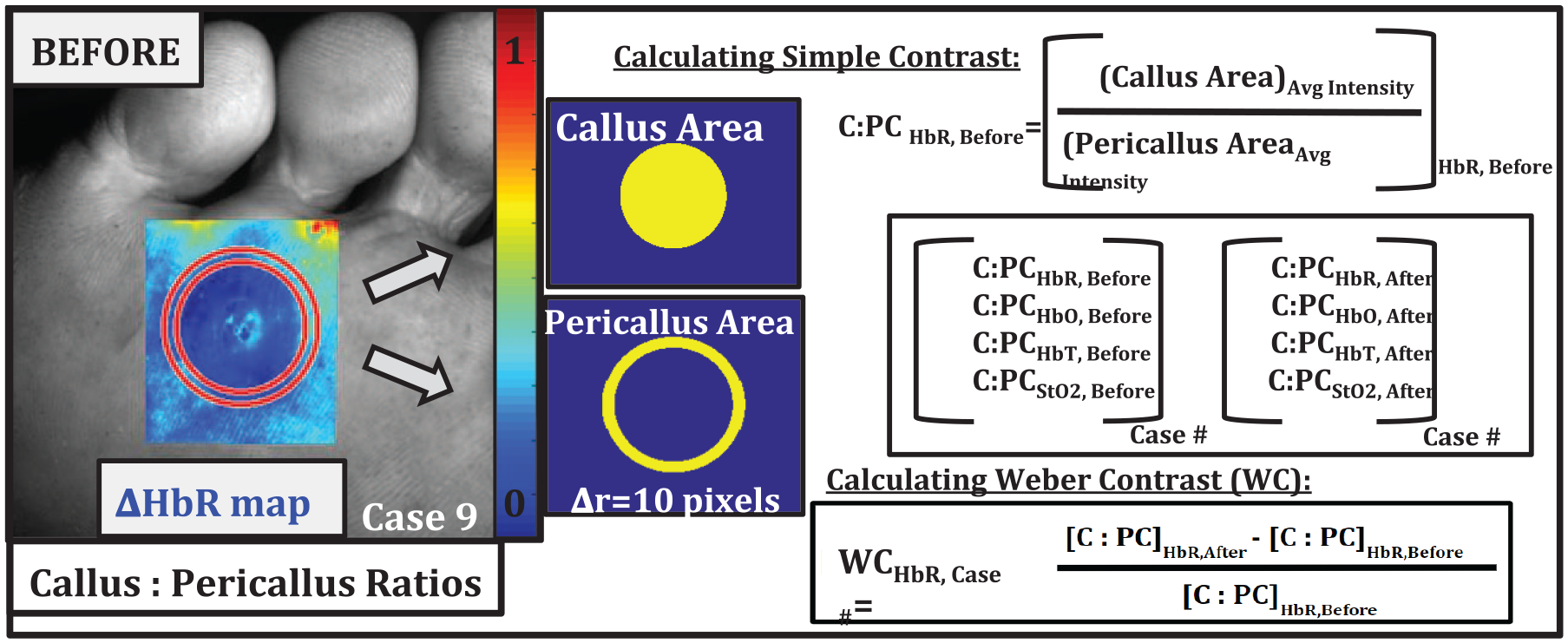
Method of callus and peri-callus area selection C:PC for one case with (i) C:PC contrast, and (ii) Weber contrast for each parameter (example case for ∆HbR shown).
Sub-callus tissue oxygenation contrast matrices
The nature of callus buildup is non-uniform and there can be irregularities in thickness across the callus surface. No two calluses will develop the same way or even at a similar rate.
48
In the previous approach (described in Section 2.4.2), Weber contrast ratios were determined for each TO parameter using the entire C:PC area (shown in Figure 5) as a single average value. Overall, a change in TO may occur but it does not imply they are significant, nor that debridement is uniform across the entire affected area. In this secondary approach, the C:PC contrast values are sampled at specific points within the callus and peri-callus tissue in each of the four quadrants (within the demarcated circular region) and statistically tested. Three locations or regions of interest (ROIs) were automatically selected within each quadrant spanning across the callus tissue (ie, at 15°, 45°, and 75°). At these three ROIs, a 6×6 pixel area was demarcated within the callus tissue and within directly adjacent, peri-callus tissue (black squares shown in Figure 6). These boxed regions or arrays were combined to form a contrast matrix between the callus and peri-callus tissue [C:PC]. Contrast matrices were reshaped into a line vector and a paired t-test performed (between before and after data) to determine if a significant difference was observed in TO contrast (for a given parameter) at that location (α=0.01). This t-test was performed at each of the three locations per quadrant, for all four TO parameters in this study (
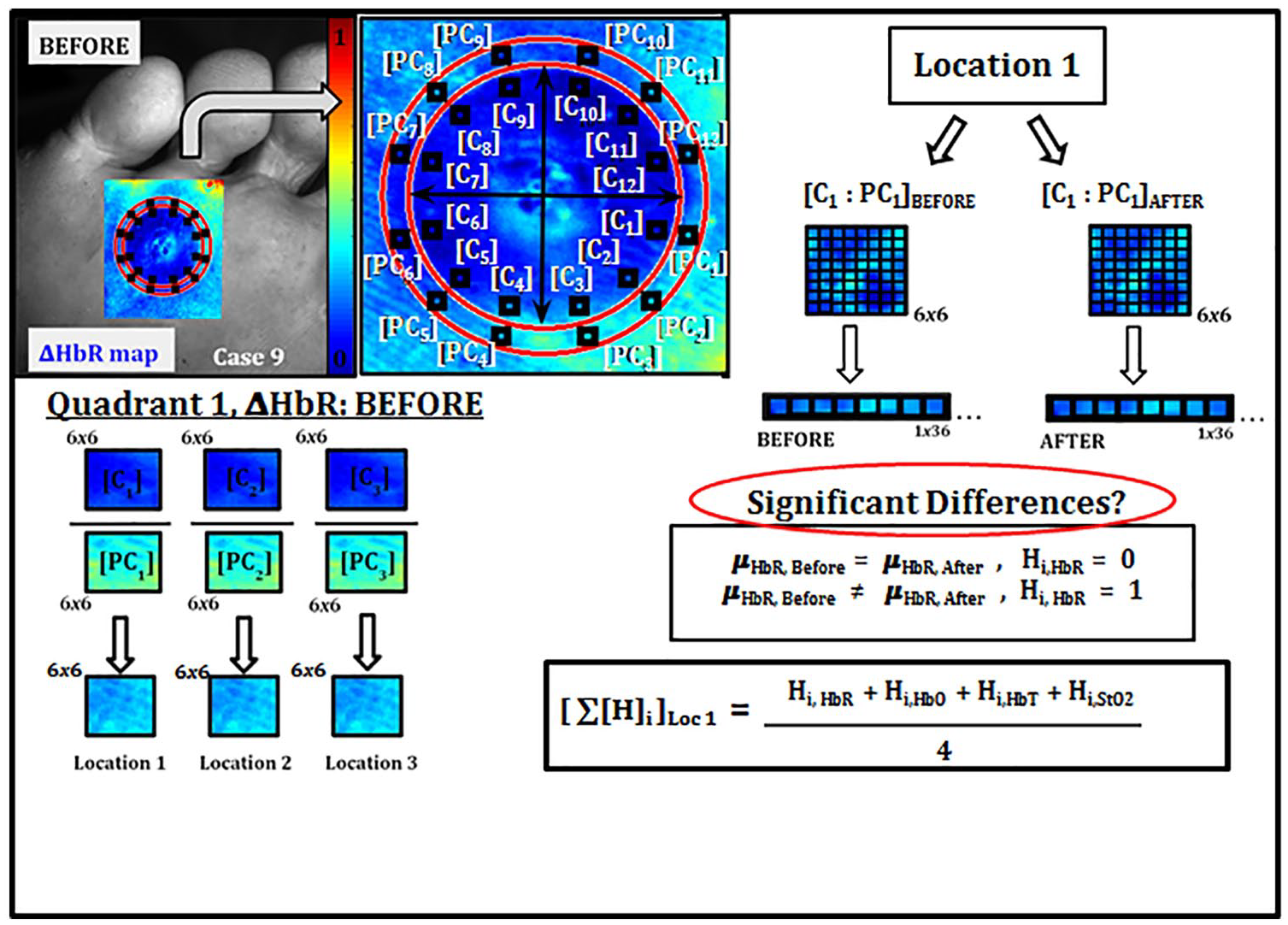
Method of [C:PC] region selection for each quadrant and quadrant significance percentage calculations for each TO parameter (∆HbR, ∆HbO, ∆HbT, ∆StO2).
Results
Subject Details
The final dataset includes results from nine subjects with diabetes with a total of 15 debridement cases (described as case #s henceforth) imaged before and after procedure. Other cases were excluded due to incomplete data collection during imaging. Six out of the 15 cases received monthly debridement, case #7 received SD every three months, and it was the first debridement visit for case #11. Subject details are given in Table 1. When no information was available, it was listed as N/A. Case #s 2, 3, 4, and 5 are marked with orange asterisks as the post-clinical assessment noted a blood clot observed beneath callus. Case #s 8 and 12 are marked with blue asterisks as incomplete debridement was suspected by clinician. All cases are classified as diabetic neuropathic and no arterial insufficiencies were noted.
Details of the Recruited Subjects with Diabetes Undergoing Scalpel Debridement (Chiropody) and the Clinical Assessment of the Process.
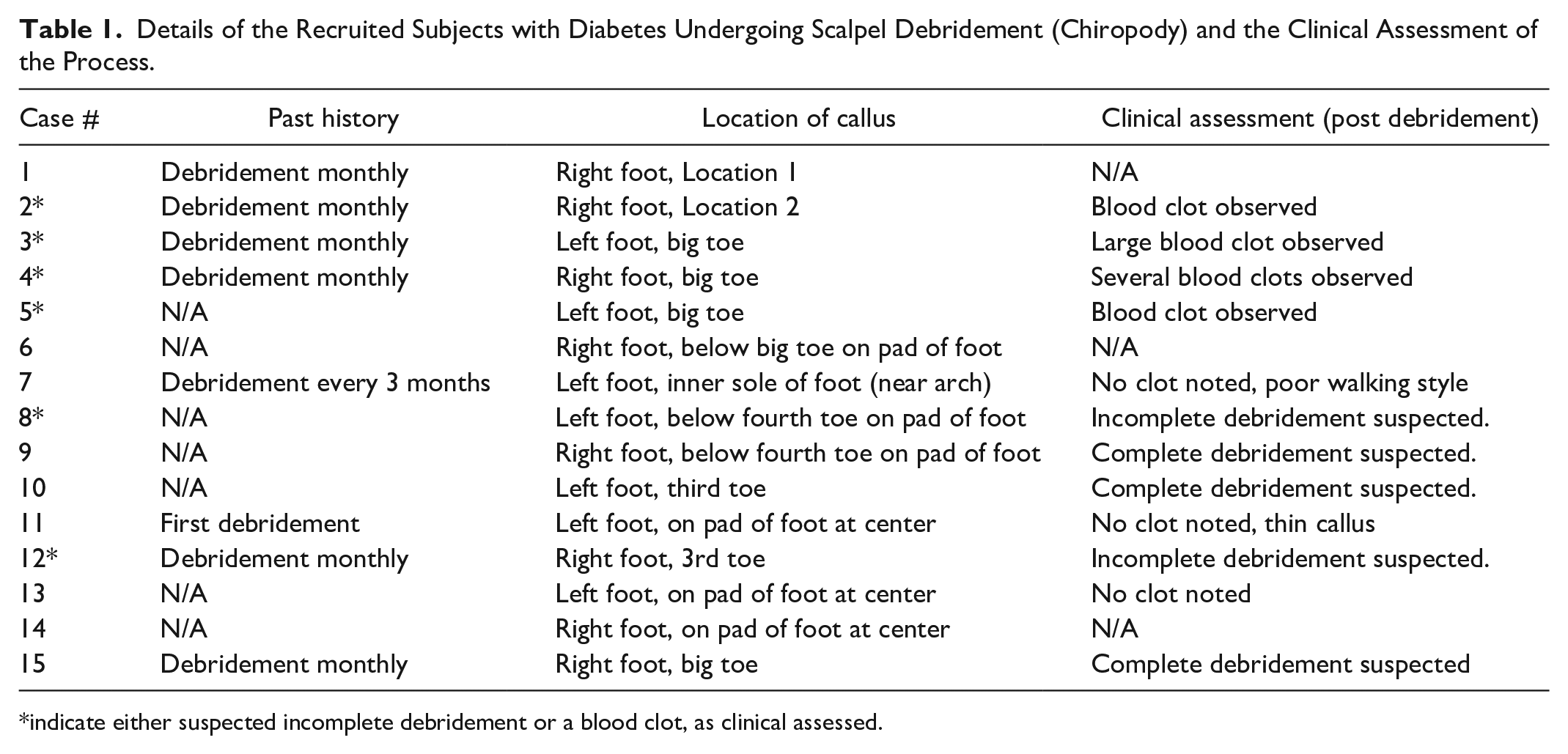
indicate either suspected incomplete debridement or a blood clot, as clinical assessed.
Tissue Oxygenation Changes in Weber Contrast
Weber (or relative) contrast results displaying a positive contrast indicates an increase in concentration (of the given parameter) within the callus region (compared to PC). A negative Weber contrast indicates a decrease in concentration (of the given parameter) within the callus region (compared to PC). The Weber contrast for all TO parameters and across all 15 cases is shown in Figure 7. It was found that contrast ratios were reduced (across the 15 cases) in terms of
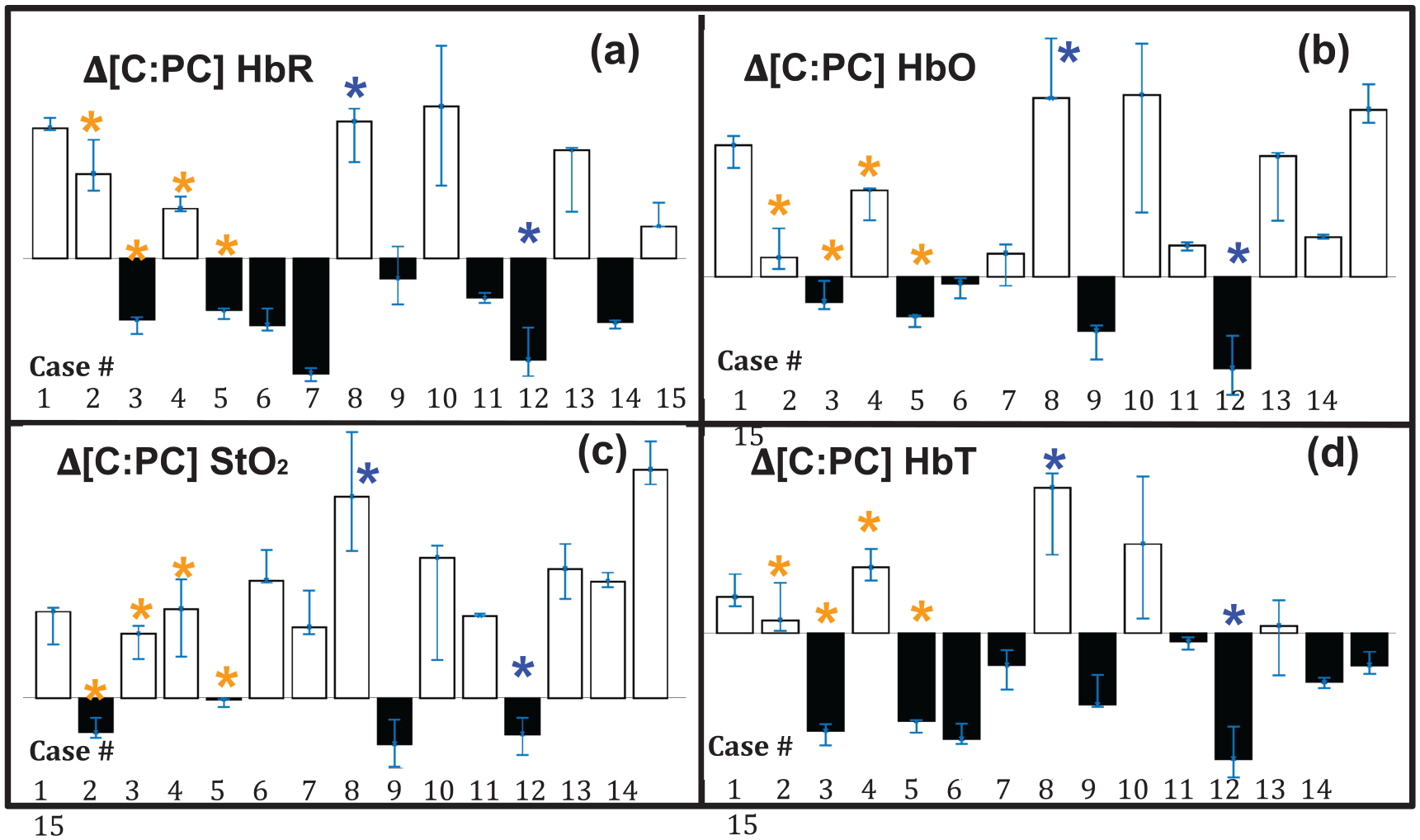
Bar plots of Weber contrast in C:PC following debridement. Results are shown for each debridement case and for each TO parameter: (a)
Sub-quadrant Percent Significance
Overall, TO in terms of the four parameters did show a difference in contrast following debridement but whether this change is significant was unclear. The callus tissue was partitioned into four sub-quadrants. For each quadrant, a percent of significance was determined as described in Section 2.4.3 and shown in Figure 6. Each quadrant’s calculated percent significance was overlain onto white light images of each debridement case and shown in Figure 8. The value 80% was chosen arbitrarily as the minimum significant change in TO following SD. Any quadrant value below 80% significance is displayed in red, while cases above this value are displayed in green, in Figure 8.
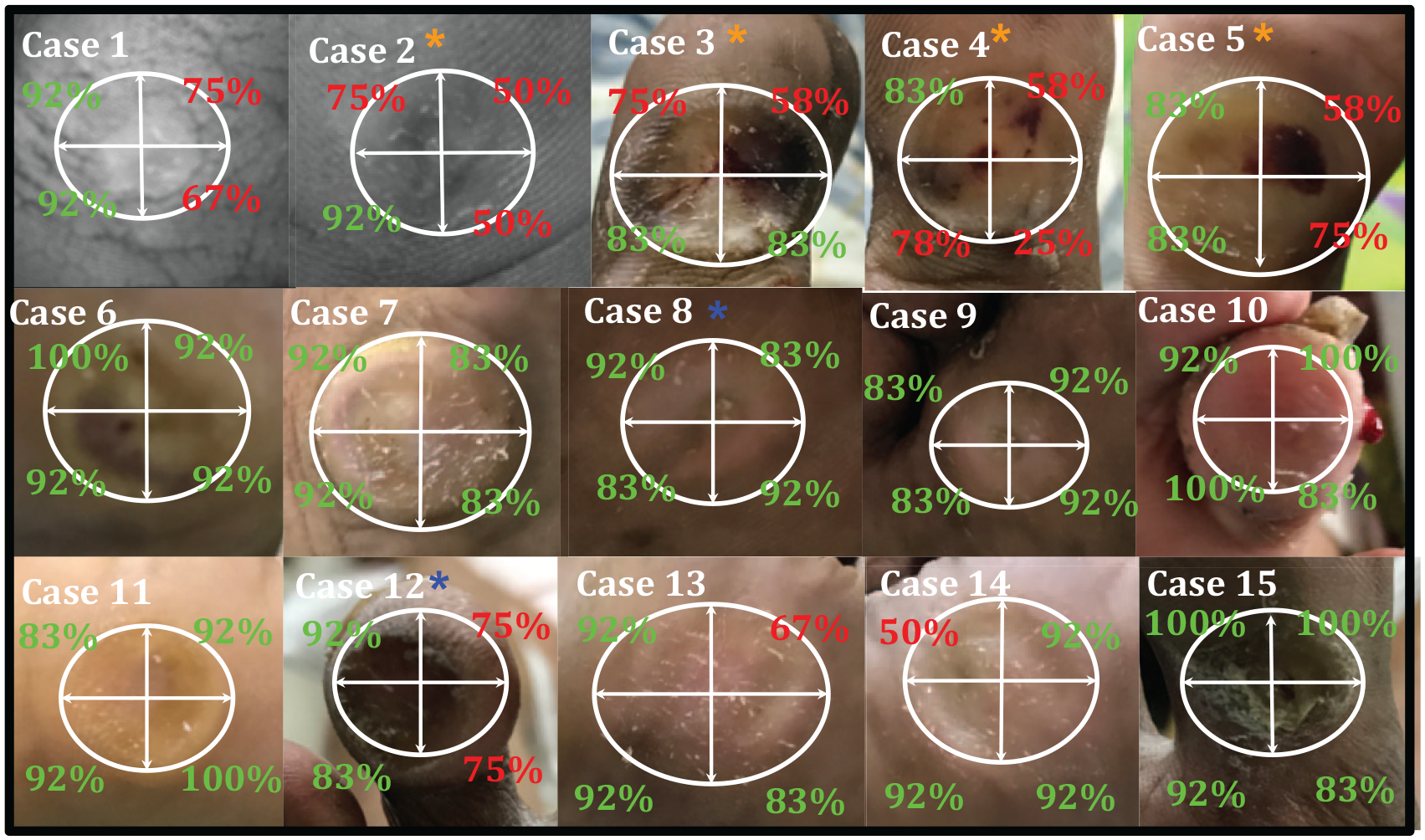
Percent significance of TO change (80% threshold) determined per quadrant for clinical representation of effectiveness in scalpel debridement from an objective physiological imaging approach. White light images were not available for cases 1 and 2 hence the percent significance results were overlain onto the NIR gray scale images (obtained at 735nm).
Nine cases (6, 7, 8, 9, 10, 11, 13, 14, 15) showed a significant change in oxygenation across at least three quadrants while six cases only had a significant change in two or less quadrants. If the sub-quadrant results from Figure 8 are displayed on a plot by taking the average of the four quadrants, it can be clearly seen from Figure 9 that cases with blood clots (case #s 2, 3, 4, and 5, shown by orange asterisks) did not indicate significant change in TO following debridement. Cases #s 8 and 12, suspected of incomplete debridement did surpass the threshold, although case 12 was close to this margin, shown in Figure 9 as the dashed, horizontal (red) line.
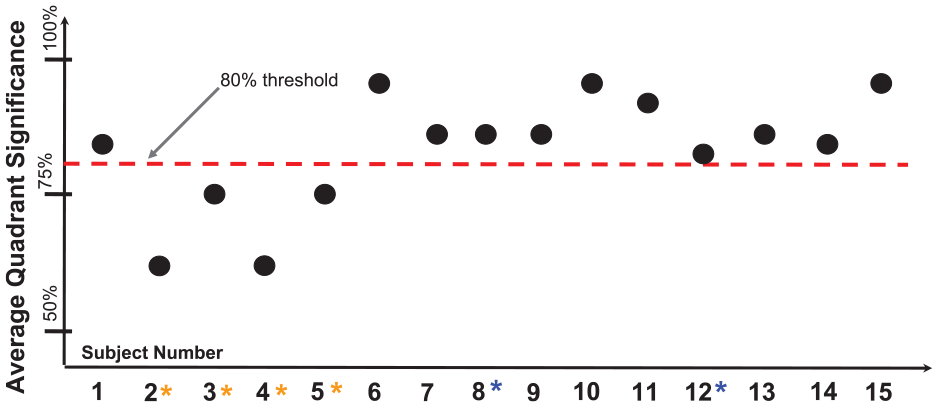
Average significance of tissue oxygenation changes within callus (compared to peri-callus) following debridement for 15 cases. An arbitrary threshold (80%) selected for this study defines the minimum average quadrant significance expected following debridement.
Discussion
In general, there are a few reasons why TO measurements may not change significantly following debridement. It is possible the debridement is incomplete and NIR light is completely absorbed as the dead cells do not allow for sufficient penetration. A previous study (using hyperspectral imaging) measuring TO in wounds reported the presence of callus causing error in obtaining accurate TO measurements. 49 Possibly the NIR signal was not capable of penetrating very deep due to dead tissue build-up and TO measurements were not obtained from beneath. Once dead tissue is removed, there should be an increase in TO. However, there may be no healthy skin underneath when a clinician encounters a buried blood clot or an ulceration appearing as dark red spots (or blood clots) at callus center, which could be an early indicator of DFU onset. 25 From a clinician’s perspective the debridement process may appear complete but from a physiological perspective there may not be any significant blood flow (or change in TO) when a blood clot is present: as observed in cases 2, 3, 4, and 5, and confirmed from post-debridement assessment. Additionally, individuals with diabetes are often affected by arterial insufficiencies. This could be another reason why TO may not be significantly improved as flow is limited in the extremity. However, none of the recruited subjects were reported with peripheral arterial disease in this small cohort. Hence, in this study the lack of significant change in TO after SD was possibly from incomplete debridement and/or presence of blood clots.
Following our TO-based assessment, it appears that the arbitrary choice of 80% cut-off to determine effectiveness of SD (from percentage of significance) is reasonable as it explicitly identified four out of the six cases exhibiting blood clot. Case 12 only suspected of incomplete debridement was identified as borderline and possibly the threshold should be increased slightly. Following further observational studies this hypothesis can be tested. Case 9 was also suspected as incomplete debridement by the clinician but was not identified by this approach. It is possible that debridement was in fact, complete from a physiological standpoint but visually appeared incomplete to the clinician. In future, more systematic studies are needed to understand how oxygenation changes occur in subjects with diabetes following SD. It is possible that this technology can be used as a tool or a guide for clinicians to administer SD and improve the effectiveness of treatment to prevent active ulceration, and thus amputation in the future. In summary, TO changes are expected post debridement and quantifying the extent of this change for each quadrant of the callus by a percentage significance value may serve as an objective approach to assess effectiveness of SD. The cut-off threshold value can be altered in future studies involving more subject cases to robustly demarcate regions of poor debridement vs successful debridement, and also monitor for potential pre-ulcerative blood clots, not visible to the naked eye. Assessment of the extent of debridement from a physiological perspective reduces variability among treatment plans. Independent of the clinician or who administers the debridement this objective imaging technique may be applied for improving pre-ulcerative diabetic foot management and also potential early-on detection of possible ulcerations (ie, DFU predictions) in future.
Conclusion
SD is a surgical procedure administered as treatment for DFU prevention. The effectiveness of each treatment is not measurable by any given standard and remains an irregular process dependent on the experience of the administering clinician. In this image analysis study, the effect of SD with respect to four parameters of TO (
Footnotes
Acknowledgements
We would like to thank the staff at Dr. Mohan’s Diabetes Specialties Centre in Chennai, India and all of the members in the Optical Imaging Laboratory at Florida International University.
Abbreviations
∆HbO, effective Oxy-Hemoglobin; ∆HbR, effective Deoxy-Hemoglobin; ∆HbT, effective Total Hemoglobin; ∆StO2, effective Oxygen Saturation; 2D, Two dimensional; C, Callus; C:PC, Callus to Peri-Callus contrast; CMOS, Complementary Metal-Oxide Semiconductor; CW, Continuous Wave; DFUs, Diabetic Foot Ulcers; DM, Diabetes Mellitus; FIU, Florida International University; GUI, Graphical User Interface; IRB, Institutional Review Board; MBLL, Modified Beer Lambert’s Law; N/A, Not Applicable; NIR, Near InfraRed; NIR, Near InfraRed; NIROS, Near-InfraRed Optical Scanner; OIL, Optical Imaging Laboratory; PC, Peri-Callus; PW, Peri-Wound; ROIs, Regions Of Interest; SD, Scalpel Debridement; TO, Tissue Oxygenation; W, Wound; W, Wound; W:PW, Wound to Peri-Wound contrast.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
