Abstract
Background:
Increased local skin temperature (hotspot) on a callus site as detected by thermography is a well-known precursor of diabetic foot ulcers. This study aimed to determine the factors associated with callus hotspots in order to predict the risk of callus hotspots and then provide information for specific interventions.
Methods:
In this cross-sectional study, 1,007 patients’ data from a diabetic foot prevention clinic between April 2008 and March 2020 were used. Data regarding patients’ characteristics, foot calluses, and callus hotspots were collected and analyzed. Callus and callus hotspot were confirmed from foot photos and thermographs, respectively. A callus hotspot was defined as a relative increase in temperature compared to the skin surrounding the callus on the thermograph. Plantar pressure was measured with a pressure distribution measurement system. A generalized linear mixed model was used to identify the factors associated with callus hotspots.
Results:
Among the 2,014 feet, 28.5% had calluses, and 18.5% of feet with calluses had callus hotspots. The factors associated with callus hotspots were number of calluses (Adjusted odds ratio (aOR): 1.540, P = .003), static forefoot peak plantar pressure (SFPPP) (aOR: 1.008, P = .001), and body mass index (aOR: 0.912, P = .029).
Conclusions:
Patients with a higher SFPPP were more likely to have callus hotspots suggesting that SFPPP might contribute to callus inflammation. SFPPP has the potential to be a useful predictor of callus hotspots in people with diabetes and at the same time provide information for off-loading interventions to prevent callus hotspots.
Introduction
Diabetic foot ulcer (DFU) is one of the major complications of diabetes. It is defined as a break of the skin of the foot that includes minimally the epidermis and part of the dermis. 1 DFUs can lead to high amputation rate, high mortality rate, and enormous economic burden to society as well as to patients and their families.2-4 Many strategies on the prevention of DFUs have been studied for the purpose of reducing the burdens DFUs bring to people with diabetes and society. Among these strategies, the monitoring of increased local skin temperature (hotspot), a sign of inflammation, has gained substantial attention over the years, and its effectiveness on reducing the risk for DFUs has been shown. 5 As a tool for visualizing skin surface temperature distribution, infrared thermography has been widespread in its use for detecting and predicting DFUs by monitoring hotspots on the sole.6-9 Previous studies focused on calluses reported that a hotspot on the callus site could be an early sign of inflammation, and the spread of increased temperature beyond the callus site could indicate callus progression to ulceration.10,11 These findings suggest the possibility of preventing or ensuring prompt treatment of callus hotspots to prevent DFUs. 12
Although numerous studies explored the causes of calluses as well as DFUs, the cause of callus hotspots remains unknown. Calluses are caused by excessive mechanical loading, 1 including plantar pressure and shear stress during gait. 13 Excessive mechanical loading can result from any combination of loss of protective sensation resulting from sensory neuropathy, foot deformities, limited joint mobility resulting from motor neuropathy, and excessive irritation from ill-fitting shoes.1,13-15 The abnormal loading on feet with calluses leads to a further increase in the loading of the underlying tissue, which eventually might lead to ulceration.16,17 However, not all calluses ulcerate. A recent cohort study reported that 11% of calluses among people with diabetes ulcerated. 18 Discovering the factors that contribute to callus hotspots, which occur in the stage preceding ulceration, could provide early risk detection of DFUs. These factors could be the worsening of existing factors that cause callus formation or completely different factors that contribute only to callus hotspots. For those factors confirmed to be included in the factors for callus formation, intervention care targeted at calluses should be effective for preventing hotspots. For factors that differ from the factors for callus formation, new intervention methods will be needed to address these hotspot-only factors.
The current study aimed to determine the factors associated with hotspots in diabetic foot calluses. We also investigated those factors associated with the presence of calluses to ascertain whether factors associated with presence of callus hotspots are also associated with calluses.
Methods
Study Design, Setting, and Participants
A cross-sectional study was conducted in a diabetic foot prevention care clinic at a university hospital located in an urban area of Japan. Participants in this study were those who visited this prevention clinic between April 2008 and March 2020. The inclusion criterion was patients who were diagnosed with diabetes. The exclusion criteria were patients who had DFUs at the first visit or were unable to stand without assistance.
Data Collection and Variables
Data used for analysis were obtained from medical records. Data from the first visit to the clinic were used for the data analysis. Data were collected between May 2020 and July 2020. Variables related to the 2 outcomes of callus and callus hotspot were selected in this study. Callus was defined as hyperkeratosis caused by excessive mechanical loading. 19 A callus hotspot was defined as a relative increase in temperature compared to the skin surrounding the callus on the thermograph. The presence of callus and callus hotspot were judged by 2 wound care management research nurses and 1 certified nurse in diabetic nursing from the diabetic foot clinic. If the judgments were divided, then the researchers would discuss until a consensus was reached.
Patients’ characteristics included age, sex, type of diabetes, duration of diabetes, HbA1c, body mass index (BMI), sensory neuropathy, motor neuropathy, autonomic neuropathy, peripheral artery disease (PAD), and history of DFUs. Callus formation related variables included dry skin, foot deformity, 20 ill-fitting shoes,13,21 and static forefoot peak plantar pressure (SFPPP).22,23 Callus hotspot related variables were the number of calluses, location, and whether the callus site matches the site of peak plantar pressure. 24
Variable Measurements and Definitions
Each variable was measured on the patients’ first visit to the diabetic foot prevention care clinic. Callus hotspot was judged from the plantar thermographic images that were taken after patients rested on a chair for more than 15 minutes to acclimate to the temperature. Three different types of thermographic devices were used at the diabetic foot prevention care clinic during different periods: Thermo Tracer TH5108ME (NEC Avio Infrared Technologies Co., Ltd., Tokyo, Japan), Thermo Tracer TH7800N, or FLIR CPA-T420A (FLIR® Systems, Inc., Wilsonville, OR, USA). Callus hotspot was judged qualitatively: the temperature on the callus was increased relative to the surrounding skin temperature after the thermal image setting unified the span (temperature range) to 12°C using InfRec Analyzer NS9200 (Nippon Avionics Co., Ltd., Kanagawa, Japan) or FLIR tools (FLIR® Systems, Inc.) (Figure 1). Because the judgment of a hotspot was done using a qualitative method of temperature comparison instead of an absolute temperature comparison, the judgment of callus hotspots was not affected and had a high inter-rater reliability (Cohen’s κ = .9160) between researchers despite using 3 different models of thermography.
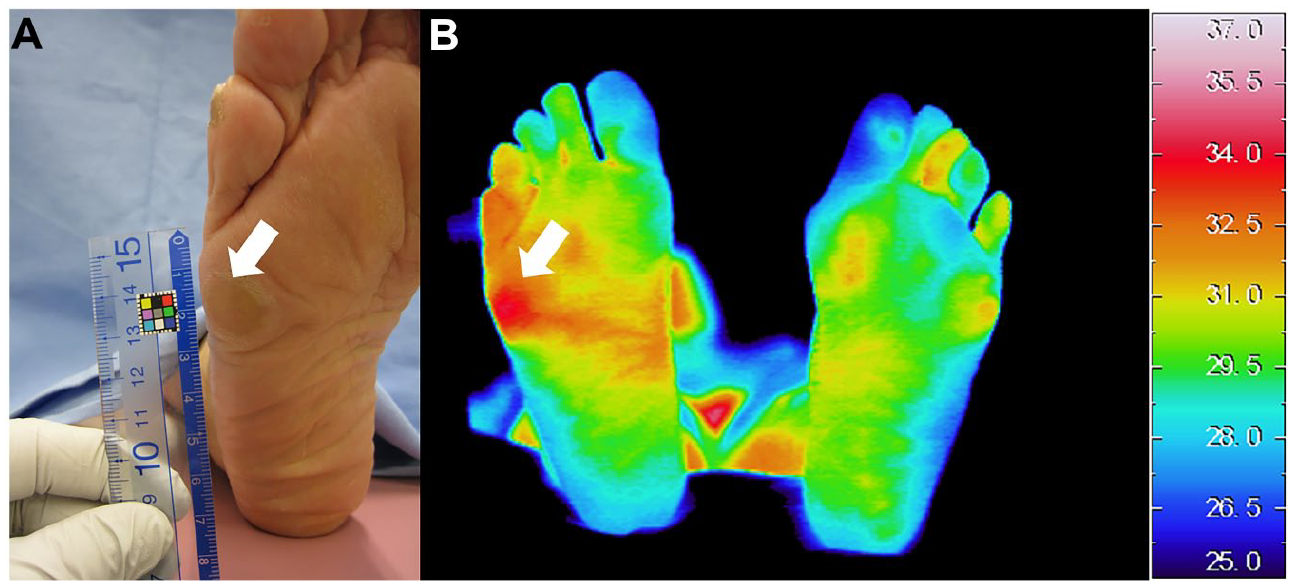
A callus hotspot in photo (A) and thermography (B). The callus denoted by the white arrow in the photography (A) matches a hotpsot on the thermograph (B).
Sensory neuropathy was judged by the results of the vibratory perception test and Semmes-Weinstein 5.07 monofilament test. Patients who had a loss of protective sensation or vibratory perception in medial malleoli less than 10 seconds were judged as positive for sensory neuropathy. 25 The presence of motor neuropathy was judged via the Achilles tendon reflex test. The presence of autonomic neuropathy was defined as the coefficient of variation R-R interval (CVRR) < 0.2. 26 The presence of PAD was defined as an ankle brachial index (ABI) ≤ 0.9 or when ABI > 1.4 and toe brachial index (TBI) ≤ 0.7. 27 CVRR, ABI, and TBI were measured with pulse-wave velocity/ABI BP 203RPEII (OMRON COLIN, Co., Ltd., Tokyo, Japan). Dry skin was defined as heel moisture level < 20% when measured with Scalar skin moisture checker MY707S (Scalar Co., Tokyo, Japan). 28 Foot deformity was defined as the presence of hammertoe, claw toes, hallux valgus, prominent metatarsal heads, or amputation. 1 An ill-fitting shoe was defined as the inside length of the shoe was not 1–2 cm longer than the foot. 19 The peak static pressure of the forefoot was measured when patients were standing barefoot on a foot pressure distribution measurement system, F-scan (Nitta Co., Ltd., Osaka, Japan), before the callus removal. The Body Pressure Measurement System Ver. 5.88c (Tekscan Inc., South Boston, MA, USA) software was used for pressure reading. Peak pressure was calculated by taking the mean of each 2-pixel by 2-pixel subregion of the plantar region of the foot and then determining the maximum of these means (Figure 2). Callus characteristics were confirmed from the recorded pictures of patients’ feet in the medical records. The number of calluses present was counted. The site of a given callus was described based on the location: toe, metatarsal head (MTH), the midfoot, or the heel.
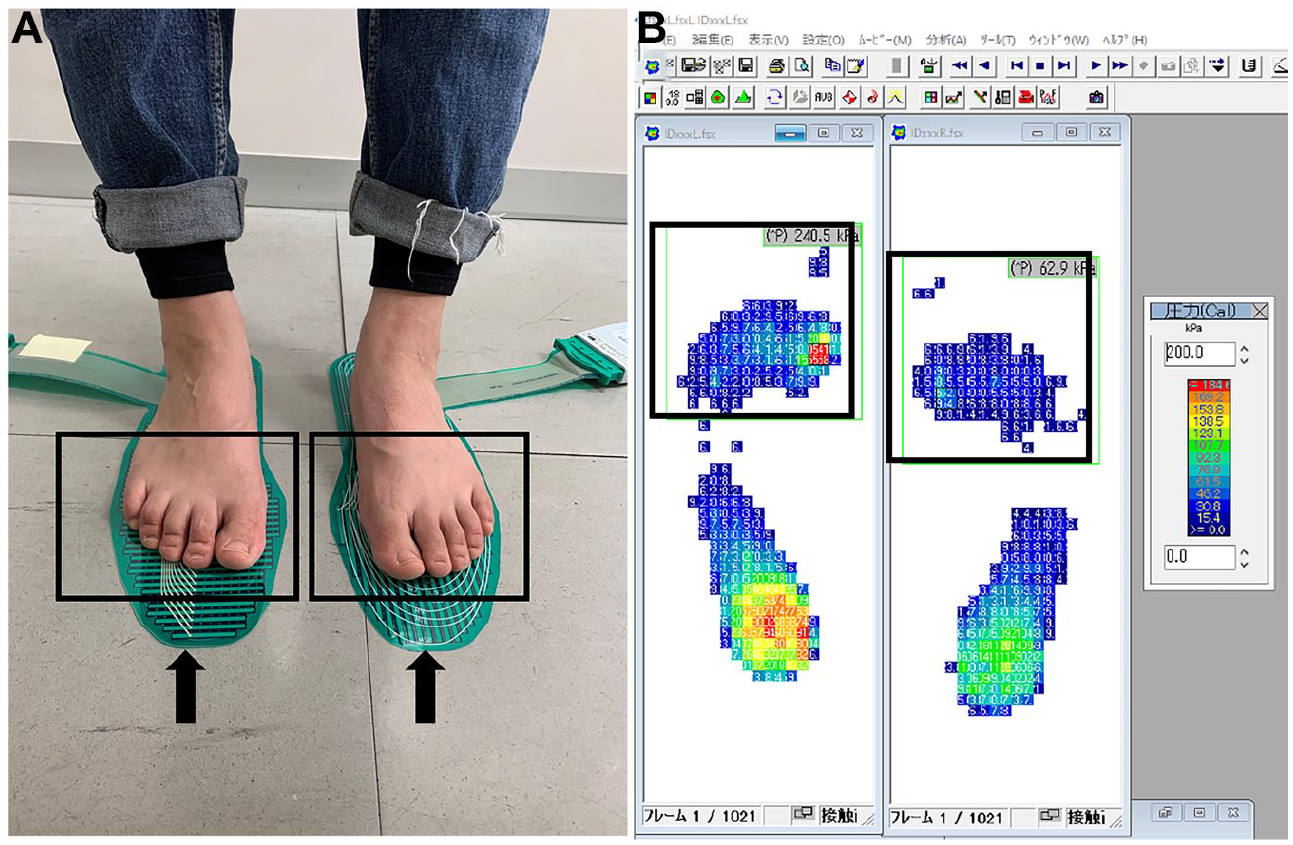
Measurement of static plantar pressure and forefoot plantar pressure reading Tekscan (black arrows) was used for the measurement of static plantar pressure. Patients were asked to stand in a comfortable position on the pressure sheets for at least 10 seconds (A). Body Pressure Measurement System was used to read forefoot (area marked by black rectangles) plantar pressure, and the peak plantar pressure was calculated as the local maximum of the mean of each 2-pixel by 2-pixel subregion inside of the black rectangle (B).
Statistical Analysis
Participants’ characteristics were presented as mean and standard deviations (SD) for continuous variables and as frequencies and percentages for categorical variables. To determine the factors associated with callus and the factors associated with the callus hotspots, tests were conducted between the following groups: feet with calluses and without; feet with callus hotspots and without. For analyses conducted between feet, the following characteristics were judged by feet: sensory neuropathy, motor neuropathy, PAD, SFPPP, dry skin, number of calluses, and ill-fitting shoes. Missing data were not input.
The chi-square test and Student’s t-test were used for each comparison between groups in categorical or continuous variables for univariable analysis, respectively. Then the generalized linear mixed model (binomial logit link function) was used to calculate the adjusted odds ratio (aOR) with 95% confidence interval for risks of callus and callus hotspots associated with patients’ characteristics. Variables with statistical significance (P < .05) in univariable analysis were used as fixed effects, and patient IDs was used as a random effect. To test the multicollinearity, the correlations between fixed effect variables were evaluated using Pearson’s r, Cramer’s v, or point-biserial correlation based on the variable type. The reason the patient ID was used as a random effect was that the same participants could be tested multiple times due to some of the data being foot-based. All analyses were conducted using IBM SPSS Statistics for Windows, Ver. 26.0 (IBM Corp., Armonk, NY, USA).
Ethical Consideration
This study was approved by the Ethical Committee of Graduate School of Medicine, the University of Tokyo (No. 3078-(6)). All subjects were provided with the opportunity to opt-out.
Results
One thousand and fifty patients were enrolled, and 1,007 patients were included in this study. Forty-3 patients were excluded because 36 patients had ongoing DFUs, and 7 patients could not stand without assistance (Figure 3).
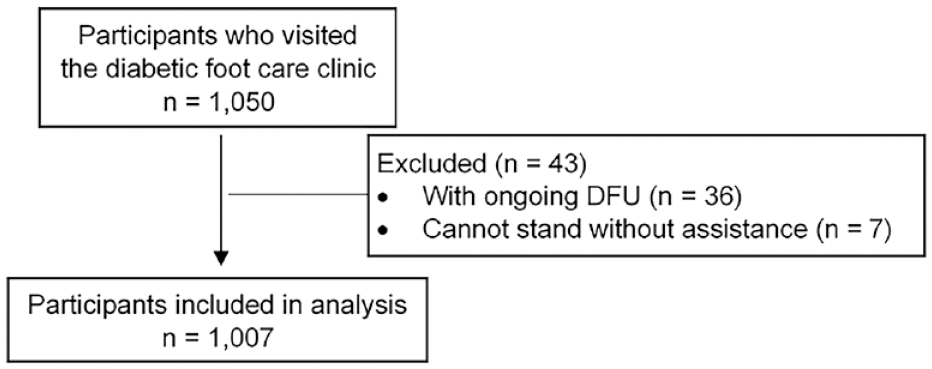
Flowchart of participant enrollment.
The participants’ characteristics are presented in Table 1. Participants were aged 67.1 ± 10.9 (mean ± SD) years old. Among them, 90.5% had type 2 diabetes, and the mean duration of diabetes was 15.1 years. HbA1c was 7.2 ± 1.1% and 3.9% of the patients had a history of DFUs.
Characteristics of Participants.
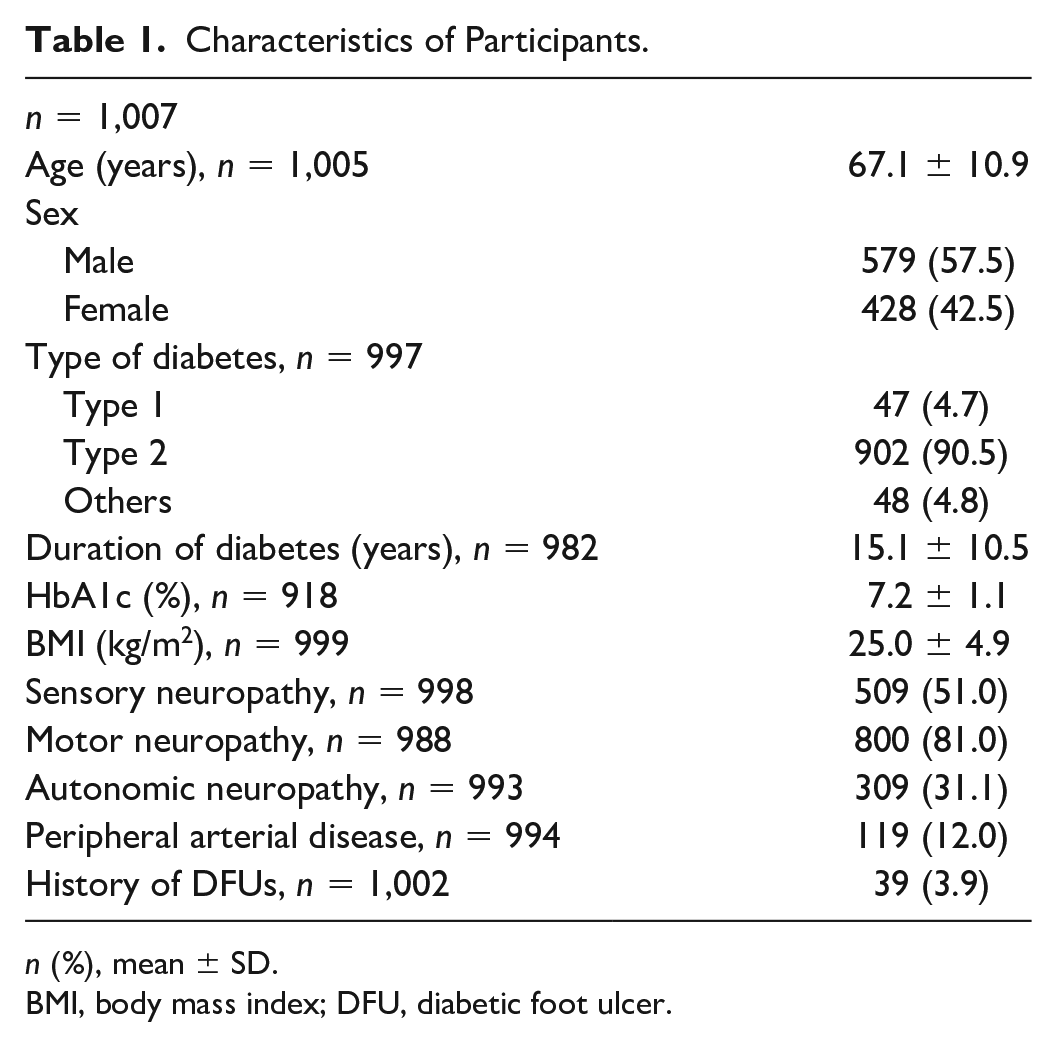
n (%), mean ± SD.
BMI, body mass index; DFU, diabetic foot ulcer.
Among the 1,007 participants, 901 calluses were counted. The top 3 most frequent sites for calluses were on the second MTH (n = 213, 23.6%), first toe (n = 195, 21.6%), and fifth MTH (n = 179, 19.9%). Hotspots were confirmed in 169 (18.8%) of the calluses. The most frequent sites for callus hotspots were the second MTH (n = 50, 29.6%), fifth MTH (n = 38, 22.5%), and first toe (n = 19, 11.2%) (Table 2).
Number of Calluses and Callus Site Distribution.
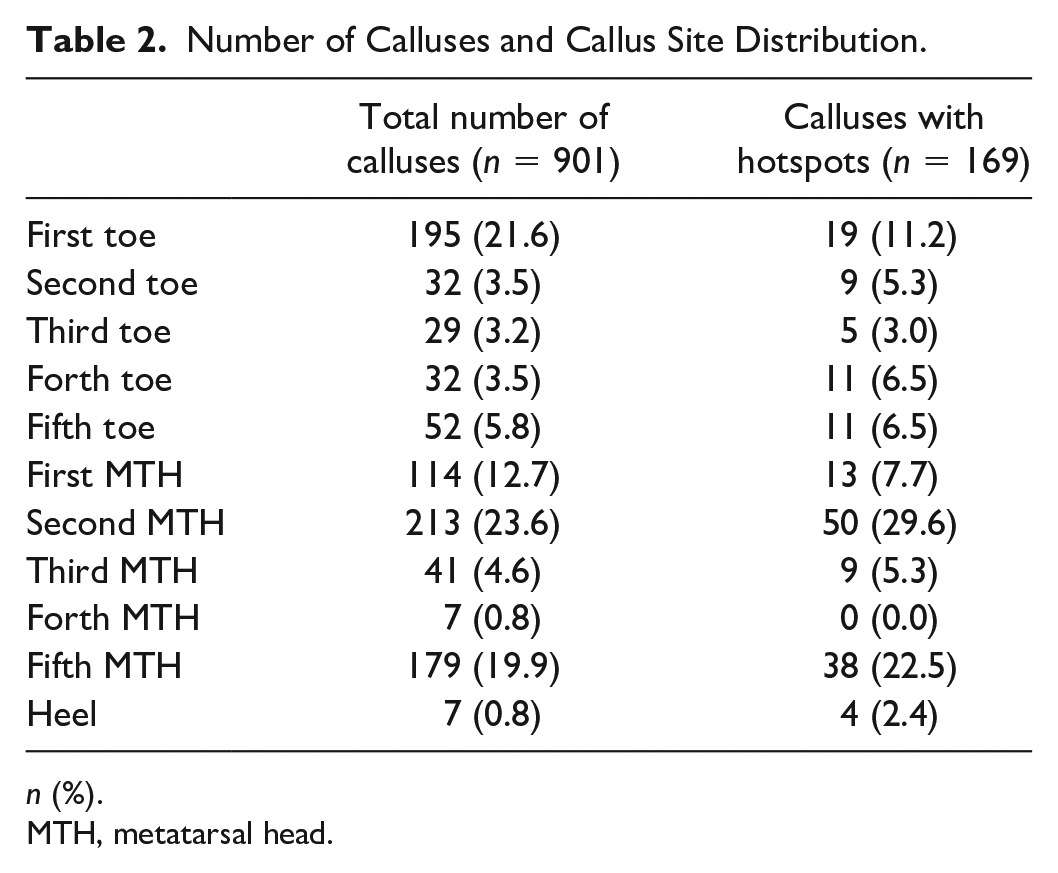
n (%).
MTH, metatarsal head.
Table 3 shows the results of the comparison between patients’ characteristics and callus-related characteristics of patients’ feet with and without callus. Data from 2,014 feet were analyzed. Among them, 574 (28.5%) feet had calluses. Feet with calluses occur more often in participants who were female, had type 1 diabetes, lower BMI, absence of autonomic neuropathy, absence of PAD, higher SFPPP, foot deformity, or dry skin. Multicollinearity was not present among the statistically significant independent variables.
Comparison of Patient and Feet Characteristics with/Without Calluses † .
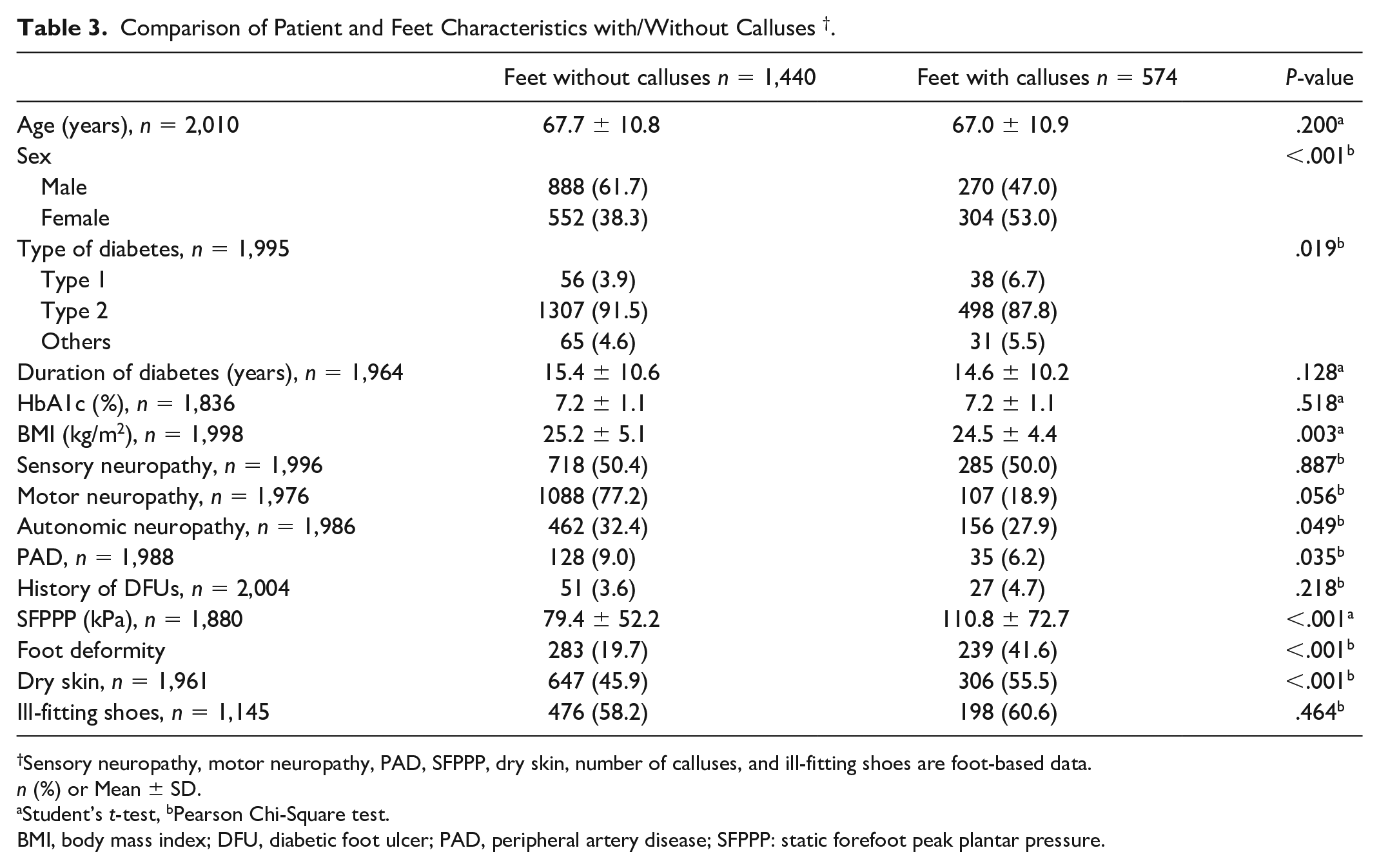
Sensory neuropathy, motor neuropathy, PAD, SFPPP, dry skin, number of calluses, and ill-fitting shoes are foot-based data.
n (%) or Mean ± SD.
Student’s t-test, bPearson Chi-Square test.
BMI, body mass index; DFU, diabetic foot ulcer; PAD, peripheral artery disease; SFPPP: static forefoot peak plantar pressure.
Table 4 shows the factors associated with the presence of calluses. In the generalized linear mixed model, the factors associated with the presence of calluses were female sex (aOR: 1.749, P < .001), higher SFPPP (aOR: 1.008, P < .001), foot deformity (aOR: 3.003, P < .001) and dry skin (aOR: 1.513, P = .007).
Factors Associated with the Presence of Calluses (n = 1,832 i ).
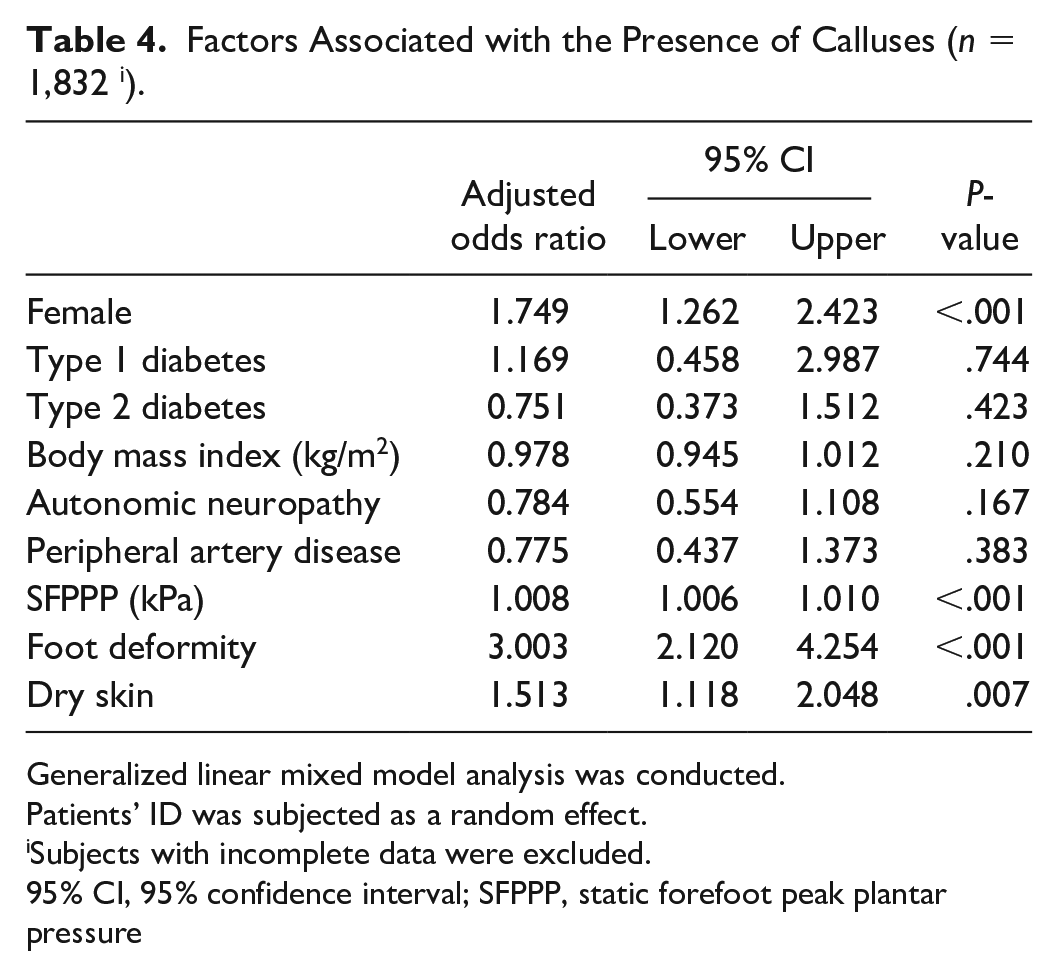
Generalized linear mixed model analysis was conducted.
Patients’ ID was subjected as a random effect.
Subjects with incomplete data were excluded.
95% CI, 95% confidence interval; SFPPP, static forefoot peak plantar pressure
The characteristics comparison between feet with calluses with hotspots and feet with calluses without hotspots is shown in Table 5. A total of 106 (18.5%) feet presented with callus hotspots. Feet that presented with callus hotspots occur significantly more often in participants who were younger, have lower HbA1c level, have lower BMI, have higher SFPPP, have more calluses, or whose peak pressure matches the callus site. Multicollinearity was not present among the statistically significant independent variables.
Comparison of Patient and Feet Characteristics with/Without Callus Hotspots † .
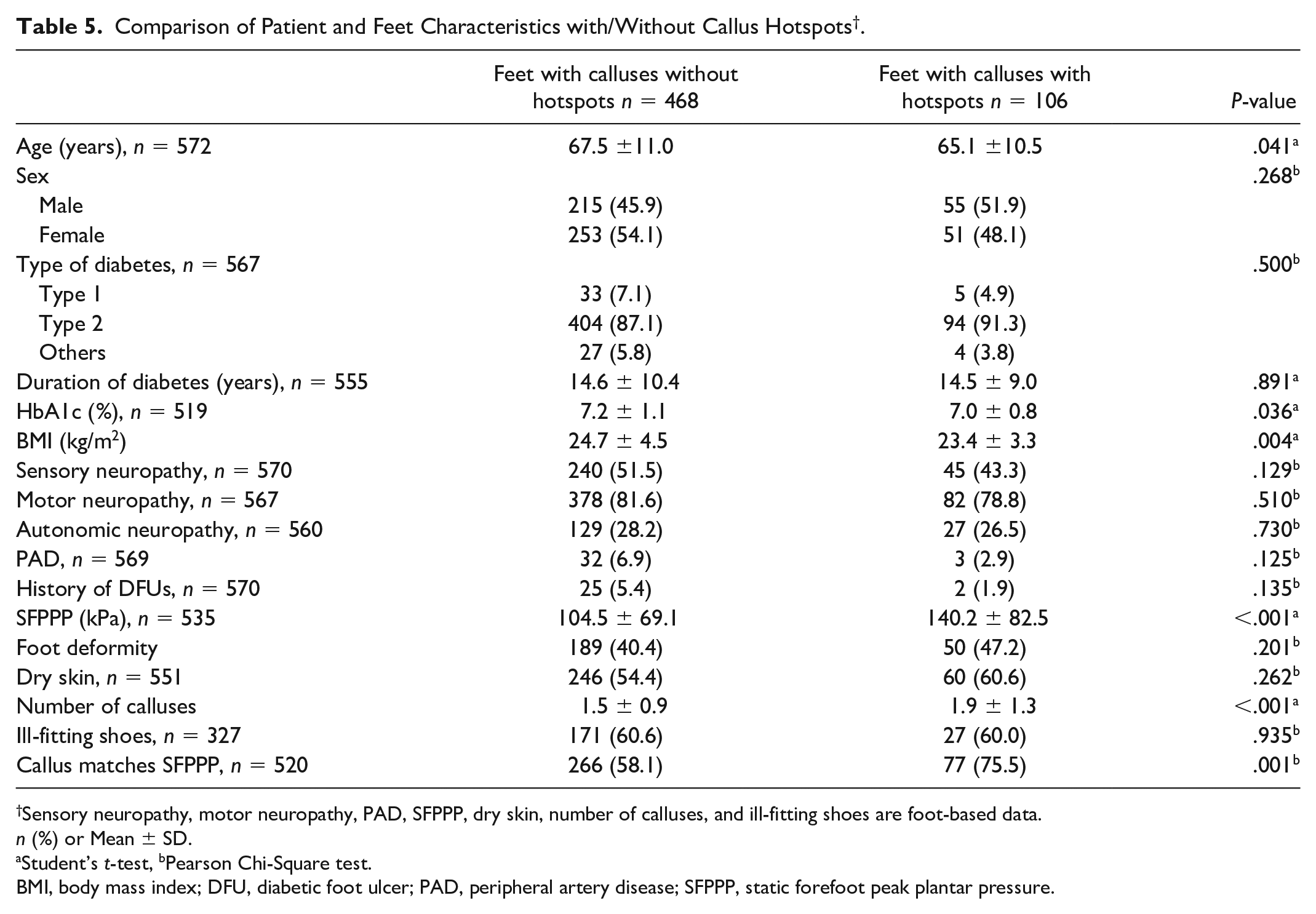
Sensory neuropathy, motor neuropathy, PAD, SFPPP, dry skin, number of calluses, and ill-fitting shoes are foot-based data.
n (%) or Mean ± SD.
Student’s t-test, bPearson Chi-Square test.
BMI, body mass index; DFU, diabetic foot ulcer; PAD, peripheral artery disease; SFPPP, static forefoot peak plantar pressure.
To determine the factors associated with the presence of calluses with hotspots, a generalized linear mixed model was constructed (Table 6). The factors associated with hotspots on calluses were BMI (aOR: 0.912, P = .029), SFPPP (aOR: 1.008, P = .001), and the number of calluses (aOR: 1.540, P = .003).
Factors Associated with Hotspot on the Callus (n=482 i ).
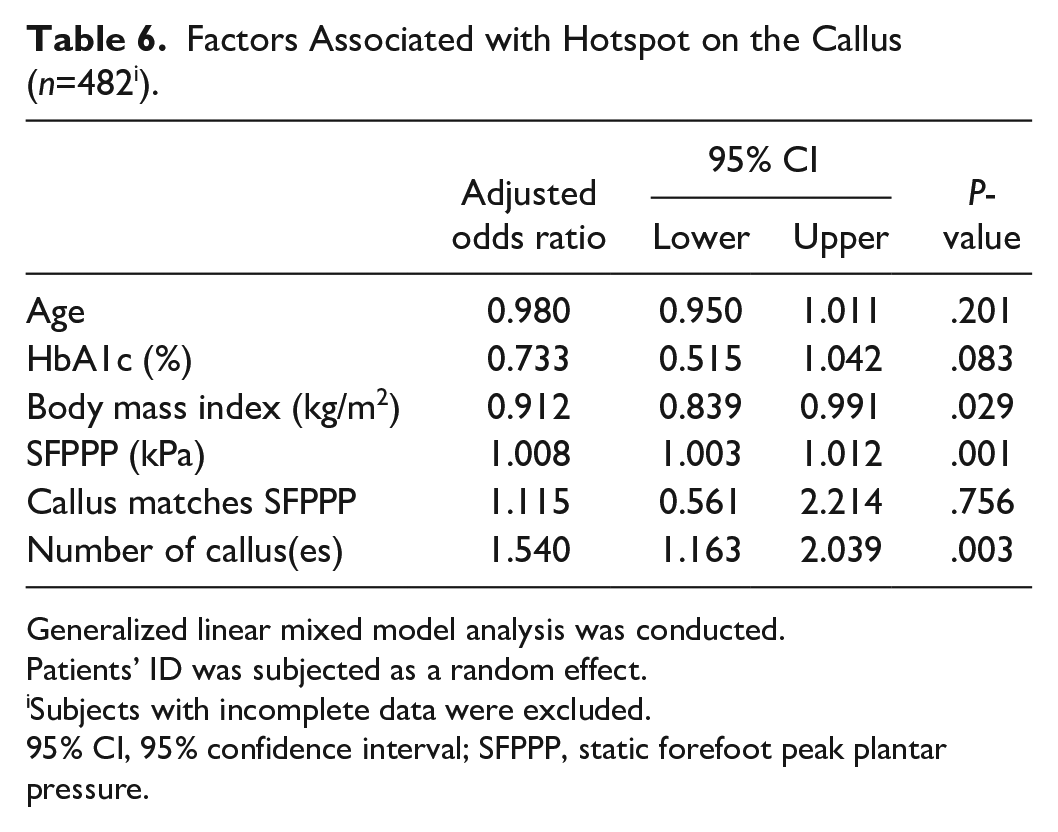
Generalized linear mixed model analysis was conducted.
Patients’ ID was subjected as a random effect.
Subjects with incomplete data were excluded.
95% CI, 95% confidence interval; SFPPP, static forefoot peak plantar pressure.
Discussion
This is the first report of the risk factors of callus hotspots. Among people with calluses, those with a higher SFPPP, lower BMI, and presence of more calluses had higher probability to present callus hotspots. SFPPP was associated with both calluses and hotspots. Therefore, providing interventions for reducing plantar pressure could prevent both callus formation and callus hotspots.
This study indicates that people with higher static plantar pressure are more likely to present a callus hotspot. Excessive plantar pressure results from 2 main factors: abnormal gait and pathological foot structure in people with diabetes. Previous studies have used dynamic peak plantar pressure to evaluate the gait dysfunction effect; however, this measurement is time-consuming and can be easily influenced by factors such as gait speed.29,30 The current study focused on the SFPPP instead, highlighting its usefulness in risk assessment and its potential for routine use in the clinical setting.
The number of calluses present was also shown to be associated with the presence of hotspots on calluses. This could be related to the severity of foot deformity. The limited joint mobility occurring at the first MTH and subtalar joint has been shown to contribute to increased plantar pressure in several studies.20,31 Unfortunately, data regarding the degree of deformities or subtalar joint mobility were not recorded. Therefore, the relationship between them is not explainable with the current data.
Additionally, lower BMI was significantly associated with the presence of callus hotspots. The lower BMI might represent a bony foot structure with less fat for reducing the pressure from excessive loading, which in turn leads to tissue breakdown under the callus.32,33 A previous study showed that tissue thickness under the MTH is related both to lower BMI and forefoot plantar pressure. 23 Furthermore, another study describes a negative relationship between BMI and peak forefoot plantar pressure. 34 However, some studies show a different result: higher BMI could increase the plantar pressure.35,36 However, the correlation between BMI and SFPPP was not found in the current study (r = .016, P = .479). Hence, there might be other pathologies that contribute to this phenomenon which could not be explained with the current data.
Surprisingly, presence of sensory neuropathy was not significantly different between people with callus hotspots and without despite this being a well-known risk factor of both callus formation and DFUs. This result contrasted with previous studies that demonstrated that lack of sensation is one of the major risk factors for ulceration among high-risk people.3,32,37 The different result in our study could be due to limitations of the measurement of vibration reception. In this study, a reception time of 10 seconds was used which can only judge the presence of vibration reception, not the severity. Therefore, information regarding sensory neuropathy lacks sufficient resolution to determine the association with callus progression.
The factors associated with callus formation are mostly consistent with previous studies, namely, being female,38,39 presence of a foot deformity,14,16 static peak plantar pressure,13,16,38 and dry skin. 17 These factors directly or indirectly contribute to excessive mechanical loading, which in turn leads to callus formation. The relationship between female patients and presence of calluses could result from the preference of higher heel and tighter head shoes when the patients were young and higher frequency of hallux valgus, which could increase the forefoot peak pressure. 39 Although ill-fitting shoes are among the well-known causes of callus formation, the relationship between ill-fitting shoes and the presence of calluses could not be detected in the current study. 14 The reason might be incomplete data regarding shoe size or the chosen measurement method not being accurate enough.
There are several noticeable limitations to the current study. The first is that this is a cross-sectional study of collected data from the past 12 years of which a lot was missing. For example, there was no standardized data regarding the daily activity level and self-care, and some of the data could not be measured due to equipment issues during certain periods. Additionally, the data we used were not multiple consecutive measurements, as used in previous studies to assess inflammation.40,41 Thus, we were unable to link any of the studied variables to underlying inflammation. Due to the study design, the causal relationship between factors and outcomes is unable to be determined or explained. Despite this limitation, it is clinically plausible to say patients with high SFPPP are at high risk of hotspot development, as callus hotspots cannot elevate SFPPP. Secondly, the data used in the current study were from a single diabetic foot care clinic at a university hospital that is specialized in diabetic foot prevention care. Patients who visit the diabetic foot prevention care clinic are those who visit the Department of Diabetes and Metabolic diseases on a regular basis and have relatively good control of HbA1c and body weight. Patients with DFUs or immobility were not included in the present study. Therefore, the generalizability of these results needs to be further confirmed in populations with more severe conditions such as critical limb ischemia. The barefoot forefoot static plantar pressure was used for analysis because it’s more convenient and easier to apply in a clinical setting. The effects of shear stress and plantar pressure during the gait were not determined in this study, despite having been previously shown as 2 of the causes of calluses. However, static pressure could still provide information for care providers to better identify the population that needs an elevated level of care even though the cutoff value of peak plantar pressure was not able to be determined in this study. Thus, for prediction of callus hotspots, a prospective study is needed in the future.
This study highlighted the importance of specific preemptive interventions on reducing the abnormal loading caused by an external force. For people with callus hotspots, stricter adherence to the use of off-loading tools is needed in terms of preventing DFUs. On the other hand, among people with calluses without hotspots, those who have high plantar pressure, low BMI, and many calluses might be at high risk of callus hotspots. Therefore, elevated attention should be paid to this population, and specific preventive interventions such as routine callus removal and the use of off-loading methods should be applied to prevent the precursors of DFUs: callus hotspots.
Conclusion
The current study suggests that among people with calluses, the presence of high plantar pressure could be related to both hotspots detected by thermography and the presence of calluses. Thus, providing off-loading interventions could be effective for prevention of both calluses and callus hotspots. Taken together, static plantar pressure could be a useful predictor of callus hotspots for the purpose of early intervention to prevent DFUs.
Footnotes
Acknowledgements
None.
Abbreviations
ABI, ankle branchial index; aOR, adjusted odds ratio; BMI, body mass index; CI, confidence interval; DFU, diabetic foot ulcer; MTH, metatarsal head; PAD, peripheral artery disease; SFPPP, static forefoot peak plantar pressure; TBI, toe branchial index.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
