Abstract
Background:
Preulcerous risk situations in patients with diabetes are often undiagnosed and care administered too late. Even with regular medical check-ups and status documentation, foot examinations have not been given enough attention. Diagnosing an individual patients’ risk of developing diabetic foot ulcers may increase vigilance for diabetic foot syndrome (DFS), and the appropriate prevention measures matching the risk involved may prevent the emergence of diabetic ulcers. The classical DFS risk factors are well established and have been extensively covered in the literature; however, there is a lack of efficient screening tools that could be used for a rapid assessment of diabetic foot ulcer risk.
Methods:
A methodical literature search was conducted to assess relevant publications for the preparation of a simple risk score for amputation related to diabetic foot ulcer. We then analyzed the risk factors for predictive value as odds ratios in foot ulcers and/or amputation. We used the available data to deduce a mean value to reflect the authors’ consensus.
Results:
In view of the current literature on the matter, we have developed a semi-quantitative scoring system using just a few items to allow rapid and visual risk assessment for diabetic foot ulcers alongside recommendations for prevention and a sensible follow-up strategy to match the risk.
Conclusion:
This relatively simple score enables rapid risk classification for patients that can ease the way for both physicians and patients in gaining an insight into individual risk situations. The score provides more effective preventative measures for high-risk patients against future complications.
Background
Patients with diabetes but without severe diabetic sequelae are mainly treated by general practitioners and family doctors in general medicine, confronting the physicians with high patient numbers and an unclear risk situation. Family doctors often lack the time and staff needed for adequate patient screening as to each patient’s individual risk of developing diabetic sequelae and providing the requisite care to preselected high-risk patients for conditions such as diabetic foot syndrome (DFS) with specific prevention or treatment programs.
The risk factors for DFS are well known, but the heterogeneity in definitions and methodologies hampers comparison. There is as of yet a lack of prospective long-term data from large patient groups for determining individual morbidity and mortality risks in patients at increased risk of ulceration and amputation. The Cochrane Library published a 33-page method paper describing all the risk factors and score systems researched worldwide for ulcer risk in DFS together with statistical methods for evaluating the heterogeneous raw data in 2014, 1 a results paper has not yet been published. A similar work was performed in the United Kingdom, resulting in a 242-page Health Technology Assessment Report on ulcer risk in DFS, which was published in 2015, 2 including a systematic overview of the literature and a meta-analysis of all known risk factors for diabetic ulcers.
The aim of the present contribution was to develop a score based on the scientific literature to provide physicians with an easy means of evaluating risk among their patients—a score for assessing individual ulcer risk that should help in early detection and preselection with clear indications for medical and nonmedical preventative measures in high-risk patients.
Epidemiology
The lifetime incidence of foot ulcers in persons with diagnosed diabetes mellitus might be as high as 25%.3,4 In view of global amputation rates, diabetes is associated with 25%-90% of all amputations.3,5 Twenty-nine thousand amputations were performed in people with diabetes in Germany in 2001 according to AOK, a German statutory health insurance organization. 6 Amputation rates showed an increasing tendency in the follow-up years of 2002 and 2003. 6 Foot ulceration is the root cause7,8 of 85% of all lower limb amputations and is associated with a marked increase in mortality at 5% within 12 months and 42% within 5 years among patients developing de novo ulceration. 9
The number of patients with diabetes, and therefore also the potential number of high-risk patients for ulcers and amputations, continues to rise sharply in the coming years according to estimates by the World Health Organization and International Diabetes Federation. 10 DFS still represents a massive economic health problem, and studies have demonstrated that amputation affects quality of life in patients with diabetes more severely than does blindness or severe renal insufficiency. 11 Amputations are closely associated with depression and anxiety disorders due to loss of independence and mobility.12,13
Methods
A methodical literature search using the keywords diabet, ulcer, and risk yielded 370 hits in PubMed. 14 We assessed and evaluated the abstracts in these studies for relevance in preparing a simple score for the risk for amputation related to diabetic foot ulcers and/or diabetes, and examined the bibliographies in the articles and the current national15,16 and international guidelines17-19 on DFS for other relevant literature sources. We then analyzed the risk factors for predictive value as odds ratios in foot ulcers and/or amputation, and their potential weighting in a score. Only studies with foot ulcer formation as a primary endpoint rather than amputation in patients without active ulceration served in further analysis. We used the available data to deduce a mean value to reflect the authors’ consensus taking clinical experience into account.
Results
Available Risk Scores
Recent years have seen efforts in developing suitable screening tools for early detection of patients at increased risk of ulceration and amputation. Four classification systems have come into common currency around the world; these systems differ substantially in the number of risk factors considered and risk-group grading, but are similar with regard to the core variables used as a basis:
Diabetic polyneuropathy (PNP)
Peripheral arterial disease (PAD)
Foot deformity
History of ulceration or amputation on the lower limb.
There are major differences in criteria for the definition of PNP and PAD, the total number of variables included, and the formulae applied in the respective scoring systems. The quality of the underlying evidence and validation of predictive significance in all of the score systems appear to be low overall; only a few studies include data on sensitivity, specificity, and the positive and negative predictive value in ulceration. Only a few cases include external data validation. 20
Risk classification according to the International Working Group on Diabetic Foot (IWGDF) has been kept simple and includes three risk stages with only one associated item: (1) PNP, (2) PAD or PNP with or without foot deformity, and (3) previous ulceration or amputation (Table 1). 21 This classification is based on the work of Lavery, 22 who proposed different combinations of major risk factors with PAD and earlier ulceration or amputation without stratification in 2008, subsequently proposing adaptation of previous risk classifications according to the Texas risk groups. 23
IWGDF Risk Classification. 22
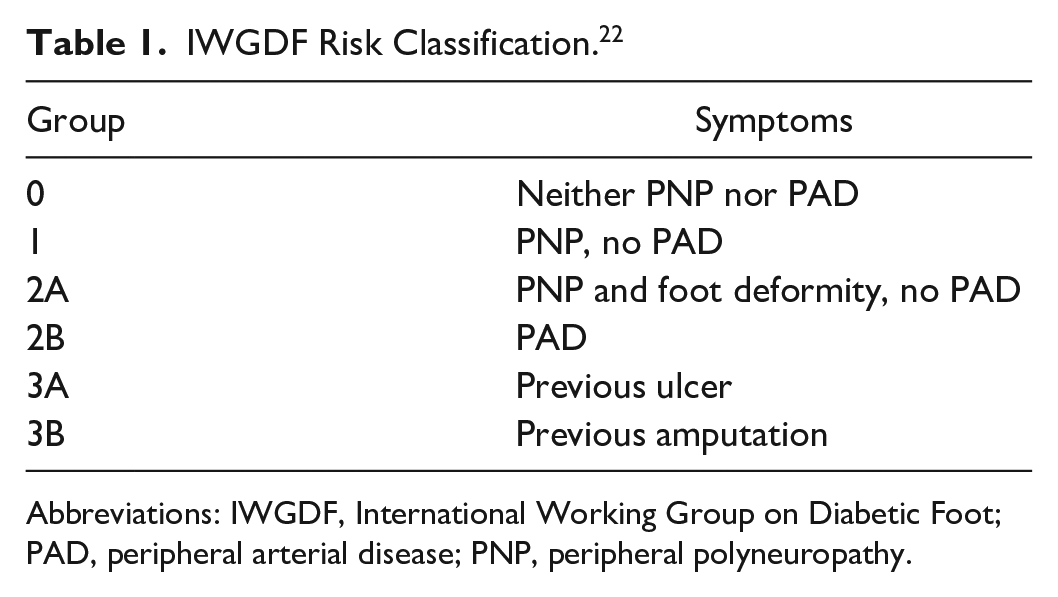
Abbreviations: IWGDF, International Working Group on Diabetic Foot; PAD, peripheral arterial disease; PNP, peripheral polyneuropathy.
Other systems developed by the American Diabetes Association, 24 Scottish Intercollegiate Guideline Network (Figure 1),25,26 and the E.J. Boyko working group27,28 include all four of the above core variables. The German Nationale VersorgungsLeitlinie medical guideline on prevention and treatment strategies of foot complications in type 2 diabetes 16 lists 4 main and 14 additional risk factors in alphabetical order (Table 2), yielding a positive data situation. This system notably groups various atherosclerotic sequelae together with examples such as renal insufficiency and stroke under the arterial disease risk factor, which appears to be very broad and open to interpretation. The Infectious Disease Society of America published a clinical practice guideline for the diagnosis and treatment of diabetic foot infections (DFI), classifying DFI into three categories according to depth of infection and presence of systemic inflammatory response syndrome. 29 A recent evaluation of this risk score 30 resulted in the suggestion to modify the assessment for DFI by including osteomyelitis and merging the moderate and severe infection categories. In a following commentary, the recommendation to include osteomyelitis into the classification of DFI was welcomed while merging of the two infection categories was not supported. 31
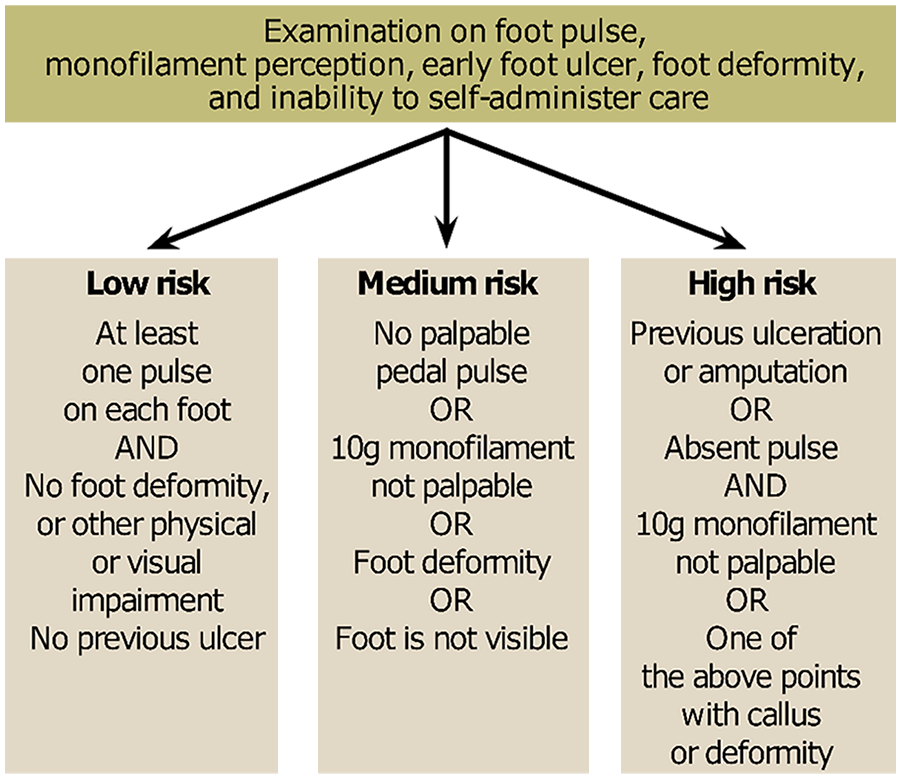
Algorithm according to the Scottish Diabetes Foot Action Group. 25
Main and Secondary Criteria for the DFS According to the Nationale VersorgungsLeitlinie medical guideline for Foot Complication Prevention and Treatment Strategies in Type 2 Diabetes. 16
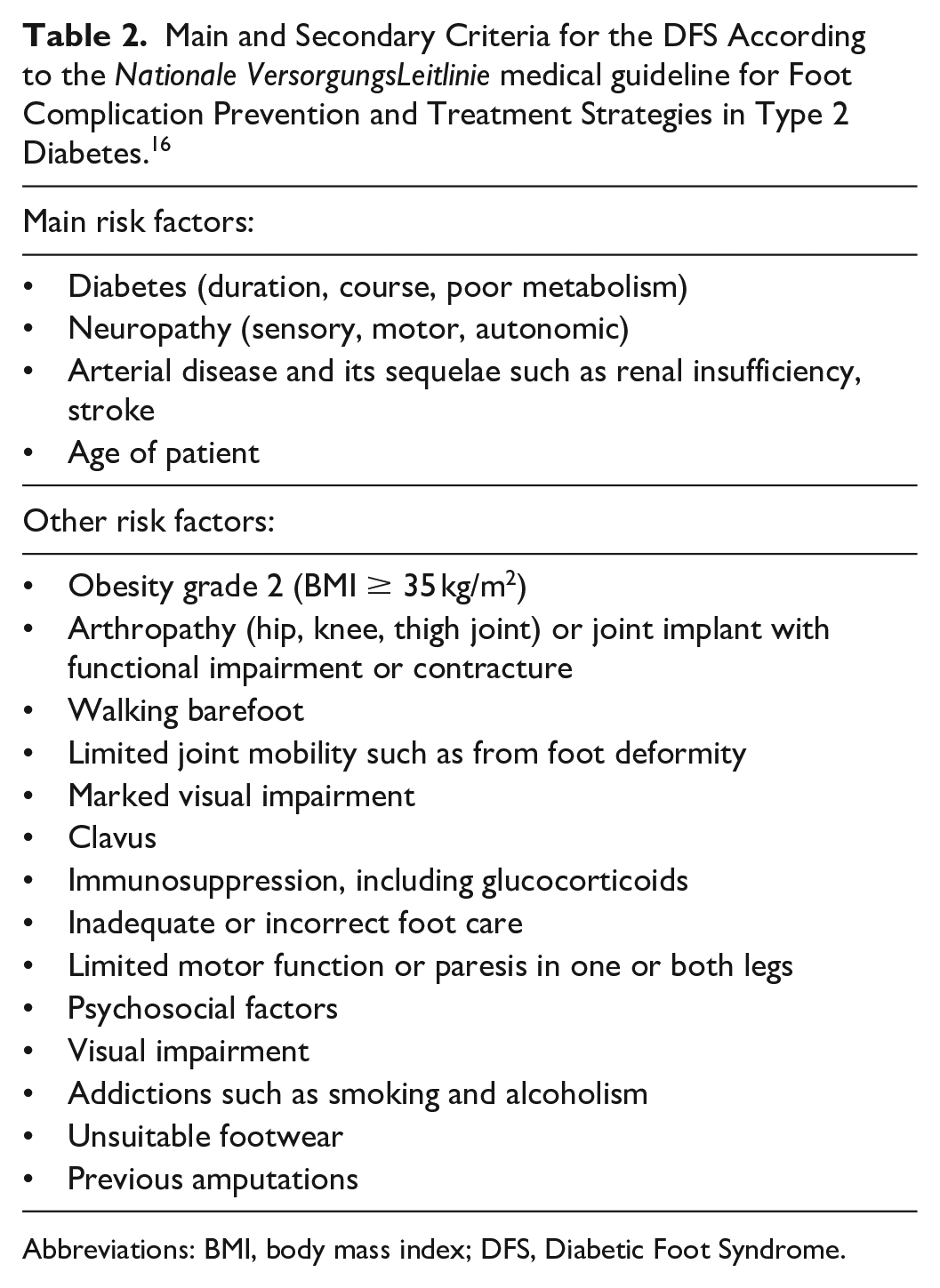
Abbreviations: BMI, body mass index; DFS, Diabetic Foot Syndrome.
A Canadian guideline for medical assistants includes a questionnaire with four risk assessment items: (1) previous foot ulceration, (2) sensory loss as measured by Semmes-Weinstein monofilament (SWMF), (3) structural or biomechanical foot abnormality, and (4) absent pedal pulses or patient history of claudication problems. At least one positive result for these criteria indicates an increased risk. After that, the respective patient’s self-sufficiency competence is evaluated from six additional questions: (5) ability to self-administer foot care or summon assistance, (6) appropriate footwear, (7) podiatric training, (8) daily foot examination by the patient, (9) own initiative in seeking professional assistance with foot problems, and (10) own initiative in risk minimization. A deficit in self-sufficiency is assumed if only one of these additional items is negative, and appropriate training programs are offered.
Additional Screening Tools
Apart from the validated scoring systems devised by international specialist diabetes organizations, screening instruments such as the 60-second foot score developed in 2004 by Inlow et al exist for physicians in low-income American countries. 32 The Inlow system includes clear instructions on further treatment for patients and specifies time intervals for follow-up examinations depending on risk situation. Orsted et al developed and published a bedside version, the simplified 60-second test, based on the Inlow test in 2009 (Figure 2). 33 The test did not take root in Germany or found its way into any European guidelines even with its extensive validation and simple implementation.
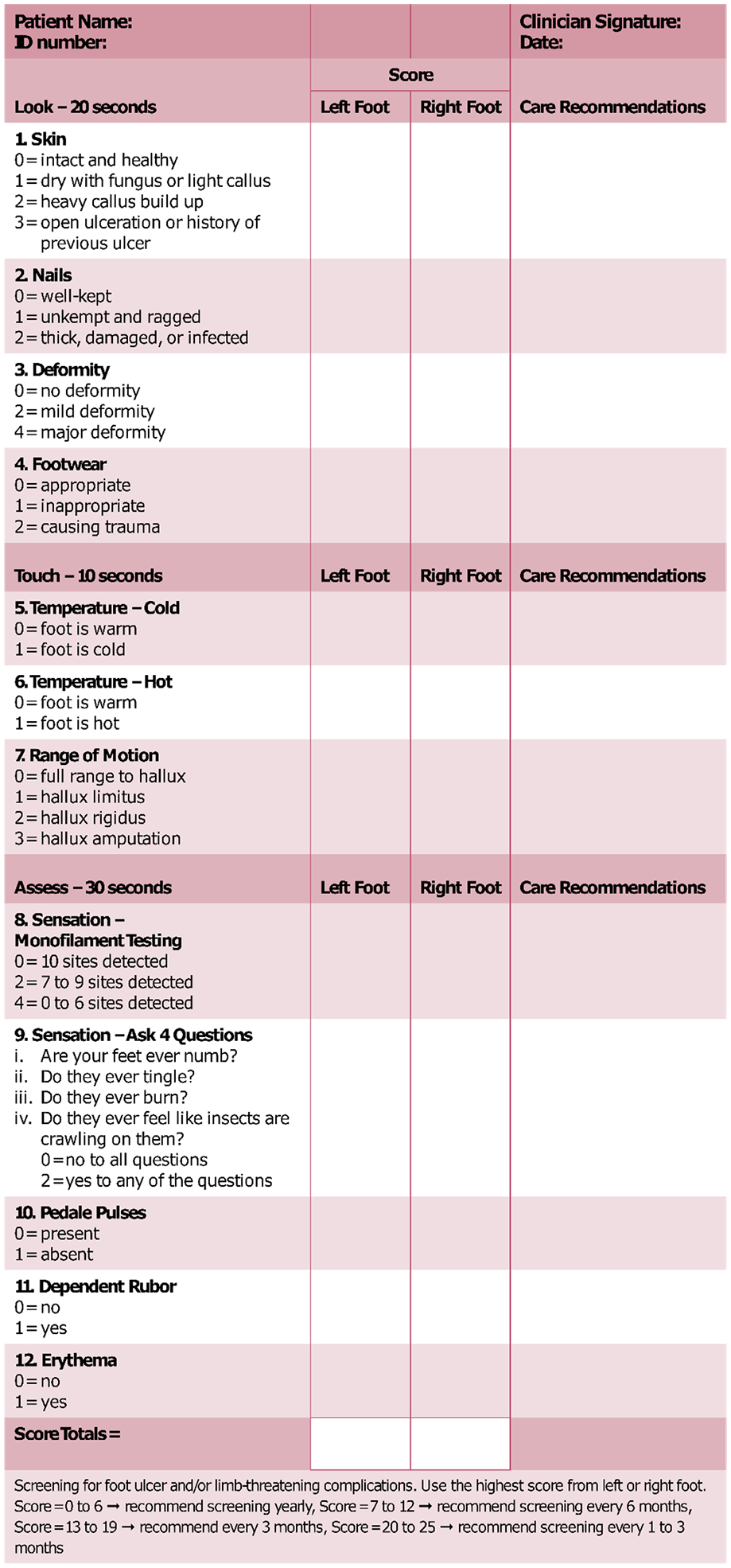
Simplified 60-second test for high-risk diabetic foot. 33
An indicator patch (Neuropad) mainly developed for diagnosing diabetic peripheral neuropathy 34 uses sudomotorism (sweat stimulation) on the foot, as this correlates with innervation by post-ganglionic sympathetic small fibers in a highly reproducible color change.35-37 This plaster test also predicts DFS38,39 and is suitable for patient self-application due to its ease of use (Figure 3). 40 Daily dermal temperature measurement on the foot represents another effective screening method of preventing ulceration for high-risk patients to apply themselves.41-43
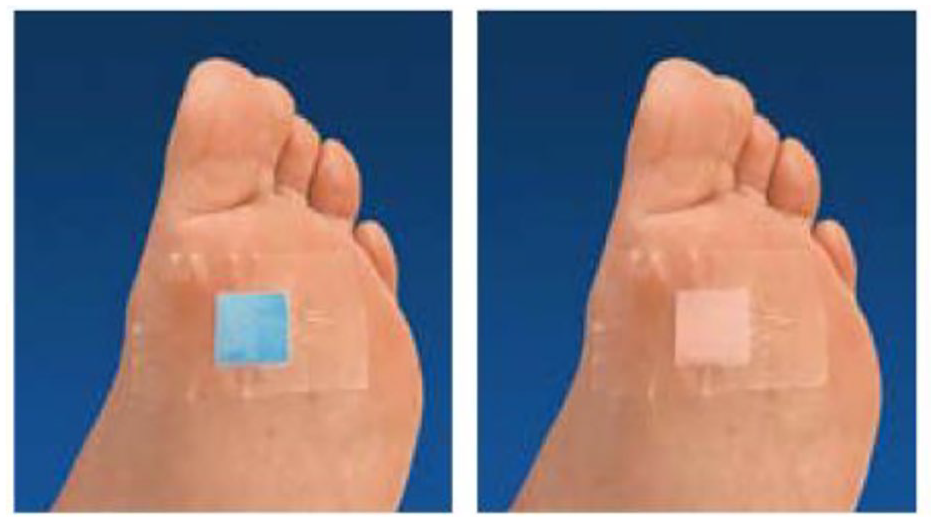
Neuropad indicator patch test, pathological result (left), and non-pathological result (right).
An Australian research team developed a software solution for risk assessment and clinical decision making in 2015 with the assistance of a commercial vendor, and tested the system in a comprehensive multistage validation process. 44 The system is mainly aimed at podiatrists and medical assistants, but we do not see the system as worthy of unreserved recommendation due to unclear definitions and subjective selection in some of the items. Other software solutions such as the Diabetes Dashboard clinical decision support were shown to improve diabetes management 45 and might thus on the long run improve outcomes with regard to diabetes comorbidities.
Prediction Score for Diabetic Foot Ulcers
The scoring system we have developed and present in this contribution includes three categories for assessing individual ulcer risk: (1) major criteria, (2) moderate criteria, and (3) minor criteria (Figure 4).
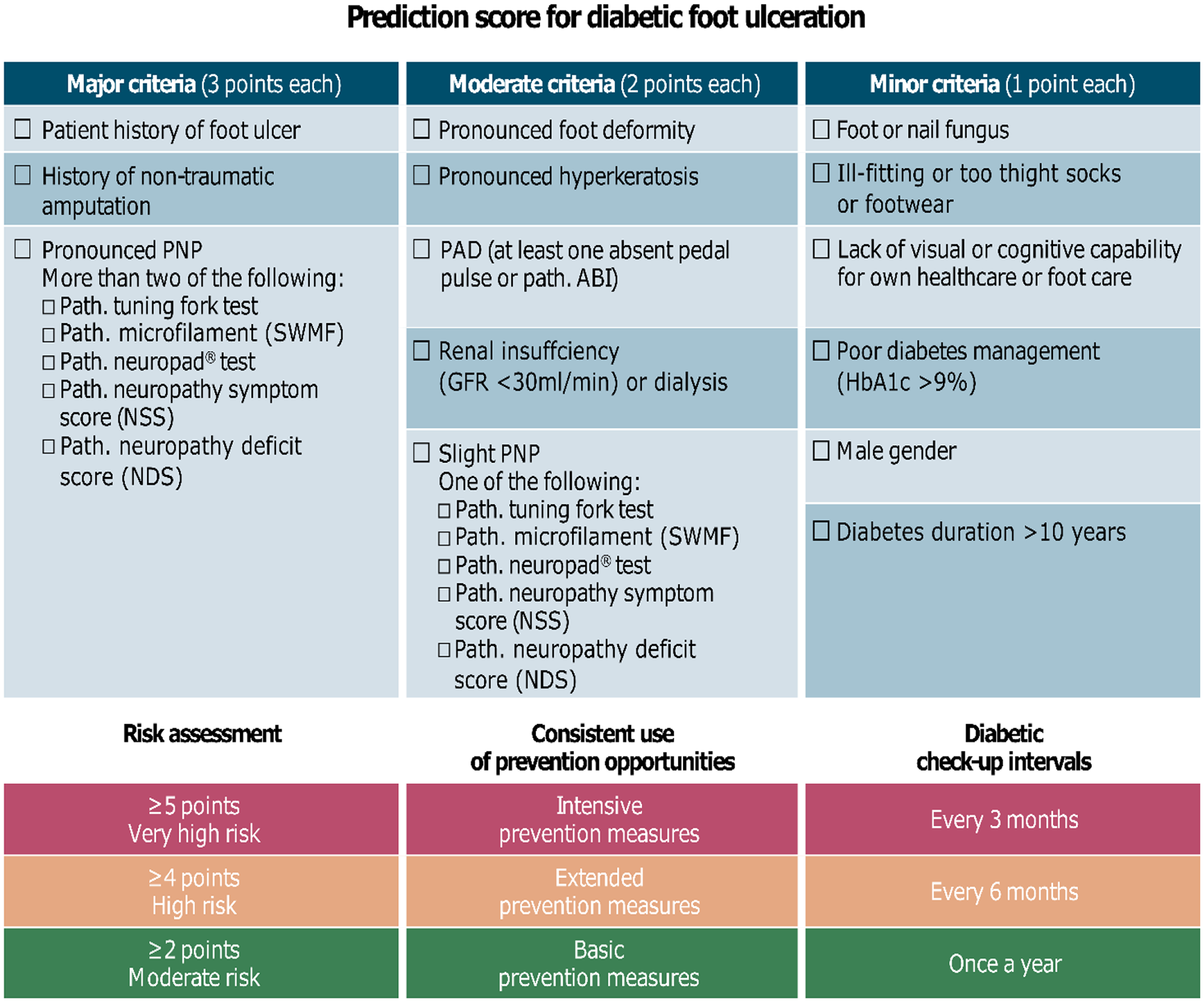
Prediction score for diabetic foot ulceration.
Classified major criteria
History of amputation or ulcer: The risk of de novo ulceration is very high after amputation or a patient history of ulceration due to DFS, PAD, or other nontraumatic causes;2,27,46-51 see Table 3 for predictive values of ulcer formation as determined in the studies.
Predictive Values of Major Risk Factors for Diabetic Ulceration.

Abbreviations: ABI, ankle-brachial index; GFR, glomerular filtration rate; HbA1c, glycated hemoglobin; OR, odds ratio; RR, relative risk.
Severe polyneuropathy: The combination of two pathological test methods for a diabetic sensorimotor or autonomic neuropathy—SWMF, 60 tuning fork, Neuropad, neuropathy symptom score (NSS), neuropathy deficit score, and similar—served as the main criterion. At least one absent pedal pulse or sensitivity disorder using SWMF measurement showed the same predictive value as moderate risk according to the foot screening tool by the Scottish Care Information (SCI) Diabetes Collaboration in a meta-analysis by the National Health Service (see Figure 1). One absent pedal pulse does not show higher predictive value for ulceration than pathological results from SWMF measurement (Figure 2).
Classified moderate criteria
Severe foot deformity: Documented charcot foot, severe hallux deformations, and hammer or claw toes show high predictive values (Table 3) and are therefore classified as moderate criteria.23,48,54,60,61
Severe hyperkeratosis at more than two millimeter thickness is an ulcer precursor associated with underestimated risk of ulceration despite its prevalence. 54 Hyperkeratosis results from mechanical stress especially increased plantar pressure load such as shoes or stockings that are too tight, or foot deformity from impaired perception of pain that prevents the necessary correction against abnormal biomechanical stress. Conversely, hyperkeratosis increases inward pressure on underlying tissues, thus promoting ulceration. 54
Slight polyneuropathy (PNP): This includes the typical clinical symptoms of PNP as listed in the NSS as well as positive test methods ideally determined using SWMF, or alternatively tuning fork or Neuropad indicator patches. A pathological test result is interpreted as slight PNP.
Documented peripheral arterial disease (PAD): The ankle-brachial index used alone for diagnosing PAD shows moderate predictive value, as does at least one absent pedal pulse (Table 3). Both factors are therefore evaluated as moderate criteria. PAD secured by diagnostic means or therapeutic intervention shows a high relative risk between 2 and 4.
Renal insufficiency, especially at a glomerular filtration rate of <30 mL/min, is a clear predictor 59 according to some studies, but is less strongly associated with de novo ulceration. 62 Dialysis is a strong independent risk factor for ulcers. 53
Classified minor criteria
Foot or nail fungus infection: The data situation for onychomycosis is somewhat more compelling than it is for tinea pedis, and only in primary ulcers in either case. 27
Glycated hemoglobin (HbA1c) more than 9%: Increased HbA1c values are associated with increased risk of ulceration,27,52,55 although most studies test for increasing values with few studies testing for threshold values. Patients with HbA1c values above 9% are mostly in the high-risk group, so we used this as a threshold value in the score. The influence of diabetes duration is well documented, and longer duration clearly associates with higher risk of ulceration. However, very few studies state whether they used threshold values such as 10 years’ duration 52 or increasing values.55,58,59,63 We selected a threshold of 10 years due to the paucity of data available—patients with longer durations were mostly in high-risk groups in the studies.
Visual or cognitive impairment in personal health or foot care: This “soft” risk factor comprises a sum of various factors that do not provide enough predictive value as independent risk factors, or escaped accurate definition although expert consensus indicates sufficient cause for at least one risk point according to the definitions we have selected, and includes factors such as visual impairment,27,48 retinopathy,28,52 dementia, 27 and immobilization. 28
Ill-fitting shoes or socks: No studies have as of yet investigated the potential relationship between shoes or socks and ulcers, but there is extensive data on the risk of ulceration from increased plantar pressure48,49,58,64 and frequent walking barefoot. 65 However, therapeutic footwear does protect high-risk patients from ulcer formation.66-68 An early publication found inadequate footwear to cause foot ulcers in 50% of patients 69 where, inter alia, the foot circumference was greater than that of the shoe. Whether or not ulceration is ultimately due to lack of perception in mechanical compression owing to marked impairment of nerve sensation on the foot remains unclear due to the absence of adequate studies. Considering clinical experience, we have therefore classified visibly unsuitable footwear—including extensive walking barefoot—or restrictive and unsuitable socks as a minor criterion.
Male gender: The data on poorer outcomes in men compared with women are extensive and consistent—Crawford states an odds ratio of 0.70 (95% confidence interval 0.60–0.92) for female gender as a protective factor. The review by Monteiro-Soares 49 cited 10 out of 34 studies on gender dependency, with significantly worse outcomes in men and none with contradictory findings.23,70,71
Nonintegrated risk factors
We have not included the following factors in the score because of insufficient or contradictory evidence: age; education; social status; marriage status; ethnicity; depression; diabetes type; limited joint mobility; plantar pressure; edema; or lack of awareness, training, or participation in screening programmes.2,49 Insulin resistance has also been associated with ulceration in older studies, but we have not included this potential factor in the score as it no longer accurately reflects the current situation—new medicines in addition to insulin have seen increasing use in therapy strategies in recent years. It should be kept in mind that therapeutic decisions with regard to new treatment options strongly depend on patient histories, for example, for people with a history of neuropathy and diabetic foot ulcer; results from clinical trials with regard to amputations should be considered.72,73
Preventative measures and screening intervals by score
IWGDF Guidance 2015 refers to prevention as identifying risk feet and their regular examination. 21 Patients, their relatives, and healthcare workers should be trained to prevent foot ulcers; apart form that, diabetes-compatible shoes should be available to match the risk, and attempts should be made toward establishing regular podiatric treatment in preulcerous risk patients.
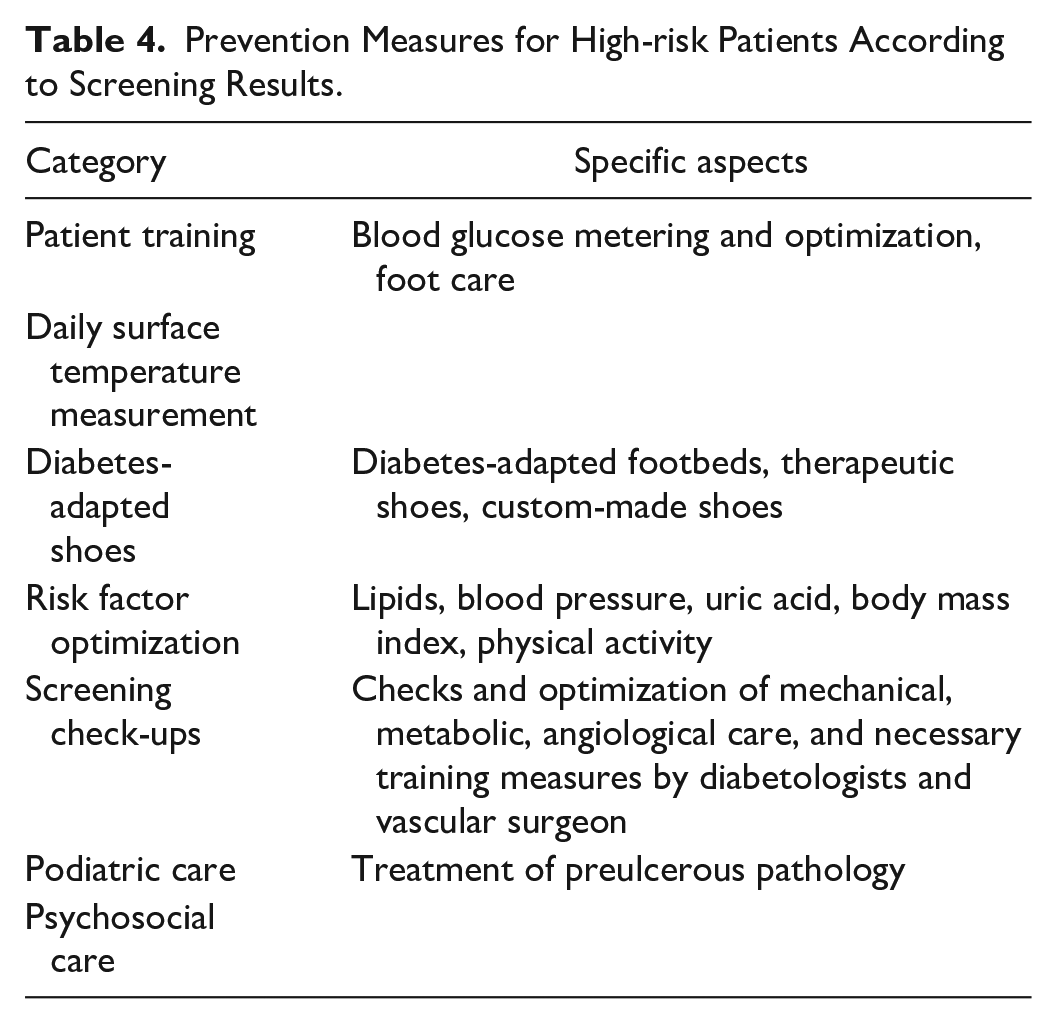
The whole repertoire of preventative measures against ulceration with documented effectiveness should be available to patients at high or very high risk. The guidelines provide detailed instructions, so we have only included a brief overview of potential prevention strategies in Table 4.
Prevention Measures for High-risk Patients According to Screening Results.
There are no evidence-based examinations on the necessary specialist medical and nursing examinations, and the recommendations available to us from national and international guidelines vary widely.16,19,21,25,50,74 The screening intervals proposed below reflect consensus among the authors on generating a very simple control scheme from the literature available.
Very high risk: Daily foot, sock, and footwear self-inspection should be self-evident among high-risk patients. Daily surface temperature measurement on the feet and medical presentation on significant temperature increases may also prevent ulcer formation. Monthly podiatric treatment is recommended for preulcerous pathology. A diabetes check-up to optimize risk factors should be carried out every three months. All of the patients should receive specific foot training and footwear designed for the condition, and the footwear should be checked every three months and replaced as necessary.
High risk: The recommendations comprise podiatric check-ups every three months, diabetes check-ups every six months, and additional consultation with a vascular surgeon once a year. All patients should be given special foot training and at least footbeds adapted for diabetes as well as therapy footwear.
Moderate risk: Recommendations comprise podiatric check-ups every six months, a diabetic check-up once a year, and angiological screening every two years. A foot bed adapted for diabetes should be prescribed and replaced annually.
Weaknesses in risk scores
Risk scores such as the European Society of Cardiology score for cardiovascular mortality 75 and the ASCVD Risk Estimator Plus of the American Heart Association, and the American College of Cardiology 76 are usually generated using long-term multinational prospective clinical trials or large-scale epidemiological data. Once developed, a score is ideally reproduced on a comparable cohort for subsequent validation at national level.
In the absence of large-scale prospective data with high research data quality for the given topic, our aim was to examine well-known risk factors by methodical analysis of existing literature and deduce an appropriate weighting; this approach extrapolates existing research data of varying quality to a certain extent of subjectivity. Typical variations in terms used in the underlying studies for elementary concepts such as polyneuropathy, foot deformity, etc. and their definitions as risk factors hamper attempts at dealing with the topic, so the results should therefore be interpreted as “expert consensus.”
Outlook and strengths in the risk score
No risk score currently offers any primary prevention opportunity against diabetic foot ulcers, especially for unspecialized general practitioners, but this score may be performed in the diabetes disease management program (DMP) in less than three minutes. Directly linking the score items with those of the DMP would be ideal and has been planned. The electronic system displays the risk category using a traffic lights color system of red, amber, and green, together with the check-up intervals indicated for the score, immediately after score result and DMP input.
This score should be tested for clinical use in a pilot study in everyday medical practice, with consecutively screened patients monitored for ulcer development over a period of two years. The score will be determined in the diabetes DMP. Every second patient will be checked strictly according to the score results using the check-up intervals outlined above; the other patients will be treated according to normal clinical practice. The suitability of the score for patient testing in adapted form along with the possible effects of stronger awareness for risk patients and the possibility of ulceration will also be evaluated.
Conclusion
Preulcerous risk situations should be diagnosed in patients with diabetes with comprehensive preventative measures taken to prevent diabetic ulceration and sequelae. We have developed a semi-quantitative risk score using methodical literature analysis toward practical and meaningful risk analysis within a few minutes using existing documentation software (from the DMP), providing doctors with recommendations for check-up intervals, and other preventative measures.
This relatively simple score provides rapid risk classification for patients, and sports a visually striking user interface to ease the way for both physicians and patients in gaining an insight into individual risk situations. The aim of this score is to provide high-risk patients with preventative measures against future complications more effectively.
The risk factors for DFS are well known, but the heterogeneity in definitions and methodologies makes comparison difficult. Our aim was to use research literature to develop a score providing physicians with a fast and simple method to evaluate their patients’ individual risk situation:
Classified major criteria: history of amputation or ulceration, pronounced polyneuropathy.
Classified moderate criteria: pronounced foot deformity, pronounced hyperkeratosis, slight polyneuropathy, documented peripheral arterial disease, renal insufficiency.
Classified minor criteria: foot or nail fungus, HbA1c >9%, diabetes duration, lack of visual or cognitive capability for self-administered healthcare or foot care, unsuitable shoes or socks, male gender.
This score should be tested for clinical use in a pilot study in everyday medical practice, with consecutively screened patients monitored for ulcer development over a period of two years.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This publication was previously published as “Kress et al 77 ” in German and is re-published with permission by Verlag Kirchheim & Co. GmbH.
SK has concluded contractual relationships, conducted studies and consultations, and received invitations to congresses with and for AstraZeneca, Bayer Vital, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Hoffmann-La Roche, Janssen-Cilag, Johnson & Johnson, Lohmann-Rauscher, Menarini/Berlin-Chemie, Merck, MSD Sharp & Dohme, Novartis, Novo Nordisk, Omniamed, Sanofi, Smith & Nephew, and Takeda. HA is a member of the supervisory board and advisory board of Sanofi, and has received grants or fees from Sanofi, MSD, Lilly, Boehringer Ingelheim, Novo Nordisk, and GlaxoSmithKline. AB is an employee at Sanofi-Aventis Germany. GF is the medical director and managing director of Institut für Diabetes- Technologie Forschungs- und Entwicklungsgesellschaft mbH, University of Ulm, Ulm (IDT), which conducts clinical studies on medical devices for diabetic therapy on its own behalf and on commission for various companies. GF/IDT have received speakers’ honoraria or consulting fees from Abbott, Ascensia, Dexcom, I-sens, LifeScan, Menarini Diagnostics, Metronom Health, Novo Nordisk, PharmaSense, Roche, Sanofi, Sensile, and Ypsomed. LH is an employee at Science Consulting in Diabetes, a member of the supervisory board and metabolic research advisory board, at Profil Institut für Stoffwechselforschung; he holds shares/capital in Profil, Neuss, and Profil, San Diego (US), and has received grants or fees from Roche, Sanofi, Cellnovo, and Abbott. UH denies any conflict of interests. BK has received grants or fees from Sanofi, Novo Nordisk, Berlin-Chemie, Roche, and Abbott. AP is an employee of Sanofi-Aventis Germany and a shareholder in Sanofi, Paris. OS has received grants or fees from Sanofi. HV is a member of the Sanofi advisory board and has received grants or fees from Sanofi, Lilly, Roche, Berlin-Chemie, Bayer, and Lifescan. CZ denies any conflict of interest with respect to the content of the article. RL has shares in funds and has received grants or fees and third-party funds and project funding from Urgo.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Preparation of this manuscript was funded by Sanofi-Aventis Deutschland, Berlin, Germany.
