Abstract
Background:
Blood glucose monitoring (BGM) is essential for glycemic control in diabetic therapy. Followingly, accurate sensors are required for both daily personal and clinical use. The frequency of sensor use in patients with diabetes facilitates the use of disposable components. However, BGM systems are not exempt from green innovation sustainability initiatives.
Methods:
Clinical study of a high-accuracy green design blood glucose monitor using an innovative optical transmission absorbance system was carried out. Venous blood samples were collected from 104 patients with type II diabetes. The heat resistance of sensor strips was evaluated by storing sensor strips at 25℃ and 60℃ for approximately 3 months. Accuracy of the BGM system was evaluated via the ISO 15197:2013 protocol.
Results:
The BGM system achieved ±7.1% accuracy in glycemic level measurement, with 84% of all measurements within ±5% of the reference values. Furthermore, the sensor strip demonstrated heat resistance for more than 3 months when stored at 60℃.
Conclusions:
A new, highly accurate BGM system was developed based on the latest optical measurement system, introducing a rare metal-free “green-strip.” The developed BGM system achieved the highest reported accuracy in clinical research, using venous blood from patients with diabetes. The sensor strip also exhibited high heat resistance, reducing limitations on storage conditions.
Introduction
Accurate blood glucose monitoring (BGM) is key to achieving tighter glycemic control during diabetic therapy. A smaller BGM systematic bias is associated with a decrease in HbA1c, lower rates of hypoglycemia, and blood glucose (BG) levels within the target range; conversely, a large bias results in worsening of these indicators.1-5 Accordingly, standards which reinforce the accuracy of such systems have been implemented, such as the ISO 15197:2013.6,7 Although various new glucose monitoring devices are being developed and continuous glucose monitoring (CGM) systems are recognized as ideal monitoring systems for glycemic control, self-monitoring of BG still plays an essential and significant role in patients with type I and type II diabetes. 8 CGM systems calibrated with high accuracy BGM systems result in better clinical outcomes. 9 Additionally, BGM systems are cost-efficient, with improved economic performance observed with the use of high accuracy devices.10-12 Due to these advantages, medical device companies are aiming to introduce highly accurate BGM entities to the market.13-16
Considering the negative environmental impacts that are associated with disposable medical devices, the development of an eco-friendly product is critical.17,18 Single-use BGM sensor strips intended for at-home sensing generate a large amount of waste, and the future of BGM systems is not exempt from “Green Innovation.” This environmental suitability initiative aims to reduce the carbon footprint of future technological developments through conserving natural resources, as well as improving waste management processes. Strategies to achieve green innovation in BGM systems include manufacturers downsizing both their products and packaging. Lowering the associated carbon footprint of these products can be achieved by reducing the size of devices, modifying the design or materials used, increasing their operational stability, and increasing the availability of recyclable devices and chemicals. Thus, accuracy and accordance with a “green” design is essential for the next generation of BGM.
In this study, we report a novel BGM system, utilizing new optical measurement technology and considering the aforementioned environmental criteria. 19 Our next generation BGM system demonstrated the highest reported accuracy for BG measurement compared with the BGM systems available on the market. Owing to the recently developed chemistries used in this optical principle-based BGM system, rare metal-based electrodes, such as gold or palladium, and rare metal complex mediators, such as osmium or ruthenium, were not employed. Furthermore, the storage stability of this BGM system was investigated at higher temperatures (60℃), well above the normal storage conditions (1~30℃).
Material and Methods
Accuracy Evaluation for the Newly Developed BGM System
The study protocol was approved by the Osaka Pharmacology Clinical Research Hospital ethics committee and written informed consent was obtained from all participants. Assessment of system accuracy was conducted at Osaka Pharmacology Clinical Research Hospital (Osaka, Japan), and completed in April 2019. Fresh venous blood was collected using a vacuum blood collection tube, and anticoagulated with heparin.
A total of 104 adult patients (≥20 years old) with type II diabetes were evaluated for system testing. The exclusion criterion were health conditions which deemed a participant unsuitable for this study, as evaluated by a healthcare professional. The classification of samples is shown in Table 1.
Classification of Blood Samples.

Optical BGM Device
A novel, highly accurate BGM device was constructed based on a recently developed optical measurement system, with a rare metal-free sensor strip. 19 The device uses an optical unit with a multiwavelength detection system and original reagents for the sensor strip to realize accurate BG measurements in 9 seconds with 1 µL of blood sample. Figures 1 and 2 are schematic diagrams of the optical measurement and original reagent systems, respectively.
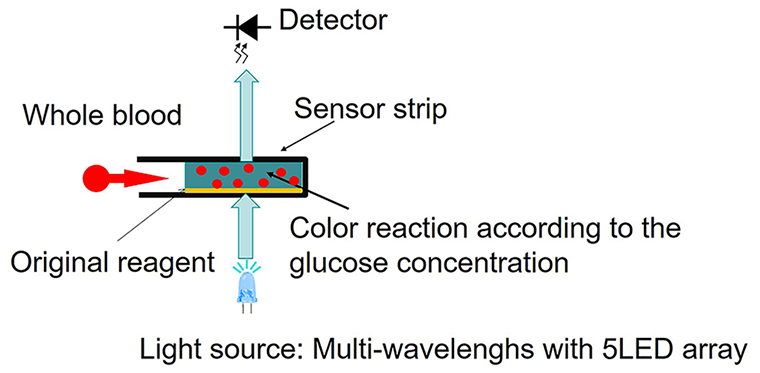
Optical unit of novel blood glucose monitoring system.
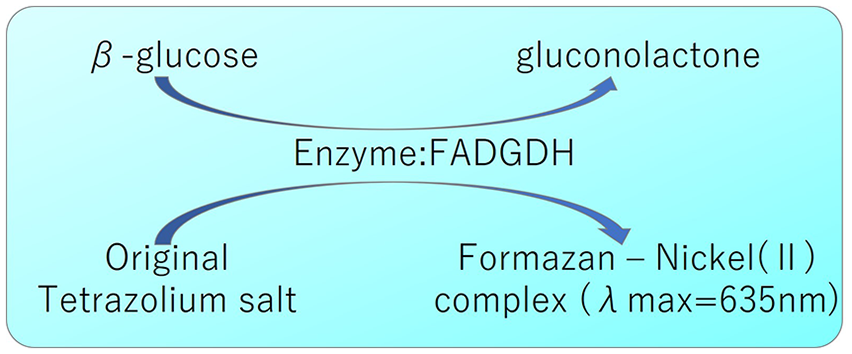
Original reagent system used to achieve a large glucose reaction signal for the blood glucose monitoring system. 19 Abbreviation: FADGDH, flavin adenine dinucleotide glucose dehydrogenase.
Experimental Procedures
All experimental procedures were conducted by trained technicians summarized as follows:
Fresh venous blood was collected with a vacuum blood collection tube and anticoagulated with heparin.
BG level was measured using a reference instrument.
The BG level was modified, if necessary.
The hematocrit level was modified to 3 groups (low, no adjustment, high).
BG level was measured with the reference instrument before BGM measurement with the developed system.
BG levels were measured via BGM system with 1 sensor strip lot (duplicate measurements per blood sample).
BG levels were measured with the reference instrument after BGM.
Procedures (1) to (7) were repeated until the required number of samples was obtained.
The BG level was modified to allow distribution over the entire measurement range, with reference to ISO 15197:2013. Whole blood samples were modified through addition of glucose or incubation to raise or lower BG levels, respectively.
Reference glucose concentrations were measured via the hexokinase (HK) standard method using a HK automatic analyzer 7700 (Hitachi High-Technologies Corporation, Tokyo, Japan). Quality control for the HK automatic analyzer was performed according to the manufacturer’s instructions each measurement day.
The hematocrit of the sample was measured as follows.
A microhematocrit capillary tube (Hirschmann Laborgeräte GmbH & Co. KG, Eberstadt, Germany) was placed near fresh venous blood, allowing blood to flow into the tube.
The tube was sealed with clay, placed in a hematocrit centrifuge, and spun for 5 minutes at 12,000 rpm (15,000 g).
A ruler was used to measure the length of packed red cells and whole blood to calculate hematocrit level (%).
Statistical Analysis
To evaluate the accuracy of the BGM system, BG values obtained with the BGM device were compared with the reference values after each measurement, according to ISO 15197:2013 criteria. BGM readings should fall within ±15 mg/dL, ±10 mg/dL, or ±5 mg/dL of the reference at BG concentrations <100 mg/dL, or within ±15%, ±10%, or ±5% at BG concentrations ≥100 mg/dL. Additionally, to confirm the accuracy when 95% of the data were included for each glycemic and hematocrit range, a consensus error grid was used to evaluate the clinical significance between the results obtained via BGM and the reference measurements.
Evaluation of Heat Stability of the Newly Developed BGM System
Heat stability was evaluated by storing sensor strips in an incubator (DRM620DA; Advantech Co., Ltd., Taipei, Taiwan) placed at room humidity (36.6~44.2%) for 0, 3, 6, 14, 21, 102 days, at both 25℃ and 60℃. At each time-point and temperature, venous blood samples were adjusted to around 100 mg/dL and 400 mg/dL of glucose, and 40% ± 1% of hematocrit. All blood samples were measured via the BGM system to obtain the measured value, and the standard method (Automatic analyzer 718; Hitachi High-Technologies Corporation) to obtain the reference value. All measurements were conducted in triplicate at room temperature and humidity. The sensitivity of our system was then calculated according to: 100×(average of measured value—average of reference value)/(average of reference value) [%]. The differences between the sensitivities at 60℃ and 25℃ for each time-point were used to assess the effect of high temperature.
Results
Accuracy Evaluation for the Newly Developed BGM System
The hematocrit level of the samples used in the study ranged from 28% to 64%. Since 104 blood samples were adjusted to 3 hematocrit levels and measured in duplicate, a total of 624 measurements were obtained. Figure 3 shows the accuracy of the BGM system; following analysis of the 624 measurements, it was confirmed that 95% of the data was within ±7.1 mg/dL or ±7.1% accuracy. The percentage of BGM readings within the specific accuracy limits of ISO 15197:2013 are illustrated in Table 2. Over the entire glucose concentration range, 100% of BGM results were within the ±15 mg/dL and ±15% limits at BG concentrations <100 mg/dL and ≥100 mg/dL, respectively. In all, 99.2% of the BGM results were within ±10 mg/dL at BG concentrations <100 mg/dL and ±10% at BG concentrations ≥100 mg/dL. Lastly, 84.0% of the BGM results were within ±5 mg/dL at BG concentrations <100 mg/dL and ±5% at BG concentrations ≥100 mg/dL. ISO 15197:2013 accuracy criteria require 95.0% of measurements to be within ±15 mg/dl and ±15% limits; therefore, these results demonstrate that the constructed BGM system meets ISO criteria.
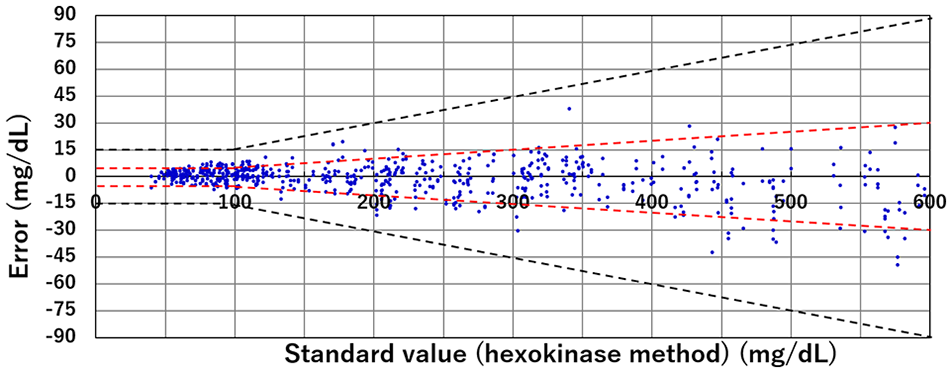
System accuracy of the new optical blood glucose monitoring system. Absolute differences between the blood glucose monitoring and reference measurement results; 624 data points are shown (312 samples measured in duplicate).
Accuracy of Blood Glucose Monitoring System.

Percentage of blood glucose monitoring readings within specific accuracy limits outlined in ISO 15197:2013.
Abbreviations: BGM, blood glucose monitoring; ISO, International Organization for Standardization.
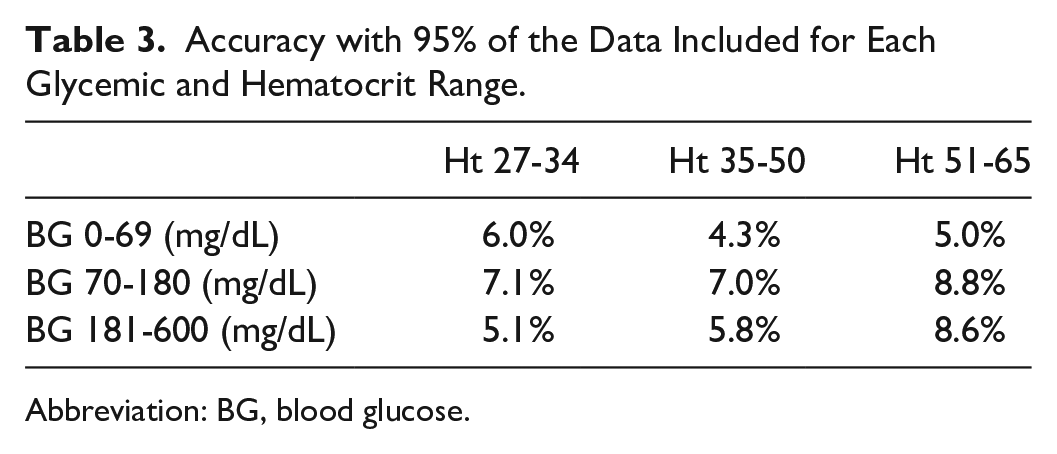
Accuracy data for 95% of the measurements within each glycemic and hematocrit range are presented in Table 3. The BGM system demonstrated higher accuracy (5.1%) in the lower hematocrit range (27%-34%), however this accuracy was reduced to 7.1% in the BG concentration range of 70 to 180 mg/dL. According to ISO 15197:2013 criteria, 99% of individually measured values should fall within zones A and B of a consensus error grid. Figure 4 shows the consensus error grid for the BGM system; all values fall within zone A, meeting the requirements of the ISO 15197:2013 criteria.
Accuracy with 95% of the Data Included for Each Glycemic and Hematocrit Range.
Abbreviation: BG, blood glucose.
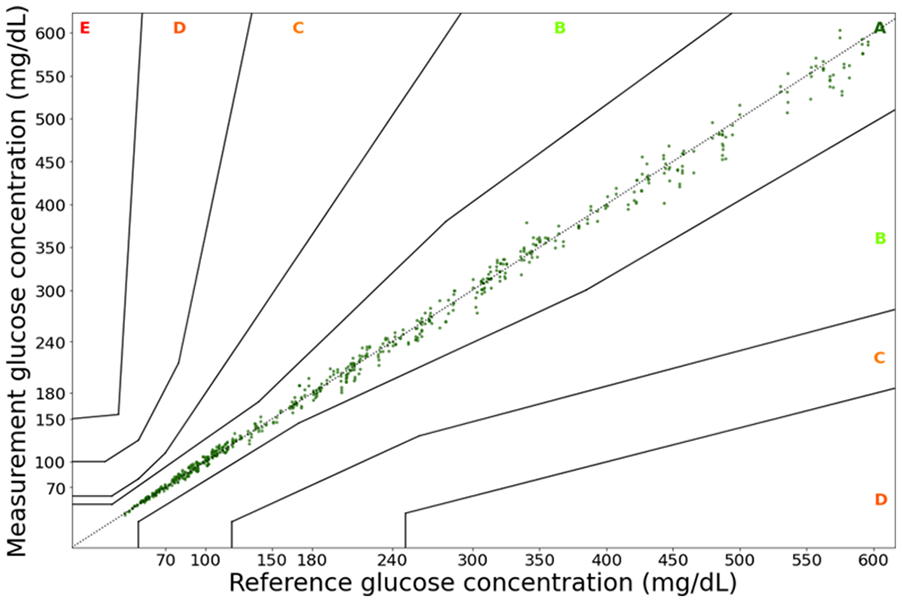
Consensus error grid of evaluated blood glucose monitoring system. (a) The horizontal axis represents the blood glucose concentrations obtained using the reference measurement (hexokinase method). (b) The vertical axis represents the glucose concentration measured via the developed blood glucose monitoring system. The consensus error grid is divided into 5 zones signifying the estimated risk to the patient. Zone A: no effect on clinical action; Zone B: altered clinical action—little or no effect on clinical outcome; Zone C: altered clinical action—likely to affect clinical outcome; Zone D: altered clinical action—may have significant medical risk; Zone E: altered clinical action—may have dangerous consequences.
Evaluation of Sensor Strip Heat Resistance within the Developed BGM System
Figure 5 presents the results of the sensor strip storage stability investigation at 60℃ and 25℃. At 100 mg/dL, the highest difference in measured values was +2.8% after 6 days of storage, and the lowest difference of -1% between measured values was observed after 21 days. Following these results, an increase to +4% was observed between 21 and 102 days.
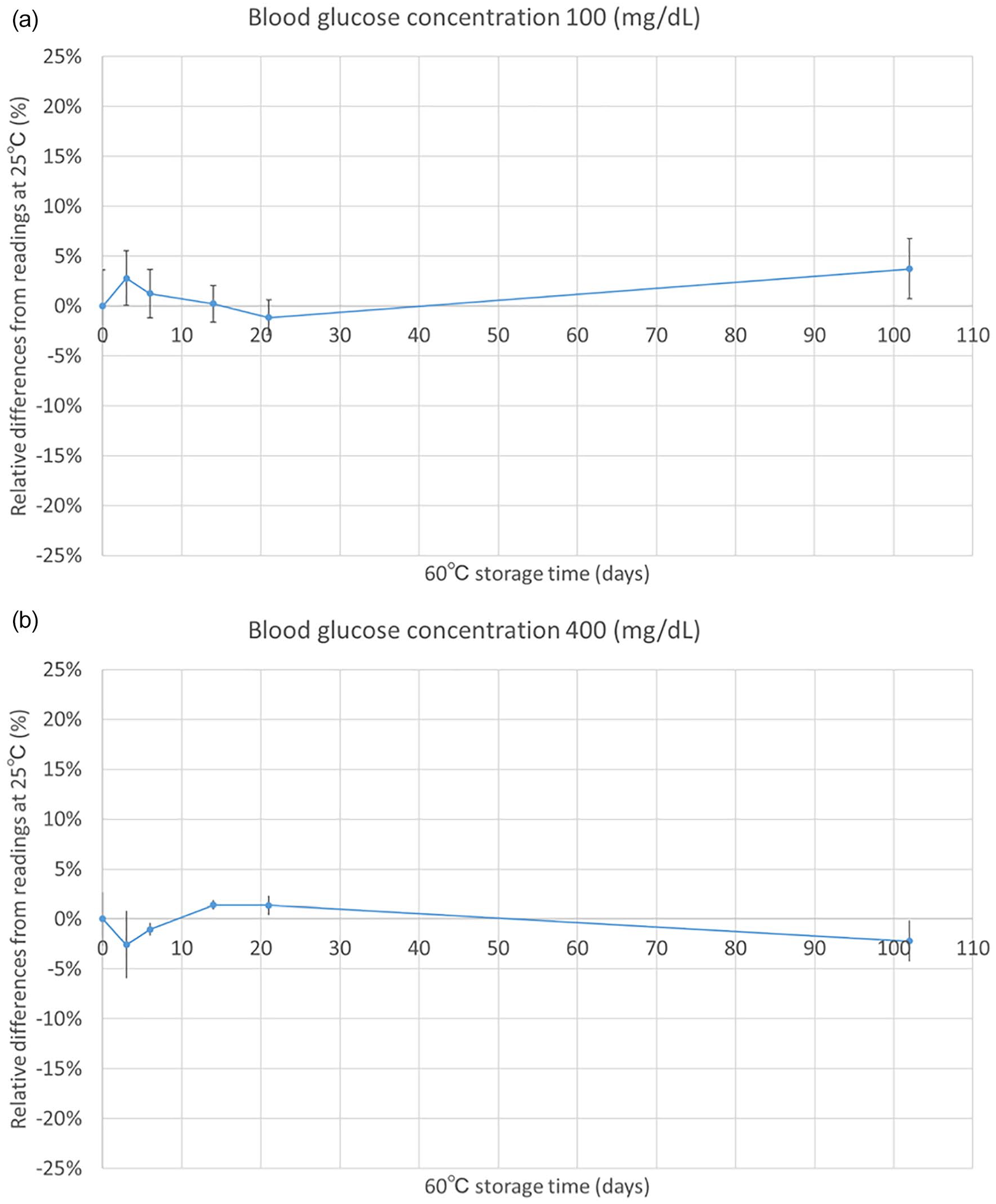
Storage stability of sensor strips at 60℃. Stability was investigated with 100 mg/dL (a) or 400 mg/dL (b), and compared with the sensor responses of those stored at 25℃. Error bars represent standard deviation.
At 400 mg/dL, the maximum difference in measured values was -2.6% after 3 days. After 14 days, an increase to +1.4% was observed; however, a gradual decrease in values was observed over the remainder of the experimental time course, reaching a final measured value of -2.2% at 102 days. For both concentrations, the differences were less than 5% at each time point, indicating that the sensor strip demonstrated high heat stability for 3.5 months at 60℃.
Discussion
In this study, we report a next generation BGM system, utilizing new optical measurement technology which employs novel chemistries combined with a multi-wavelength (green, orange, red, infrared [below 800 nm], infrared [over 900 nm]) optical unit that is accurately capable of detecting hematocrit signal, glucose signal, and scattering nose of red blood cells. To the best of our knowledge, this new BGM sensor achieved the highest accuracy among currently reported BGM systems.14-16,20-22 In all, 95% of the data measured with this BGM system was within ±7.1 mg/dL or ±7.1% accuracy. Accuracy in the hypoglycemic region, which is of particular importance in patients with diabetes, was less than ±6%. Conversely, in the high BG (≥181 mg/dL) and low hematocrit range, sensor accuracy was highest at ±5.1%. The data show a gradual decrease in accuracy as the hematocrit value is increased. This phenomenon is attributed to an increase in scattering noise caused by red blood cells, the amount of which increases with increased hematocrit. Nevertheless, despite the effects of hematocrit level, the clinical accuracy is considered to be sufficiently high. 22
Owing to the new chemistries employed in this optical principle-based BGM system, rare metal-based electrodes, such as gold or palladium, and rare metal complex mediators, such as osmium or ruthenium, were not utilized. The constituents of this sensor have thus met the philosophy of the Green Diabetes Initiative. 17
It has been also confirmed that the reagent system is not affected by interfering substances such as acetaminophen, uric acid, and bilirubin. 23
Furthermore, the stable chemistries promoted resistance to degradation when stored at high temperatures. Functional evaluations at 60℃ revealed that the performance of the product remained stable for more than 3 months. One explanation for the observed high heat resistance is the high heat stability of the enzyme. The enzyme utilized in this sensor is an engineered fungi derived glucose dehydrogenase (GDH) harboring flavin adenine dinucleotide (FAD) as the cofactor (FADGDH), which is stable at high temperatures. Similar to other reported fungi derived FADGDHs, 24 this FADGDH is specific to glucose, and there is no reactivity toward maltose. There is some reactivity with D-xylose, however, only about 10% reactivity compared to β-D-glucose. Additional cause for the high heat resistance is the low moisture content of the packaged reagent—a result of the desiccant contained in the packaging material—thus preventing enzyme deterioration. This high heat resistance can eliminate the limitation of conventional storage temperatures (30℃ for commercially available BGM is suggested due to deterioration of the reagents).
Currently, strict temperature control during product delivery is necessary due to the instability of the sensor strips. Additionally, there are situations during at-home use where storage temperatures can exceed the recommended limit, causing deterioration of reagents.25-27 For example, in warmer climates or seasons, storage in a car—or even at home—exposes the reagents to temperatures greater than 30℃. Since the newly developed sensor strip is stable long-term at 60℃, it will maintain accuracy, even when unexpectedly exposed to high temperatures. This thereby contributes to enhanced usability and decision making for both patients with diabetes and their medical providers. Moreover, the high heat resistance may reduce both the distribution costs and environmental burden, since unlike conventional sensor strips, distribution of the fabricated sensor strips does not require temperature control. The outstanding thermal stability significantly reduces the energy required for sensor strip storage, which also fulfills the Green Diabetes Initiative philosophy. 17
Future research will investigate algorithmic noise reduction of scattering light. Our sensor strips demonstrated heat resistance at 60℃ for more than 3 months, suppressing the thermal deterioration of sensor strips caused by unrecommended storage (e.g storage of sensor strips in a car during summer), and thus enhancing usability. The sensor strips are also low-cost and eco-friendly, since refrigeration or strict temperature control is not necessary. Additionally, surveillance has become a major safety issue in recent years. 28 This optical sensor addresses the issue by maintaining performance throughout the product’s lifecycle and by providing stable glucose measurements for patients with diabetes. 29 Therefore, our BGM system regulates glycemic control and reduces environmental burdens, which are essential in building a sustainable society.
Conclusions
In this study, we report a clinical study using a next generation BGM system, which utilizes a new optical measurement technology through employing novel chemistries combined with a multiwavelength optical unit. This sensor represents the world’s most accurate BGM system which does not employ rare metal components, and exhibits high thermal storage stability. Therefore, the current requirements to develop more sustainable and environmentally conscious disposable BGM devices for at-home use are met, adhering to the philosophy of the Green Diabetes Initiative.
Footnotes
Acknowledgements
The authors would like to thank Ms. Ellie Dewese Wilson, PhD student at the Joint Department of Biomedical Engineering, UNC Chapel Hill and North Carolina State University, for English proofreading of this article.
Abbreviations
BGM, Blood Glucose Monitoring; ISO, International Organization for Standardization; CGM, Continuous Glucose Monitoring; HK, Hexokinase; BG, Blood Glucose; Ht, Hematocrit.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Takeyuki Moriuchi, Yuto Otaki, Hiroya Satou, Fumihiko Chai, Yuma Hayashida, Ryokei Aikawa and Takayuki Sugiyama are full time employees of Terumo Corporation. Koji Sode is a full-time employee of the University of North Carolina at Chapel Hill. Sode’s research projects at UNC are partially financially supported by Terumo Corporation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Terumo Corporation.
