Abstract
Introduction:
Self-monitoring of blood glucose (BG) is important in diabetes management, allowing people with diabetes (PWD) to assess responses to diabetes therapy and to inform if they are attaining their glycemic targets. This study assessed the accuracy and user performance (UP) of a new blood glucose monitoring system (BGMS), CONTOUR®PLUS ELITE, according to International Organization for Standardization (ISO) 15197:2013 criteria and also more stringent criteria.
Methods:
In laboratory Study 1, capillary fingertip blood samples from 100 PWD were evaluated using the new BGMS. In clinical Study 2, 130 PWD had Yellow Springs Instrument (YSI) analyzer reference measurements against subject-obtained fingertip and palm blood, and trial staff-obtained venous blood. The new BGMS was tested with test strips from three different lots. A UP questionnaire assessed ease of use.
Results:
Study 1: 100% of combined accuracy results fulfilled ISO criteria (±15 mg/dL at BG <100 mg/dL; ±15% at BG ≥100 mg/dL); 99.8% fulfilled more stringent criteria (±10 mg/dL at BG <100 mg/dL; ±10% at BG ≥100 mg/dL). Error grid analysis showed that 100% of results were within zone A. Study 2: >98% of subject- and 100% of trial staff-obtained performance results met ISO criteria. Most subjects (>96%) found the BGMS easy to use.
Conclusion:
The new BGMS exceeded minimum ISO 15197:2013-specified standards for both accuracy and UP criteria, along with the more stringent accuracy criteria. These data show that this new BGMS can be a useful tool in managing glycemic control for PWD.
Keywords
Introduction
Self-monitoring of blood glucose (BG) plays a key role in diabetes management, by allowing people with diabetes (PWD) to assess their responses to diabetes therapy and to inform if they are attaining their glycemic targets. 1 The International Organization for Standardization’s (ISO) standard 15197:2013 defines criteria for assessing accuracy and user performance (UP) of a blood glucose monitoring system (BGMS). 2 The accuracy evaluation (section 6.3) assesses the analytical accuracy of a BGMS under controlled conditions with trained operators. 2 The UP evaluation (section 8) assesses if intended users can obtain accurate BG values when operating the BGMS, given only the instructions and training materials routinely provided. 2
A new BGMS, CONTOUR®PLUS ELITE, has been developed for use with currently available CONTOUR®PLUS test strips and is designed to synchronize with the CONTOUR®DIABETES app on a smartphone or tablet. It is intended as an over-the-counter device for measurement of fingertip capillary blood and blood from the palm (alternate site testing [AST]) by PWD or healthcare professionals (HCPs), and venous blood by HCPs only.
In this study, the accuracy of the new BGMS was assessed by trained operators and PWD under optimal operating conditions according to the analytical performance evaluation outlined in ISO 15197:2013. Results were expressed using these and more stringent criteria (±10 mg/dL at BG <100 mg/dL and ±10% at BG ≥100 mg/dL). The accuracy of the BGMS can be a critical component in reducing hypoglycemia and HbA1c 3 and in calibrating continuous glucose monitors when required. 4
Study Design and Methods
Study 1 included 100 PWD; Study 2 included 130 PWD. An institutional review board approved the study protocols, and all subjects completed the informed consent process.
A New Blood Glucose Monitoring System
Key features of the new BGMS are listed in Table 1. Visual and functional testing were conducted on all meters to verify proper functioning prior to study start.
Summary of the New BGMS Technology and Features.
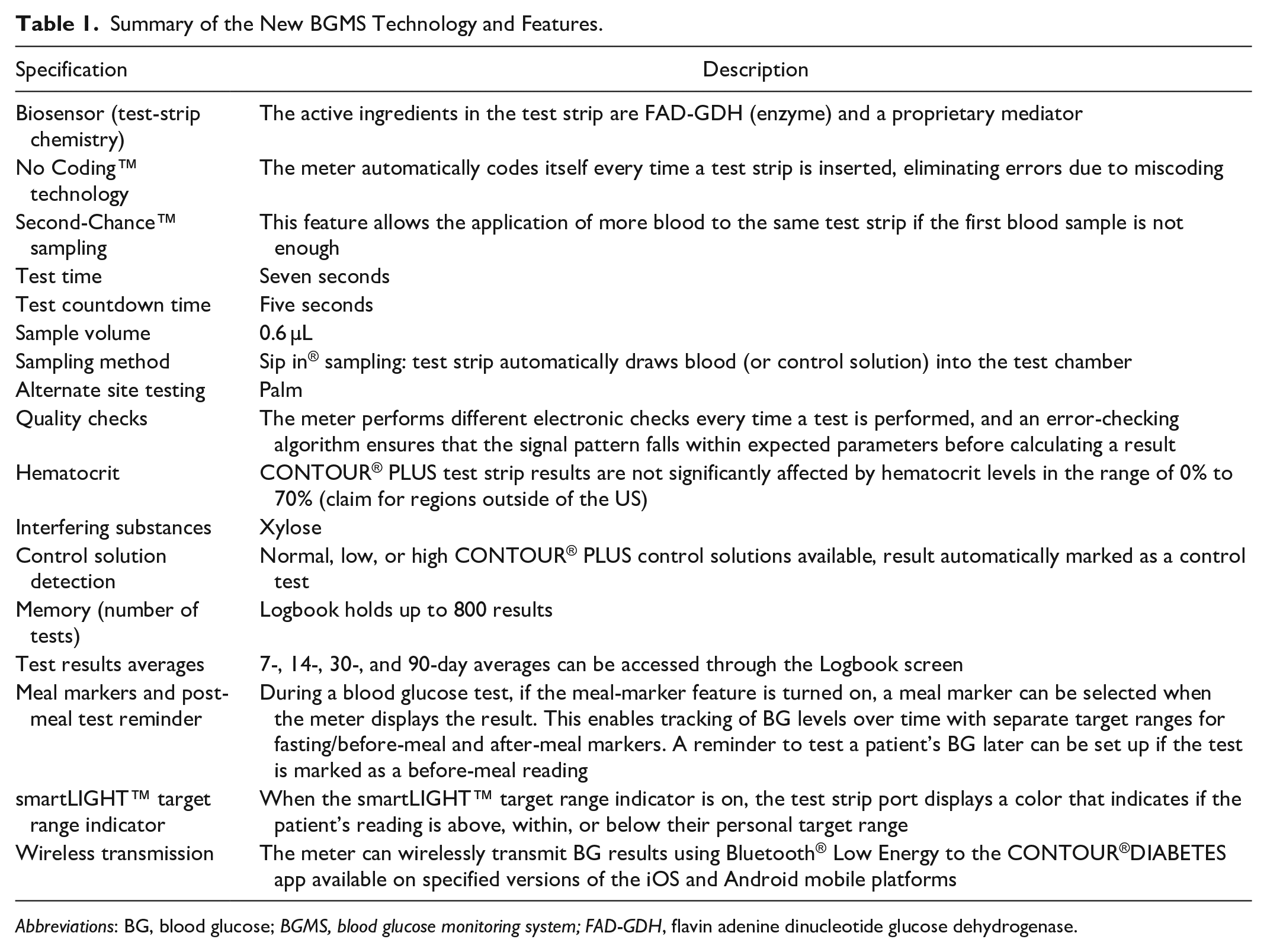
Abbreviations: BG, blood glucose; BGMS, blood glucose monitoring system; FAD-GDH, flavin adenine dinucleotide glucose dehydrogenase.
Reference Glucose Measurement
Separated plasma samples were tested in parallel on a Yellow Springs Instrument (YSI) 2300 Stat Plus Glucose and Lactate Analyzer (YSI Inc., Life Sciences, Yellow Springs, OH, USA). The precision and accuracy of the YSI Analyzer was validated throughout the study. After each sample was run on the YSI Analyzer, a serum control level with a target closest to the sample BG reading was run as a quality check to ensure proper YSI Analyzer function. The serum-based controls were produced by Bio-Techne (Minneapolis, MN, USA) and were National Institute of Standards and Technology traceable to Standard Reference Material 917. If samples were altered, only one BG reference result was obtained. Altered samples required either spiking with glucose to a higher level, or glycolyzing in a water bath. Once the sample reached the desired BG concentration via one of these processes, both the YSI and meter testing were performed within 15 minutes. Since this blood sample is outside of the body, the rate of glycolysis within this time frame should be minimal. Therefore, a second YSI run/sample was not necessary. Two YSI measurements were performed on native samples.
Study 1: Accuracy Analysis
Accuracy of the new BGMS was assessed at a single study site. A trained operator tested the BG of the PWD with the new BGMS and the YSI Analyzer. Data from fingerstick testing were combined to achieve 100 independent capillary samples within the specified BG distribution as per ISO 15197:2013 (Table 2). Each sample was tested with three different lots of test strips, with duplicate readings per lot. Meters were disinfected between subjects. ISO 15197:2013 states that if the study population does not provide sufficient samples with very low and very high glucose concentrations, modified samples in which the glucose concentration has been raised or lowered may be substituted to achieve the required distribution. To ensure a sufficient number of samples within specified concentrations, modified blood samples were prepared as described in the ISO 15197:2013 protocol. 2
Distribution of BG Concentrations of Samples for the New BGMS Accuracy Evaluation as Specified in ISO 15197:2013.

Abbreviations: BG, blood glucose; BGMS, blood glucose monitoring system.
Accuracy was evaluated using ISO 15197:2013 criteria by calculating meter results falling within ±15 mg/dL of the reference value for BG <100 mg/dL or within ±15% of the reference value for BG ≥100 mg/dL. More stringent criteria were also tested (±10 mg/dL at BG <100 mg/dL and ±10% at BG ≥100 mg/dL). Parkes consensus error grid analysis evaluated the clinical significance of deviations of results from the new BGMS from laboratory BG results. 5
Study 2: User Performance Analysis
In total, 130 PWD, who had never used the new BGMS previously were enrolled in the study at a single clinical site. Subjects were given the new BGMS User Guide (UG) and Quick Reference Guide (QRG) to learn how to use the system with no additional training. Three test strip lots were used, with each subject testing strips from a single lot.
All subjects performed one fingerstick and one palm AST. Study staff also performed Tenderlett® lancing (Accriva Diagnostics, San Diego, CA, USA) of the subject’s fingertip to obtain a capillary YSI sample, as well as a meter test using the subject’s fingertip blood. All capillary meter results were compared with the YSI laboratory reference results of subject capillary blood from a separate Tenderlett® fingerstick (Accriva Diagnostics, San Diego, CA, USA). Subjects had venipuncture and venous blood tested with the new BGMS by study staff. All venous meter results were compared with YSI results from venous blood. The new BGMS is not limited to fingerstick testing by the PWD or the HCP, but is extending its indication for use to AST (by lay users [PWD] or HCPs) and venous blood (by HCPs only), which are evaluated as per ISO 15197:2013 standards.
User Performance Feedback
At study completion, PWD were asked for feedback (via a user performance questionnaire [UPQ]) on the ease of use/features of the new BGMS, as required by ISO 15197:2013. The goal was that 90% of the responses be rated as ≥3 on a five-point (Likert) scale: 1 = strongly disagree, 2 = disagree, 3 = neutral, 4 = agree, 5 = strongly agree.
Results
Study 1: Accuracy Analysis
In total, 600 new BGMS readings were collected using fingerstick specimens from 100 PWD; BG range 21-521 mg/dL, hematocrit range 29.5-52.0%. Of 100 samples, 78 met the BG distribution requirements naturally; 22 samples were modified to meet the reference range (Table 2).
When grouped according to BG levels <100 mg/dL or ≥100 mg/dL, 100% of results for the new BGMS fell within ±15 mg/dL or ±15% of the reference value, as summarized in Table 3. Regression and bias plots, showing ISO 15197:2013 limits, are included in Figures 1(a) and 1(b). Parkes consensus error grid analysis showed 100% (600/600) of test results within Zone A (Table 3 and Figure 2).
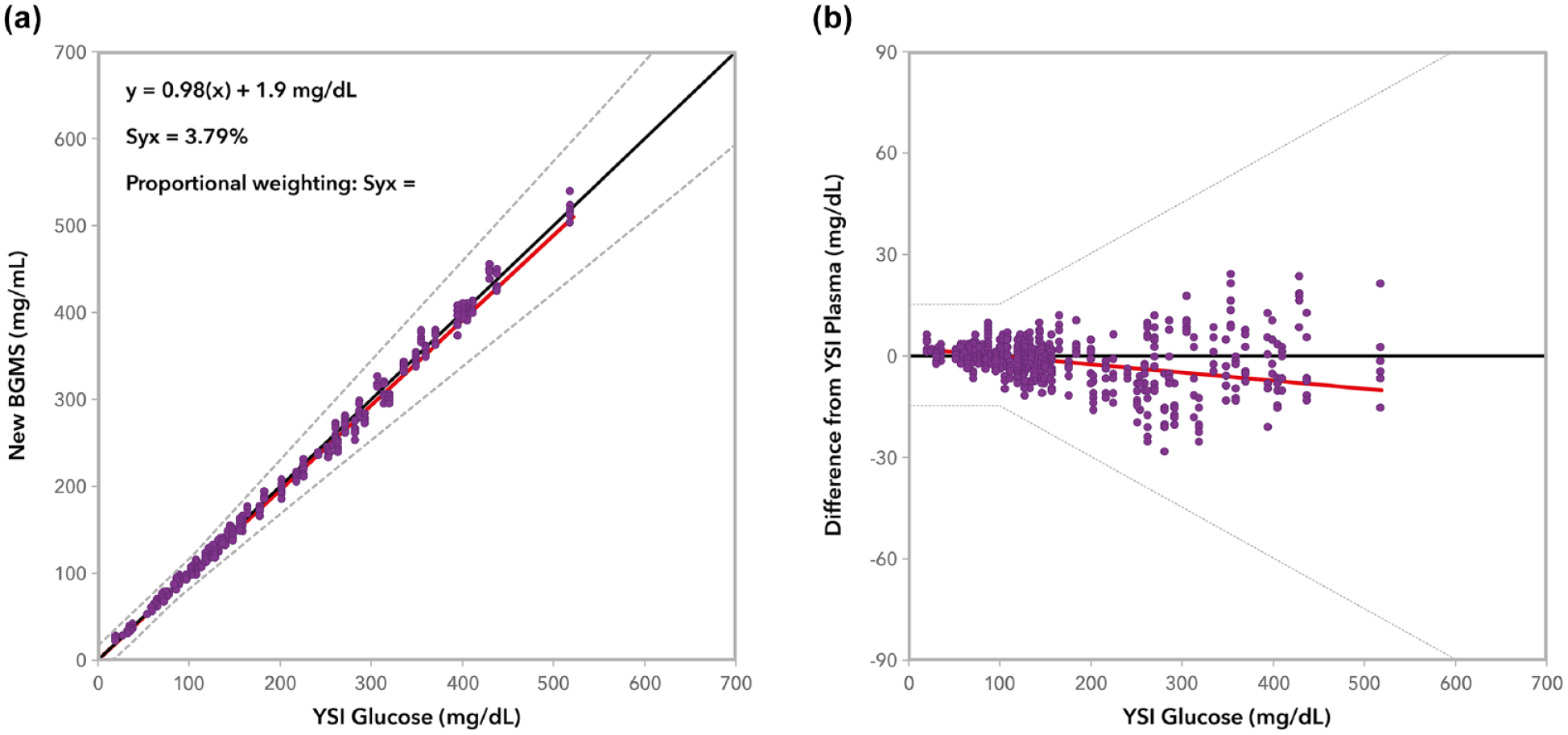
Regression (a) and bias plots (b) for the new BGMS according to ISO 15197:2013 limits. The more stringent criterion is included as part of the regression plot and is the standard error of the estimate and this indicates the variation around the regression line.
The New BGMS Accuracy Results (Trained Operators) Fall Within Minimum Acceptable ISO 15197:2013 Criteria and More Stringent Criteria.
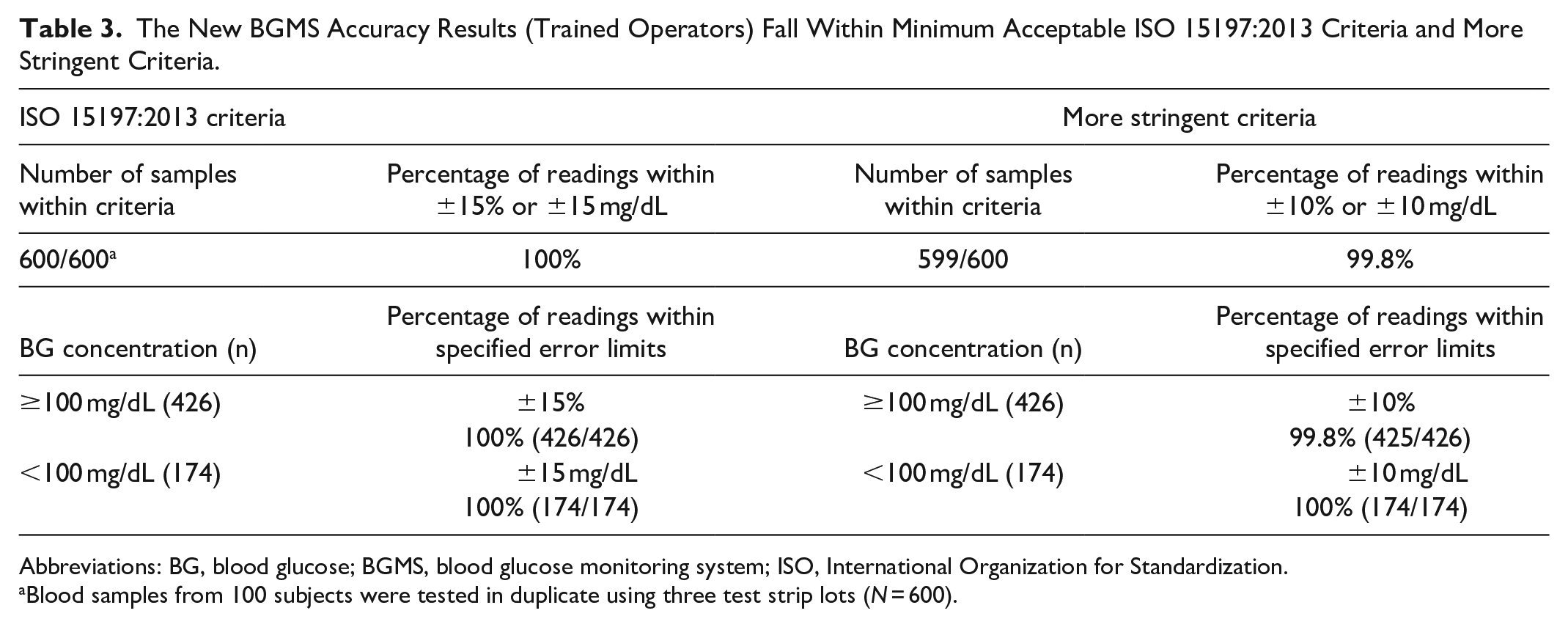
Abbreviations: BG, blood glucose; BGMS, blood glucose monitoring system; ISO, International Organization for Standardization.
Blood samples from 100 subjects were tested in duplicate using three test strip lots (N = 600).
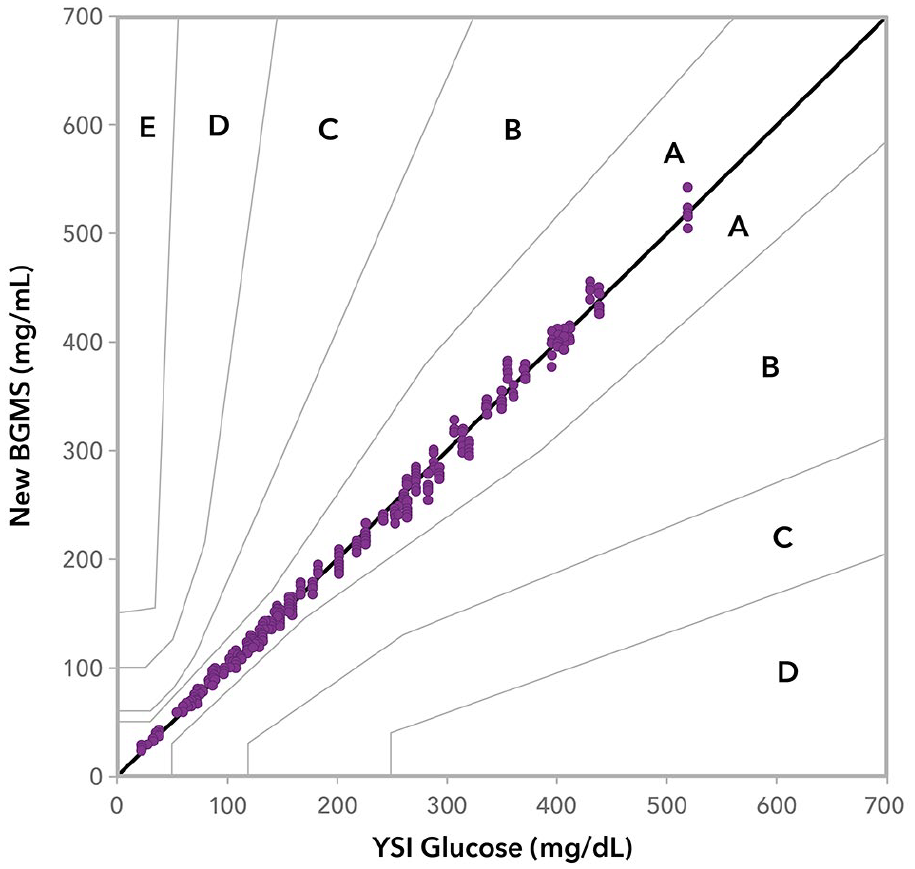
Parkes consensus error grid analysis of the new BGMS (100% in A).
Additionally, the new BGMS met more stringent accuracy requirements with 99.8% of combined results within ±10 mg/dL or ±10% of the reference value (Table 3).
Study 2: User Performance Analysis
In total, 130 PWD completed informed consent; 129 completed the study, one withdrew consent. Subject demographics are shown in Table 4. PWD demonstrated that they could operate the new BGMS and obtain valid results with no training, by using only the UG and QRG to learn to use the system. Blood testing spanned the range of the system with naturally occurring BG levels ranging from 66 mg/dL to 514 mg/dL by YSI measurement.
Subject Demographics of PWD in Study 2 (N = 130).
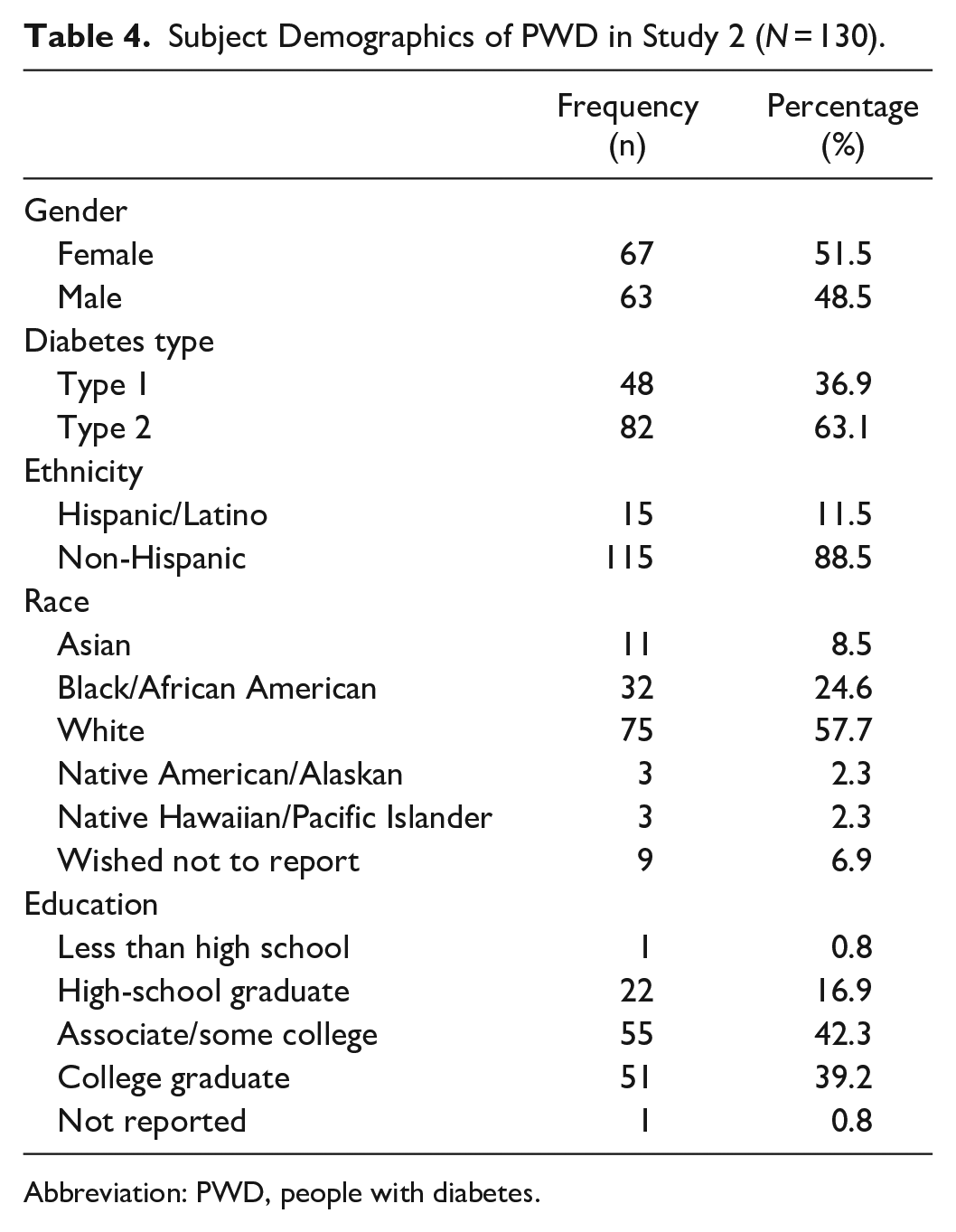
Abbreviation: PWD, people with diabetes.
PWD performing self-testing from the fingertip using the new BGMS achieved >99% of BG results within ±15 mg/dL for YSI reference BG <100 mg/dL, and within ±15% for YSI reference BG ≥100 mg/dL (Table 5). Figures 3(a)-(c) show the modified Bland-Altman difference plots for subject fingertip tests and palm AST, as well as for staff testing of venous blood. All plots showed a similar distribution and were symmetrical with respect to positive/negative errors (no trend was observed). Figure 4 shows the radar plot for subject-generated capillary results. Results from the subject fingerstick and AST palm and trained operator venous tests all met ISO criteria. Furthermore, 95% of subject fingerstick results were within 8.4 mg/dL or 8.4%. The study goal was met for the UPQ; >96% of subjects responded with a score of ≥3 for each statement (Table 6).
The New BGMS Performance Results (PWD Self-Test and Study Staff) Fall Within Minimum Acceptable ISO 15197:2013 Criteria.
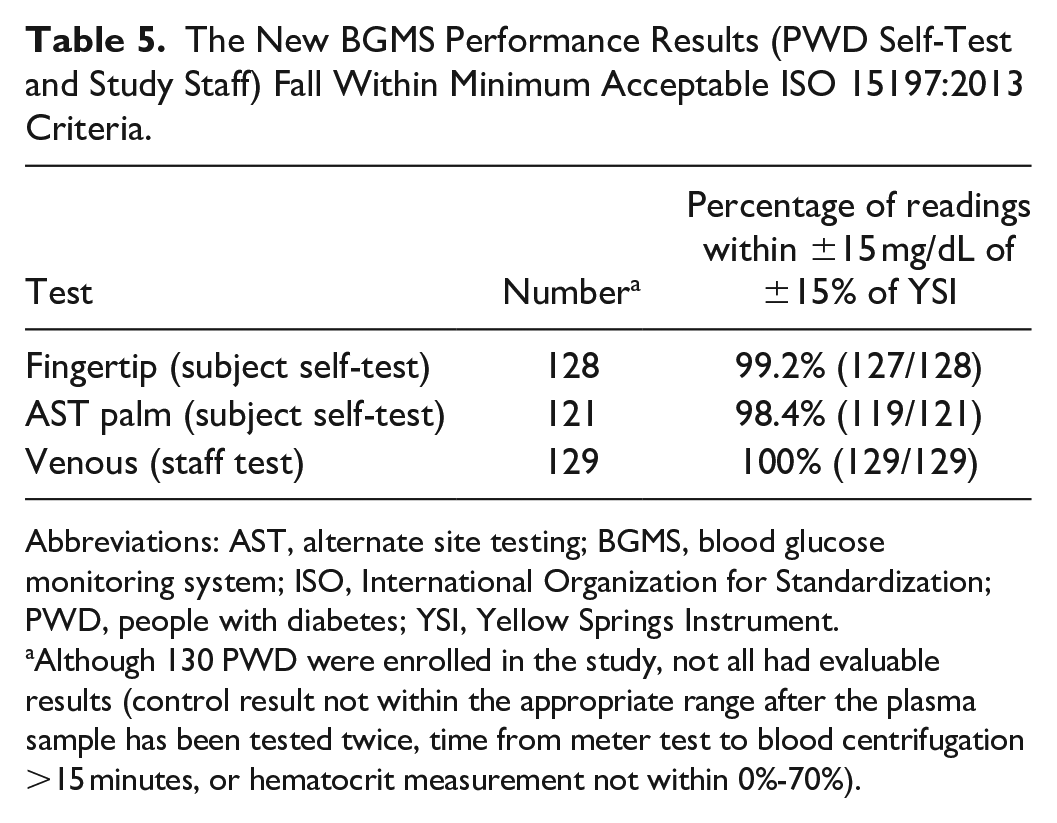
Abbreviations: AST, alternate site testing; BGMS, blood glucose monitoring system; ISO, International Organization for Standardization; PWD, people with diabetes; YSI, Yellow Springs Instrument.
Although 130 PWD were enrolled in the study, not all had evaluable results (control result not within the appropriate range after the plasma sample has been tested twice, time from meter test to blood centrifugation >15 minutes, or hematocrit measurement not within 0%-70%).
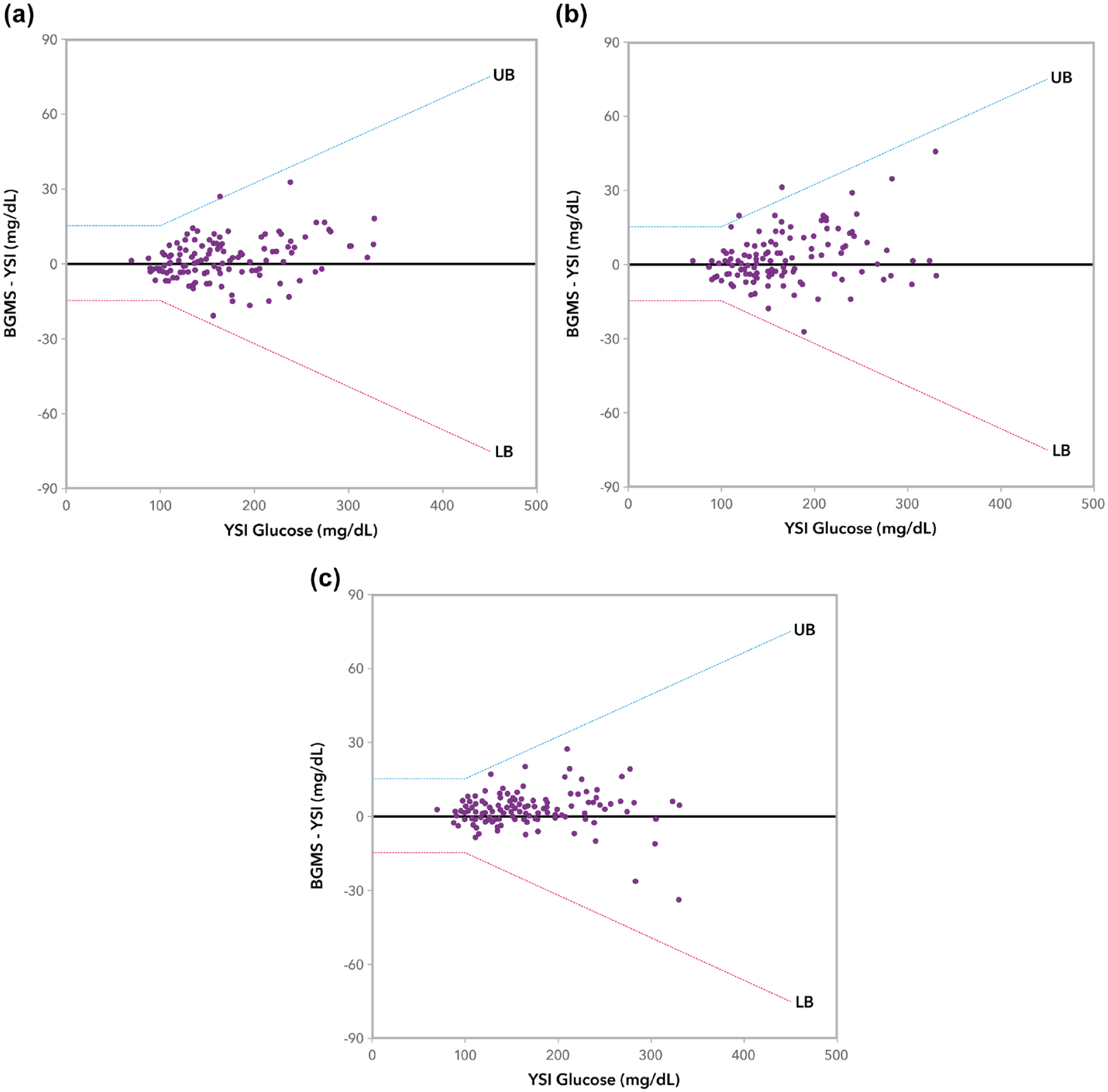
Modified Bland-Altman plot of the new BGMS: (a) fingertip subject self-test, (b) AST palm subject self-test, and (c) venous staff test. The lower and upper bounds (LB and UB) are either ±15 mg/dL (BG <100 mg/dL) or ±15% (BG ≥100 mg/dL). Data below or above the LB and UB, respectively, are outliers.
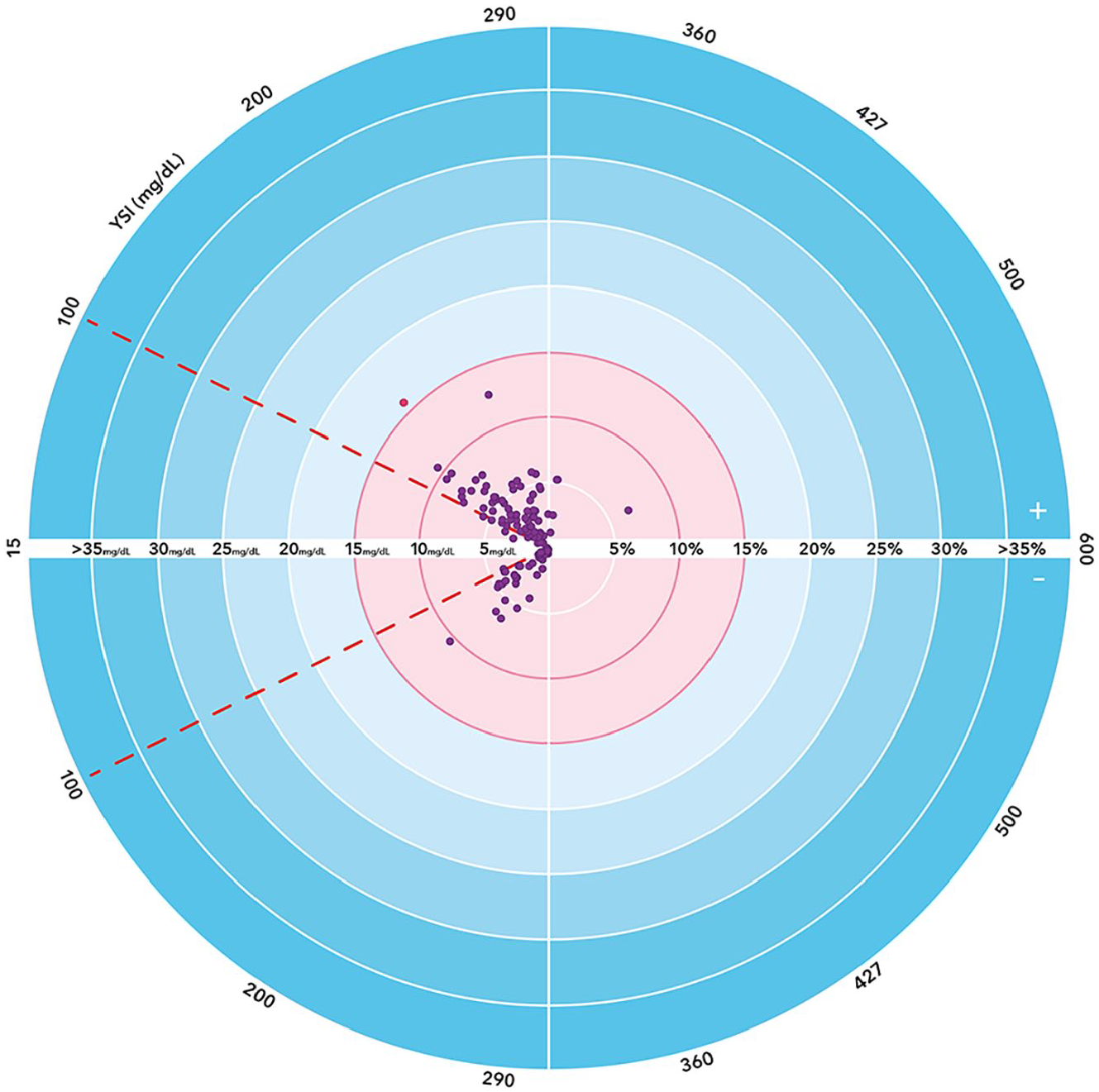
Radar plot: PWD-generated capillary results of the new BGMS. Pink points fail the ISO 15197:2013 accuracy criteria; purple points satisfy the ISO 15197:2013 criteria.
UPQ Results (N = 129).
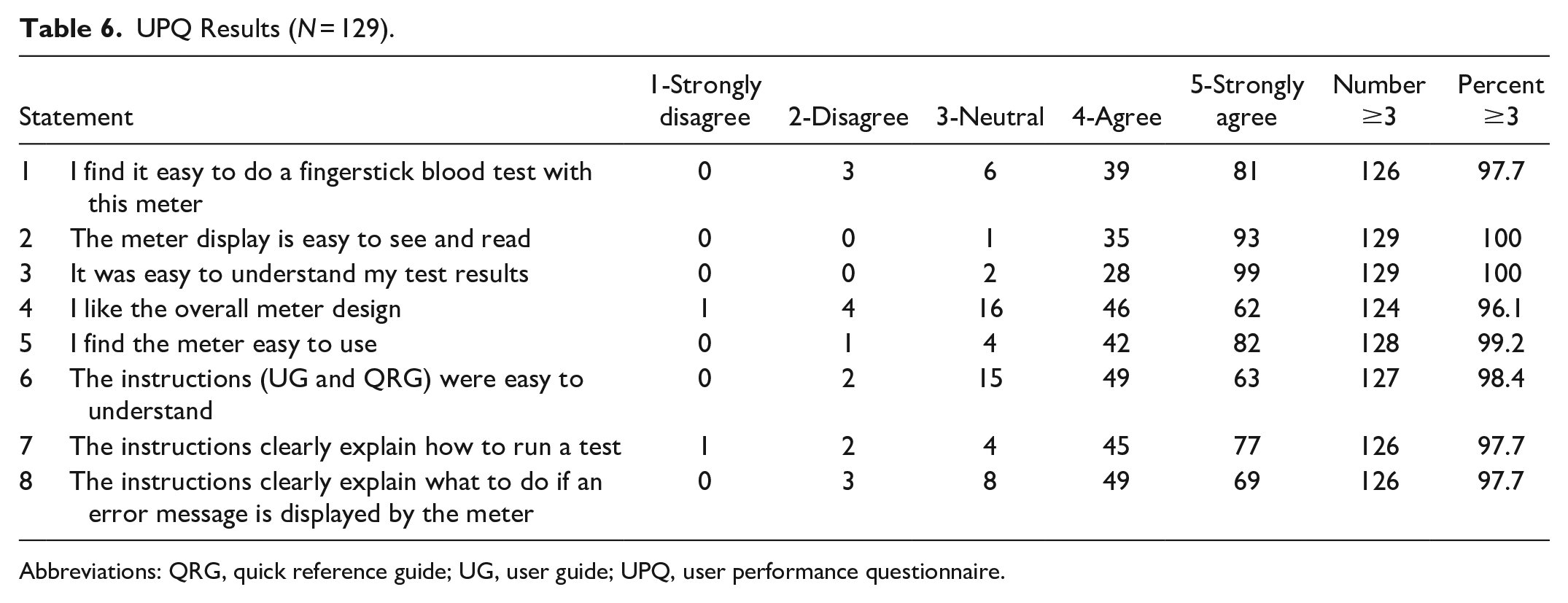
Abbreviations: QRG, quick reference guide; UG, user guide; UPQ, user performance questionnaire.
Discussion
The accurate testing of BGMS is required especially where frequent adjustment of insulin doses is needed to achieve near normal BG control with minimal hypoglycemia. Two studies, one European and one US, have previously investigated a system accuracy evaluation of marketed current-generation BGMS as specified by international ISO standards.6,7 Klonoff et al tested the accuracy of 18 commercially available BGMS approved by the US Food and Drug Administration. Products were sourced from consumer outlets and tested in three triple-blind studies. Only six out of 18 BGMS met the predetermined accuracy standard in all three studies (within 15% of a reference plasma value [for BG ≥100 mg/dL or within 15 mg/dL]). Disappointingly, only five BGMS met the accuracy standard in two studies, three met it in one study, while four did not meet it in any of the three studies. 6 Similarly, Pleus et al carried out a study on the accuracy of 18 current-generation BGMS available in Europe, selected based on market research data. In total, 14 BGMS had ≥95% of results within ±15 mg/dL or ±15%, while three BGMS had ≥95% of results within ±10 mg/dL or ±10% of the results obtained with the comparison method. 7 Results from both evaluations show that not all BGMS approved in Europe and the US fulfill the ISO accuracy requirements reliably.6,7 Patients with diabetes, especially type 1 disease, rely on their BGMS for insulin dose adjustment and for prediction, diagnosis, and treatment of hypoglycemia. Therefore, the accuracy and precision of a reliable BGMS is a critical element of successful diabetes management.
The new BGMS described here is an easy-to-learn and easy-to-use meter, and displays excellent accuracy and performance specifications according to ISO 15197:2013 standards. Results were similar to those described in other published studies of the CONTOUR® family of BGMS.8-15 For trained operator tests, all BG fingertip results were within acceptable accuracy limits by delivering 100% of results within ±15 mg/dL (<100 mg/dL) or ±15% (≥100 mg/dL) of reference results. These results were reflected in the consensus error grid analysis, which showed that 100% of the measurements were within zone A. Furthermore, when data were analyzed using more stringent accuracy criteria of results being within ±10 mg/dL or ±10% of reference results, >99% of results fell within the limits. The UP results from the PWD fingerstick, AST palm, and trained operator venous tests all met the ISO 15197:2013 criteria. PWD were able to learn to use the system from the UG and QRG alone.
The addition of Intermittent, Second-Chance™ sampling is also an important design feature of the new BGMS, as it allows the application of more blood to the same test strip if the first sample is insufficient. This leads to cost savings through reduced wastage of test strips.
Both accuracy and cost savings benefits combined should be considered by primary care providers and HCPs when deciding on which BGMS may be best suited for therapy.
Conclusion
The evaluations here show that the new BGMS exceeds current ISO 15197:2013 standards for both accuracy and UP, and the more stringent accuracy criteria. The new BGMS can be a useful tool in managing glycemic control for PWD, while offering cost savings through reduced wastage of test strips. Due to the high accuracy of the new BGMS, it may be possible to better detect hypoglycemic events and prevent complications as part of the self-management regimen of PWD.
Footnotes
Acknowledgements
Medical writing assistance was provided by Madano and funded by Ascensia Diabetes Care Holdings AG.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JMR is a full-time employee of Ascensia Diabetes Care Holdings AG, Basel, Switzerland, and SP, PS, AWS, PV, and DZ are full-time employees of Ascensia Diabetes Care, Valhalla, NY, USA. LK is an employee of Rainier Clinical Research Center, Renton, WA, USA, and has received research support from Ascensia Diabetes Care, Abbott Diabetes Care, Eli Lilly and Company, Medtronic plc, Novo Nordisk A/S, Pfizer Inc, REMD Biotherapeutics Inc., F. Hoffman-La Roche Ltd, Xeris Pharmaceuticals, Inc, and Zealand Pharma A/S.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These studies were supported by Ascensia Diabetes Care, Parsippany, NJ, USA.
