Abstract
Background:
Despite a marked increase in polypharmacy in patients with diabetes there have been no thorough evaluations of the impact of polypharmacy on the accuracy of any current blood glucose monitoring (BGM) system. This study evaluated the accuracy of a BGM test-strip with respect to polypharmacy using a large clinical registry dataset.
Methods:
Medication profiles were analyzed for 830 subjects (334 with type 1 [T1D] and 496 with type 2 diabetes [T2D]) attending three hospitals. Blood samples were analyzed to determine clinical accuracy of the BGM test-strip compared to a laboratory comparator.
Results:
Across the 830 subjects, 473 different medications (41 diabetes and 432 nondiabetes) were recorded. Patients took on average 6.5 (
Conclusions:
This comprehensive analysis for this specific test-strip platform demonstrated no evidence of interference and robust clinical accuracy of this test strip, irrespective of the prescription medication status of patients with diabetes.
Introduction
The burden of polypharmacy (the usage of multiple medicines for one or more conditions) is a growing concern for the wider population and for people living with diabetes. A multicenter population-based study showed a marked increase in medication usage and polypharmacy in those aged over 65 in the United Kingdom in the last 20 years, with the number of people taking five or more medicines quadrupling from 12% to 49%. 1 An analysis of the burden imposed by polypharmacy in people with diabetes mellitus found that people with type 1 (T1D) and type 2 diabetes (T2D) were prescribed on average five (T1D) or eight (T2D) different drug compounds per day. 2 Despite this rising number and variety of medications, data on potential interference of home blood glucose monitoring (BGM) and continuous glucose monitoring (CGM) systems with even the most commonly prescribed medications has not been extensively evaluated. In terms of BGM performance, a comprehensive literature review 3 demonstrated a continued focus on a narrow subset of interferents (eg, maltose, dopamine, or mannitol) that had the potential to affect test-strip performance, while a separate in vitro study challenged BGM systems with up to 30 drug substances commonly used in a hospital setting. 4 However, studies on BGM interferents, as mandated by EN ISO 19157:2015 and CLSI EP07 guidance, 5 only advocate evaluation of system performance in the presence of a single substance or single medication but not the impact of medication combinations or multiple interferents and therefore do not reflect the exposure of glucose monitoring systems (BGM or CGM) to real-world clinical circumstances. With respect to recently approved CGM systems, a pilot study in 19 healthy drug-naïve subjects without diabetes concluded there was some evidence of CGM interference, in terms of the accuracy of sensor readings, to a limited range of medicines which included lisinopril, albuterol, acetaminophen, and atenolol. 6 Although multiple factors (eg, environmental, human, physiological) contribute to analytical performance of BGM 7 and CGM 8 systems there remains scant information on the impact of the full range of prescription medications that patients are exposed to on a daily basis in terms of the reliability of their glucose readings. This study endeavors to address this gap in information for a widely available BGM test-strip by leveraging a unique UK hospital-based registry containing the current medication profiles of 830 people with diabetes, combined with the in-clinic strip performance results obtained from blood samples from the same 830 patients, to enable exploration of associations between BGM accuracy and prescription medications.
Patient Registry
The patient registry was launched by the BGM test-strip manufacturer in May 2016. All patients attending one of the manufacturer’s three UK National Health Service (NHS) clinics (The Highland Diabetes Institute, Inverness; The Royal Infirmary of Edinburgh; Birmingham Heartlands Hospital) must first enroll in the registry with any subsequent testing performed under UK (Scotland) Research Ethics Committee approval (10/S1103/2). The registry collects anonymized demographic, medical history, and study participation data electronically within a Medidata RAVE system (Medidata Solutions, NY). Initially, self-reported details on medications taken by each patient are provided to trained facilitators and then verified against the subject’s NHS medical records before being recorded within the registry. This data can therefore be cross-referenced with in-clinic patient test-strip performance data to evaluate any impact of medications on strip performance or other patient factors. If a patient makes a repeat visit to the clinic, they are then asked to restate their current medications and the patient registry revised accordingly.
Clinic Blood Glucose Test-Strip Method
Performance of test-strips, manufactured by LifeScan Inc. (Wayne, PA), is routinely monitored through clinical accuracy assessment of representative production batches. Only clinical data specific to the OneTouch Verio Strip platform was included in this assessment. Inclusion criteria require participants to have completed informed consent, a diagnosis of diabetes, and a hematocrit of 20%-60%. During clinical assessment, the participant’s finger is lanced (finger-stick) by the site staff and a drop of blood applied to up to 18 blood glucose (BG) test-strips. Hematocrit levels are recorded as the percentage volume of red blood cells as a proportion of the total sample volume. Reference glucose values were determined from the centrifuged plasma fraction of a 200 µL capillary blood sample collected by Microvette and analyzed, within 30 minutes, on two separate YSI STAT PLUS BG analyzers (Yellow Springs Instrument Co. Inc., OH). If meeting acceptance criteria, the mean of the duplicate comparator values was used. Participants may have visited a clinic site on multiple occasions and been tested with more than one strip lot per visit, thus the clinical dataset is not entirely composed of unique donors. Each strip lot was assessed on a minimum of 100 separate subjects with all tests being performed by trained staff. The clinical accuracy of each BGM value within the patient registry dataset was evaluated against the EN ISO 15197:2015 definition of accuracy: A minimum of 95% of BGM values to be within ±15 mg/dL (<100 mg/dL) or ±15% (≥100 mg/dL) of comparator. Data were also evaluated by surveillance error grid (SEG).
Methods
Medication and Test-Strip Performance Dataset
Over the assessment period (January 2017 to July 2018) the dataset included 830 evaluable patients. Given that individual patients may have visited a clinic more than once during this period, and on each occasion may have provided a blood sample for accuracy evaluation of several production batches, the number of medications and associated BG reading combinations numbered over 13 000. The maximum number of medications assigned to a patient was considered as visit-specific within this assessment (if subjects took at least one medication from a class, they were assessed against that class). Clinic testing was scheduled during standard daily operating hours and it would reasonably be expected that dosage of medications was as per prescribed therapeutic levels. However, factors such as time of administration, dosage, pharmacokinetic properties, and other patient-specific factors were not controlled with patients routinely recruited directly from the diabetes clinic waiting areas. All medications reported by the patient were first classified as either a diabetes medication or nondiabetes medication and then classified according to their intended physiologic or therapeutic action into 15 medication classifications. Medications were then either individually assessed with respect to test-strip performance or assessed with respect to cohorts of patients taking certain medication classes (eg, antihypertensive, lipid lowering, or antidepressants).
Analysis
Each OneTouch Verio test-strip batch was assessed against the EN ISO 19157:2015 definition of clinical accuracy: A minimum of 95% of all BGM system values must be within ±15 mg/dL of reference at BG values <100 mg/dL or ±15% at BG values ≥100 mg/dL as described earlier. Analyses were performed using SPSS Statistics v21 software (IBM Corp., Armonk, NY) with independent statistician verification.
Results
Subject Demographics
Of the 830 patients, a greater proportion were male (54.7%), with significant variation in age range (15-88 years) with an overall mean of 55.8 years (Table 1). A high proportion of the T2D patients in our cohort (311/496; 62.7%) reported using insulin, compared to typical patients from community or primary care settings, which reflects the fact that hospital-based clinics often have more difficult to manage or complex patients referred to them. Noninsulin users accounted for 193/830 (23.3%) of the cohort. Patients had significant durations of diabetes averaging 19 years with a range of 1-62 years.
Subject Demographics of Patients Tested Using Verio Strips.
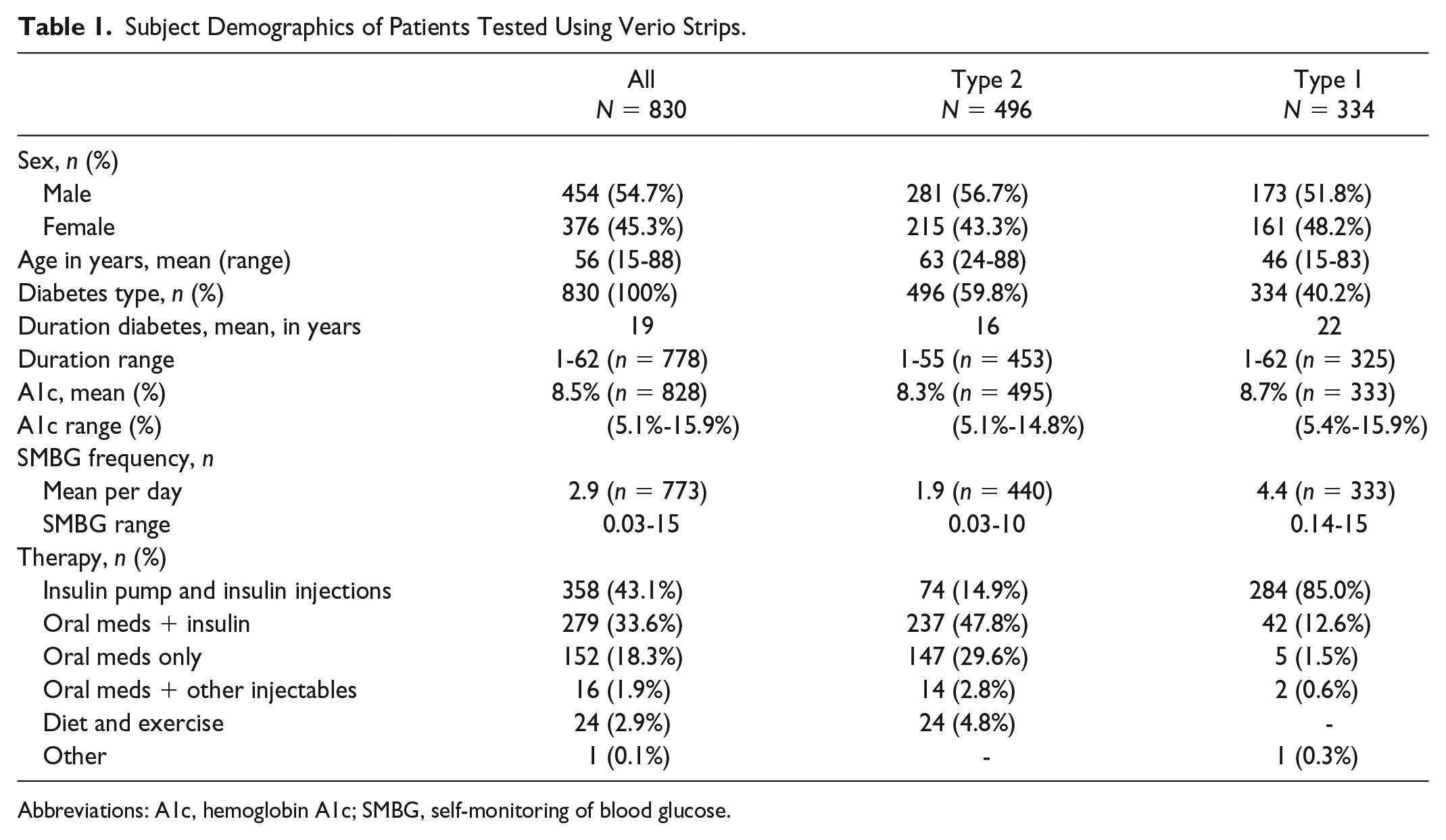
Abbreviations: A1c, hemoglobin A1c; SMBG, self-monitoring of blood glucose.
Medication Classifications
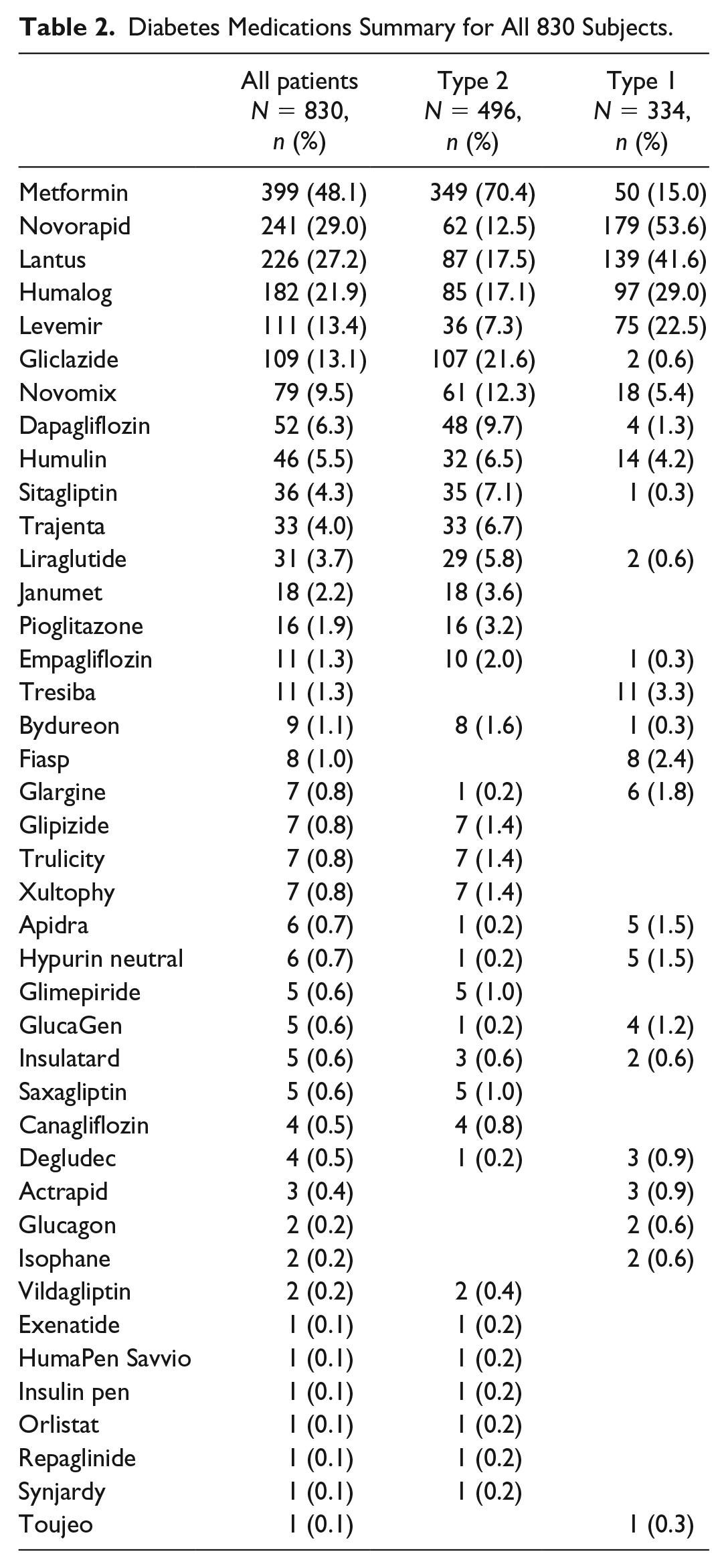
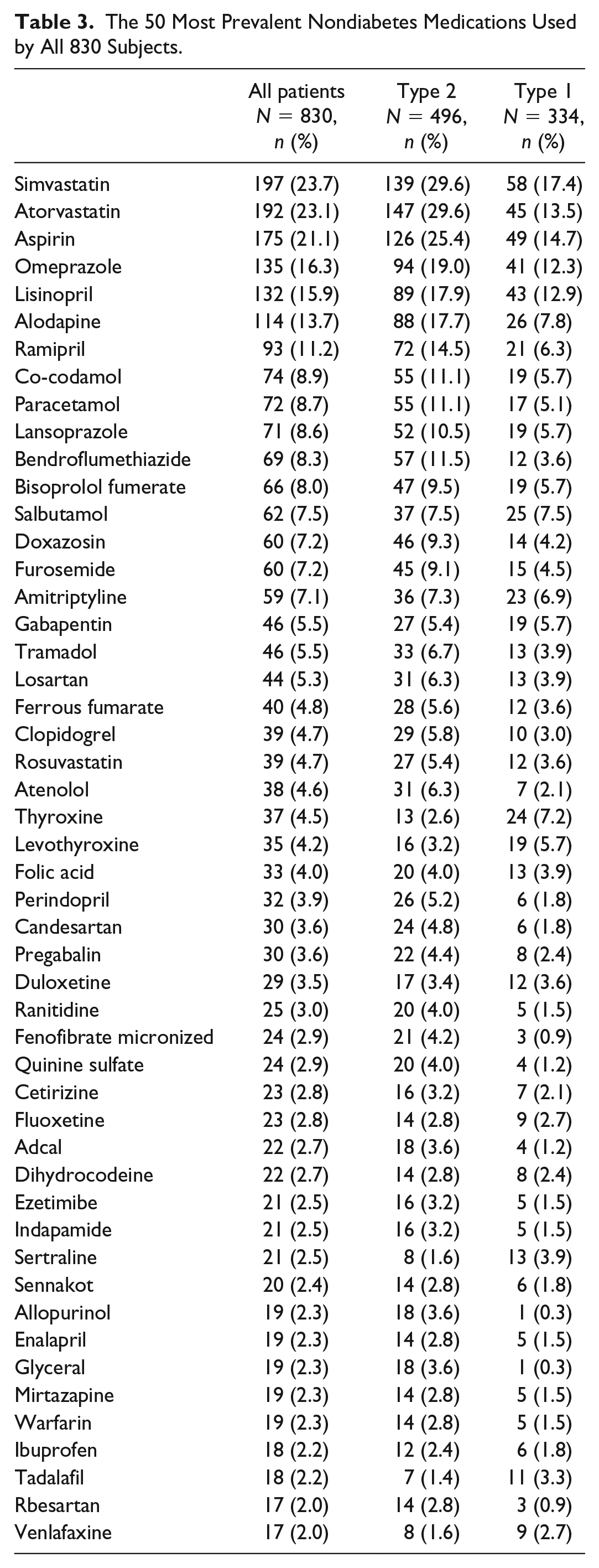
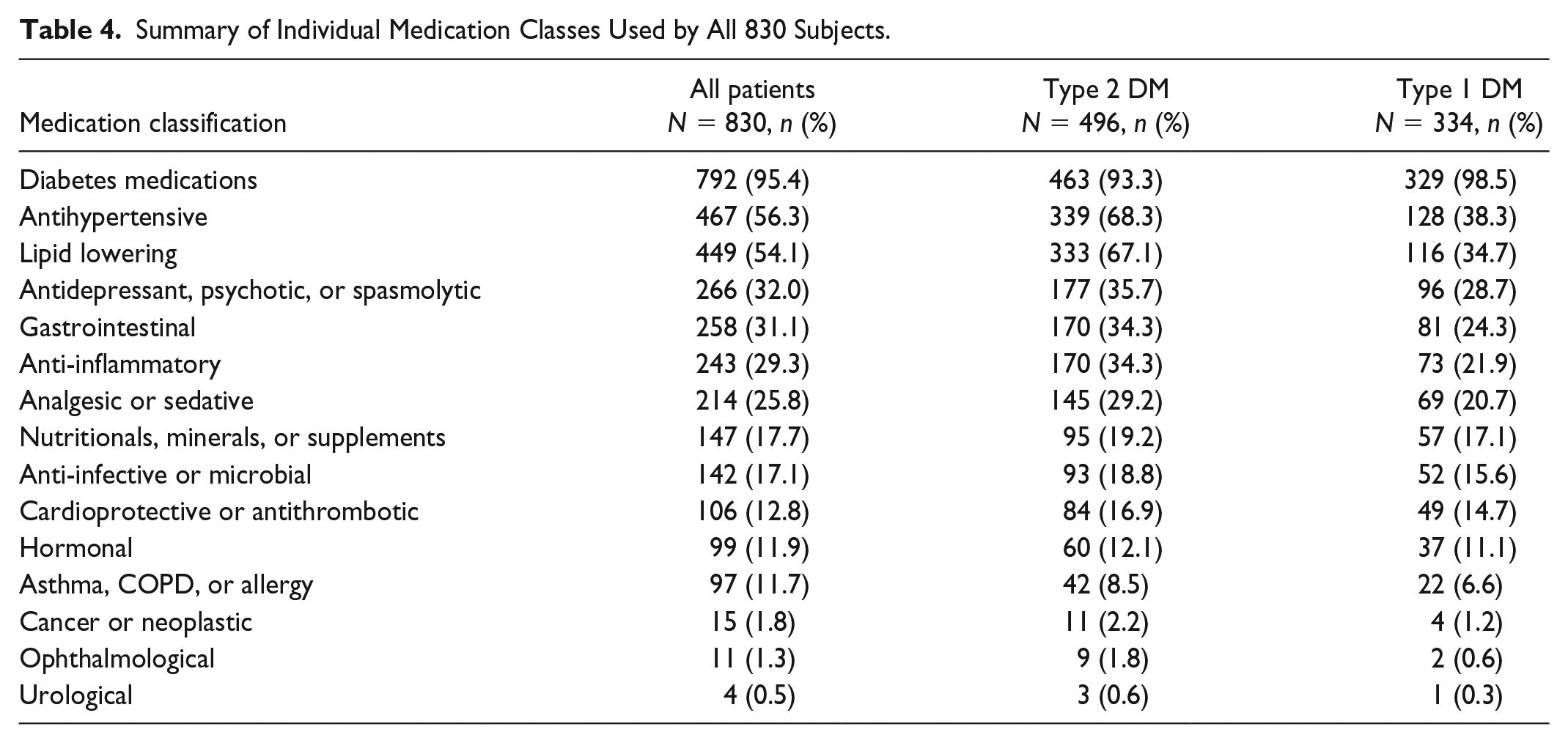
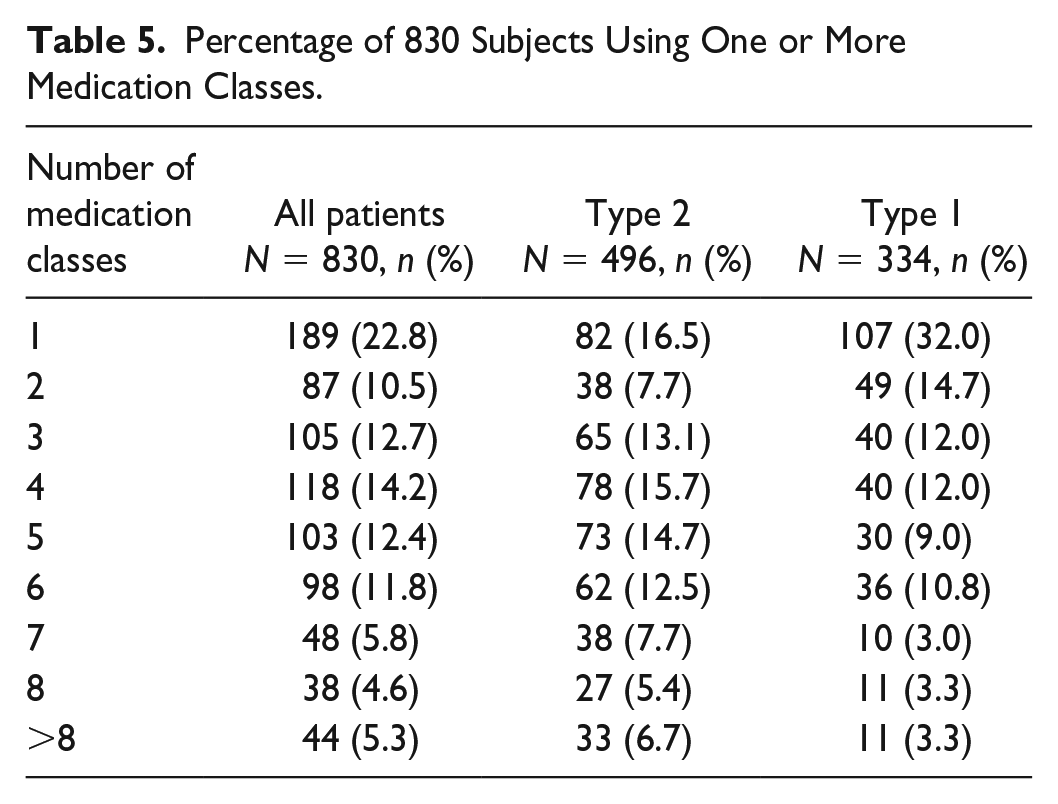
A total of 473 unique medications were recorded across all 830 patients. This included 41 individual diabetes medications (Table 2) and 432 nondiabetes medications. Only the 50 most prevalent nondiabetes medications are listed in Table 3. All 473 medications were then classified according to intended physiologic or therapeutic action into 15 medication classifications. The number of patients taking at least one medication from each of the 15 medication classes is shown in Table 4. The highest percentage of patients took diabetes medications (95.4%), antihypertensive (56.3%), lipid lowering (54.1%), or antidepressants (32.0%). The average number of medication classes taken by all, patients with T1D, and patients with T2D were 4.0, 3.3, and 4.4, respectively. Nearly a quarter of patients (22.8%) used only one class of medication whereas 27.5% used six or more medication classes (Table 5). The average number of individual medications taken by all, patients with T1D, and patients with T2D were 6.5, 5.2, and 7.3, respectively, with a range of 1-23 across all subjects (Figure 1).
Diabetes Medications Summary for All 830 Subjects.
The 50 Most Prevalent Nondiabetes Medications Used by All 830 Subjects.
Summary of Individual Medication Classes Used by All 830 Subjects.
Percentage of 830 Subjects Using One or More Medication Classes.
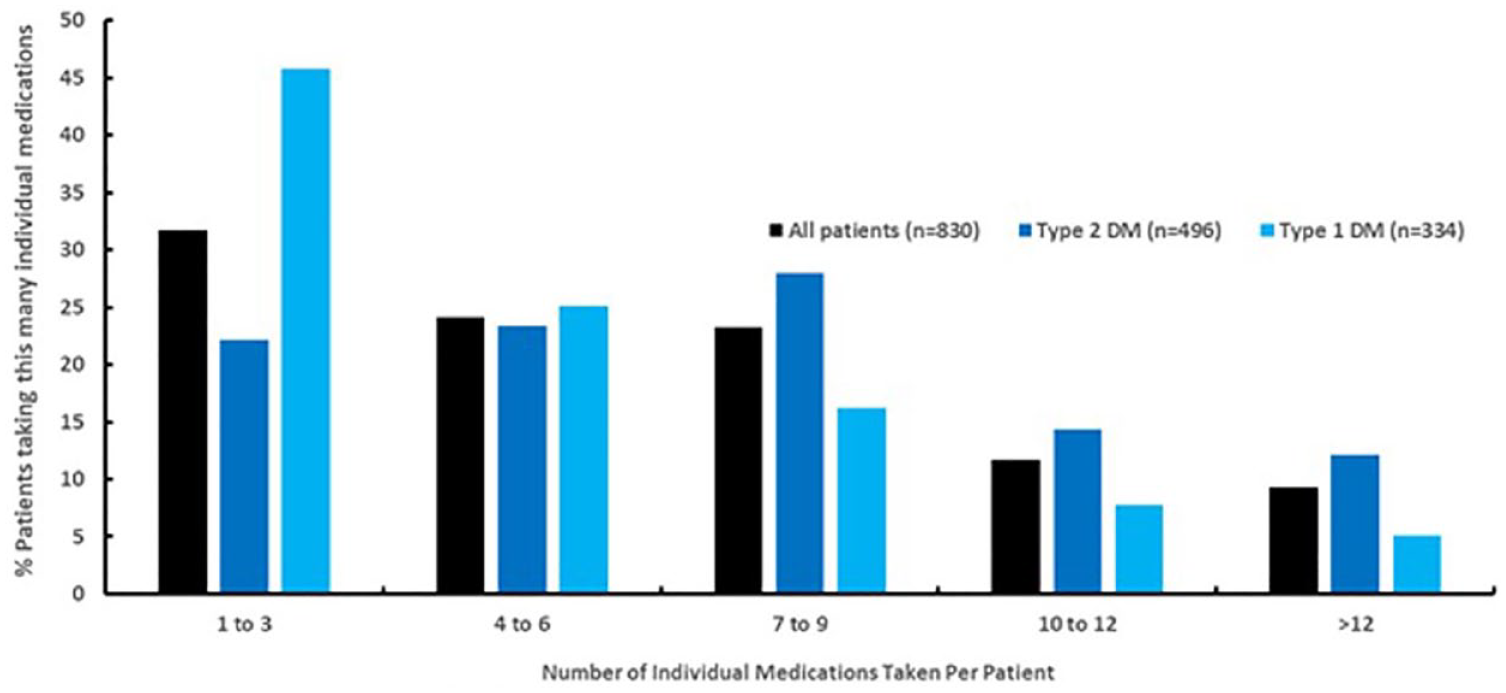
Average number of medications used by all 830 subjects.
Clinical Accuracy of Verio Test-Strip With Respect to Medications
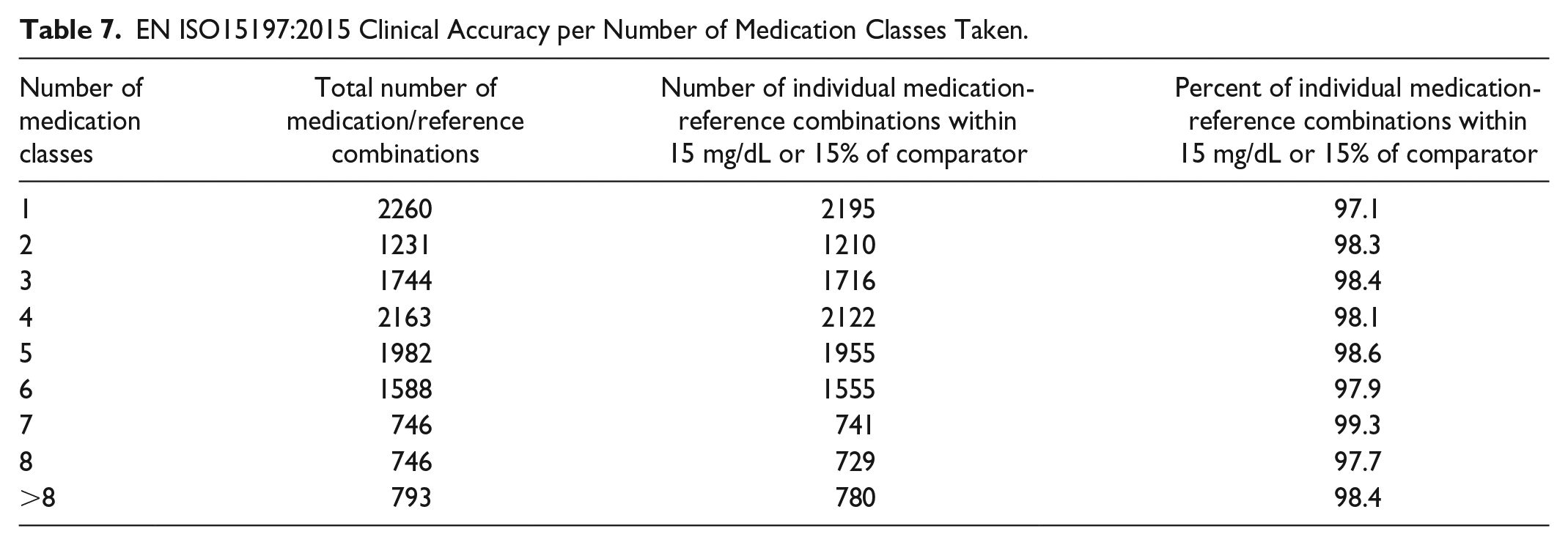
Clinical accuracy was assessed for all medications within each medication classification (Table 6). Each accuracy subset (eg, 8200 readings for antihypertensives) represents strip accuracy data from subjects who confirmed they were taking antihypertensives but such subjects would reasonably also be taking other concomitant medications. Therefore, each subset is defined by the fact that all subjects in that grouping are taking at least that specific medication, as a minimum. For all combinations of individual medication-reference paired readings, at least 95% of glucose values were within ±15 mg/dL (<100 mg/dL) or ±15% of comparator (≥100 mg/dL) with the exception of the urological class (94.5%) with this lower percentage value potentially being influenced by the small sample size. Table 7 lists clinical accuracy categorized by number of medication classes taken by patients. No systematic effect on clinical accuracy was evident with increasing number of medication classes taken. Furthermore, there was no impact on clinical accuracy based upon increasing average number of individual prescription medications taken per subject, categorized from 1 to 4, 5 to 8, 9 to 12, and greater than 12 taken (97.7%, 98.4%, 98.1%, and 98.5%, respectively).
EN ISO15197:2015 Clinical Accuracy for All 15 Medication Classes.
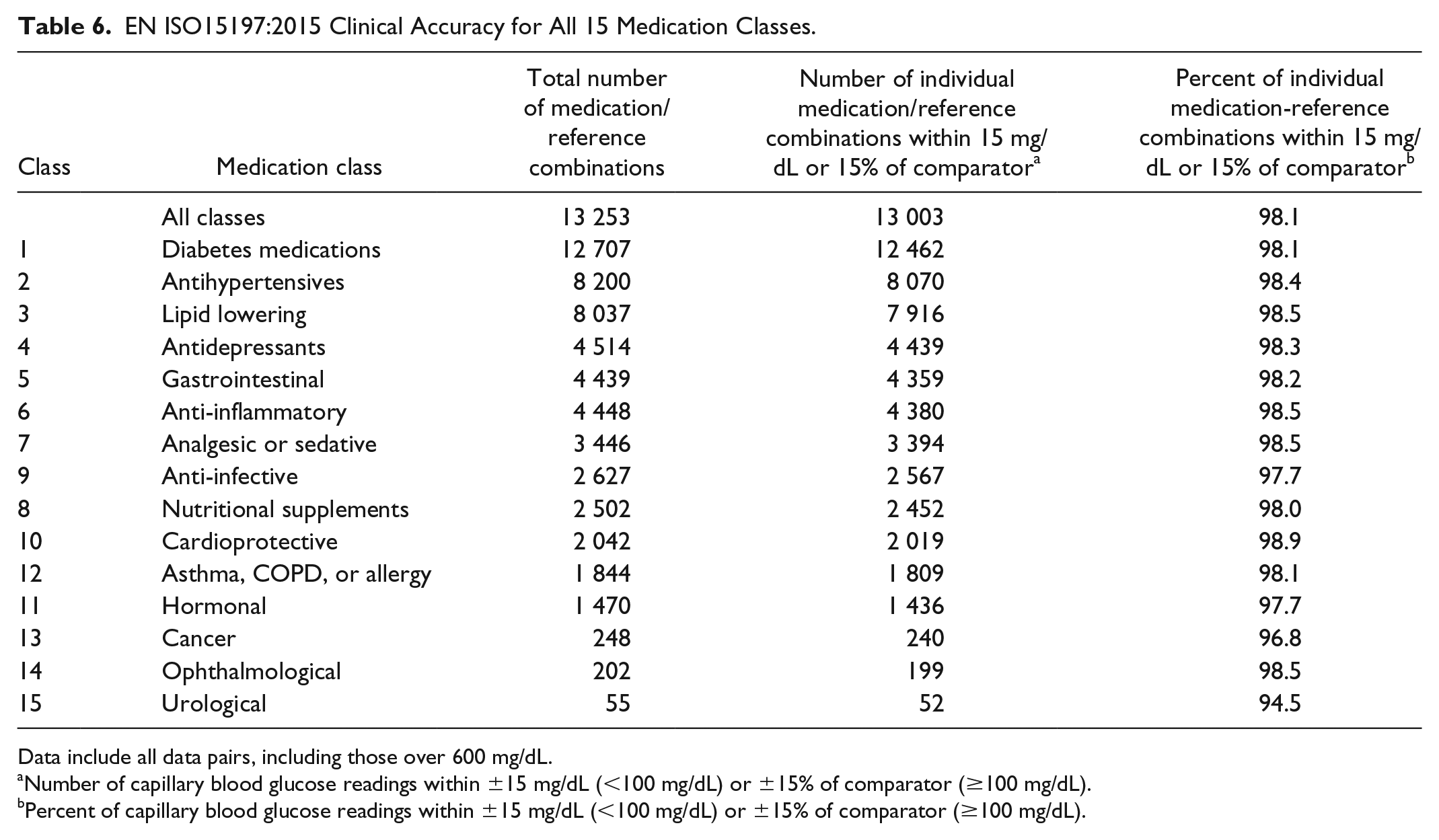
Data include all data pairs, including those over 600 mg/dL.
Number of capillary blood glucose readings within ±15 mg/dL (<100 mg/dL) or ±15% of comparator (≥100 mg/dL).
Percent of capillary blood glucose readings within ±15 mg/dL (<100 mg/dL) or ±15% of comparator (≥100 mg/dL).
EN ISO15197:2015 Clinical Accuracy per Number of Medication Classes Taken.
Evaluation of Clinical Accuracy by Surveillance Error Grid
The SEG classifies data into 15 zones according to an assigned level of risk.
9
The SEG plot for all individual medication-reference paired readings is shown (
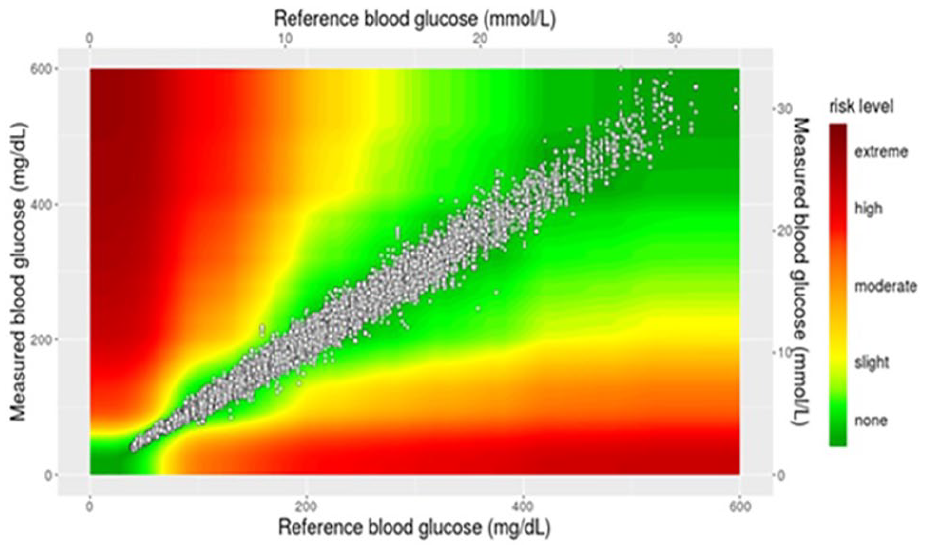
Surveillance error grid plot of all individual paired medication-reference readings from the clinical registry.
Surveillance Error Grid (SEG) Data Categorized for All 15 Medication Classes.
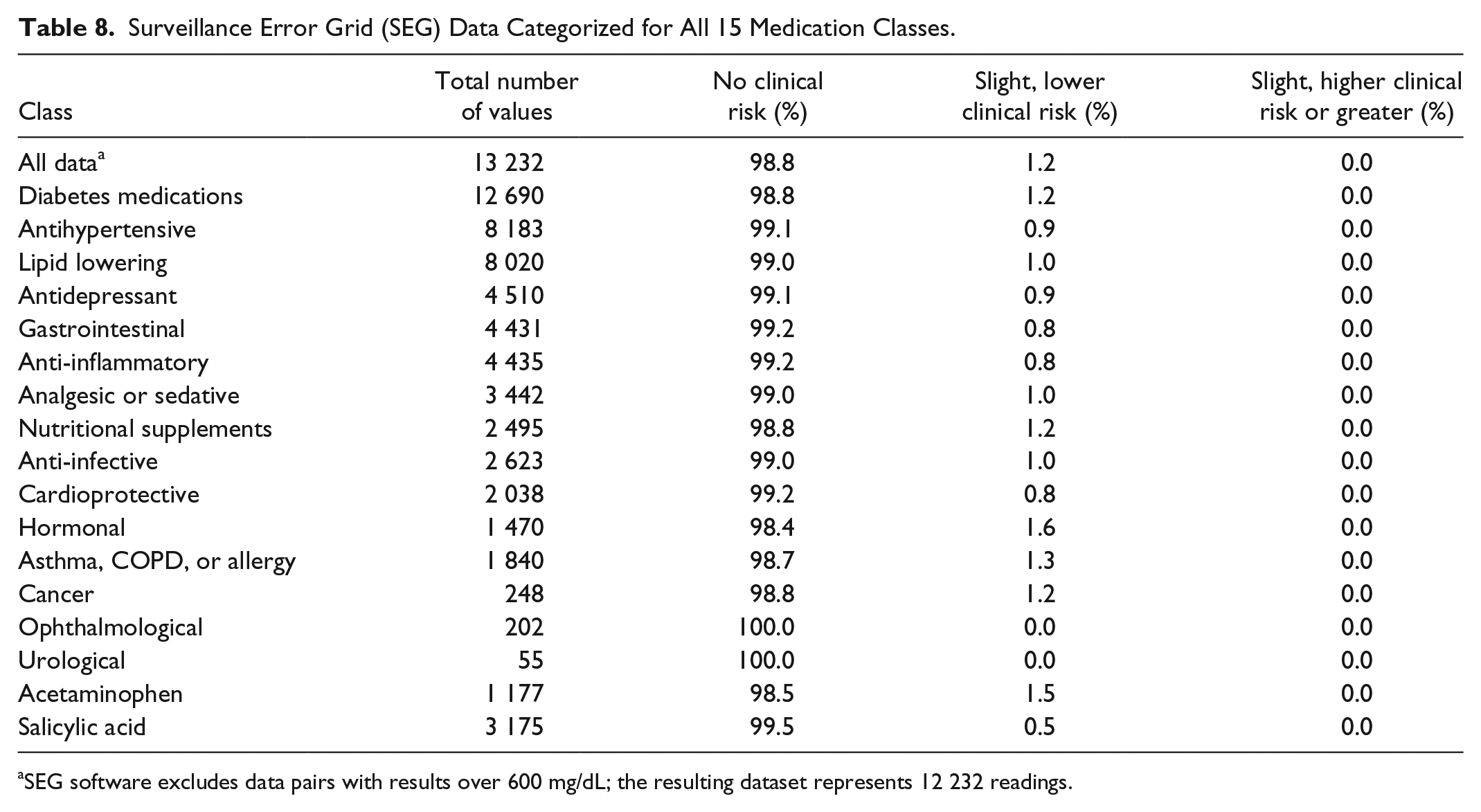
SEG software excludes data pairs with results over 600 mg/dL; the resulting dataset represents 12 232 readings.
While the SEG data indicated that no individual glucose values presented a risk level greater than slight, certain medications may present a greater risk to BG measurement inaccuracy. The test-strip design under investigation is based on electrochemical transduction, thus potentially it may be influenced by the presence of electroactive medications that may directly electrochemically oxidize at the test-strip working electrode. Acetaminophen, a common electroactive medication, was recorded as being taken by 72 subjects (1177 glucose readings) but was not associated (Figure 3) with any systematic effect on the accuracy of Verio test-strips by SEG analysis (no clinical risk: 1159/1177 [98.5%]; slight, lower clinical risk: 18/1177 [1.5%]; slight, higher or greater clinical risk: 0/1177 [0.0%]). A similar outcome was recorded for salicylic acid.
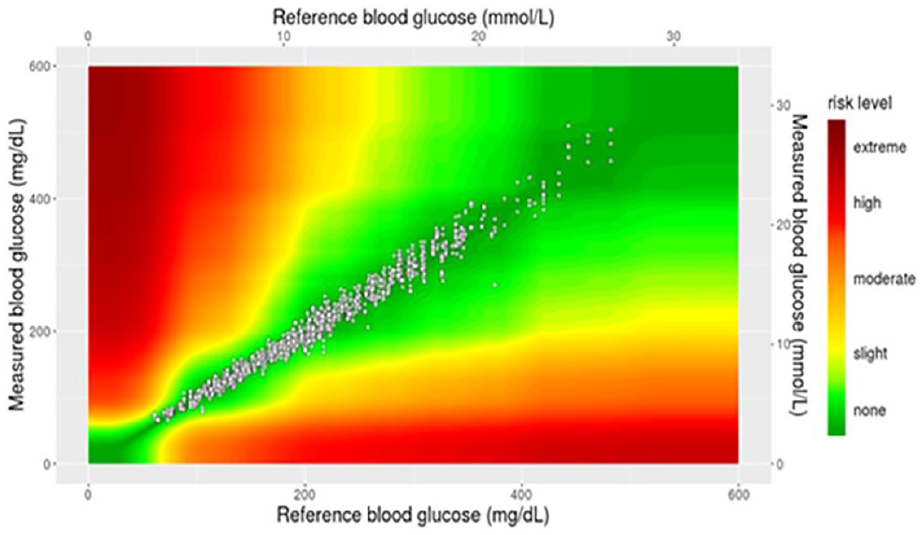
Surveillance error grid plot of all paired medication-reference readings where the patient reported using acetaminophen.
Discussion
Analysis of a comprehensive dataset gathered for the Verio BGM test-strip found that the clinical accuracy of this strip platform was unaffected by 15 distinct classes of medication as defined by EN ISO 15197:2015 accuracy criteria. There was also no apparent association between the clinical accuracy of this test-strip and the average number of individual medications taken by subjects. Furthermore, there was no evidence that any class of medication adversely affected the level of clinical risk to the patient, with respect to SEG analysis. These findings are reassuring for patients for whom polypharmacy is part of their daily routine when managing diabetes in conjunction with other long-term health conditions such as cardiovascular disease, hypertension, or depression.
Our dataset confirmed the findings of Gao et al who showed that polypharmacy has been increasing, with the number of patients over 65 taking five or more medications quadrupling in the last 20 years. 1 Despite focusing on a younger group of subjects (mean age of 56 years) our dataset showed that around 70% of people with diabetes were taking more than four medications per day. It is not only the number of medications which is sobering, but the number of distinct classes of medication taken is symptomatic of the complex health issues faced by people with diabetes; 40% of our dataset were taking more than five distinct medication classes and it is probably not surprising that patients with T2D were taking a higher proportion of medications across all 15 classes than those with T1D. Notably usage of antihypertensive (68.3%; 38.3%), lipid lowering (67.1%; 34.7%), and, to a lesser extent, antidepressants (35.7%; 28.7%) was far higher in patients with T2D than T1D, respectively. Despite being managed in specialty hospital-based clinics, neither our T1D nor T2D patients were in good glycemic control with respect to the guidelines of the American Diabetes Association, which recommends an A1c of <7.0% for nonpregnant adults. 10 In contrast, our T1D and T2D subjects had mean A1c values of 8.3% and 8.7%, respectively. Furthermore, over 75% of our cohort were on some form of insulin therapy. In these circumstances where patients are faced with daily decisions on the dose and/or timing of insulin therapy it is paramount that the results from their BGM are accurate under a range of physiologic conditions.
Prior analysis of the Verio strip platform showed that it was consistently unaffected by hematocrit level (20%-60%) over seven years of product surveillance in 736 production lots released to the market 11 and is also unaffected by 32 commonly cited interferents in routine product testing including tetracycline, uric acid, and ascorbic acid.5,12 However, it would seem timely to expand on the definition of interference given the reality of polypharmacy to which patients are exposed, with so many new medications reaching the market, with a tacit assumption that the accuracy of all BGM systems has been substantiated for the myriad medication combinations patients are prescribed. The scale of our dataset enabled us to address such questions and demonstrate there was no association between increasing numbers of medications (from 1 to >12 per day) or increasing number of medication classes (from one to more than eight) in terms of the accuracy of the Verio strip platform. We felt it was appropriate to explore whether there was a “polypharmacy” effect due to the impact of increasing medication burden on the accuracy of a BG test-strip because ultimately that was the open question that has not been previously addressed in the literature.
The dataset may be considered robust in that it draws from a large patient base (830 subjects), from three distinct clinic locations representing differing socioeconomic backgrounds, varying diabetes type, subject age, duration of diabetes, and medications taken (473 unique medications). All medication classes, when assessed against the EN ISO 19157:2015 clinical accuracy specifications, were clinically accurate, with the exception of the urological medication class. However, it is highly likely this lower number is driven by the small sample size (
To our knowledge few reports have investigated the impact of commonly used medications on CGM performance. In one such study, the Dexcom G4 Platinum and Medtronic Guardian Sof-Sensor were both subject to varying degrees of interference from medications, including lisinopril (antihypertensive), albuterol (antiasthmatic), and acetaminophen (analgesic). 6 The most thorough investigation to date involved the Eversense CGM (Senseonics) where testing was performed in accordance with CSLI guidance (EP07-A2) against 41 substances, with only two substances (tetracycline and mannitol) not meeting the ISO 15197:2013 (EN ISO 15197:2015) accuracy limits within the therapeutic levels of each substance. 13 However, unlike BGM systems that are routinely required to be tested against CSLI guidance, there are no reports of other major CGM manufacturers performing such a robust analysis (against CSLI guidance) with so many common substances. On the contrary, if we take acetaminophen specifically, some CGM systems (Freestyle Libre Pro, Dexcom G4 and G5, Medtronic Enlite, and Guardian systems) were reported to be impacted by interference with acetaminophen.14-18 Notably, Dexcom G6 and Eversense CGMs have published subsequent evidence demonstrating these systems are unaffected by therapeutic levels of acetaminophen.13,19 In the present study, we specifically analyzed the data for 72 subjects taking acetaminophen and found no detrimental impact on the accuracy of the Verio test-strip. Similarly, when we focused solely on data from the 467 subjects (56.3%) taking antihypertensives (such as lisinopril, ramipril, perindopril, and enalapril) or the 97 subjects (11.7%) taking antiasthmatics (such as salbutamol or salmeterol) we saw no deterioration in accuracy of the Verio test-strip.
Given the nature of this type of analysis there are a number of limitations to this study. First, the patients initially self-reported all their current medications when they attended the diabetes clinic and our staff recorded these within the registry. To validate these self-reports of medication usage, our LifeScan clinic staff were able to access patient national health service (NHS) medical records to validate the subjects’ self-reported medication notes. It is possible that errors or omissions within the NHS patient record or the accuracy of the patient record at the time of transcription could affect the integrity of our database. Furthermore, it is possible that patients were taking medications that were not recorded on the database at the time of each clinic visit (eg, over-the-counter medications or supplements that were not prescribed and not within a patient’s NHS medical record) and although we routinely asked patients to advise us of such additional medications or substances we must reasonably assume that this was not always the case. In addition, it is important to recognize that patients often do not adhere to their prescribed medications. Vietri et al reported that approximately 30% of US T2D patients treated using oral medications missed at least one dose within a four-week recall period, indicating that missing doses of oral medication is widespread. 20 Therefore, whether it is diabetes or nondiabetes medications, we have to assume that a number of patients attending our clinic may not have been actively taking their prescribed medications or perhaps not to the extent that therapeutic levels were in their system at the time that blood samples were provided. Clearly, despite the fact that all of these factors compromise the study results it is incumbent upon us to recognize these limitations and to also recognize that in this type of pragmatic, real-world evaluation we have made our best efforts to collate such an extensive dataset and to associate the patient data with verifiable in-clinic performance data.
Conclusion
This comprehensive analysis for this specific test-strip platform demonstrated no evidence of interference and robust clinical accuracy of this test-strip, irrespective of the prescription medication status of patients with diabetes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MG, HC, SP, GS, and SS are all employees of LifeScan Scotland.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by LifeScan Scotland.
