Abstract

With directive 15197, 1 the International Organization for Standardization’s (ISO) directive provides harmonized procedures and guidelines to ensure international quality standards for blood glucose monitors (BGMs). As a continuous quality assessment of BGMs is not only of utmost clinical importance, but also a normative argument for evidence-based decision making for manufacturers and users, we here present a premarketing evaluation of the CE-marked GL49 BGM’s system accuracy (Beurer, Ulm, Germany).
System accuracy was assessed in accordance with the requirements stipulated in ISO 15197 at the Institute für Diabetes Karlsburg, Germany, in July 2020 in compliance with normative and legislative requirements. Reference measurements were performed with an YSI 2300 Stat Plus Analyzer. The blood glucose monitor (by virtue of the declarations of conformity and equivalence also model GL48) and sensor strips from three lots (TD20H514-C0E, TD20H814-C0F, TD20H914-C0D) were supplied by the sponsor. Study personnel collected the capillary blood sample from the subject’s fingertip. Measurements of whole blood glucose were performed for all three lots in duplicate with two BGM using sensor strips from the same vial and compared with duplicate reference plasma values (prior and after BGM testing). Environmental conditions during the study period met the manufacturer’s requirements (20.4°C-22.1°C, 28%-36% rH). The hematocrit was determined reading the packed cell volume using a nomogram alignment chart.
A total of 102 subjects were enlisted in order to obtain 100 evaluable data sets. Blood glucose concentrations were distributed in accordance with stipulated sampling requirements and ranged from 35.5 mg/dL to 625 mg/dL, using native and manipulated blood (by either glycolysis or glucose supplementation). Results obtained with the BGM were found to be in full compliance with the normative acceptance criteria (see Table 1, indicated by italics), showing negligible across-lot variation: between 99.3% and 100% measurements reside within ±15 mg/dL / ±15% of the reference for glucose concentrations <100 mg/dL and ≥ 100 mg/dL, respectively. All measurements reside within 10 mg/dL / ±10% for glucose concentrations <100 mg/dL. Blood glucose levels above 100 mg/dL were correctly determined within ±10 mg/dL in 88.4%–91.1% (across lot average 89.9%) of measurements. All measurements reside within zone A of the consensus error grid. These results fall in line with previous findings on the performance of GL49 in hypo and hyperglycemic blood glucose ranges. 2
Results of System Accuracy Evaluation of BGM GL48/49.
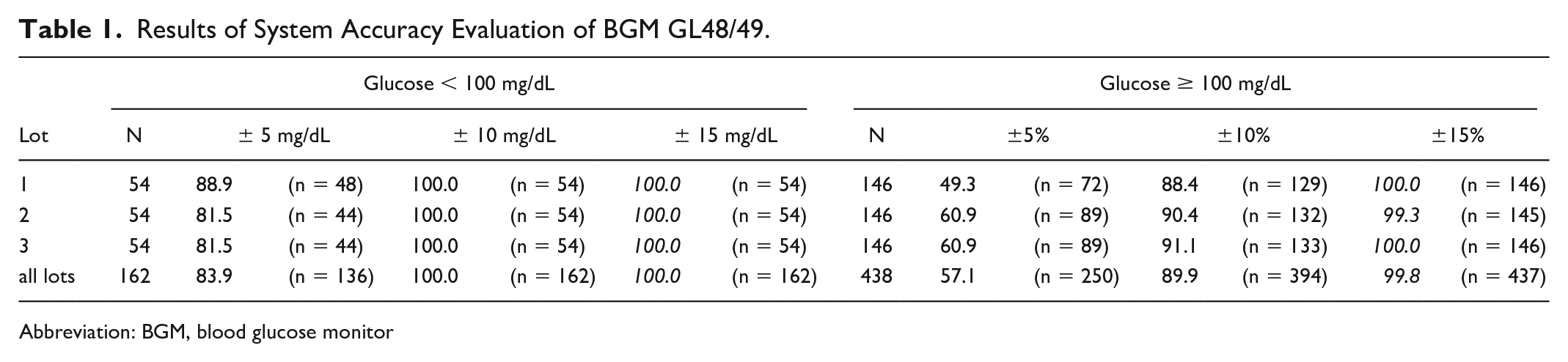
Abbreviation: BGM, blood glucose monitor
Footnotes
Acknowledgements
The authors thank the personnel of the IDK for providing technical help, intellectual input, and feedback in conducting the studies, and preparing the manuscript. All parties were permitted to review and comment on the manuscript; final decision on content was retained by the authors.
Abbreviations
BGM, blood glucose monitor; CE, Conformité Européenne; CEG, consensus error grid; ISO, international organization for standardization.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of the Institute of Diabetes Karlsburg GmbH, Germany, which carries out studies evaluating blood glucose meter systems on behalf of various clients.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Beurer GmbH, Ulm, Germany.
