Abstract

One of the main goals of modern diabetes therapy is to reach and keep normoglycemic blood glucose levels in order to avoid long term complications associated with the disorder by preventing hypo- and hyperglycemic episodes. Blood glucose monitoring systems for personal use that allow a constant monitoring of the metabolism are an integral component of the disease’s medical management, enabling and facilitating fast adjustments of a therapy, which is of particular importance in the administration of a medication such as Insulin.
According to the international classification of diseases ICD-10, hyperglycemia is defined as blood glucose concentrations above the renal threshold for glucose of 180 mg/dl. Hypoglycemia (asymptomatic) is defined as blood glucose concentrations below 70 mg/dl. While normative guidelines for a standardized quality assurance of blood glucose monitors (eg, ISO 15197) generally, but not exclusively, center clinical acceptance criteria on normoglycemic blood glucose concentrations of 100 mg/dl, it is the threshold values of hypo- and hyperglycemia that are increasingly important in a continued performance assessment (eg, FDA guidance 2019, 1 for hospital use).
Along these lines, we analyzed the system accuracy of 4 personal blood glucose monitors (GL44, GL48/49, and GL50 evo form Beurer GmbH, and Gluco-A+ from Microlife Corporation) in accordance with ISO 15197:2015 2 and ADA’s 2019 3 protocols and requirements with emphasis on the upper and lower threshold concentrations.
Duplicate glucose determination of native as well as manipulated capillary blood samples (ie, stored in shaking water bath at 37°C for 3-4 hours to lower blood glucose levels, addition of a 40% glucose solution) were performed using the blood glucose monitor and the glucose oxidase based YSI2300 STAT PLUS (YSI Incorporated, Yellow Springs, Ohio, USA) as well as Cobas c111 (Roche) plasma glucose reference method according to standard protocols outlined in the respective directives. Trueness and precision of the comparison assay were verified using bioanalytical standards and manufacturer issued controls. Based on in-/exclusion criteria framed by the manufacturer and appendix A of ISO 15197, 400 subjects totaling 2400 measurements were included in the analysis.
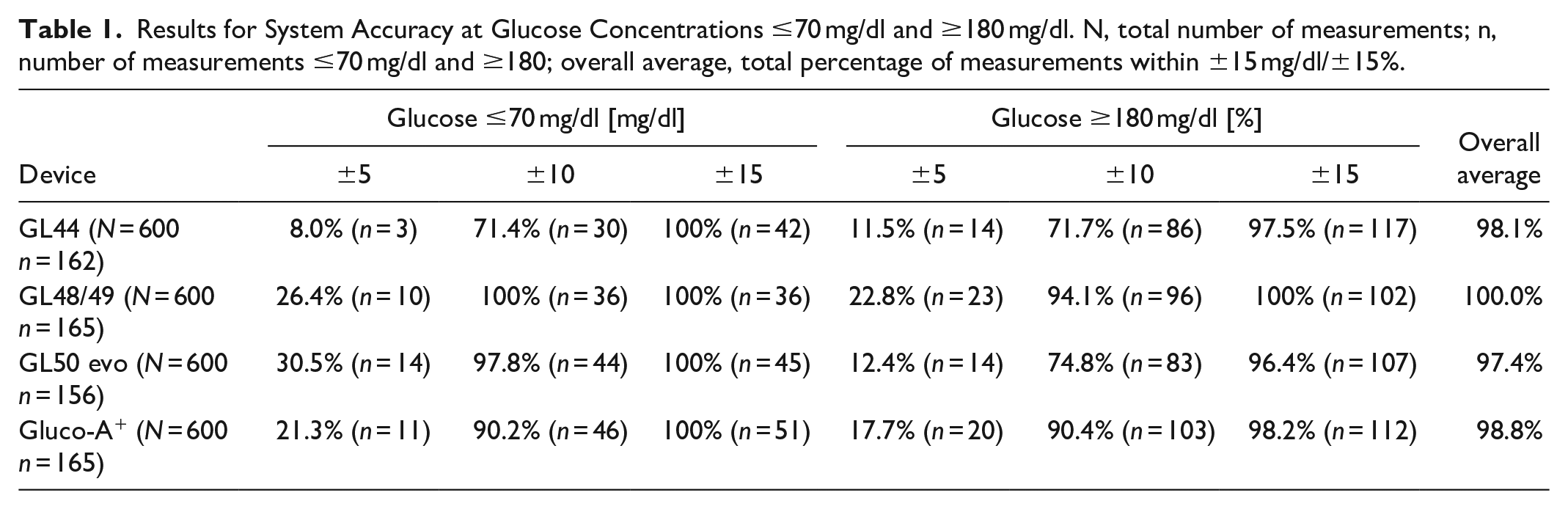
All devices fulfilled the acceptance criteria pursuant to ADA guidelines for extreme values. Between 96.4% and 100% of measurements fell in the range of ±15 mg/dl of reference measurements for glucose concentrations ≤70 mg/dl and ±15% for glucose concentrations ≥180 mg/dl, respectively (Table 1). Between 71.4% and 100% of values fell in the range of ±10 (mg/dl/%), and only 8 to 30.5% of values showed deviations of less than ±5 (mg/dl/%) to reference measurements. All devices performed marginally better in lower concentrations (except GL44 for ±5). Altogether however, the blood glucose monitor GL48/49 performed the most precise measurements across all concentrations.
Results for System Accuracy at Glucose Concentrations ≤70 mg/dl and ≥180 mg/dl. N, total number of measurements; n, number of measurements ≤70 mg/dl and ≥180; overall average, total percentage of measurements within ±15 mg/dl/±15%.
Footnotes
Acknowledgements
Beurer GmbH and Microlife Corporation were permitted to review and comment on the manuscript, final decision on content was retained by the authors.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of the Institut für Diabetes Karlsburg, Germany, which carries out studies evaluating blood glucose meter systems on behalf of various companies.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Beurer GmbH and Microlife AG.
