Abstract
Objective:
To compare healthcare utilization, costs, and incidence of diabetes-specific adverse events (ie, hyperglycemia, diabetic ketoacidosis, and hypoglycemia) in type 1 diabetes adult patients using real-time continuous glucose monitoring (rtCGM) versus traditional blood glucose monitoring (BG).
Methods:
Adult patients (≥18 years old) with type 1 diabetes in a large national administrative claims database between 2013 and 2015 were identified. rtCGM patients with 6-month continuous health plan enrollment and ≥1 pharmacy claim for insulin during pre-index and post-index periods were propensity-score matched with BG patients. Healthcare utilization associated with diabetic adverse events were examined. A difference-in-difference (DID) method was used to compare the change in costs between rtCGM and BG cohorts.
Results:
Six-month medical costs for rtCGM patients (N = 153) increased from pre- to post-index period, while they decreased for matched BG patients (N = 153). DID analysis indicated a $2,807 (P = .062) higher post-index difference in total medical costs for rtCGM patients. Pharmacy costs for both cohorts increased. DID analysis indicated a $1,775 (P < .001) higher post-index difference in pharmacy costs for rtCGM patients. The incidence of hyperglycemia for both cohorts increased minimally from pre- to post-index period. The incidence of hypoglycemia for rtCGM patients decreased, while it increased marginally for BG patients. Inpatient hospitalizations for rtCGM and BG patients increased and decreased marginally, respectively.
Conclusions:
rtCGM users had non-significantly higher pre-post differences in medical costs but significantly higher pre-post differences in pharmacy costs (mostly due to the rtCGM costs themselves) compared to BG users. Changes in adverse events were minimal.
Keywords
Introduction
Personal-use continuous glucose monitoring (CGM) technology, also called real-time CGM (rtCGM), has been revolutionary for patients with insulin-dependent diabetes or type 1 diabetes who are not meeting glycemic targets or experiencing frequent hypoglycemia episodes. 1 Unlike traditional blood glucose monitoring (BG) systems which require patients to perform multiple fingersticks per day for “point-in-time” glucose values, the modern rtCGMs measure glucose levels in real time (as often as 5 minutes, 24 hours per day) and alert patients to the rate and trend of their glucose level to help make better-informed diabetes management decisions.2,3 The most recent generation of rtCGMs does not require fingerstick calibration, which further reduces the burden of use and the expense of necessary ancillaries than the previous generations. 4 The advances in accuracy, reliability, and usability of rtCGMs have demonstrated favorable results on hypoglycemia risk, quality of life, and hospitalization rate in randomized control trials.5-12 The rtCGMs are now being recognized as a critical component of glycemic management in type 1 diabetes patients. 3
The financial burden of rtCGM use in type 1 diabetes patients is high. 3 Several studies have compared the cost-effectiveness of rtCGM versus BG using multiple daily insulin injections (MDI) or continuous subcutaneous insulin infusion (CSII).13-17 For example, a study conducted by Wan et al. 16 reported a total 6-month costs of $11,032 for rtCGM use versus $7,236 for BG use in type 1 diabetes patients using MDI. They also reported the incremental cost-effectiveness ratios (ICERs) ranging from $33,459 per quality-adjusted life year (QALY) gained to $98,108 per QALY for rtCGM. They concluded that rtCGM is cost-effective in real-world use at the willingness-to-pay threshold of $100,000 per QALY. More recently, a meta-analysis conducted by Pease et al. 15 compared the cost-effectiveness of health technologies in adults with type 1 diabetes. The evidence suggested that rtCGM combined with MDI was cost-effective, whereas the cost-effectiveness for combined rtCGM and CSII was less clear. 15 The less cost-effective rtCGM and CSII combination may be impacted by the rtCGM costs and the initial and ongoing costs of CSII use.3,17
Other researchers have conducted retrospective studies to examine the effect of rtCGM on healthcare utilization. Parkin et al. 3 and Gill et al. 18 used health plan administrative data to assess the effect of rtCGM versus BG in commercially insured adult patients with type 1 diabetes. Parkin et al. reported that the pairing of rtCGM and MDI use was associated with improvements in hemoglobin A1C (HbA1C) compared with rtCGM and CSII. Parkin et al. also reported that rtCGM use was associated with reductions in diabetic-specific hospital and ED admissions; however, only diabetic-specific ED admissions were associated with higher costs. Gill et al. continued to explore the associated health care spending using another large repository of health plan administrative data and reported that rtCGM use was associated with lower total health costs, fewer hospital admissions, and lower HbA1C versus the BG use. These studies did not evaluate the cost for rtCGM in their study populations. Our research explores the costs for rtCGM and BG, and other associated health care spending using a large national health plan administrative database. The objective of this study compares the healthcare utilization, costs, and incidence of diabetes-specific adverse events (ie, hyperglycemia, diabetic ketoacidosis, and hypoglycemia) in adult patients with type 1 diabetes using rtCGM versus BG.
Methods
Data Source, Population Selection and Study Period
A retrospective cohort study was conducted using a subset of a large U.S. national administrative claims data from January 1, 2013 to December 31, 2015. The database includes over 12 million current and formal health plan members, which is comprised of patient enrollment, demographics, medical (inpatient and outpatient) and pharmacy claims for fully insured Medicare Advantage Prescription Drug (MAPD) and commercial plan members. Members’ records were de-identified and linked using a unique patient identifier. The database followed the Health Insurance Portability and Accountability Act (HIPPA), and the study was approved by the Institutional Review Board (IRB) from the University of Texas at Austin.
Inclusion criteria specified the need for at least one inpatient or outpatient medical claim for type 1 diabetes as the primary medical diagnosis (Appendix I) and at least one pharmacy claim for insulin. To determine patients who used real-time continuous glucose monitoring devices (rtCGM) or traditional blood glucose monitoring devices (BG), patients with at least one prescription involving rtCGMs (eg, DEXCOM G4 or G5) or BGs (eg, FREESTYLE, ONETOUCH) were identified using the generic drug name variable in the pharmacy data. Patients were also included as rtCGM users if they had CPT code A9276, A9277, or A9728, given the fact that patients might have received the rtCGMs through their health care providers during the study period. 19 The first prescription or service date for rtCGM or BG was defined as the index date. In addition, patients were included if they were at least 18 years old at the time of index date, had at least 6 months of continuous health plan enrollment in both pre-index (baseline) and post-index (follow-up) periods, had at least one pharmacy claim for insulin in both pre-index (baseline) and post-index (follow-up) periods. Other exclusion criteria included: patients with diagnoses of type 2 diabetes, using oral or injectable prescription drug products for type 2 diabetes, patients with CPT code 95250 or 95251 for professional CGMs, 19 patients with diagnoses of gestational diabetes, HIV/AIDS, metastatic cancer, or patients who were dual eligible for Medicare and Medicaid. Patients were divided into 2 groups (rtCGM and BG) according to their index device identified. The final number of rtCGM and BG patients prior to propensity score matching were 153 and 6,991, respectively. Figure 1 illustrates the attrition based on inclusion/exclusion criteria and number of patients included in the analysis.
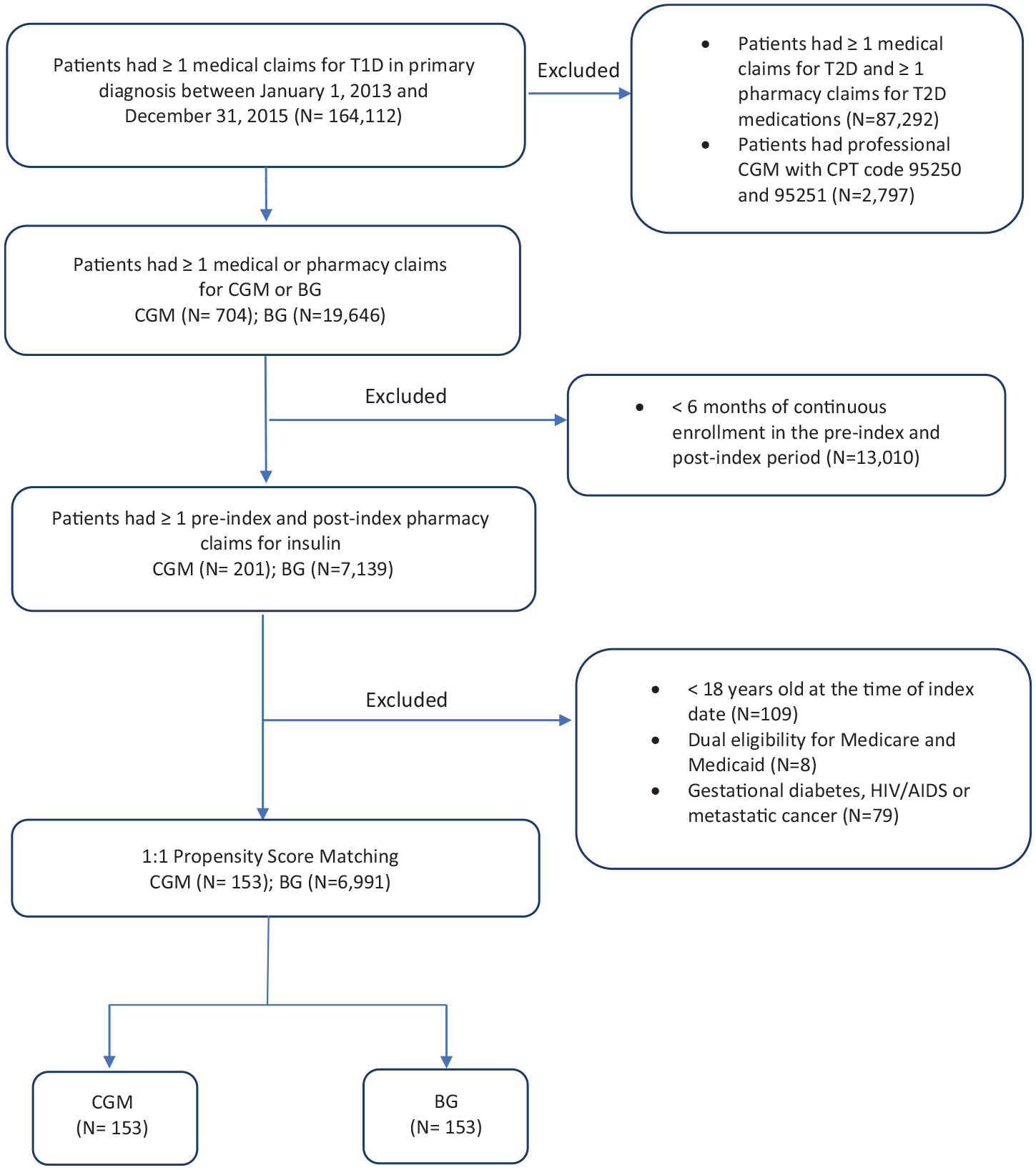
Patient selection flowchart.
Baseline Patient Characteristics and Study Outcomes
Demographic and clinical characteristics were measured during the 6-month baseline period. These variables included age, gender, insurance type, comorbidities, and severity of diabetes complications. Comorbidities were characterized using a composite measure of overall disease severity based on the presence of selected conditions using ICD-9-CM and ICD-10-CM diagnosis codes, which is measured by the Charlson Comorbidity Index (CCI). 20 In addition, the severity of diabetes complications was classified by incorporating 7 categories of diabetes complications, including cardiovascular disease, nephropathy, retinopathy, peripheral vascular disease, stroke, neuropathy, and metabolic diseases, based on the presence of selected conditions using ICD-9-CM and ICD-10-CM diagnosis codes. The overall degree of severity of diabetes complications, defined as the Diabetes Complications Severity Index (DCSI), 21 is the sum of the severity index scores among the 7 disease categories. Patients were followed up to 6 months after the index date. The primary outcomes were health care costs and utilization for the 2 patient groups. Direct health care costs were defined as overall medical and pharmacy costs based on all medical and pharmacy claims for each patient. Medical costs were categorized into non-diabetes (Non-DM), and diabetes (DM) related costs. Diabetes (DM) related costs were further subcategorized into adverse events costs, such as hyperglycemia, diabetic ketoacidosis (DKA), hypoglycemia, and others. Pharmacy costs were categorized into real-time continuous glucose monitoring device (rtCGM), traditional blood glucose monitoring device (BG), insulin-related (eg, insulin, monitoring supplies, ancillaries, test strips, and insulin delivery devices), other diabetic medications (eg, injectable for type 1 diabetes), and other non-diabetic medication costs. Healthcare utilization associated with adverse events was defined as the number of inpatient hospitalizations, emergency department (ED) or outpatient visits involving hyperglycemia, DKA, and hypoglycemia. 22 A claim for an outpatient visit or inpatient hospitalization associated with the adverse events within 5 days was treated as a single event. In addition, inpatient hospitalizations or ED visits associated with diabetes-specific cause were also characterized. A claim for an ED visit resulting in a subsequent inpatient hospitalization on the same day was treated as an inpatient hospitalization.
Statistical Analysis
Descriptive statistics were used to describe patient demographics and clinical characteristics. A 1:1 propensity score method was used to create a matched pair for the rtCGM and BG groups with similar baseline characteristics. The propensity scores were estimated based on logistic regression with covariates of age, gender, comorbidities (CCI score), and insurance type (Medicare versus commercial). The caliper was set as 0.5 of pooled standard deviation of logit of the propensity score. Patient demographics and clinical characteristics between rtCGM and BG were compared before and after matching. A difference-in-difference (DID) method was used to compare the change in costs over time between the rtCGM and BG groups while controlling the impacts of both observed and unobserved confounding. 23 The DID costs were estimated based on unadjusted repeated measures linear regression. The magnitude of the DID coefficient is the post-index cost difference between rtCGM and BG groups. All statistical tests were conducted using SAS software, version 9.4 (SAS Institute Inc. Cary, NC), with two-sided statistical tests at a 0.05 significance level.
Results
From January 1, 2013 through December 31, 2105, a total of 20,350 patients (704 rtCGM and 19,646 BG patients) with at least one medical claim for type 1 diabetes and one medical or pharmacy claim for rtCGM or BG were identified. There were 7,144 patients (153 rtCGM and 6,991 BG patients) in the study after applying the exclusion criteria (Figure 1). Baseline patient characteristics of each group before and after matching are described in Table 1. Before propensity score matching, the mean age (SD) of rtCGM patients was significantly (P < .001) younger [46.9 (14.6) years] compared with BG patients [68.2 (12.4) years]. The mean CCI score (SD) of rtCGM patients was significantly (P < .001) lower [2.16 (1.52)] compared with BG patients [3.79 (2.19)]. The number of BG patients [N = 6,665 (95.2%)] on Medicare Advantage MAPD was significantly higher (P < 0.001) than rtCGM patients [N = 42 (27.5%)]. After propensity score matching, patient characteristics were balanced and not significantly different between the 2 groups. The variables of DCSI score and number of DM complications were included in Table 1 to demonstrate the patient characteristics before and after matching. They were not the covariates used in propensity score matching. There were 153 matched pairs in the study.
Demographic Characteristics.
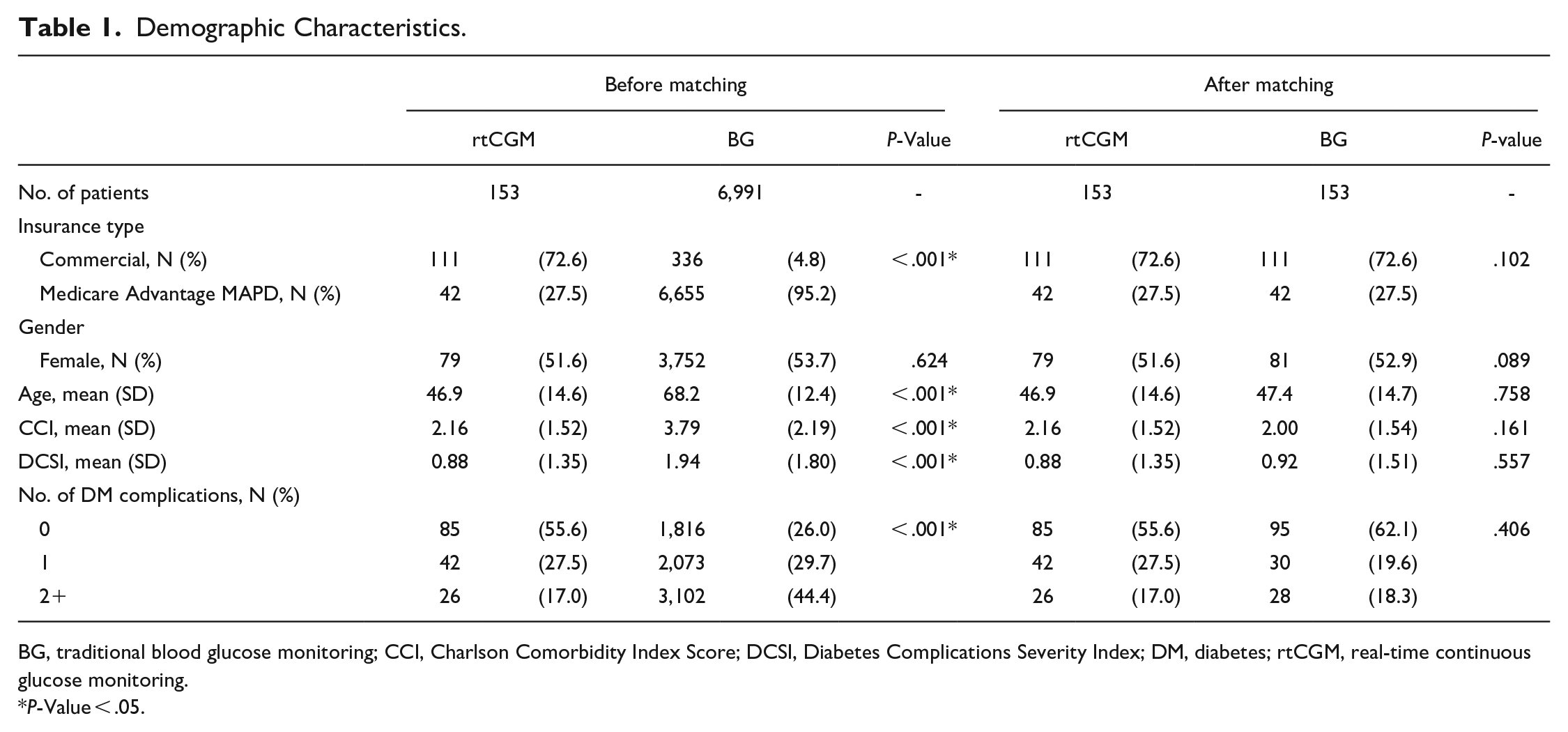
BG, traditional blood glucose monitoring; CCI, Charlson Comorbidity Index Score; DCSI, Diabetes Complications Severity Index; DM, diabetes; rtCGM, real-time continuous glucose monitoring.
P-Value < .05.
Table 2 shows the breakdown of 6-month pre-index and post-index medical/pharmacy costs and the DID cost comparisons for rtCGM and BG patients. The total 6-month medical costs [$ (SD)] for rtCGM patients increased from pre-index to post-index period [$6,416 (15,887) to $7,928 (17,644)], while medical costs decreased for BG patients [$6,691 (13,063) to $5,397 (12,381)]. For DM-related medical costs, including hyperglycemia, DKA, hypoglycemia, and others adverse events, rtCGM patients increased from pre-index to post-index period [$1,358 (2,501) to $3,345 (5,848)], while the costs decreased for BG patients [$1,927 (6,518) to $1,756 (7,466)]. The DID analysis indicated a $2,807 (SE = 1,495.92, df = 304; t = 1.88; P = .062) non-significantly higher post-index difference in total medical and a $2,158 (SE = 722.22, df = 304; t = 2.99; P = .003) significantly higher DM-related medical costs for rtCGM patients. The total pharmacy costs and insulin-related costs for rtCGM patients increased after receiving rtCGM [total pharmacy costs: pre-index $4,235 (5,377) to post-index $7,000 (8,858); all insulin-related costs: pre-index $2,465 (1,234) to post-index $2,681 (1,297)]. The total pharmacy costs and insulin-related costs for BG patients also increased after receiving BG [total pharmacy costs: pre-index $4,322 (8,235) to post-index $5,313 (9,785); insulin-related costs: pre-index $2,514 (1,691) to post-index $3,189 (1,926)]. The DID analyses indicated a $1,775 (SE = 493.88, df = 304; t = 3.59; P < .001) significantly higher post-index difference in total pharmacy costs and a $459 (SE = 173.76; df = 304; t = −2.64; P = .009) significantly lower post-index difference in all insulin-related costs for rtCGM patients. The primary driver of the total pharmacy costs increase in rtCGM patients was due to the cost of rtCGM itself.
Medical/Pharmacy Costs during 6-month Pre-Index and 6-month Post-Index Periods and Difference-in-Difference Cost Comparisons (N = 153 for both groups).
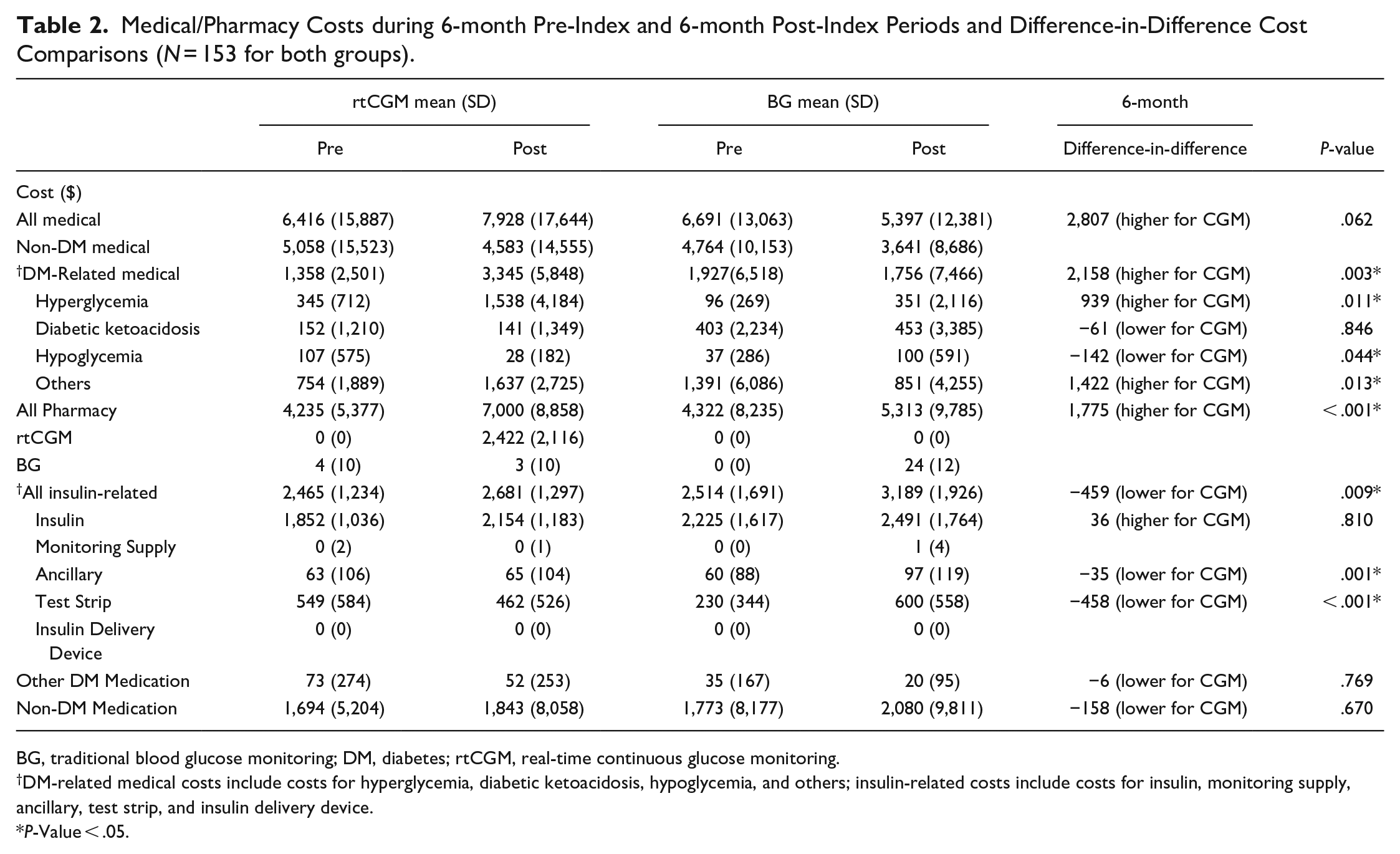
BG, traditional blood glucose monitoring; DM, diabetes; rtCGM, real-time continuous glucose monitoring.
DM-related medical costs include costs for hyperglycemia, diabetic ketoacidosis, hypoglycemia, and others; insulin-related costs include costs for insulin, monitoring supply, ancillary, test strip, and insulin delivery device.
P-Value < .05.
Table 3 shows the average number of adverse events (ie, hyperglycemia, DKA, and hypoglycemia), ED visits, and inpatient hospitalizations associated with diabetes during pre-index and post-index periods. Of the 153 rtCGM and 153 BG patients, DKA and hypoglycemic events were rare in both groups. The number of hyperglycemic events for rtCGM and BG patients increased minimally from pre-index to post-index period [rtCGM: 196 events (mean = 1.28 per patient) to 238 events (mean = 1.56 per patient); BG: 61 events (mean = 0.40 per patient) to 77 events (mean = 0.50 per patient)]. In addition, ED visits and inpatient hospitalizations associated with diabetes were also rare.
Average Number of Adverse Events, Emergency Room Visits, and Inpatient Hospitalizations during 6-month Pre-Index and 6-month Post-Index Periods (N = 153 for Both Groups).
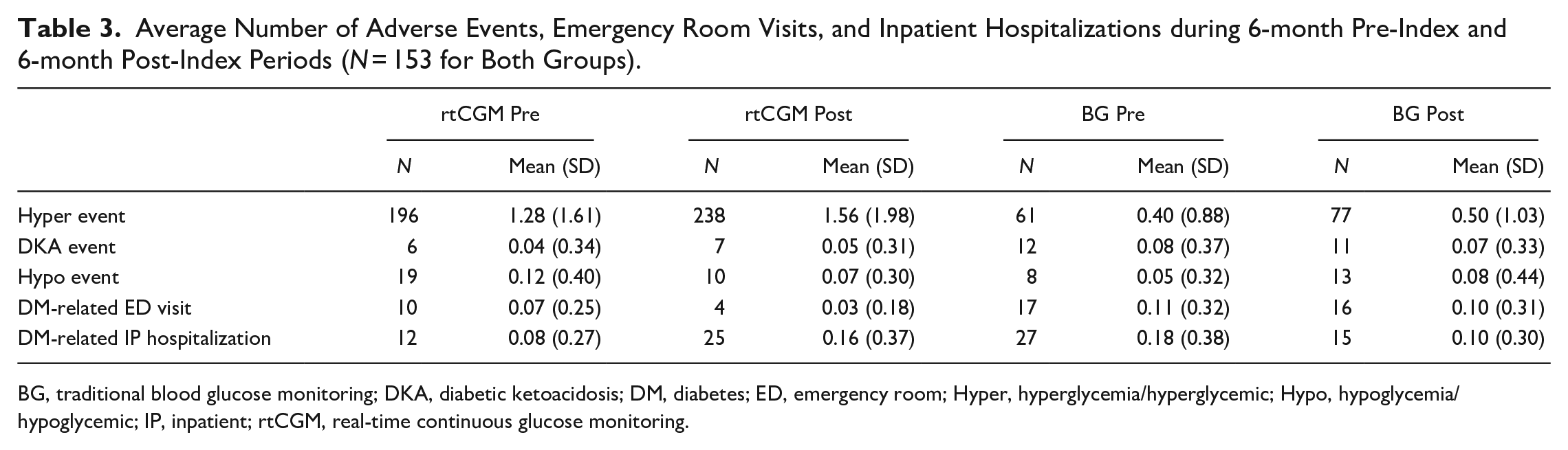
BG, traditional blood glucose monitoring; DKA, diabetic ketoacidosis; DM, diabetes; ED, emergency room; Hyper, hyperglycemia/hyperglycemic; Hypo, hypoglycemia/hypoglycemic; IP, inpatient; rtCGM, real-time continuous glucose monitoring.
Discussion
This study demonstrated when comparing BG patients with similar demographics and clinical characteristics, the use of rtCGM in type 1 diabetes adult patients was associated with higher medical and pharmacy costs. The increased medical costs in the post-index period of rtCGM was partly due to some patients receiving the initial or subsequent rtCGM through their health care providers during their treatment journey. We conducted a sensitivity analysis (data not included in Table 2) by removing the patient insurance claims associated with the rtCGM costs and found that the average medical costs decreased marginally [from $7,928 (17,644) to $7,224 (17,296)]. The medical costs of rtCGM in the post-index period remained higher than the costs in the pre-index period, suggesting that patients who received the rtCGM through their health care providers were not the main driver of the increased medical costs. While the increased costs of insulin and increased utilization of diabetes technology (eg, rtCGM and insulin pumps) were described in the literature,24,25 our study found the increase in total pharmacy costs was primarily due to the utilization of CGM technology. The use of rtCGM was associated with lower insulin-related costs compared with BG use.
Additionally, our study found the incidence of DKA and hypoglycemia were rare, as well as inpatient hospitalizations and ED visits associated with diabetes, in both rtCGM and BG patients. However, when comparing the incidence of hypoglycemia between pre- and post-index period, the incidence for rtCGM patients decreased marginally [from 19 events (mean = 0.12) to 10 events (mean = 0.07)], while the incidence for BG patients increased marginally [from 8 events (mean = 0.05) to 13 events (mean = 0.08)]. The benefit of decreased hypoglycemia risk after patients using rtCGM aligned with the findings reported in randomized control trials.5,8-10 Our study also found when comparing the incidence of hyperglycemia between pre- and post-index period, the incidence for rtCGM and BG patients increased minimally. We acknowledged the fact that patients who received the rtCGM through their health care providers might have a diagnosis code for hyperglycemia, diabetic ketoacidosis, or hypoglycemia during the rtCGM visit. We conducted a sensitivity analysis (data not included in Table 3) by removing the patient medical insurance claims associated with rtCGM and found that there was little to no impact on the incidence of adverse events. The incidence of hyperglycemic events in the rtCGM post-index period decreased minimally [from 238 events (mean = 1.56 per patient) to 218 events (mean = 1.46 per patient)] and was marginally higher than the incidence of hyperglycemic events in the pre-index period [196 events (mean = 1.28 per patient)]. There was no clear evidence to demonstrate the improvements of hyperglycemic incidence after using rtCGM in our study. Furthermore, when comparing the incidence of inpatient hospitalizations associated with diabetes between pre- and post-index period, the incidence for rtCGM and BG patients increased marginally [from 12 events (mean = 0.08) to 25 events (mean = 0.16)] and decreased marginally [from 27 events (mean = 0.18) to 15 events (mean = 0.10)], respectively. Our findings were different than those from studies conducted by Parkin et al. 3 and Gill et al. 18 , which reported fewer all-cause or diabetes-specific inpatient hospitalizations associated with rtCGM use; however, our study reported similar trend as Parkin et al. regarding fewer diabetes-specific ED visits associated with rtCGM use. Parkin et al. reported that rtCGM patients had a lower number of all-cause and diabetes-specific inpatient and ED admissions; however, only the diabetes-specific ED admissions were associated with higher costs. Gill et al. reported that rtCGM patients had fewer all-cause inpatient and ED admissions with lower associated costs. Several distinctive methodological differences of our study compared to other studies by Parkin et al. and Gill et al. included: (1) our study examined the total medical and pharmacy costs, as well as many other cost subcategories including insulin supplies and ancillaries; (2) our study population focused on patients receiving their rtCGM or BG devices through their pharmacy plans; (3) our study used the index period of 6 months before and after the first claim for the initiation of rtCGM or BG for cost estimates. Before matching, the patient groups showed some striking differences, as the BG group was mostly composed of older patients with Medicare insurance. This may be due to differences in insurance coverage decisions.
Limitations
Several limitations of this retrospective claims-based study should be considered when interpreting the results. First, our study had a small sample size after propensity matching and large standard deviations associated with costs. A larger sample size would have provided a more robust assessment of the impact of rtCGM use on healthcare utilization and costs. Second, our study design was limited by the lack of prior diagnostic encounters for type 1 diabetes that would allow us to better characterize the disease duration of the population, given the analysis used a subset of a large US national administrative claims data from 2013 to 2015. While the database contained a linked laboratory sub-database for the HbA1C measures during clinical encounters, not all the patients with the cost data also had lab results during the analysis periods. This is an inherent limitation of the database; hence, we did not include the baseline HbA1C values as a covariate in our analysis. Third, our study had a follow-up period of 6 months which might not be long enough to reflect the real impact of rtCGM use on glycemic control and healthcare utilization. A longer follow-up period would further confirm our findings for the incidence of DKA, hypoglycemia, inpatient hospitalizations, and ED visits associated with diabetes in these patients. Furthermore, like other retrospective analyses, our study inherently included unmeasured variables that could have affected the results, despite the use of propensity score matching. Although our study demonstrated the association of rtCGM use with healthcare utilization and diabetic-related adverse events, causal relationships cannot be established. Our study outcomes might also be impacted by the possibility of selection bias in which patients receiving rtCGM as the intervention might be intrinsically different from those who used BG. Furthermore, we excluded patients who had any claims for professional CGM (CPT code 95250 or 95251) to ensure patients only used personal CGM or rtCGM in the study.1,19 In the study timeframe of data, we acknowledged the fact that some patients might receive their initial or subsequent personal CGM or rtCGM through their health care providers (CPT code A9276, A9277, A9728). We included these patients in our study to accurately capture the medical costs. We could not confirm the specific products that were used during the personal CGM visits since these products were documented in the medical claims as CPT code. Also, it is important to know that there were no intermittently scanned CGM (isCGM), that is, Freestyle Libre, identified in the timeframe of our pharmacy data. Patients who had a prescription for CGMs were all rtCGM users, that is, Dexcom G4 or G5. They were still required to use fingersticks to calibrate their rtCGM devices and were likely to have a prescription for BG in our study timeframe. Finally, our administrative claims database contained only reimbursement costs for medical and pharmacy claims. The pharmacy spending did not include rebates.
Conclusions
Type 1 diabetes remains a major burden for patients and healthcare systems. Real world evidence from this study indicated that rtCGM use by type 1 diabetes patients was associated with higher medical and pharmacy costs. Higher medical costs for rtCGM users were partly due to some patients receiving their rtCGM through their health care providers; however, higher pharmacy costs were primarily driven by the cost of rtCGM. The incidence of DKA, hypoglycemia, ED visits, and inpatient hospitalizations associated with diabetes were rare. Changes in adverse events, ED visits, and inpatient hospitalizations were minimal in both rtCGM and BG groups. A longer follow-up period is needed to further confirm these findings.
Supplemental Material
sj-pdf-1-dst-10.1177_19322968211031519 – Supplemental material for Healthcare Utilization, Costs, and Adverse Events of Real-Time Continuous Glucose Monitoring (rtCGM) versus Traditional Blood Glucose Monitoring (BG) among US Adults with Type 1 Diabetes
Supplemental material, sj-pdf-1-dst-10.1177_19322968211031519 for Healthcare Utilization, Costs, and Adverse Events of Real-Time Continuous Glucose Monitoring (rtCGM) versus Traditional Blood Glucose Monitoring (BG) among US Adults with Type 1 Diabetes by Benjamin Wong, Yalin Deng and Karen L. Rascati in Journal of Diabetes Science and Technology
Footnotes
Author Contributions
Study concept and design: Benjamin Wong, Karen L. Rascati. Acquisition of data: Karen L. Rascati. Analysis and interpretation of data: Benjamin Wong, Yalin Deng, Karen L. Rascati. Drafting of the manuscript: Benjamin Wong, Yalin Deng. Revising it for intellectual content: Benjamin Wong, Yalin Deng, Karen Rascati. Final approval of the completed manuscript: Benjamin Wong, Yalin Deng, Karen L. Rascati.
Abbreviations
A1C, hemoglobin A1C; CCI, Charlson Comorbidity Index; DCSI, Diabetes Complications Severity Index; DID, difference-in-difference; DKA, diabetic ketoacidosis; DM, diabetes mellitus; ED, emergency department; HIPPA, Health Insurance Portability and Accountability Act; Hyper, hyperglycemia/hyperglycemic; Hypo, hypoglycemia/hypoglycemic; IP, inpatient; IRB, Institutional Review Board; MAPD, Medicare Advantage Prescription Drug.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
