Abstract
Objective:
To assess the accuracy of continuous glucose monitoring (CGM) in medical intensive care unit (MICU) patients.
Methods:
A Medtronic Enlite® sensor accuracy was assessed versus capillary blood glucose (CBG) and plasma glucose (PG) using the mean absolute relative difference (MARD), surveillance error grid (SEG) analysis and modified Bland-Altman plots.
Results:
Using CBG as a reference, MARD was 6.6%. Overall, 99.7% of the CGM readings were within the “no risk” zone. No significant differences in accuracy were seen within vasopressor subgroups. Using PG as the reference, MARD was 8.8%. The surveillance error grid analysis showed 95.2% of glucose readings were within the “no risk” zone. There were no device-related adverse events.
Conclusion:
The CGM sensor showed acceptable accuracy in MICU patients, regardless of vasopressor use.
Introduction
Hyperglycemia in critical care is common and associated with adverse outcomes. 1 Intermittent measurement of blood glucose by point-of-care testing is the only clinical method routinely used in the ICU to make adjustments to insulin therapy. Frequent glucose testing ensures the safety of insulin therapy but increases the workload of the nursing staff. 2 Continuous glucose monitoring (CGM) might alleviate the nursing workload. This study aimed to assess the accuracy of glucose levels measured using the Medtronic Enlite® subcutaneous CGM sensor compared with capillary blood glucose (CBG) and plasma glucose (PG) levels in medical intensive care unit (MICU) patients.
Methods
We conducted a prospective, single-arm, open-label study. We included patients aged >18 years, who were admitted to the medical ICU with CBG levels ≥180 mg/dL. Patients with type 1 diabetes, pancreatic diabetes mellitus, diabetic ketoacidosis, and hyperglycemic or hyperosmolar coma were excluded. Patients with a contraindication for CGM insertion or receiving acetaminophen more than 1000 mg per day were also excluded. The study was approved by the Institutional Review Board of Chulalongkorn University, and registered at the Thai Clinical Trial Registry (clinicaltrials.in.th: TCTR20180615001).
CGM sensor (Medtronic, Enlite® sensor) was inserted into subjects’ subcutaneous tissue in the abdominal area. CGM readings were collected in a blinded fashion and CGM calibration was done retrospectively using capillary glucose results every 12 hours.
Capillary blood glucose levels were measured by glucose dehydrogenase PQQ modified by site-directed mutagenesis (Mutant Q-GHD) enzyme (Roche Accu-Chek Performa II). The diagnostic performance was defined as within an acceptable range mandated by ISO15197. An inter-assay variability between each strip lot ranged from 3.1% to 4.4% at blood glucose levels of 30 to 400 mg/dL. The frequency of the capillary glucose tests was every 1 to 2 hours in the first 24 hours and at least every 6 hours thereafter.
Plasma glucose concentrations were collected in sodium fluoride tube and measured by the hexokinase/G-6-PDH method (Architect Abbott). Inter-assay variability was 2.67% to 3.33%. We collected plasma glucose samples every 12 hours and repeat testing was performed if the CBG was <40 or >300 mg/dL. The insulin infusion rate was adjusted according to the local protocol, which is based on capillary glucose results. 3
We collected demographic data, reasons for ICU admission, comorbidities, severity of the illness, and adverse reactions. Glycemic control parameters including CBG at insulin initiation and at the timepoints described in the previous paragraph, coefficient variation, and percentage of glucose values within ranges according to standard recommendations were also recorded. Glucose results were collected until 6 days after ICU admission, or until the subjects were transferred out of the ICU.
All procedures performed in this study were consistent with the ethical standards of the Institutional and Thai National Research Committees and with the 1964 Helsinki declaration and its later amendments. Written informed consent was obtained by either the patient or a family member for all patients recruited to the study.
Statistical Analysis
The sample size was calculated based on the Bland-Altman method, and the precision of the 95% limits of agreement (LOA) according to the formula which estimates the standard error:
The accuracy of CGM was assessed by comparing the Mean Absolute Relative Difference (MARD) between CGM and the CBG and PG measurements. Surveillance error grid (SEG) analysis and Modified Bland-Altman plots were calculated using a web-based tool at http://www.diabetestechnology.org/seg/. MARD of 14 or less and 97% of glucose readings within “no risk” zones in the surveillance error grid are considered acceptable accuracy for CGM use in a critical care setting.4,5 Also, CGM accuracy will be analyzed in vasopressor subgroup.
P values of less than .05 were considered statistically significant. The statistical analysis was conducted by SPSS version 22.
Results
A total of 12 subjects with 353 paired CGM and CBG glucose values and 125 paired CGM and plasma glucose values were included in the analysis. The clinical characteristics of the subjects are summarized in Table 1.
Baseline Characteristics.
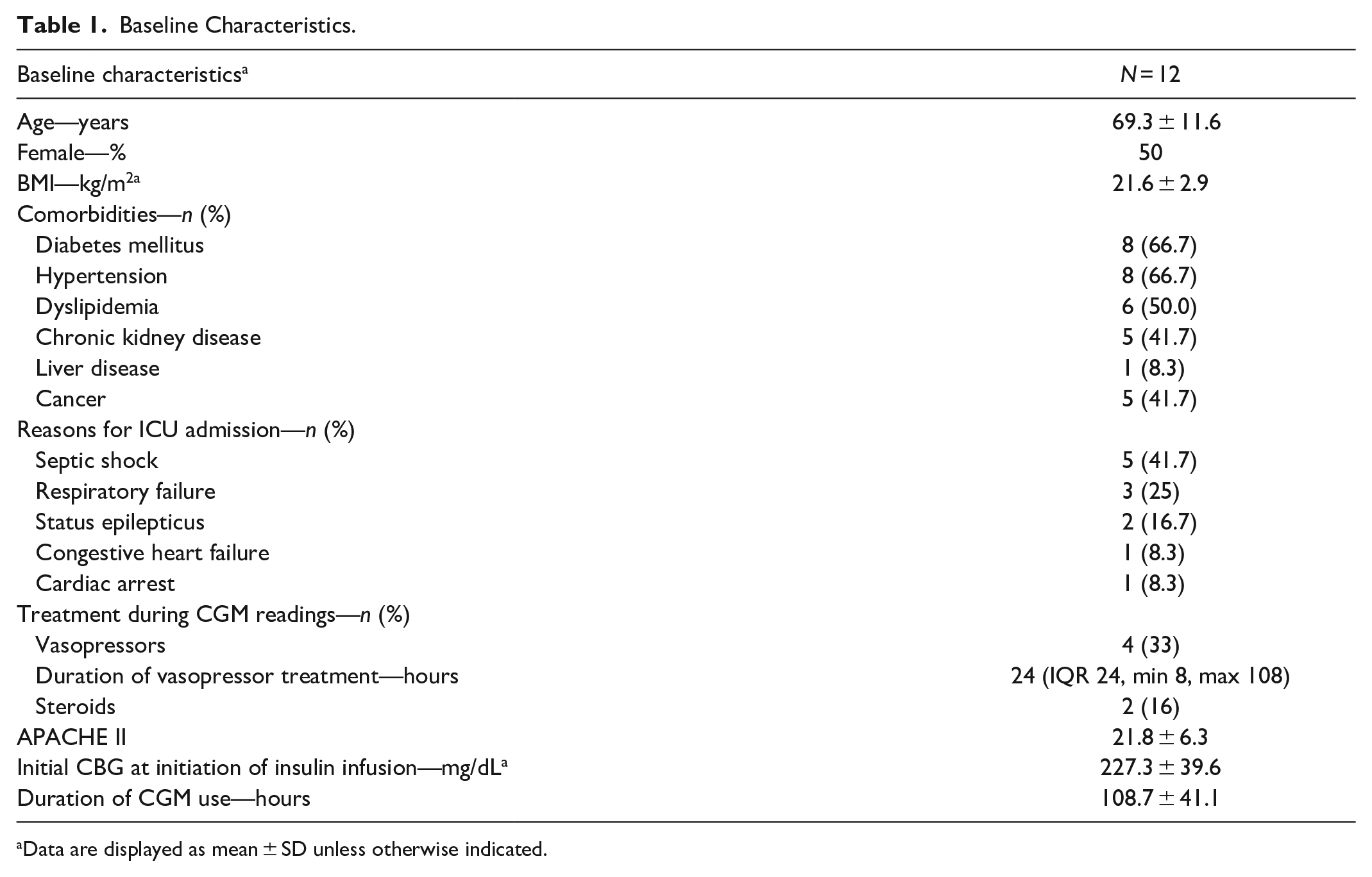
Data are displayed as mean ± SD unless otherwise indicated.
There was no difference between the mean glucose values from CGM, CBG, and PG. The minimum, maximum values, %CV and percentage of glucose reading in various ranges were comparable between CGM values and plasma glucose results (Table 2).
Glucose Values.
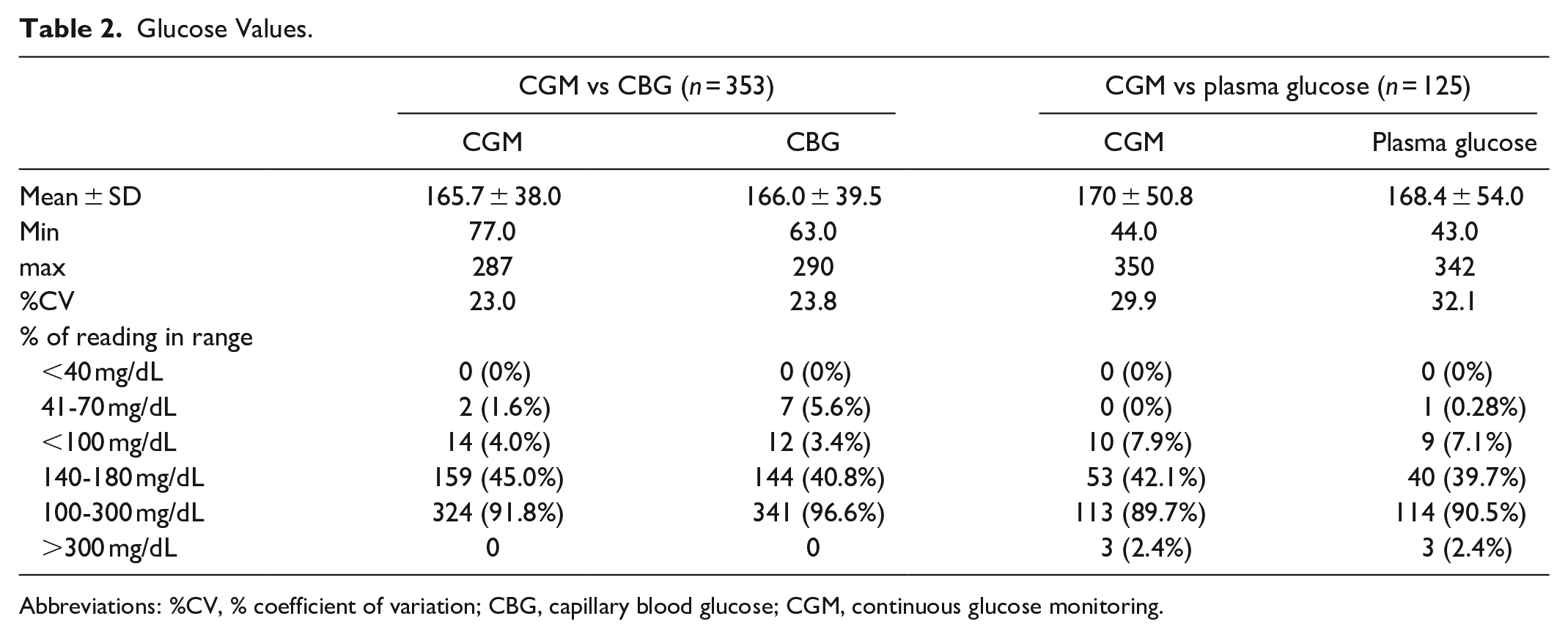
Abbreviations: %CV, % coefficient of variation; CBG, capillary blood glucose; CGM, continuous glucose monitoring.
We did the analysis using CBG as the reference standard. In 353 paired glucose results, MARD was 6.6%. Overall, 99.7% of the CGM readings were within the “no risk” zone. The modified Bland-Altman plot showed 95% of the limit of agreement was −18.4% to 19.2%, with a bias of +0.4% (Figure 1A and B).
CGM accuracy using capillary glucose value as a reference. (A) Modified Bland-Altman plots. (B) SEG.
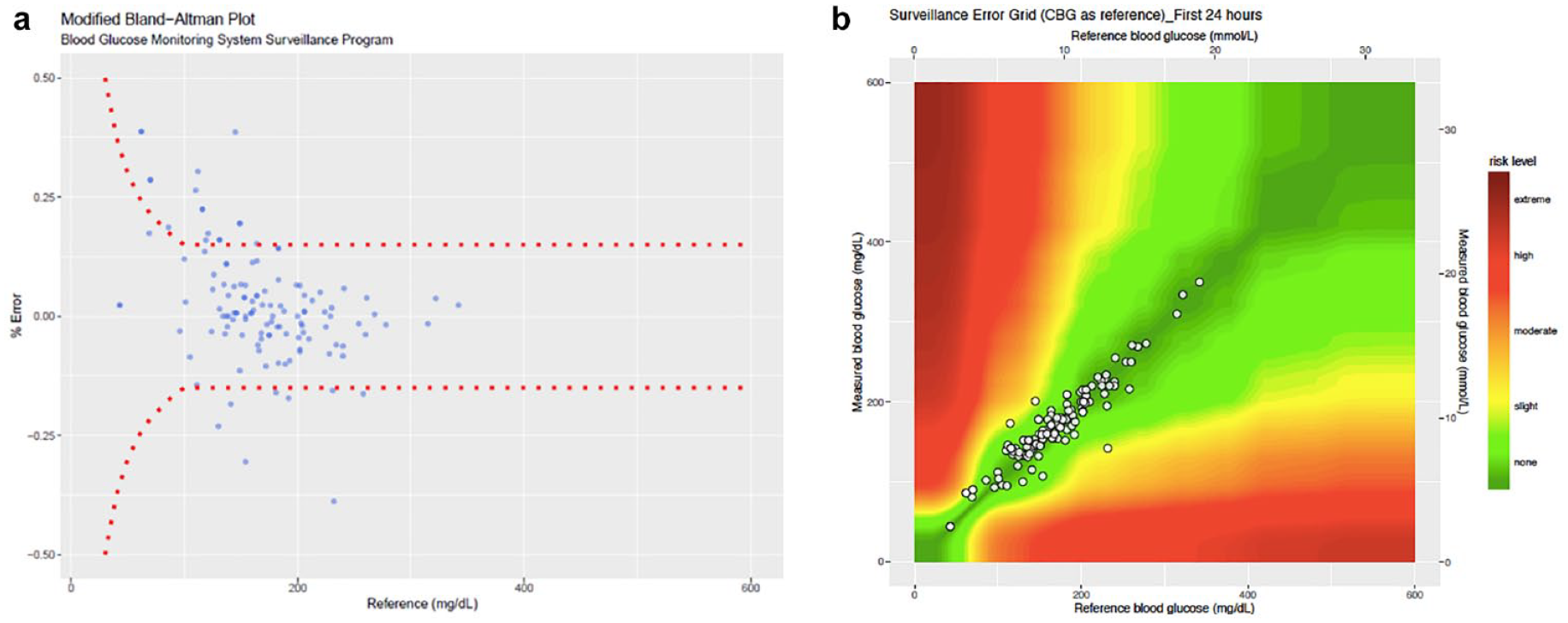
There were 144 paired glucose results obtained during the first 24 hours after CGM insertion, and another 209 paired results after 24 hours. MARD was 7.2% in the first 24 hours and 6.1% after 24 hours of use. During the first 24 hours, all glucose readings lay within the “no risk” zone and 99.5% of readings lay within “no risk” zone after 24 hours (Supplemental Figure 1A and B). Modified Bland-Altman plots showed comparable accuracy between levels the first 24 hours and the period after 24 hours (Supplemental Figure 2A and B). The LOA in the first 24 hours was 19.3% to 20.6%, with a bias of +0.7% and −17.8% to 18.3%, and bias of +0.3% in the period after 24 hours.
We also did the analysis using PG as the reference standard. In 144 paired glucose results, the overall MARD was 8.8%. SEG and modified Bland-Altman plot also showed acceptable accuracy. The surveillance error grid analysis showed 95.2% of glucose readings were within the “no risk” zone. The Modified Bland-Altman plot showed 95% LOA of −22.6% to 28.2%, and a bias of +2.8% (Figure 2A and B).
CGM accuracy using plasma glucose value as a reference. (A) Modified Bland-Altman plots. (B) SEG.
Total of 134 paired glucose measurements were available for assessing a potential effect of vasopressors. MARD was not affected by vasopressor use (MARD 6.1% in the vasopressor group vs MARD of 6.8% in group not using vasopressors, P = .35). The surveillance error grid analysis showed 100% of readings in the “no risk” zone in the vasopressor group. The Modified Bland-Altman plot also showed comparable accuracy between the 2 groups (95%LOA = −15.6%-17.1%, bias +0.8% in vasopressor group vs, 95%LOA of −20.0% to 20.4%, and bias of +0.2% in the non-vasopressor group (Figure 3A and B).
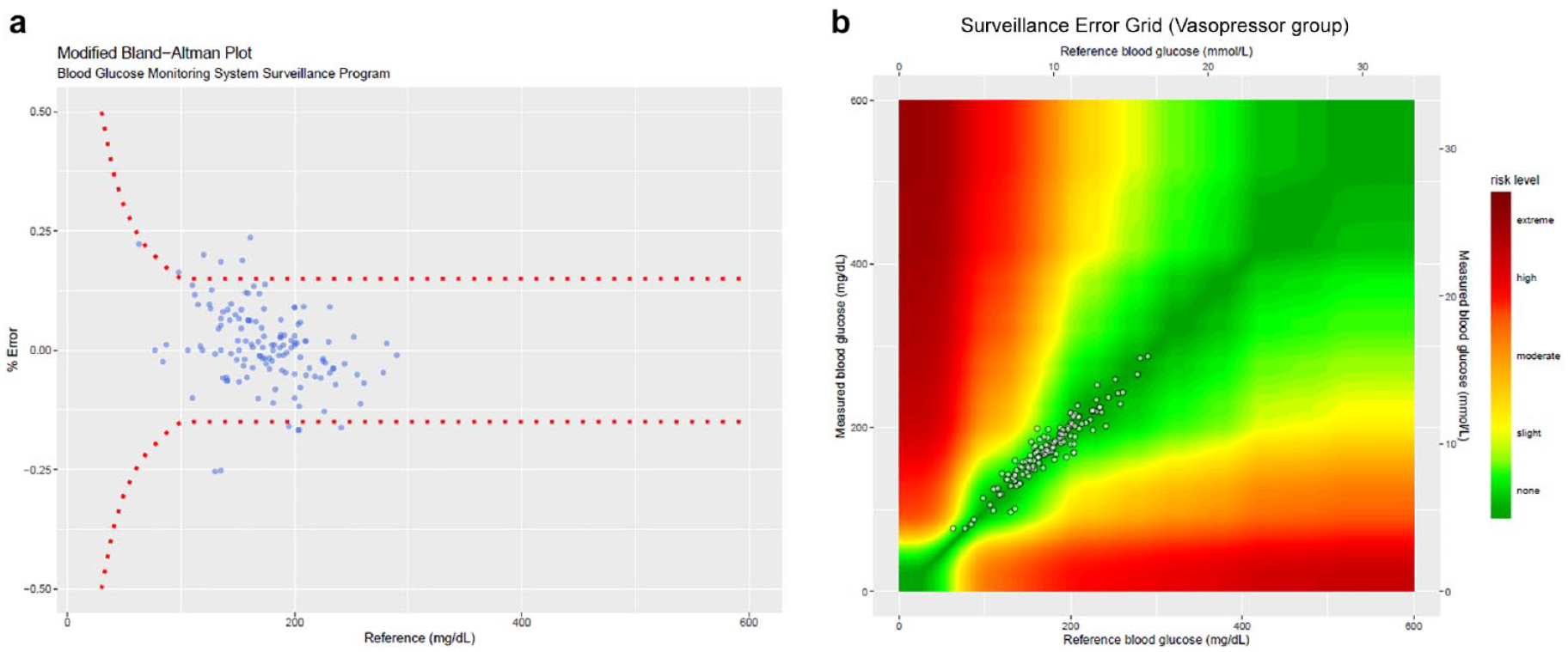
Effect of vasopressor. (A) Modified Bland-Altman plots. (B) SEG.
Plasma glucose measurement detected significantly more episodes of hypoglycemia (41-70 mg/dL) than CGM readings (1.6% vs 5.6%, P = .03), whereas no difference in hypoglycemia detection was found between CGM and CBG (0% vs 0.28%, P = .32) (Table 2). All hypoglycemia was mild, and could be easily managed according to the insulin infusion protocol. No severe hypoglycemia (≤40 mg/dL) occurred in this study. No other adverse events related to CGM insertion occurred during the study.
Conclusion
Using various assessment methods, the subcutaneous CGM sensor showed acceptable accuracy in MICU patients, in both the vasopressor and non-vasopressor groups. However, this study does not include the patients with diabetes emergencies such as DKA and HHS, which CGM accuracy needed to be explored. In conclusion, the accuracy of CGM in MICU patients was comparable to those observed in non-critical care settings and has potential benefits in critically ill patients.
Supplemental Material
sj-pdf-1-dst-10.1177_19322968211027590 – Supplemental material for The Accuracy of Continuous Glucose Monitoring in the Medical Intensive Care Unit
Supplemental material, sj-pdf-1-dst-10.1177_19322968211027590 for The Accuracy of Continuous Glucose Monitoring in the Medical Intensive Care Unit by Wannita Tingsarat, Patinut Buranasupkajorn, Weerapan Khovidhunkit, Patchaya Boonchaya-anant and Nitchakarn Laichuthai in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors thank all of the medical ICU staff during the study periods, all the patients in the medical ICUs who participated. The authors also thank Medtronic, Thailand for providing sensor transmitter and technical support during the study period and Assistant Professor Pornpoj Pramyothin for being part of a project committee, Dr. Yuda Chongpison for statistical consultation and Professor Stephen J Kerr for manuscript editing.
Abbreviations
%CV, coefficient of variation; CBG, capillary blood glucose; CGM, continuous glucose monitoring; LOA, limits of agreement; MARD, mean absolute relative difference; MICU, medical intensive care unit; PG, plasma glucose; SEG, surveillance error grid.
Author Contribution
W.T. and N.L. generated the data. P.B., P.B., W.K., and N.L. reviewed and revised the manuscript. N.L is the guarantor of this work and had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Quality Improvement Fund, King Chulalongkorn Memorial Hospital development program, The Thai Red Cross Society [grant number 1-62-30101-A-11].
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
