Abstract
Diabetes Technology Society hosted its annual Diabetes Technology Meeting on November 12 to November 14, 2020. This meeting brought together speakers to cover various perspectives about the field of diabetes technology. The meeting topics included artificial intelligence, digital health, telemedicine, glucose monitoring, regulatory trends, metrics for expressing glycemia, pharmaceuticals, automated insulin delivery systems, novel insulins, metrics for diabetes monitoring, and discriminatory aspects of diabetes technology. A live demonstration was presented.
Introduction
On November 12 to November 14, 2020, Diabetes Technology Society (DTS) gathered healthcare professionals (HCPs), industry representatives, academicians, researchers, and United States (USA) regulatory officials for the Diabetes Technology Meeting. This three-day meeting included two workshops, 11 sessions, and a keynote presentation by the Director of the United States Food and Drug Administration (FDA) Center for Devices and Radiological Health (CDRH), all covering current research and emerging topics in diabetes technology. Table 1 presents the agenda for the meeting, with a list of the workshop and session topics. This meeting report summarizes the key points of each speaker’s presentation and for each session, provides a consensus to answer the question posed in the session topic.
Agenda of the Meeting, with a List of the Workshop and Session Topics.
Workshop 1: Novel Software; Panel 1: Artificial Intelligence
Moderators
B. Wayne Bequette, PhD
Rensselaer Polytechnic Institute, Troy, New York, USA
Piotr Ladyzynski, PhD
Nalecz Institute of Biocybernetics and Biomedical Engineering of the Polish Academy of Sciences, Warsaw, Poland
CGM-AI: From Pattern Recognition to Automatic Control
Boris Kovatchev, PhD
University of Virginia, Charlottesville, Virginia, USA
Diabetes Data Science is emerging as a method to decipher the complex and voluminous diabetes-related data sets, exemplified by but not limited to, continuous glucose monitoring (CGM) time series;
In this context, a variety of artificial intelligence (AI)-based analytical approaches are applied to the interpretation of the CGM data streams;
We illustrate several of these analytical approaches, ranging from straightforward pattern recognition, clustering and classification to problem-solving and learning used in automated glucose control.
Clinical Uses of Artificial Intelligence
Moshe Phillip, MD
National Center for Childhood Diabetes, Schneider Children's Medical Center of Israel, Petah Tikva, Israel
The digital clinic and AI
Decision support for insulin-treated people with diabetes
Outcome of prospective randomized studies
Use of Artificial Intelligence in the Design of Decision Support Systems
Peter G. Jacobs, PhD
Oregon Health and Science University, Portland, Oregon, USA
Review the difference between data driven vs. physical models and advantages and disadvantages of each.
Discuss how mathematical models and metabolic simulators can be used to design new decision support dosing recommender algorithms.
Discuss how AI can be used for predicting hypoglycemia in both the short-term and long-term (eg, overnight).
An Artificial Intelligence Augmented Mobile APP
Mercedes Rigla, MD, PhD
Parc Taulí Sabadell University Hospital, Barcelona, Spain
The app, SineDie, provides support to gestational diabetes mellitus patients and performs changes in diet to correct hyperglycemia and ketonuria without medical intervention.
The SineDie expert system integrates a machine learning (ML) automatic glucose classifier (C4-5 decision tree learning algorithm), whose functionality is to assign an appropriate mealtime and “moment of measurement.”
The clinicians use the professional SineDie app designed to facilitate the detection of those patients showing non-optimal results and points out the patients with treatment proposals awaiting validation.
There were four major topics of CGM AI that were presented: (i) Defining CGM-AI, (ii) An Ecosystem of Diabetes Devices, (iii) Rationalizing the Diabetes Data Space: Metrics, Patterns, Clustering and Classification, and (iv) Process Control: the Artificial Pancreas.
The definition of AI from Investopedia (https://www.investopedia.com/terms/a/artificial-intelligence-ai.asp) 1 is:
“The ideal characteristic of artificial intelligence is its ability to rationalize and take actions that have the best chance of achieving a specific goal. Algorithms often play a very important part in the structure of artificial intelligence, where simple algorithms are used in simple applications, while more complex ones help frame strong artificial intelligence. The applications for artificial intelligence are endless. The technology can be applied to many different sectors and industries. AI is being tested and used in the healthcare industry for dosing drugs and different treatment in patients, and for surgical procedures in the operating room.”
1
CGM-AI was then defined as the ability to rationalize CGM data and take actions that have the best chance of achieving optimal glycemic control. Algorithms play an important part in CGM-AI. Simple algorithms include computing summary metrics, estimating hemoglobin A1c (HbA1c), and linear near-term glucose forecasting. More complex examples including pattern recognition, clustering and classification or process control.
An ecosystem of diabetes devices includes signals (self-monitoring of blood glucose [SMBG], CGM, motion, heart rate, geolocation) and actuators (insulin pumps and pens), and in-between is an “AI algorithm” that could include advisory/decision support or closed-loop control. Figure 1 presents the ecosystem of devices that feed data into CGM-AI, which are then used for a variety of clinical services.
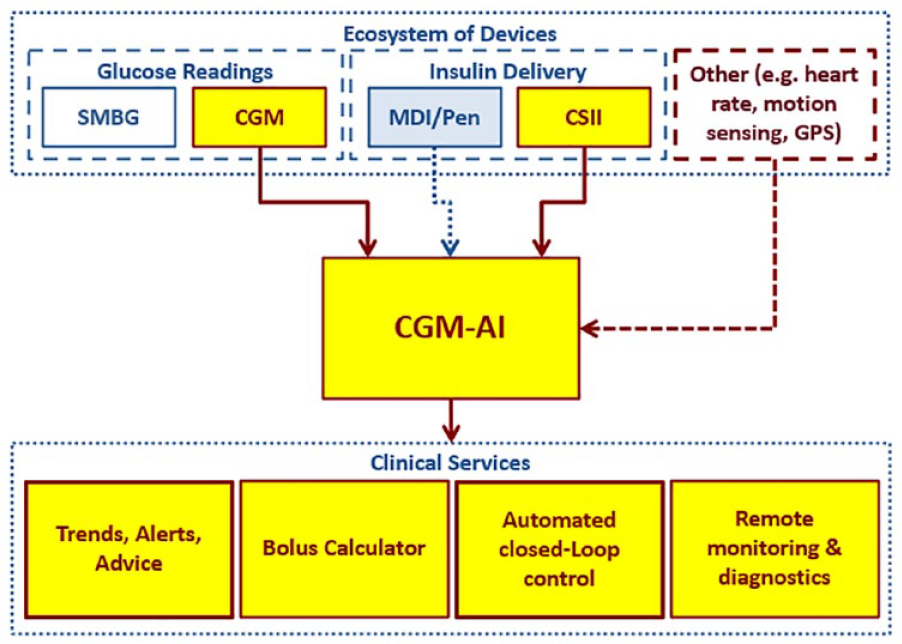
The ecosystem of devices that feed data into CGM-AI, which are then used for a variety of clinical services. Figure provided by Boris Kovatchev, PhD, University of Virginia, Charlottesville, Virginia, USA.
But how does one derive a decision based on the multitude of data available? A number of metrics can be used, such as time in range (TIR) or HbA1c (okay), patterns including daily CGM profiles (better), or CGM-AI with personalized analytics (best). 2
Three classes of CGM profiles were proposed: (1) tight control/intensive treatment, (2) glucose volatility/hyperglycemia, (3) intermediate/average control. These can be color-coded and easily displayed over a certain period of time to show daily control patterns. The “clusters” can then be summarized in a table comparing the three clusters (1, 2, 3), including the number of days, the mean CGM, and TIR (70-180) for each cluster.
For closed-loop control, an overview of the MiniMed 670G (Medtronic Diabetes, Northridge, California, USA) was provided, including sensor type and calibration, and the proportional–integral–derivative controller algorithm used in auto-mode. 3 Then, Tandem Control-IQ (Tandem Diabetes Care, San Diego, California, USA) was summarized, including the calibration-free Dexcom G6 (DexCom, Inc., San Diego, California, USA) and the t:slim X2 insulin pump (Tandem Diabetes Care). Unique algorithm components include gradually intensified control overnight, and adjustments for exercise and sleep.4,5
The definition on Wikipedia for AI was also considered. 6 The Wikipedia definition of AI is: “the term ‘artificial intelligence’ is often used to describe machines (or computers) that mimic ‘cognitive’ functions that humans associate with the human mind, such as ‘learning’ and ‘problem solving’.” 6
There is an information paradox where, in spite of improved technology (increased pump and CGM use), metabolic control has worsened. The Diabetes Control and Complications Trial published in 1993 indicated that better communication and advice from diabetes experts could lead to improved glucose control. A major challenge is the limited access to diabetes experts. This motivates having AI-based decision support to mimic diabetes team members. Phillip et al. 7 reviewed the first outpatient studies using the MD-Logic, fuzzy-logic, closed-loop approach.
The advisor-pro decision support system was also discussed. This is a system that mimics the way a physician thinks, to provide advice on changing basal rate, carb-insulin ratio, and correction factor.8,9 A large six-month trial was conducted to compare decision support with clinical advice. There were no differences between the decision support system and clinicians in HbA1c and TIR outcome metrics. Thus, AI can be used in closed-loop and in decision support systems.
Activities from the AI for Medical Systems Lab (AIMS) at Oregon Health and Science University was presented, including (i) the integration of insulin, glucagon and pramlintide delivery to prevent exercise-induced hypoglycemia, (ii) developing decision support systems for multiple dose injection (MDI) therapy, (iii) miniaturization of drug delivery with glucose sensing (glucose-sensing cannula), and (iv) using modeling and big-data sets to develop smarter control and prediction algorithms.
Two systems AIMS has built using AI are: (i) iPancreas: a closed-loop multi-hormone automated delivery system, and (ii) Daily Dose: decision support system, adjusting carb ratio and correction factor for MDI users (using the k-nearest neighbors, KNN, algorithm). Also included are nocturnal hypoglycemia prediction (using support vector machine) and short-term hypo prediction (using long short-term memory) components.
Three types of models for the use of AI in decision support systems include: (i) used in control algorithms, (ii) used in simulators, and (iii) that can be used to design new decision support algorithms. There are differences between physical models (compartment, differential equations) (white box), and data-driven (black box) models. Physical models are often used in models of type 1 diabetes (T1D), including virtual patient populations. A quick primer on compartmental models and their relationship to differential equations was presented, followed by a block diagram and description of how a virtual patient population of 100 individuals was developed. 10 In addition, a “digital twin” could be used to match with individuals in clinical studies.
Simulators can be used to design decision support systems. For example, Tyler et al 11 determined carb ratios and correction factors by simulating a variety of incorrect basal insulins and carb/correction ratios and training a KNN algorithm to make optimal recommendations. The KNN approach includes a heuristic algorithm component. Decision support recommendations agreed with physicians about as much as physicians agreed with themselves.
Another AI tool that has been created is the mobile app, SineDie. SineDie was developed to provide support to gestational diabetes mellitus patients without increasing the health care providers' workload.12-14 The SineDie expert system integrates a ML automatic glucose classifier (C4-5 decision tree learning algorithm), whose functionality is to assign an appropriate mealtime and “moment of measurement.” It performs changes in diet to correct hyperglycemia and ketonuria without medical intervention. Additionally, if the system detects that insulin therapy should be initiated, it then notifies the HCP along with recommendations for the insulin type and insulin dosage. The patient receives a text message explaining the new situation and a medical appointment is scheduled.
To validate the system, a randomized controlled trial (RCT), involving 119 women using the web-based initial version, was performed. Both metabolic control and neonatal outcomes were similar between the study groups. The mean number of face-to-face visits was 6.2 in the control group and 3.6 in the SineDie group. Patient questionnaires showed high satisfaction with the system.
A few weeks before the coronavirus disease 2019 (COVID-19) pandemic started, a RCT with the SineDie mobile version had started.15,16 After the lockdown was mandatory in Spain, it was decided to discontinue the ongoing clinical study and offer the mobile app to any patient referred because of gestational diabetes mellitus. Patients download glucose data from the glucometer, and the platform enables doctors to automatically determine which patients have adequate glycemic control and, therefore, do not need face-to-face visits. The clinicians use the professional SineDie app designed to facilitate the detection of those patients showing non-optimal results and points out the patients with treatment proposals awaiting validation. When the system generates a recommendation about insulin therapy, a proposal of daily insulin prescription is generated: amount and type of insulin, and moment of administration. When a treatment change is made, it is automatically notified to the patient with a pop-up message.
Workshop 1: Novel Software; Panel 2: Digital Health and Telemedicine
Moderators
David Ahn, MD
Mary & Dick Allen Diabetes Center at Hoag, Newport Beach, California, USA
Jennifer K. Raymond, MD, MCR
Children’s Hospital Los Angeles, University of Southern California, Los Angeles, California, USA
Diabetes Telehealth: We’re Doing It. . . But How Do We Make It Better and Easier?
Aaron Neinstein, MD
UCSF, San Francisco, California, USA
The COVID-19 pandemic has expedited a pre-existing trend toward increased use of telehealth for diabetes care delivery, leveraging changes in delivery system infrastructure and workflow, federal reimbursement policy, and patient and provider expectations.
Next steps needed to improve video visit quality and experience are better workflows and technologies to ensure pre-visit device data connectivity and availability, in-visit screen sharing and annotation, and electronic health record (EHR)-integrated diabetes device data to facilitate efficient provider review, documentation, and reimbursement.
Ultimately, care models, technologies, and workflows are needed to support continuous diabetes care, including personalized follow-up check-ins between visits and population based patient identification and outreach.
Integrated Personalized Diabetes Management (iPDM) in an Open Ecosystem: Paving the Way for Advanced Outcomes in Diabetes Therapy
Rolf Hinzmann, MD, PhD
Roche Diabetes Care, Mannheim, Germany
Integrated Personalized Diabetes Management (iPDM) from Roche Diabetes Care (Mannheim, Germany) is a holistic, patient-centric approach to diabetes management, also involving caregivers, healthcare professionals, and payers.
Enabled by an open ecosystem, iPDM comprises collection, integration, and analysis of relevant data from Roche and partner solutions, shared with consent of the patient.
Insights obtained by the data analysis using algorithms, AI, and ML, will improve treatment decisions.
Digital Health Tools to Improve Adherence to Blood Glucose Monitoring
Elizabeth Holt, MD, FACE
LifeScan, Malvern, Pennsylvania, USA
People with diabetes are receptive to using digital health tools in their diabetes management.
Data analysis tools will help healthcare providers see patterns in blood glucose readings.
Gamification and digital health coaches can motivate patients, leading to behavior changes.
The Benefits of a Multi-Sensor Platform for Diabetes
Mark Clements, MD, PhD
Glooko, Mountain View, California, USA
Comprehensive digital health for diabetes enables remote care for caregivers and people with diabetes.
The Multi-Sensor Platform includes four factors: (1) digital therapeutics, (2) telehealth and remote patient monitoring, (3) advanced insights, and (4) EHR integrations.
It is important to mobilize the therapeutic alliance for better outcomes, with digital tools to facilitate the Patient-HCP connection.
Telemedicine to Support Personalized Care
Ronald Dixon, MD
Remedy, Austin, Texas, USA
An overview of a hybrid model of telemedicine care
Enabling primary care physicians to manage risk
Leveraging software to improve the clinician experience
Prior to March 2020, crucial groundwork in digital health and telemedicine in diabetes had been laid, but the COVID-19 pandemic exponentially increased interest in and the adoption of multiple aspects of virtual care. Many diabetes and endocrinology clinics transitioned the majority of their care to telehealth, at rates higher than other medical specialties.
Despite the fact that diabetes management is relatively well-suited for virtual care, multiple challenges remain. Furthermore, the COVID-19 pandemic has highlighted disparities in care and inequities in our current healthcare systems. Thankfully, there are existing and developing models to help address these gaps.
Historically, patients with diabetes have not been empowered to access nor interact with their diabetes data, but this is changing. Roche Diabetes Care has developed iPDM through an open ecosystem that allows people with disease to share data from Roche and third party sources for algorithm-based analysis to create insights and medical value to improve clinical outcomes. 17 New tools, such as those from LifeScan (Malvern, Pennsylvania, USA), provide interpretation of patterns for those living with diabetes and providers caring for people with diabetes. 18 Traditionally, diabetes data has lived in device-specific platforms, but Glooko (Palo Alto, California, USA) is focusing on pulling together these data into one platform. 19 This allows people with diabetes and providers to see all data in one place. Finally, innovative diabetes platforms, such as those from Onduo (Newton, Massachusetts, USA), allow providers and medical practices to see and manage risk for their patient population while also improving the provider’s experience with reviewing diabetes data. 20
For broad implementation of these innovative systems, we need substantial changes in our current clinical and reimbursement models. Clinical barriers to using digital health platforms can include integration with the EHR, data safety and security, different technologies and guidelines in different geographical areas, and processes for reimbursement. Patient barriers can include digital disparities, lack of trust in the system, and insufficient or no insurance coverage. Diabetes stakeholders need to come together to address barriers on the patient, clinical, and system front to truly achieve innovative, patient-centered digital diabetes care. Table 2 presents a summary of top 10 tips for successfully implementing diabetes telehealth. 21
Summary of Top 10 Tips for Successfully Implementing Diabetes Telehealth. 21
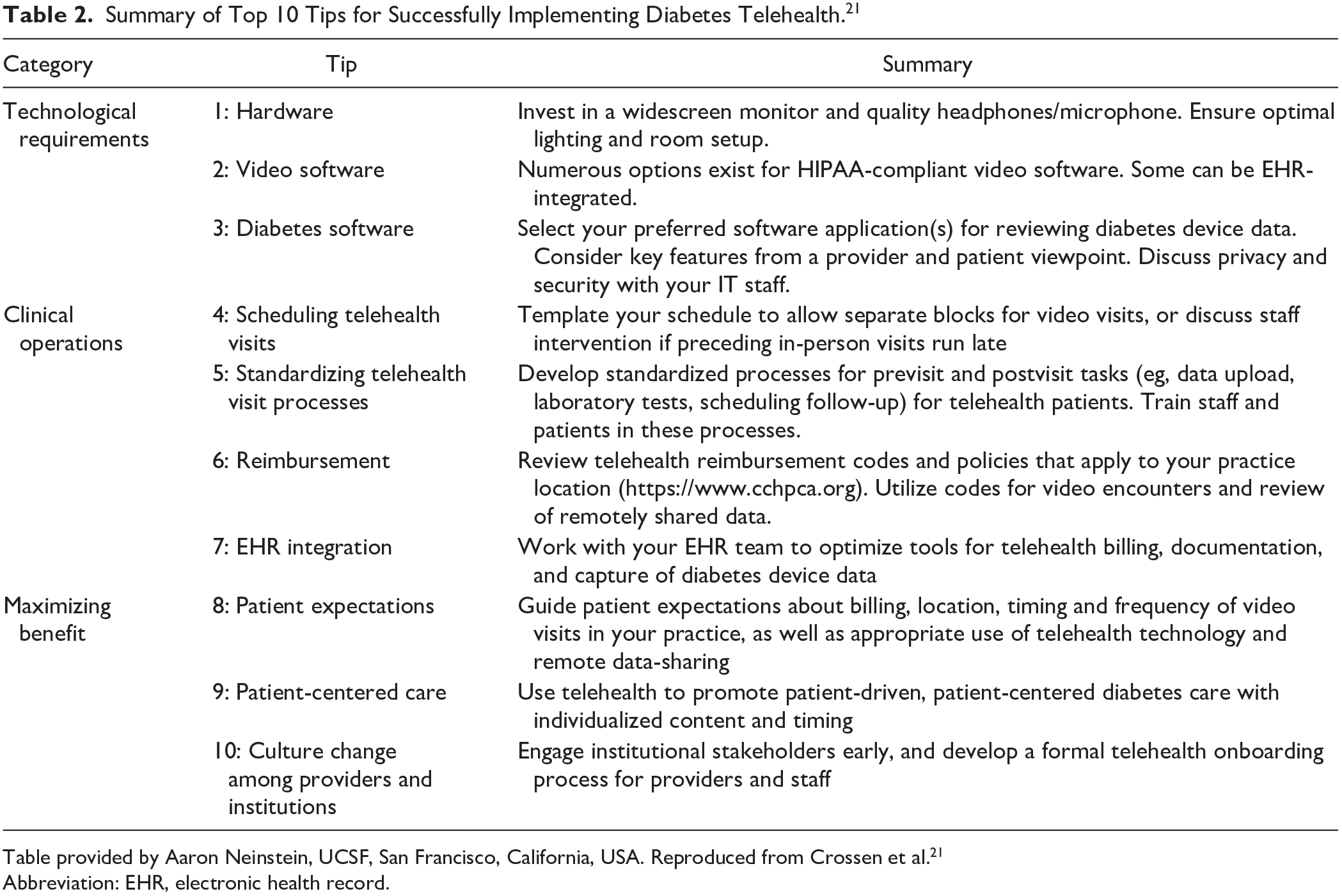
Table provided by Aaron Neinstein, UCSF, San Francisco, California, USA. Reproduced from Crossen et al. 21
Abbreviation: EHR, electronic health record.
Workshop 2: Glucose Monitoring; Panel 1: Noninvasive
Moderators
Gerard Coté, PhD
Texas A & M University, College Station, Texas, USA
Brian Frier, BSc (Hons), MD, FRCP(Edin), FRCP(Glas)
University of Edinburgh, Edinburgh, Scotland
Fundamental Issues That Limit Successful Non-Invasive Glucose Measurements
Mark Arnold, PhD
University of Iowa, Iowa City, Iowa, USA
Overall objective: Describe the key elements of noninvasive measurements that impact success, realizing no successful noninvasive glucose sensing device has been developed to date. My presentation offers a general discussion related to all types of measurements—spectroscopy, impedance, indirect signals, and so on.
○ Define noninvasive glucose measurements
○ Describe direct vs indirect method
○ Discuss issues of measurement selectivity
○ Explain background variance and its importance
○ Illustrate how variations in background variance impact glucose concentration measurements
Optical Methods for Non-Invasive Glucose Sensing: Breakthrough or Broken Promises Again?
H. Michael Heise, PhD
South-Westphalia University of Applied Science, Iserlohn, Germany
Fundamental aspects of vibrational spectroscopy—spectral data for glucose, water and interferents for mid-infrared, near-infrared and Raman spectroscopy, aspects of sensitivity and selectivity
Current status and achievements of different research teams based on the three vibrational spectroscopies—critical assessment, limitations and device miniaturization
Future developments and expectations, for example, from photoplethysmography implementations
Detection of Hypoglycemia with Non-Invasive Sensors in Subjects with Impaired Awareness of Hypoglycemia
Kåre I. Birkeland, MD
University of Oslo and Oslo University Hospital, Oslo, Norway
The relationship between p-adrenaline, sweating and electrocardiogram signals, and their association with hypoglycemia
Will a combination of non-invasive sensors for detection of sweating, heart rate, and QT-time detect hypoglycemia in patients with T1D?
What is the impact of impaired awareness of hypoglycemia on the ability to detect hypoglycemia with non-invasive sensors?
Tear Glucose: Fact or Fiction?
Christopher Wilson, PhD
Listerdale Lifescience BV, Nijmegen, Netherlands
Summary of the history
Why did a small company succeed where the giants failed?
Our clinical data
Skin-Interfaced Wearable Sweat Biosensors
Wei Gao, PhD
California Institute of Technology, Pasadena, California, USA
Non-invasive sweat analysis
Fully-integrated wearable biosensors
Device evaluation using human studies for personalized healthcare
The panel had a frank discussion of the various methods being explored to measure glucose noninvasively and their promise as well as their limitations. The workshop began by describing what is considered to be a noninvasive glucose measurement and the two main measurement methods, namely, direct (where the signal is from the glucose molecule) and indirect (where the signal is from a source other than glucose but may be correlated). 22 The presentations included nearly every type of biomedical sensor investigated to date. Two methods were featured that directly measure glucose noninvasively using samples of tears or sweat coupled with electrochemical biosensors. It was observed that for these direct measurements, the collection methods for sweat and tears strongly influence the ability to obtain reproducible glucose concentration results within and across subjects. For example, using a capillary tube that touches the eye can cause wide variation as it collects more than just tear fluid; an alternative method of collection in the lower pocket of the eye appears to be more reproducible. 23 Further, reverse iontophoresis has been used to extract interstitial fluid, which is arguably not even noninvasive as it can cause bruising or skin abrasion. 24 However, iontophoresis with the use of a drug in conjunction with a properly designed microfluidic chamber leaves no marks on the skin and can provide sweat continuously for prolonged periods. The effects of the drug on the collection of sweat samples and the physiological correlation between the concentrations of glucose in blood as well as in either sweat and tear fluid has yet to be fully determined.
The other main topic included optical approaches that can be considered direct or indirect depending on the approach. The direct optical approaches include those that measure spectral changes directly associated with the glucose molecule (eg, near-infrared to infrared absorption, Raman scattering, mid-infrared photothermal). There was also discussion of the importance of capturing background variance in a calibration model. Figure 2 presents an example of building and testing a calibration model. It was pointed out that a lack of specificity for the glucose molecule can be a primary limitation for certain optical approaches including methods that rely on visible and short-wave near infrared absorption and scatter, and this lack of specificity could make this approach appear more like an indirect measurement and thus more challenging. Background variance 22 and the heterogeneity of tissue 25 were two further important limitations to measuring glucose noninvasively. An example was presented to illustrate how the tissue spectral background can change dramatically over time periods of minutes and how such changes adversely impact measurement accuracy. A reduction of tissue background complexity can be achieved to some extent by using photoplethysmography, particularly with spectral information collected over either the combination or first overtone bands within the near-infrared spectrum. 26 A novel indirect approach was discussed by which physiological responses to autonomic stimulation were measured noninvasively and related to insulin-induced hypoglycemia. Measurements included heart rate, heart rate variability, skin temperature and sweat production (ie, galvanic skin response). While changes indicated the onset of hypoglycemia in adults with T1D who possessed normal symptomatic awareness, the method was unable to identify hypoglycemia in people with impaired awareness. 27
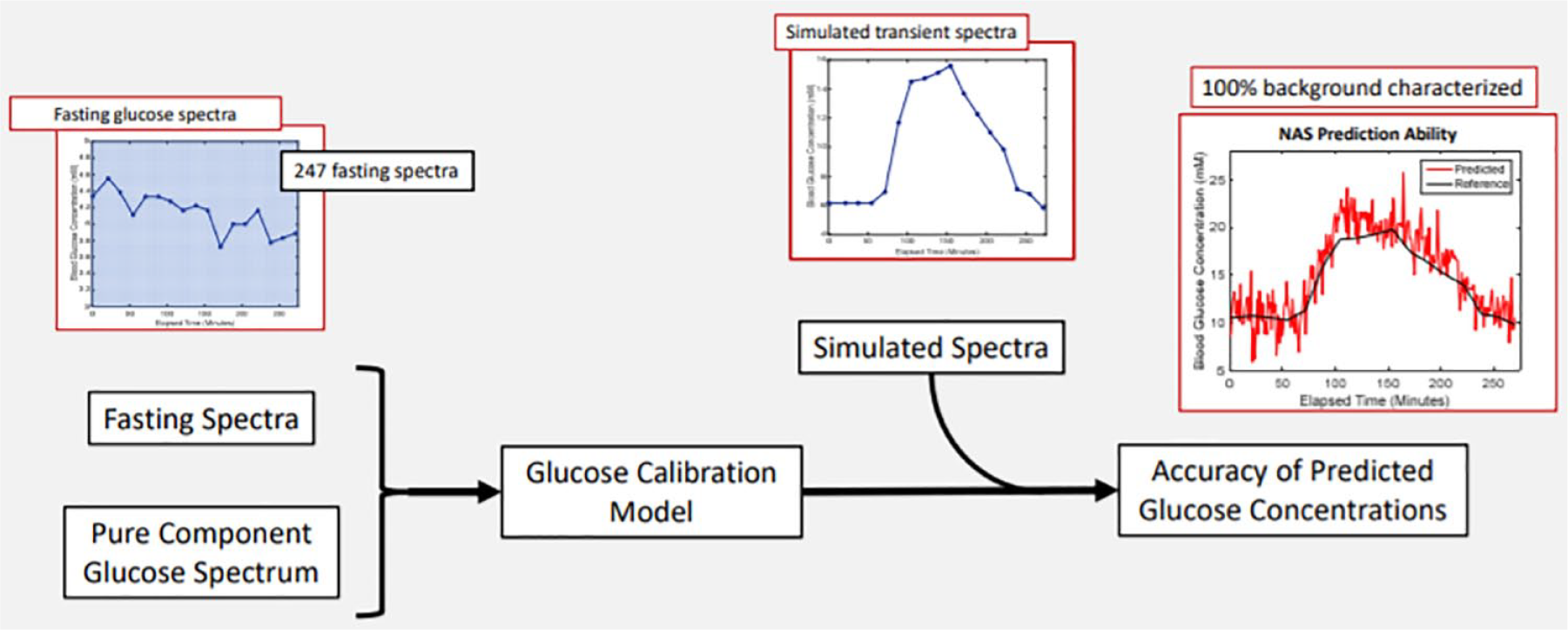
An example of building and testing a calibration model. Figure provided by Mark Arnold, PhD, University of Iowa, Iowa City, Iowa, USA.
Workshop 2: Glucose Monitoring; Panel 2: Novel Technologies in CGM Systems
Moderators
Jennifer L. Sherr, MD, PhD
Yale University, New Haven, Connecticut, USA
Hubert Vesper, PhD
Centers for Disease Control and Prevention, Atlanta, Georgia, USA
CGM Usage for the Management and Reversal of Type 2 Diabetes
Faz Chowdhury, PhD
CEO, Nemaura Medical, Loughborough, United Kingdom
Intermittent CGM usage
Diet management with coaching for Diabetes reversal
Patient psychology and CGM usage
Senseonics Eversense CGM System
Francine Kaufman, MD
Senseonics, Germantown, Maryland, USA
Andreas Stuhr, MD, MBA
Ascensia Diabetes Care, Parsippany, New Jersey, USA
The Eversense CGM System from Senseonics (Germantown, Maryland, USA) is the only long-term, implantable CGM device available to patients on a global basis—it is currently available in the USA and selective European countries. While the present system offers many innovations, future updates hold promise for not only extending the life of the sensor, but also its functionality. In addition, the recent global partnership between Senseonics and Ascensia Diabetes Care (Parsippany, New Jersey, USA) should allow for an expanded patient base to use this groundbreaking technology now and into the future.
The fully-implantable Eversense CGM System features a long-term sensor approved for use up to 90 or 180 consecutive days (USA and European Union, respectively), direct transmission of data from a smart transmitter to the user’s smartphone (with share capability), and on-body vibratory alerts, as well as other innovations. (A pivotal trial for a 180-day product in the USA with reduced calibration was recently submitted to the FDA.)
Senseonics believes that the path of future innovation is extremely robust, with the possible extension of sensor life up to 365 days, a marked reduction in calibration (up to once weekly), flash or swipe capability to combine CGM/flash glucose monitoring functionality in one device, and multiple device integration.
The partnership with Ascensia Diabetes Care, mutually beneficial to both organizations, will enable the Eversense CGM System to reach more people globally.
Waveform Technologies
Mihailo Rebec, PhD
WaveForm Technologies, Wilsonville, Oregon, USA
Provide update of the GLUCODAY 21 clinical trial results conducted on the Cascade CGM from WaveForm Technologies (Wilsonville, Oregon, USA) that demonstrated 21 day extended wear and reduced calibration frequency
Reviewed earlier clinical trials that extended device wear times to 15 days
Performance of a Next Generation iCGM System
Marc Taub, PhD
Abbott Diabetes Care, Alameda, California, USA
14 day integrated continuous glucose monitoring (iCGM) system performance in adult and pediatric populations
Real-time glucose alarms with readings every minute
Dexcom Technologies
Peter Simpson, MS
DexCom, Inc., San Diego, California, USA
Improvements to G6 system
Expanding the impact of CGM
Next generation technologies
Integrated CGM Systems: Enabling Technologies for Usability and Comfort
Akhil Srinivasan, PhD
Medtronic Diabetes, Northridge, California, USA
Medtronic is investing in technologies to enable usability, comfort and flexibility for patients utilizing CGM as well as those on or making the transition to pump therapy.
Integrated disposable design with a simple insertion process
Miniaturization through a new sterilization process for a one-piece transmitter and sensor device
Merging CGM with insulin delivery systems
In a highly stimulating session that focused on six different sensor technologies, a clear theme emerged: sensors provide the breadth and depth of data necessary to guide treatment optimization for persons with diabetes. A variety of sensing technologies were discussed including implantable (Senseonics), transcutaneous (Nemaura Medical, Loughborough, United Kingdom), and subcutaneous (WaveForm Technologies; Abbott, Chicago, Illinois, USA; DexCom, Inc.; and Medtronic Diabetes) devices, with many of the companies seeking to increase the duration of device use in future generations of the technology. Notably, impressive accuracy was reported from the pilot and pivotal trials presented with mean absolute relative differences (MARDs) of <10% for most devices.28-34 Continued innovations are planned, including: (1) decreased physical footprint of these devices, (2) reduction or elimination of calibrations, (3) assessment of the devices in broader populations with alternate sites of insertion, and (4) the insertion devices for many devices will be made easier to use. Finally, with the COVID-19 pandemic, the FDA will not object to the application of certain sensors in hospitalized persons with diabetes given the extraordinary circumstances,35,36 and use of these devices has now been shown to be feasible in the inpatient setting. 37 Furthermore, use of CGM based metrics has helped guide optimization of therapy during telehealth encounters.
Device use entails more than just application of the technology and needs to be grounded in detection of patterns and trends to guide clinical decision making. Indeed, those with type 2 diabetes (T2D) can achieve disease reversal, whereby HbA1c levels are reduced to 6% even when medications for treatment of diabetes are withdrawn. To facilitate the integration of sensor technology into the T2D population, Nemaura Medical has developed SugarBEAT, which is a transcutaneous sensor that has a re-usable transmitter and disposable sensors that measures the interstitial glucose without requiring needle insertion.38,39 MARD is reported to be 12.4% with one calibration per day. 39 With its low cost for manufacturing of the sensors, this device may be cost effective in its application to the T2D population and may be used either continuously or intermittently.
Transitioning to discussion of implanted sensors, the panelists shared a wide breadth of information spanning real-world data of the Eversense sensor, next generation technologies, and how training has been altered in the post-COVID-19 era. The Eversense sensor is a fully implanted sensor with an on-body transmitter that has vibratory alerts/alarms, which currently requires twice daily calibrations and is approved for 90-days of wear in the USA 40 and 180-days outside the USA. 41 Real world data (RWD), initially from 205 users and reinforced with a larger sample of 1656 users, show TIR of those using the device to be ~62% with median sensor wear time of ~84%.42,43 Those with the greatest improvements in TIR tended to be those who were sensor naïve. 42 Recently, a premarket approval (PMA) application has been submitted to extend duration to 180-days, with essentially one calibration per day, based on data from 181 participants. 44 Future generations of this technology will seek to extend duration to 365 days, reduce calibration to once weekly, and allow the device to have flash capability. With COVID-19, health care providers are now being successfully trained virtually on how to place the sensor.
The Cascade CGM from Waveform Diabetes with a 21-day duration of wear was then discussed. This sensor extends the currently Conformitè Europëenne (CE) marked 14-day sensor, which provides glucose levels every minute. Ten participants wore two sensors for 21 days’ time with frequent sample testing to assess the sensor performance. MARD over the course of the study was 9.6%. Out of 20 placed sensors, six did not last for their intended duration mostly because of issues with adhesive. Future studies will assess the use of alternate sites as well as expanded populations (pediatrics and pregnant women) in addition to obtaining regulatory approval of the 21-day device.
Attention then turned to flash glucose monitoring including the products from Abbott. This past year, the FDA has approved use of the FreeStyle Libre 2 system with an indication for use of the device in persons with diabetes ages 4 and up. 45 This device offers sensor glucose readings every minute, optional real-time glucose alarms, and improved accuracy with no user calibration. Of note, accuracy has especially been improved in the lower range of glucose readings (i.e., <70 mg/dL). 45 Ascorbic acid in high doses is considered an interfering substance, but this would be unlikely to occur with dietary intake alone. 31 Additionally, there has been a recent CE Mark for the Libre 3, which has a 70% reduction in volume and a one-piece applicator, with the greatest change in the device being streaming of the glucose data to the reader/app. All of this is achieved with the same accuracy noted with the FreeStyle Libre 2. 46
Returning to real time CGM systems, the Dexcom G6 is a sensor with an expected duration of 10-days, no requirement for calibrations and overall MARD of 9.0%. 33 Building on this, the company now has the Dexcom G6 PRO, which allows for a single use disposable professional sensor. 47 The Dexcom G6 has received a CE Mark for the arm as an insertion site and approval for use in women who are pregnant,48,49 as well as has improved device reliability. The Dexcom G6 has been integrated with a variety of different pump systems, connected pens, EHRs, and automated insulin delivery (AID) systems. Most recently, the FDA has said that they would not object to the use of the Dexcom G6 in the hospital setting during the COVID-19 pandemic. 35 With integration of the sensor in the inpatient unit, less time in patient rooms for those who are COVID-19 positive accompanied by reduced need for personal protective equipment and improvements in glycemic control have been noted. Finally, data from the Dexcom G7, which will be >60% smaller than the G6 with faster start-up, pilot trial data was shared, which showed an overall MARD of 8.7%, with slightly better data achieved with arm insertion.
The importance of ease of insertion was addressed, with a next generation product from Medtronic planned to only require three steps for insertion. It will have a disposable transmitter attached and a symmetrical design with a significantly thinner profile. Critical to achieving this improved insertion were new sterilization techniques that are being employed and were reviewed in detail. The USA pivotal trial of this new sensor device has completed enrollment. Finally, discussion turned to advances in infusion set technology. Recently, an extended duration wear site has now received a CE Mark and is in pivotal trials in the USA. With the advent of a 7-day infusion site, attention is now turning to developing an integrated CGM infusion set. Initial feasibility testing has shown that sensor glucose readings will not be impacted by being in close proximity to insulin delivery. In short, with the plethora of CGM systems available and with the advent of the next generation of devices, sensor use is expected to continue to rapidly grow. Indeed, with consensus guideline setting targets for TIRs, CGM provides a richer data-set than HbA1c measurements upon which to alter therapies, and with the technological advances that are to come, the use of sensor technology in all persons with diabetes, who are amenable to this therapy, is warranted.
Keynote: Regulatory Trends at the FDA in Diabetes Devices
Timothy Stenzel, MD, PhD
Director, Office of In Vitro Diagnostics and Radiological Health at the FDA, Silver Spring, Maryland, USA
The FDA is working toward interoperability between iCGM systems, alternate controller enabled (ACE) pumps, and controllers.
Since the last Diabetes Technology Meeting in 2019, the FDA has authorized the Tandem Control-IQ, a medical development tool called the Insulin Dosing Systems: Perceptions, Ideas, Reflects, and Expectations (INSPIRE) questionnaire, and the Medtronic MiniMed 770G.
Current trends in diabetes technology are smart insulin pens, CGM based decision support apps, glucose trending apps, and more advanced CGM systems.
The FDA hopes to make iCGM systems, ACE pumps, and controllers truly interoperable devices. They have had success on improving the interoperability between these devices through mixing and matching, and they hope for more applications to come to their office. They would like to do away with glucose meters and believe interoperability will spur device innovation. New regulatory pathways make accessing glucose and insulin data as inputs into digital platforms more streamlined. There are supports integrating data from different devices into multiple differently configured systems, and these supports enable consolidated digital solutions that offer real time device data access, device control, and decision support in one interface. Regulatory advantages may incentivize additional technology development for accuracy and reliability.
Recently, there have been regulatory pathways for iCGM systems, which are CGM systems that are able to integrate with other devices. The FDA requires that iCGM systems have reliability and accuracy and a safe communication process to connect compatible devices. The FDA also requires transparency with certain info in the labeling. They believe that there is potentially room for over-the-counter iCGM systems if manufacturers can show that people are able to use the device when provided with the instructions.
Notable updates in the diabetes technology space from the FDA since the last Diabetes Technology Meeting in November 2019 start with news from December 2019. In December 2019, the FDA authorized the Tandem Diabetes Care Control-IQ system. The Tandem Diabetes Care Control-IQ is an interoperable glycemic control system which adjusts insulin delivery based on connecting to an ACE and iCGM. 50 This can allow this interoperable system to be used as a full AID system. In June 2020, the FDA authorized a medical device development tool called the INSPIRE questionnaire. 51 It can be used to qualitatively evaluate the psychosocial effects on youth with T1D using the AID system, their parents/guardians, as well as adults with T1D using AID systems and their partners. 52 The FDA hopes that with this questionnaire, they can use patient and caregiver input into regulatory decisions. In August 2020, the FDA authorized the Medtronic MiniMed 770G, and it became the first AID system for 2 to 6 year old children. 53
The FDA observes that current trends in diabetes technology include smart insulin pens, CGM based dose decision support apps for patients and clinicians, glucose trending apps, and glucose readings directly on mobile platforms and “follow” apps. The next steps involve standalone algorithms that will provide more flexibility in selecting a pump and CGM, insulin pump (remote bolusing from phones, insulin/therapeutics), mobile interfaces for AID systems, more available devices, and more devices for T2D patients, working toward a fully closed loop system. The FDA is aware of the formation of a collaborative community around suicidal ideation in patients with diabetes. The FDA is interested in anything that can reduce risk of self-inflicted injury and suicide and any role that they can play. The FDA has great interest in having a collaborative community that they can recognize.
Session 1: What Are the Best Metrics for Expressing Glycemia in CGM?
Moderators
Jessica Castle, MD
Oregon Health Sciences University, Portland, Oregon, USA
Guillermo Arreaza-Rubin, MD
NIDDK/NIH, Bethesda, Maryland, USA
Time-in-Range or HbA1c: Why Not Both?
Lutz Heinemann, PhD
Science Consulting in Diabetes GmbH, Neuss, Germany
HbA1c will remain the standard parameter for estimating risk for development of late complications due to diabetes.
TIR is a valuable supplement to HbA1c, but it has limitations and is not a replacement for HbA1c.
In clinical practice: look at the CGM profile first, put the numbers second.
Metrics for Evaluation of Quality of Glycemic Control
David Rodbard, MD
Biomedical Informatics Consultants LLC, Potomac, Maryland, USA
Multiple metrics are available to evaluate glycemic control.
○ M
○ Q-Score, Comprehensive Glucose Pentagon (CGP), Personal Glycemic State (PGS) calculated from mean glucose, %TIR, hypo- and hyperglycemia, glycemic variability
○ CGM Index—based only on %TIR (50%), %time below range (TBR) (35%), standard deviation (15%)
%TIR (70-180 mg/dL) is not sufficient to characterize glycemic control from CGM tracings. It is necessary to also examine %TBR (<54 mg/dL and <70 mg/dL) and %time above range (TAR) (>180 mg/dL and >250 mg/dL) by time of day.
Only two metrics are needed to describe glycemic control: a measure of average level of glycemia (HbA1C, mean glucose, %TIR, or %TAR) and a measure of hypoglycemia (%TBR, low blood glucose index, or rate of hypoglycemia).
Efficacy (average level of glycemia) and safety (hypoglycemia) can be plotted together to make comparisons of different interventions.
Essential Metrics: The Principal Components of a CGM Daily Profile
Boris Kovatchev, PhD
University of Virginia, Charlottesville, Virginia, USA
A variety of CGM metrics exists, many of which reflect the same glycemic feature in different ways.
To streamline the multitude of metrics, a principal component analysis can be used aiming to derive the main features of a CGM daily profile that explain most, if not all, metrics of glycemia.
TIR and risk for hypoglycemia represent well the two principal dimensions of glycemic control.
Interconnected Relationships of Metrics in the AGP
Richard Bergenstal, MD
International Diabetes Center, Minneapolis, Minnesota, USA
The glucose management indicator (GMI) is the bridge between HbA1c and %TIR.
The three critical metrics to focus on are: %TIR (70-180 mg/dL), %time 54 to 69 mg/dl, and %time <54 mg/dL.
For clinical care we need to move from analysis to action. The analysis of CGM data can be simplified from nine steps into three steps:
○ Step one—more green less red (MGLR) = more TIR and less TBR
○ Step two—flat, narrow, and in range (FNIR) glucose profile
○ Step three—2, 4, 6 to 8; titrate, titrate, titrate (often takes 2 sensor sessions to reduce hypoglycemia and then you need to follow with 4 sessions to reduce hyperglycemia to achieve FNIR after 6 to 8 sensor sessions.)
Data is only useful if it can be used to improve the lives of those living with diabetes.
There are multiple metrics available to assess glycemic control. These include M

Only two metrics are required for characterization of quality of glycemic control: a measure of safety and one of efficacy. 54 LBGI, Low Blood Glucose Index (or Low Glucose Index); %TAR, %time above range; %TBR, %time below range; %TIR, %time in range. Figure provided by David Rodbard MD, Biomedical Informatics Consultants LLC, Potomac, Maryland, USA
With the exception of the low blood glucose index, multiple CGM metrics are highly correlated. This multicollinearity calls for principal component analysis. Figure 4 presents a principal component analysis of CGM metrics. A CGM daily profile is quantified by two essential metrics or principal components: TIR and risk of hypoglycemia as assessed by low blood glucose index. These two essential metrics explain >90% of the variance of all glycemic metrics. HbA1c and TIR reflect the same underlying process, but HbA1c depends on individual glycation rates and red blood cell clearance. As demonstrated using data from two International Diabetes Closed-loop trial protocols, %TIR and HbA1c can be used interchangeably provided that an appropriate model of individual glycation and red blood cell clearance is used to reconcile the two. 59
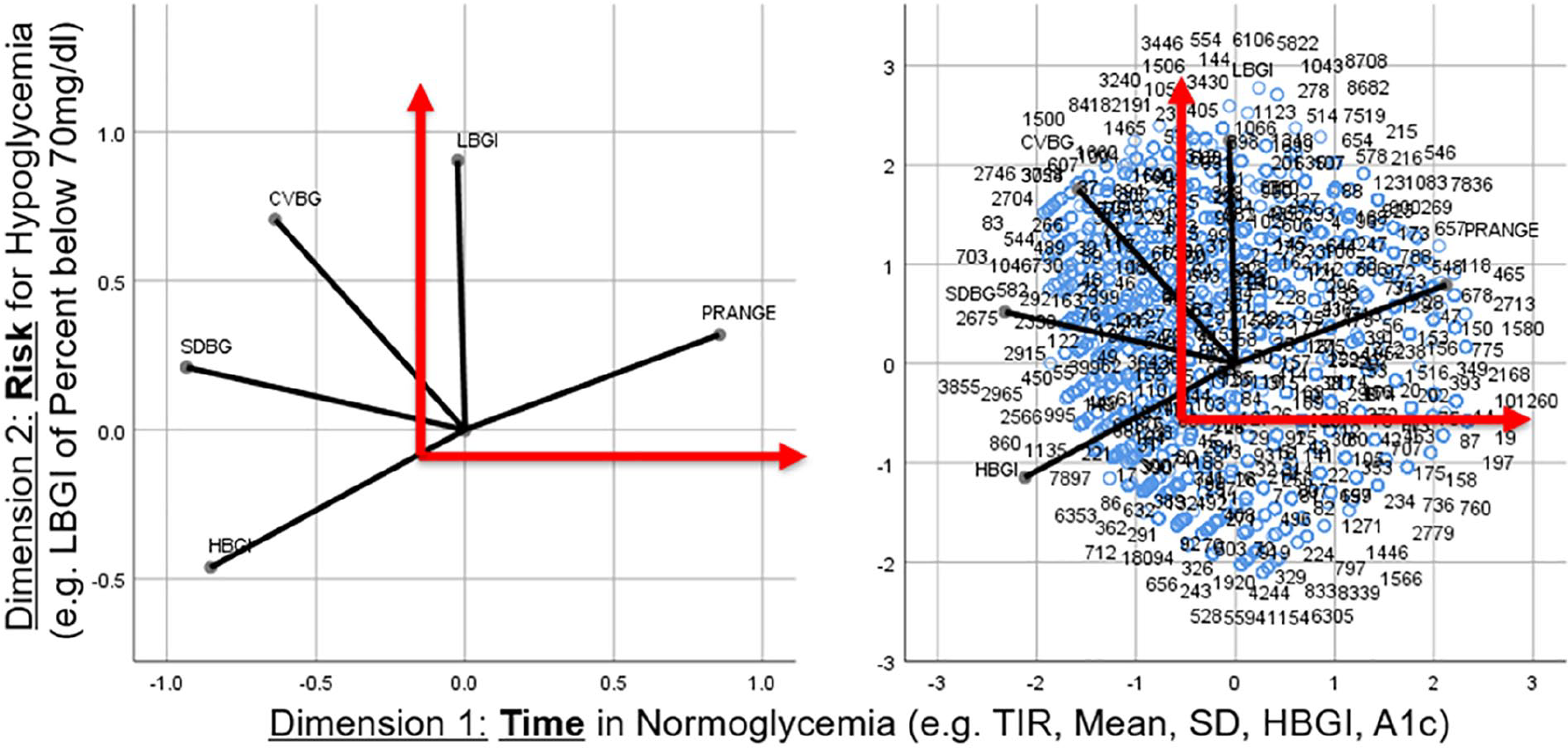
A principal component analysis of CGM metrics. Figure provided by Boris Kovatchev, PhD, University of Virginia, Charlottesville, Virginia, USA.
As new measures are proposed to assess glycemic control, the upside and downside of each parameter should be weighed. Historically, HbA1c has been the glycemic metric of choice as the primary outcome for clinical trials. There are multiple issues with using HbA1c, including that it does not provide information about hypoglycemia or glycemic variability and it is impacted by factors other than glucose such as ethnicity and hemoglobinopathies. There has been a push to move away from HbA1c in favor of TIR. However, there are also issues with using TIR as the primary metric for glycemic control. For example, different CGM systems may be used to estimate TIR and it has been demonstrated that the use of different CGM systems in the same participants can result in markedly different estimates in % time in hypoglycemia (<70 mg/dL). 56 Without standardization and traceability, it is not possible to get a reliable estimation of TIR.
With the downsides of HbA1c measurement being considered, the field of diabetes is moving from the HbA1c management era to the CGM management era. The bridge between the two is the ambulatory glucose profile, and specifically the GMI. The nine steps of analyzing CGM data 60 can be simplified to three. Step one is to assess if there is a problem using %TIR with a goal of “more green” and %time in hypoglycemia (54-69 mg/dL and <54 mg/dL) with a goal of “less red.” If there is a problem, step two is to assess the glucose profile to determine where the problem is and work toward a flat and narrow profile in the target range. Step three is a CGM-guided titration strategy with six to eight sessions over three months as intensive titration is typically what is required. It is critical to leverage the tools we have available within the ambulatory glucose profile to improve the quality of life of those living with diabetes. Glycemic metrics are only valuable if they improve the patient experience.
Session 2: What Role Does BGM Have in the Future?
Moderators
Robert Gabbay, MD, PhD
Chief Scientific and Medical Officer, American Diabetes Association, Arlington, Virginia, USA
John Pickup, MA, BM, DPhil, DSc, FRCPath
King's College London, London, United Kingdom
BGM in the 21st Century: A Perspective
Robert Schumm, MBA
President & CEO Ascensia Diabetes Care, Basel, Switzerland
Traditional blood glucose monitoring (BGM) will continue to play an important role in safely and effectively managing diabetes, especially:
○ As an active partner and supplement to CGM
○ As a cost effective and accurate option for needs of emerging markets
○ As a proven cost effective as well as simpler solution for many other people with T2D
Applications of BGM
Richard Bergenstal, MD
International Diabetes Center, Minneapolis, Minnesota, USA
Standardizing the ambulatory glucose profile (AGP) Report: BGM
Intermittent real time CGM and BGM
The key metric: number of tests per day or number of adjustments per week?
Is BGM Dead?
Steven Edelman, MD
Veterans Affairs Medical Center, UC San Diego, La Jolla, California, USA
CGM is available to only a small minority of people with diabetes worldwide.
BGM is not dead and will be around for many years/decades to come.
BGM used sparingly, wisely and individualized can be of tremendous benefit.
Blood Glucose Assessment in a Digitalized Sensor Ecosystem Environment: Future Perspectives
Andreas Pfützner, MD, PhD
Pfützner Science and Health Institute, Mainz, Germany
Past: Glucose is the one and only decision marker (for example: for determination of insulin doses).
Today: New invasive and non-invasive technologies start to provide relevant complimentary information.
Future: Glucose is only one component to determine treatment measures as part of a digitalized health care approach.
Trends in Capillary BGM
Irl Hirsch, MD
University of Washington School of Medicine, Seattle, Washington, USA
Local, regional, national, and international look at BGM
BGM is predicted to grow for another eight years internationally, although not as quickly as the rate of CGM growth.
BGM is not going away, rather it will continue to grow. There is an increase in incidence and prevalence, but it is still an economic challenge.
BGM has an illustrious history, with the patent for the first blood glucose meter filed in 1968 and SMBG going on to become an important component of the Diabetes Control and Complications Trial and intensive insulin therapy over the last 40 years. Although CGM use is now outgrowing SMBG in higher income countries, as it becomes the standard of care—an estimated compound annual growth rate of about 32% vs 5% (CGM vs SMBG) over the next few years 61 —the market for test strips in North America and many parts of the world will still be substantial in the foreseeable future and will likely increase steadily, albeit at a lesser rate than with CGM. Since the number of people with diabetes in the world continues to increase, the panelists believe that BGM will continue to be part of the future of diabetes care.
BGM is an important tool for taking and staying in control for people with both T1D and T2D. Although the increasing use of CGM will lead to a decline in BGM, the latter is certainly not dead, as emphasized, for example, by the recent World Health Organization’s Study on Global Ageing and Adult Health (SAGE), which assessed technology use in adults with T1D in five world regions (Latin America, West and Eastern Europe, Middle East and Asia). 62 Globally, fingerstick BGM was used by 92% of people with diabetes. Even in Western Europe where CGM use was highest (46%), BGM use was 83%. 63 However, glycemic control remains poor, with only 24% globally in the SAGE Study having an HbA1c <7%. 62 The same message of poor control comes from the 2010 T1D Exchange data in the United States where only ~30% had an HbA1c <7%, and control is even worse when real-world patients are studied. 64 BGM must therefore be used more effectively.
Traditional BGM, or SMBG, will continue to play a role in safely and effectively managing diabetes in three important ways. Firstly, accurate BGM is and will be needed as a partner or supplement to CGM, 65 most importantly when symptoms do not match readings,66,67 but also for calibration (routine or otherwise), when there is no value or trend arrow, when there is rapid glucose change, after recovery from hypoglycemia or when taking "sensor vacations."31,68,69 Secondly, BGM continues to be recommended as a simpler solution for T2D, where it has been shown to be cost-effective, particularly in insulin-users and when used to modify behaviour and pharmacological treatments.70,71 Thirdly, in emerging markets in low-income countries where access to CGM is restricted because of affordability and availability, BGM is a rational and cost-effective alternative for glycemic monitoring. 72
While current BGM devices will certainly continue over the next years, it was also pointed out that future developments in BGM are likely to fit into a new model of ‘integrated care’ where an encrypted, cloud-based patient database is fed disease monitoring information (eg, concerning diabetes, hypertension, coronary heart disease, respiratory conditions and so on) from multiple locations such as the home, hospital, pharmacy, ambulance and so on.73,74 The current focus on glucose and HbA1c for diabetes monitoring will likely be replaced in the future by a more holistic, "big data" approach with parallel assessment of glucose, by both established and new methods, and by direct and indirect technologies (eg, devices based on glucose-induced changes in osmotic pressure, or vascular or electroencephalographic signals), together with the monitoring of many other biochemical and physiological variables, made by multiple sensing systems in different body compartments, and supported by cloud-based AI for patient management. Figure 5 presents examples of other parameters that may be considered as part of a more holistic view of the body for patient management.
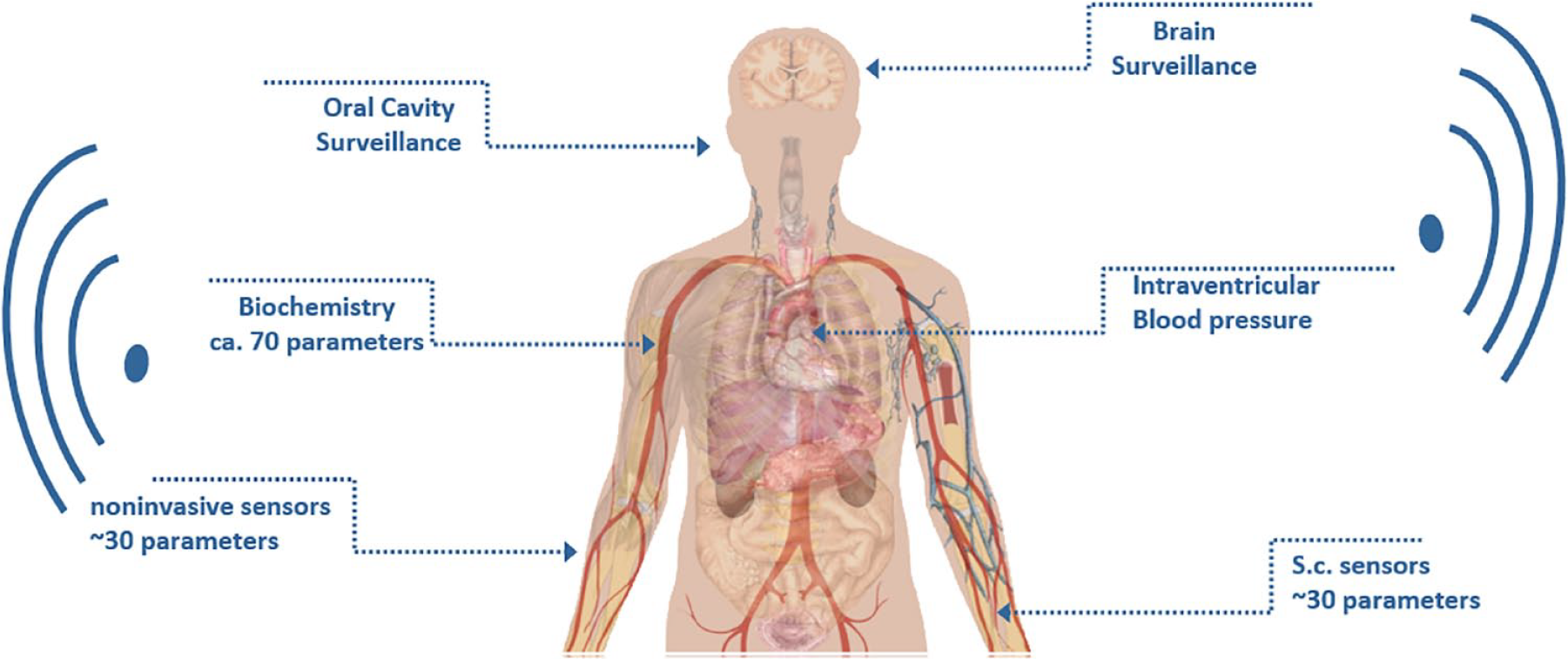
Examples of parameters that may be considered as part of a holistic view of the body for patient management. Figure provided by Andreas Pfützner, MD, PhD, Pfützner Science and Health Institute, Mainz, Germany.
It was pointed out that BGM will remain useful only if there is a plan to support and adjust lifestyle and medications according to patterns. Rational BGM testing strategies such as 7-point profiles, and paired and staggered testing, are underutilized in real-world diabetes management. The person with diabetes and the team must engage in interpreting and acting upon BGM data, and the key to increasing the rational use of BGM is probably to organize glucose data so that it is standardized, consistent and therefore easier to act upon. In this respect, BGM can learn from CGM in adopting the one-page AGP report for BGM devices. Furthermore, when continuous CGM use is precluded by affordability and availability, intermittent CGM can be used to point to when BGM needs to be done. 75
Session 3: What Are Recent Developments in Regulatory Science for Diabetes Devices?
Moderators
Alberto Gutierrez, PhD
NDA Partners, LLC, Rochelle, Virginia, USA
Yarmela Pavlovic, JD
Manatt, Phelps & Phillips, LLP, San Francisco, California, USA
Insulin Dosing Software
Naomi Schwartz, MS
FDA, Silver Spring, Maryland, USA
Landscape of current diabetes (software) devices (relevant regulations, special controls)
How different software devices fit into the interoperable ecosystem of diabetes devices
Upcoming developments (personal mobile devices as platforms for more diabetes software devices)
Automated Insulin Dosing Systems
Yiduo Wu, PhD
FDA, Silver Spring, Maryland, USA
The traditional PMA approach is not one size fits all.
The current, modular approach of iCGM system, ACE pump and iAGC (interoperable automated glycemic controller) allows interoperable systems to come under 510(k).
What it means for device developers and patients
Cybersecurity
Suzanne Schwartz, MD, MBA
FDA, Silver Spring, Maryland, USA
Criticality of FDA evolving along with the evolving landscape of device and healthcare cybersecurity.
Importance of Adoption of Total Product Lifecycle (TPLC) Approach across entire ecosystem
Imperative to advance and customize communications related to cybersecurity to suit specific stakeholder needs
Real World Evidence
Kenneth Quinto, MD, MPH
FDA, Silver Spring, Maryland, USA
Describe the main elements of FDA’s real-world evidence (RWE) Program
List FDA considerations for evaluating the use of real-world data to generate RWE for regulatory decisions
Explain program items the Agency plans to address in the RWE Program
In recent years, diabetes devices have rapidly evolved, warranting updates to their regulatory frameworks. Diabetes devices are increasingly using software driven by AI/ML algorithms. These components facilitate more personalized treatment and could be used for therapeutic and diagnostic purposes, predicting meals and activity and identifying the onset of diabetes. With respect to the regulatory requirements for algorithm-based devices, traditional regulatory frameworks necessitate frequent regulatory submissions because of the iterative nature of AI/ML. To address this issue, FDA released a draft discussion paper in 2019 that describes an updated regulatory framework allowing manufacturers to make previously agreed changes to an algorithm without requiring a new regulatory submission. 76 By formulating a predetermined change control plan (PCCP) with FDA, companies can propose types of allowable changes, the protocols for carrying out the changes, and the degree of evidence required to accept those changes. 76
Additionally, the session described regulatory advancements for other types of devices. With the expansion of the diabetes device marketplace, there has been a push for greater device interoperability, relying on wireless communication between components, mobile devices, and cloud-based services. CGM systems and controllers have historically been class III devices, and changes to any of these components within an automated dosing system has necessitated a new PMA application for both the individual component and the integrated system, which is taxing both for industry and FDA. Since 2014, the Agency has been exploring avenues for greater interchangeability of device components, and has created three new sets of class II device categories via the de novo pathway—the iCGM system, ACE pump, and iAGC. This will both incentivize device development in the diabetes space, including for smaller, less established companies, and will reduce the volume of submissions to the FDA.
While interoperable ecosystems streamline diabetes monitoring and management for people with diabetes and their healthcare providers, they also require reliance on wireless communication. Therefore, cybersecurity must be prioritized to protect the safety and privacy of patients. FDA views cybersecurity as a matter of patient safety and, therefore, has found 510(k) devices not substantially equivalent and PMA devices not approvable if there are cybersecurity concerns. FDA acknowledges that cybersecurity will continue to evolve and has consequently ensured that policies allow for flexibility and iteration. Significant importance has been placed on evaluating risks through the total product life-cycle. The Agency has also made a point to work with other organizations to explore cybersecurity options for legacy devices and to make effective use of software bills of materials to evaluate cybersecurity vulnerabilities for each device component. Additionally, FDA has encouraged the adoption of coordinated vulnerability disclosure policies to facilitate cybersecurity vulnerability assessment and disclosure. 77
Efforts have additionally been made by FDA to streamline development and approval of drugs and biologics through the RWE program. RWE is the clinical evidence pertaining to the use of a medical product and is derived from RWD, which can be collected from a variety of sources, including the EHR, claims and billing data, and patient-generated data. The concept of RWE was introduced under the 21st Century Cures Act to address approval of drugs for new indications and to satisfy post-approval study requirements. RWE can inform drug effectiveness but must be fit for use, as data can vary in terms of quality, consistency, and relevance to the scientific question at hand. FDA leverages principles from its 2013 guidance document on electronic healthcare data to assess RWD and the resulting RWE. The Agency is continuing to develop guidances pertaining to the use of RWD, including electronic informed consent, as well as a roadmap for RWE data standards. Additionally, FDA has implemented an RCT Duplication project with the goal of replicating clinical trials to determine whether findings can be reproduced when utilizing RWE. 78 Figure 6 presents a framework for evaluating RWD and RWE for use in regulatory decisions.
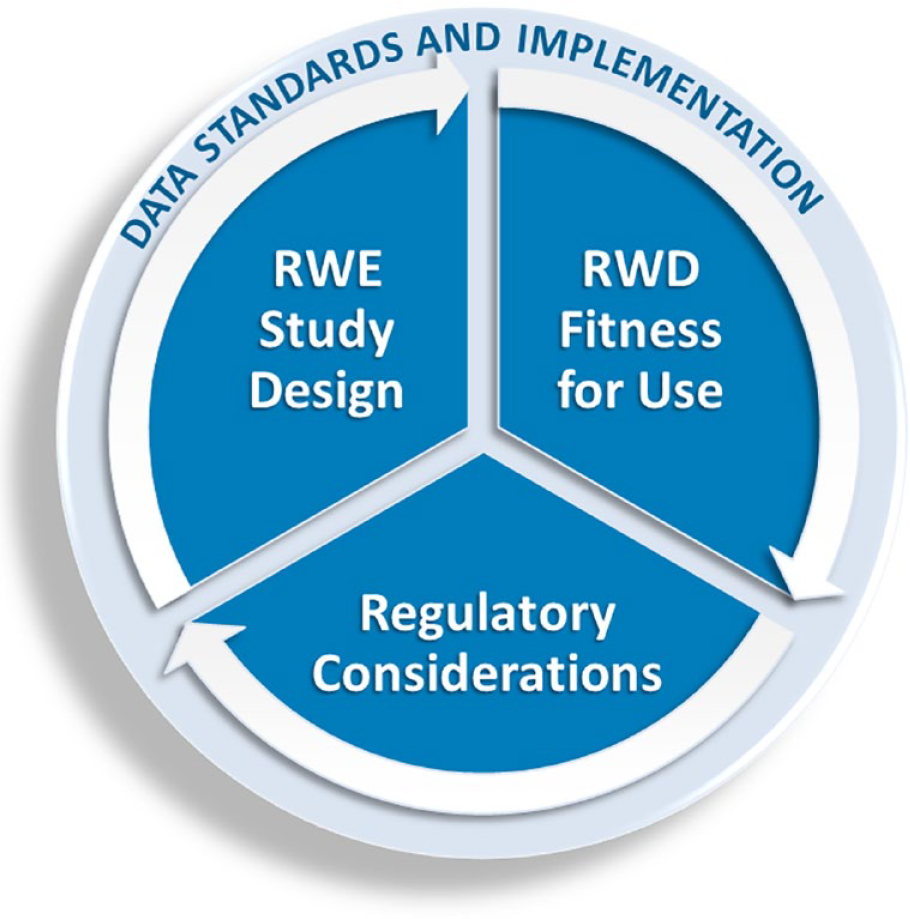
Framework for evaluating RWD and RWE for use in regulatory decisions. Figure provided by Kenneth Quinto, MD, MPH, FDA, Silver Spring, Maryland, USA.
Finally, FDA has developed the MyStudies platform to facilitate collection of patient-recorded outcomes using a mobile app on patients’ own mobile devices, principally for studies of new drug products. Several clinical studies utilizing the MyStudies platform are in progress or planned. In the context of the COVID-19 pandemic, MyStudies can additionally be used to collect informed consent remotely and has been utilized for this purpose in 79 trials to date. 79
Session 4: What is the Role of Pharma in Digital Health?
Moderators
Juan Espinoza, MD, FAAP
Children’s Hospital Los Angeles, University of Southern California, Los Angeles, California, USA
Matthew Taylor, CFA
US Medical Supplies & Devices Analyst, UBS, New York, New York, USA
Diabetes is a Wicked Problem
David Kerr, MBChB, DM, FRCP, FRCPE
Sansum Diabetes Research Institute, Santa Barbara, California, USA
Diabetes and COVID-19 is a Syndemic.
What are metric of success for digital health?
Digital health will drive personalized prescribing.
Unleashing the Potential of Connected Insulin Pens
Howard Wolpert, MD
Lilly Cambridge Innovation Center, Cambridge, Massachusetts, USA
Turning insulin data into actionable insights: the development of insulin dosing metrics
Understanding the behavioral and attitudinal factors underlying insulin dosing behavior
Communication with the patient with diabetes: ensuring that insulin data empowers and fosters engagement in self-care, and does not become a demotivating report of errors and failure
New Components for the Virtual Digital Diabetes Clinic That Will Improve Outcomes and Efficiency
Anders Dyhr Toft, MD, PhD, eMBA
Novo Nordisk, Copenhagen, Denmark
Clinicians need data for decision-making, both on blood glucose and insulin dosing. Smart pens and blood glucose data from connected BGM systems and CGM systems will provide reliable data that can be leveraged in the patient/HCP dialogue to improve outcomes.
Clinicians should spend more time on high-risk patients and less time on low-risk patients. Triaging of patients into high- and low-risk groups will be possible based on fluctuations of blood glucose as well as data on insulin injection adherence.
There is insufficient time for titration of insulin in both primary and secondary care. Digital therapeutics can safely and effectively titrate patients to blood glucose target—with minimal or no HCP intervention.
Levels of Complexity and Reward from Digital Health
Christopher Boulton, BSc
Sanofi, Bridgewater, New Jersey, USA
Digital health is becoming more and more a reality, accelerated by COVID-19.
What are the different options and approaches to digital health available for pharma?
What does a solution look like for diabetes in the near future?
Digital Health Changes How Patients Are Using Drugs: A New Parameter in Drug Development
Sam Collaudin, PhD, jMBA
Independent consultant, Marburg, Germany
Digital health is becoming a key element in diabetes management.
Digital health introduced new metrics as TIR and new tools that influence the way patients interact with their drugs.
Drug development evolves to integrate these new tools in clinical trials and to partner with device and digital health technologies to combine medical benefits.
First described in 1973 by Rittel and Webber, 80 the concept of a wicked problem refers to a complex and challenging issue with no obvious solution and often involving multiple domains, ranging from behavior and science to public policy and economics. 80 Climate change, criminal justice reform, and poverty are all often described as wicked problems. Wicked problems are particularly difficult to address because of missing and often contradictory information, and the fact that addressing one component can worsen another. Diabetes has been described as a “wicked problem,” given the interconnectedness of pathophysiology, social determinants of health, behavioral and environmental factors, and outcome inequities. 81 Diabetes and COVID-19 together have created a syndemic, “conditions that are clustered within social groups according to patterns of inequality deeply embedded in our societies.” 82
Telehealth use has risen rapidly since March 2020. However, it is not clear that all patients have equitable access to the technologies needed to participate in digital health, nor that all components of diabetes care are being delivered. Diabetes education is sometimes being omitted, which is critical to engaging patients and improving outcomes, particularly among patients with limited health literacy. 83 Additionally, in the United States, 1 in 4 Medicare beneficiaries lack digital access. 84 The disparities go deeper than that in diabetes; even though people of color shoulder a disproportionate burden of disease, they are a tiny proportion of patients who are included in the research and development process, and few of the clinicians that care for them are themselves from underserved communities. 85
One approach is for pharma to offer technologies that can be integrated equitably. These technologies need to be usable, accessible, reliable, interoperable, existential (in other words, context aware, that the implementation does not aggravate another problem), and episodic. Digital health can take existing products and expand their use into new markets. Additionally, pharma should focus on technologies that do not add to the time burden of managing disease. Finally, pharma as an industry should consider metrics of success that consider not just genetics and biology, but also behavior, psychology and society. 86 The concept of pharmacoadherence—whether patients will actually take a medication—should be elevated to the same status as the more traditional pharmacokinetics (PK)/pharmacodynamics (PD) in evaluating the effectiveness of drugs. 87
Technology can also help drive innovation, providing new insights into personalized health and opportunities for drug discovery. For example, CGM systems can be used to discover patterns of dysglycemia that may be amenable to pharmaceutical intervention. 88
One panelist’s view is that the role of pharma is to create a digital ecosystem in which devices, data, applications, and communication are integrated to create interoperable platforms that facilitate personalization of care, patient support, and better health outcomes. 87 Figure 7 presents a diagram of this ecosystem. In order to achieve this, it will require a shift from “a priori” thinking, designing systems that we hope patients will use, to “a posteriori” thinking—developing tools from empirical observations of real world utilization patterns and behaviors of patients.
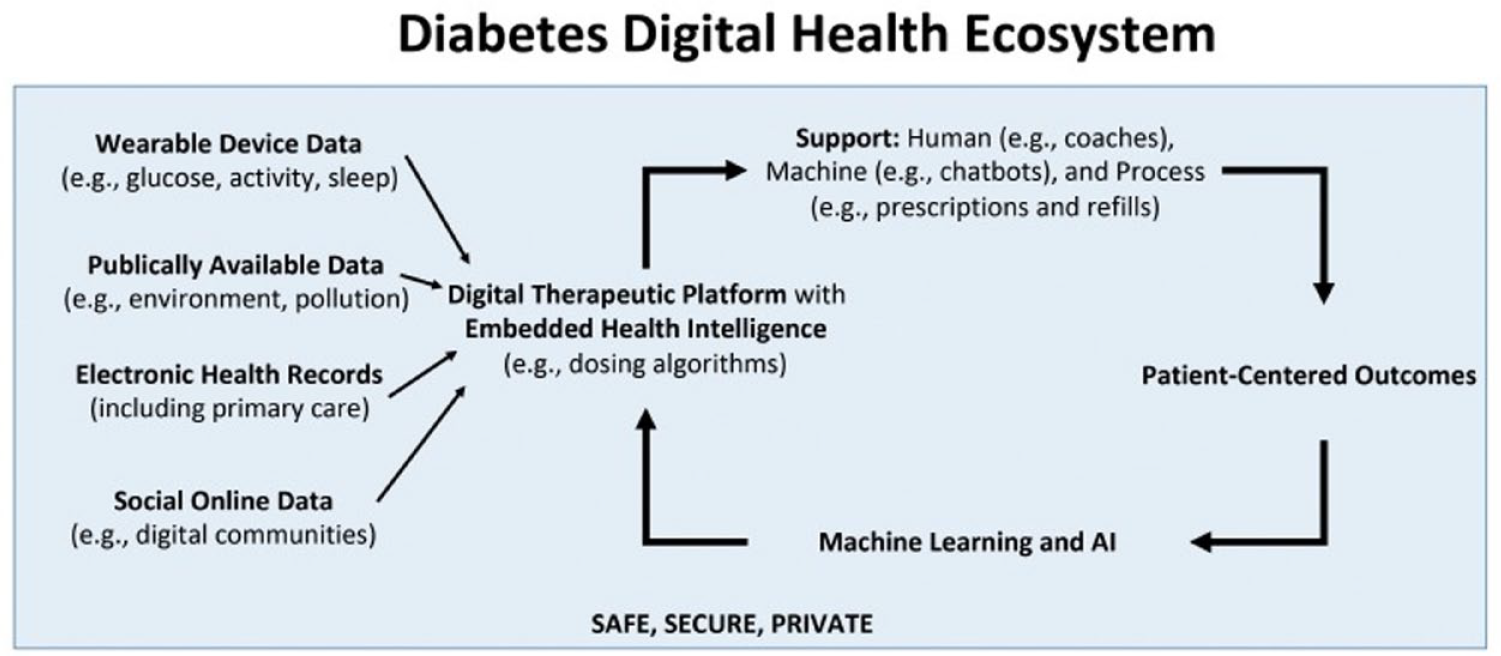
A diagram of a diabetes digital health ecosystem. Figure provided by David Kerr, MBChB, DM, FRCP, FRCPE, Sansum Diabetes Research Institute, Santa Barbara, California, USA.
The proliferation of CGM has led to multiple clinically significant metrics for glucose control being defined in recent years.2,89,90 Connected or smart insulin pens have the potential to provide the data necessary to optimize insulin therapy. 91 Improving insulin use can decrease TAR, minimize TBR, and ultimately maximize TIR for patients. A recent study of connected pens demonstrated their value in understanding patient behaviors around missed insulin doses, patient fears, and emotional engagement. 92
One challenge of this new data source is that interpreting diabetes data generated by devices is time consuming and requires clinical expertise. The goal for pharma should be to simplify insulin data into clinically meaningful and actionable metrics that can be prioritized, implemented by a wide range of providers, and understood by patients. An important consideration is that these data should not be seen as “compliance reports,” but rather an opportunity to facilitate conversation between the patient and provider and review, discuss, troubleshoot, and celebrate their care.
Connected pens can provide data about a number of dosing practices, and potential metrics that can be derived from a connected insulin pen are presented in Table 3. Comparing CGM and connected pen data can help identify a variety of dosing patterns and behaviors. As more data is gathered about these new tools, it will be important to develop a simple and standardized set of Insulin Metrics to prioritize clinical problems.
Potential Metrics That Can be Derived From a Connected Insulin Pen.

The concept of the virtual digital diabetes clinic is a data driven approach to the remote management of patients with diabetes. This may include data from CGM systems, connected pens, the EHR, glucometers, and other data sources. The goal is to present a comprehensive picture of the patient that can facilitate disease management by surfacing risk stratification, behavioral insights, and pharmacoadherence. Novo Nordisk (Bagsværd, Denmark) recently did a small study in Sweden of their NovoPen 6 connected insulin pen for patients that already were using CGM. After nine months of study, participants had on average two more hours a day in target range, this coming mainly from less TAR but also less TBR. The reusable NovoPen has a dose log with 800 events, fits all Novo Nordisk insulin cartridges and uses near field communication and open standards to transmit data. Novo Nordisk is also developing a Bluetooth add-on device that can be used with existing prefilled pens to count doses.
From an interoperability perspective, Novo Nordisk is championing an open-partnership strategy across the industry, making their connected pen data available on the platforms of other manufacturers and third party aggregators. This approach is intended to be easier on patients and providers who can continue to rely on the platforms they already use, but also creates an opportunity to increase our understanding of patient behaviors by sharing data.
Pharmacoadherence was again brought up during this presentation. Several studies have demonstrated that digital dosing support can improve glucose control.93-96 This seems to be driven by adherence to prescribed dosing regimens. Finally, it was proposed that connected pen data, alongside CGM data and other relevant sources, could be used to risk-stratify patients to help guide clinical management, identify potentially at-risk patients.
Digital therapeutics have been a growing trend that accelerated in 2020 because of the COVID-19 pandemic. Although promising, there are still significant questions about the role of digital therapeutics. The development of digital therapeutics is distinct from traditional drugs, including shorter development timelines, lower research and development costs, low to medium market protection, and low to medium revenue potential. Another major difference is the approach to development. Traditional drug development is similar to the classic “waterfall” software engineering methodology, in which incremental changes lead to the final product, but the interim steps often do not meet or satisfy the user’s needs. Instead, pharma needs to embrace an Agile methodology, in which each stage of development is intended to maximize utility for users, even if it is not the final product. As an industry, it is important to nurture and support new technologies, particularly as they are traversing the wide chasm between early adopters and the general population.
A recent survey by the Association of Diabetes Care & Education Specialists (ADCES) showed that patients have difficulty with tracking information effectively. Almost two thirds of people with diabetes wished it was simple to track their insulin use and glucose levels. And while 95% of respondents said they knew that it is important to track their insulin use, 62% reported having been too busy and or forgotten to log their insulin use. 97 Digital health, including devices and mobile applications, may be able to address these gaps. Survey respondents that connected insulin devices could help give them more personalized understanding of their diabetes (79%), make tracking insulin use less time consuming (78%), and make them feel more empowered when it comes to managing their diabetes. The ultimate goal of pharma-developed digital health solutions should be to contribute to a patient-centered connected ecosystem that empowers patients and providers to use technology and data to personalize care and deliver the right care at the right time and right place. While there may be significant complexities in achieving this goal, the rewards should be well worth it for patients, providers, and industry.
Digital health is being incorporated into every aspect of diabetes care, with a proliferation of telehealth, mobile apps, connected devices, and online platforms. Interestingly, the use of technology is changing the way patients use their treatments. In an analysis of the 2017 to 2018 T1D Exchange registry, patients who used CGM systems took 15% more boluses per day than patients who did not. 98 Figure 8 presents the data from this 2017 to 2018 T1D Exchange registry. CGM systems are becoming more common as metrics in clinical trials and clinical development.
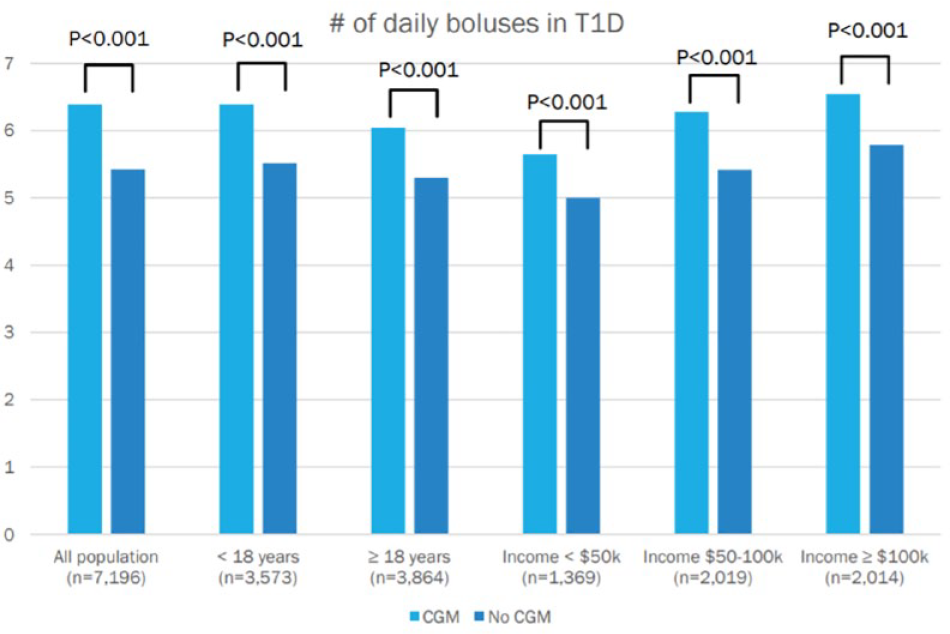
Analysis of data from the 2017 to 2018 T1D Exchange Registry comparing the number of daily boluses in patients with T1D using insulin pumps with or without a CGM. Figure provided by Sam Collaudin, PhD, jMBA, Independent consultant, Marburg, Germany.
A notable example of the impact of digital health on drug development is the case of the development of Pramlintide and insulin combinations. This combination results in improved postprandial hyperglycemia and glycemic variability. 99 Different companies are developing fix combinations of pramlintide and prandial insulin with two different strategies. The first strategy is to use regular human insulin, the prandial insulin with a PK that fits a slow increase of glucose in the blood because of pramlintide. This solution bears the flaws of human insulin with insulin that stays active in the blood hours after injection. 99 The second strategy is to combine rapid-acting insulin or ultra-rapid acting insulin. The use of digital technologies as automatic insulin delivery algorithms allows to leverage the benefits of these rapid insulins as it adapts bolus injections to avoid early hypoglycemia. 100 This case study is one example of how new digital technologies can influence the design during drug development.
Session 5: Artificial Pancreas (What Do Patients Like and Dislike About Their Automated Insulin Delivery Systems?)
Moderators
Bruce Buckingham, MD
Stanford University, Stanford, California, USA
Alexander Fleming, MD
Kinexum Services, LLC
Harpers Ferry, West Virginia, USA
During this session, four people discussed their experience with four different AID systems, providing real-life perspectives compared to the more theoretical or data-oriented presentations about these systems. The four different AID systems discussed were the CamAPS FX (CamDiab, Cambridge, United Kingdom), Insulet Omnipod (Insulet Corporation, Acton, Massachusetts, USA), Tandem Control-IQ, and Medtronic 780G.
One panelist, whose daughter uses the CamAPS FX, discussed how one of his family’s favorite aspects about the AID system is the control of the system overnight, allowing for them to sleep without worrying if their daughter has high or low blood glucose levels. He also mentioned that it has been helpful for his daughter to have one less thing to worry about while she has been busy with schoolwork. However, one concern he has about the system is when the AID system delivers insulin that leads to a drop in blood glucose that is just slightly low, there are constant alarms that are triggered until the glucose levels stabilize. In addition, the parent mentioned that his daughter did not like that the CamAPS FX can only be used with an Android phone, making it inconvenient for her to bring two phones since she is an Apple iPhone user. Overall, the system has helped his daughter to have less highs and better HbA1c levels compared to when she used standard pump therapy.
The next panelist, whose daughter uses the Insulet Omnipod system, echoed much of the benefits mentioned by the panelist representing the CamAPS FX system. She discussed their family being able to sleep better at night, and not having to worry about highs and lows as much. One negative she brought up was having to also remember to bring around the wireless Omnipod Personal Diabetes Manager (PDM). However, even if the PDM is not with the user, it is still able to adjust insulin levels. They also mentioned the benefit of not having to do as many boluses throughout the day, and an improved TIR.
The panelist using the Tandem Control-IQ system discussed how much he has also benefited from the system, highlighting how helpful it is to wake up with normal blood glucose levels. He discussed how since the Tandem Control-IQ system modulates basal rates, it reduces the number of times he is hypoglycemia as well as the severity of hypoglycemia. Another positive he mentioned is not needing to worry as much about overtreating when he is in hypoglycemia, or think about how much insulin to give himself when he is in hyperglycemia. He also felt that the app and the automatic data upload aspects of the system were very convenient. His TIR has improved as well. He points out that one factor to consider when using AID systems overall is ensuring that one’s settings, including the range of basal profile, carbohydrate ratios, and insulin sensitivities, are all correct in order to have the best experience with the system.
Finally, the panelist using the Medtronic 780G discussed how helpful the system has been in reducing the lows and highs, which used to be something she struggled with prior to using the system. The system has mitigated the problems of forgetting to bolus. She has rarely had problems overnight because the system has helped manage her glucose levels well. Her HbA1c has also improved after starting to use the system.
Session 6: Can We Build a Fully Automated AP Without Pre-Meal Announcements?
Moderators
Jeffrey Joseph, DO
Thomas Jefferson University, Philadelphia, Pennsylvania, USA
Laurel H. Messer, PhD, RN, CDCES
Barbara Davis Center for Childhood Diabetes, Aurora, Colorado, USA
Anticipating Glycemic Disturbances to Control Unannounced Meals and Exercise
Marc Breton, PhD
University of Virginia, Charlottesville, Virginia, USA
Glycemic disturbances such as meals and exercise impact glycemia much faster than current insulin kinetics and for hours to come, simple feedback systems are unlikely to be sufficient to appropriately control them.
The combination of multisensory arrays, personalized pattern recognition, and realtime pattern identification can provide early information to an AID system in order to fend off such disturbances.
Early in-silico and clinical trials indicate such AID system are safe and improve glycemic control.
Fully Automated AP Without Pre-Meal Announcements
Ali Cinar, PhD
Illinois Institute of Technology, Chicago, Illinois, USA
Meal estimation from CGM data (incremental estimation of carbohydrate, detection of meal consumption, mini-boluses given incrementally) use a different method than manual entry or historical data.
○ Meal prediction from historical data, extracting trends and habits of the individual
○ Use systems engineering, ML, AI and multivariate statistical techniques to detect meal consumption from CGM data in real time and estimate carbohydrate consumed from CGM data and physiological models. Deliver successive mini boluses proportional to carbohydrate estimated.
○ The second strategy serves as safety net for people who prefer manual entry when forgetting to enter meal info or estimating incorrect carbohydrate.
Accuracy of the meal estimate is critical for manual control (pumps, pens and injections). However, in AID, the feedback control system will make additional corrections in insulin dose (minibolus or basal) when the meal information is less accurate.
The automated meal estimation is embedded in our multivariable artificial pancreas (AP). By interpreting physiological data reported in real time from a wearable device, the reason for a glucose concentration increase (meal, high-intensity physical activity or acute psychological stress), the multivariable AP optimizes insulin dosing decisions. The multivariable AP can mitigate both meal and physical activity effects without any manual entry.
Eliminating Meal Announcements
Roman Hovorka, PhD, FMedSci
University of Cambridge, Cambridge, United Kingdom
What is possible with current insulin analogues
What is not possible with current insulin analogues
Way forward—pharmaceutical and algorithmic developments
Multivariable Control for the Artificial Pancreas
Frank Doyle, PhD, CPGS
Dean, Harvard John A. Paulson School of Engineering & Applied Sciences, Cambridge, Massachusetts, USA
Topic for this talk is “multivariable” feedback control for the AP—looking beyond the current paradigm of one-action (insulin) and one-measurement (glucose)
Enhanced measurements could include sensors related to physical activity, alternate metabolites, and inferential signals for food
Will discuss a system design approach to a truly multivariable controller, including the possibility of an unbalanced number of inputs and outputs
Current AP systems require the user to announce carbohydrate content prior to consumption of meals in order to initiate sufficient bolus insulin delivery, rendering systems reliant on user input. This is because of the limitations in current subcutaneous rapid acting insulin PK and PD, and to glucose sensors measuring glucose in five-minute intervals, potentially delaying vital feedback to closed-loop systems. In order to progress to a fully automated system, novel approaches are needed to autonomously contend with meals and the resultant glycemic disturbances.
One approach to this problem is the inclusion of new sensor inputs and ML algorithms to anticipate meal disturbances. Novel sensors could include accelerometers, galvanic skin response, skin temperature, or heart rate data, and may be collected passively without user input, increasing automation and decreasing user burden. 101 An incremental approach may be to qualitatively announce meals without a carbohydrate count, or use photography-based input to estimate the anticipated glycemic disturbances. 101 All of these inputs may be used to create more sophisticated model-predictive-control algorithms that incorporate pattern recognition and meal detection.102,103
A second approach would be to innovate on rapid acting insulin delivery with faster PK/PD profiles. 104 In addition to new formations of subcutaneous insulin, alternative delivery pathways (eg, intraperitoneal) may increase the PK/PD for bolus insulin delivery. This would decrease the need for new models and new inputs to detect meal events, as the faster acting agents could more easily contend with sudden increases in glucose levels.
Finally, multi-hormone approaches to fully closed loop AP could deliver safe and effective insulin delivery in the absence of meal announcements. The inclusion of glucagon allows for a “foot on the gas and foot on the brake” approach, though applicability may be limited to sub-populations who are interested in actively managing 2 hormones, as well as the limited commercial availability of a shelf-stable formulation.105,106 Pramlintide is another promising approach, which can be co-formulated with insulin, and could effectively slow the glycemic disturbance to better match the insulin action needed.107,100
Session 7: What Is the Role of Ultra Short Acting and Ultra Long Acting Analog Insulins?
Moderators
Gerold Grodsky, PhD
University of California San Francisco, San Francisco, California, USA
Tim Heise, MD
Profil, Neuss, Germany
The Clinical Need for Ultra Short Acting and Ultra Long Acting Analog Insulins
Anne Peters, MD, FACP, CDE
University of Southern California, Los Angeles, California, USA
Use of CGM has increased awareness of the limits of subcutaneous insulins.
Newer agents are available that could potentially improve outcomes.
Real world experience varies but for some there is true benefit.
Ultrarapid Insulin Aspart Injection (Fiasp Insulin). What Will Be Its Role?
Athena Philis-Tsimikas, MD
Scripps Whittier Institute for Diabetes, UC San Diego, San Diego, California, USA
Understand the mechanism of action for faster aspart
Brief review of key clinical study outcomes for T1D and T2D
Identify which patients might benefit from transition to faster aspart and how to do this
Ultrarapid Lispro (Lyumjev) Insulin. What Will Be Its Role?
Leslie Klaff, MD, PhD
Rainier Clinical Research, Renton, Washington, USA
PK and PD of ultra-rapid lispro (URLi), also known as Lyumjev insulin developed by Eli Lilly and Company (Indianapolis, Indiana, USA)
Key clinical trial data of URLi insulin
Possible role in treatment of T1D
Hepatic Directed Insulin
Bruce Bode, MD
Atlanta Diabetes Associates, Atlanta, Georgia, USA
Physiological endogenous pancreatic insulin is released directly to the liver, primarily in bolus amounts at mealtime, allowing the liver to take up 30% to 65% of mealtime glucose and to convert that glucose to glycogen for storage. Mealtime hepatic glucose uptake is a primary mechanism for preventing peripheral hyperglycemia, and the hepatic conversion of glycogen back to glucose for release into peripheral circulation is the primary mechanism for preventing routine hypoglycemia.
Injected peripheral insulin is non-physiological because disproportionate exposure of insulin to adipose and muscle leaves the liver insulin deficient, enhancing post prandial hyperglycemia and rendering the liver unable to make glycogen and subsequently release glucose to prevent hypoglycemia.
Designed to provide hepatocyte targeting of commercial insulin, Hepatic-Directed Vesicle insulin (HDV) targets a percentage of injected insulin to the liver, allowing for glycogen formation leading to less hypoglycemia with no change in peripheral insulin PK.
Inhaled Insulin for Pulmonary Delivery
Eda Cengiz, MD, FAAP, MHS
Yale University School of Medicine, New Haven, Connecticut, USA
Unique pharmacokinetic and pharmacodynamic characteristics of inhaled insulin
Inhaled insulin’s role in mitigating post-prandial hyperglycemia during open and closed-loop treatment
Brief summary of what is on the horizon for inhaled insulins
Weekly Basal Insulin Icodec. . .the Way of the Future!
Julio Rosenstock, MD
Dallas Diabetes Research Center at Medical City, Dallas, Texas, USA
Once weekly insulin icodec has three amino acid substitutions that lead to greater molecular stability, reduced enzymatic degradation, and reduced receptor mediated clearance. It also has the removal of B30 and a C20 Icosane Fatty Diacide.
Phase 2 trials have shown that weekly Icodec is as effective as daily Glargine U100, and sometimes shows statistically significantly better results than Glargine U100.
Weekly Insulin Icodec show promise in improving diabetes management compared to currently used Glargine U100 insulin, and will need to undergo Phase 3 trials.
Despite all improvements in insulin therapy and glucose monitoring, less than 50% of patients with T1D achieve HbA1c-levels <7.5%. Patients often suffer from high postprandial glucose values, even when they do adequate carb counting, keep an injection-meal interval and have a good understanding of insulin dose adjustments. Other patients experience shortcomings in basal insulin therapy with high glucose values in the morning because of a dawn-phenomenon or hypoglycemia between meals. Therefore, newer insulins are needed, both longer-acting for basal and faster-acting for prandial insulin therapy. Eventually, differently acting insulins that are closer to endogenous insulin, in particular insulins responding to glucose levels, will be needed to achieve optimal control.
The different kinds of insulin that were discussed were ultrarapid insulin aspart injection (Fiasp Insulin), URLi (Lyumjev) Insulin, HDV, Inhaled insulin (Afrezza), and Weekly Basal Insulin Icodec.
Fiasp, developed by Novo Nordisk, is one of the new ultrafast insulins that are now available on the market. Because of new excipients (niacinamide, arginine), the onset of appearance of Fiasp in the bloodstream is twice as fast, and the insulin exposure within the first 30 minutes is almost twofold higher with Fiasp than with insulin aspart.108,109 In clinical studies, Fiasp improved postprandial glucose levels110-112 and, to some extent, also HbA1c-values vs insulin aspart when administered immediately before the meal.110,111 With postprandial administration, Fiasp achieved non-inferior glucose control to insulin aspart injected before the meal. 110 However, elevations in glucose levels were observed sometime after dinner which led to slightly higher HbA1c-values in one study. 112 Overall, ultrafast insulins like Fiasp might help patients to get a bit closer to target, but the timing of injection in relation to a meal and, at least in some patients, also basal insulin therapy has to be adapted. The option of post-prandial administration will help patients to control postprandial glucose values even when they have forgotten to take their insulin before the meal.
Similarly to Fiasp, URLi (Lyumjev) which was developed by Eli Lilly and Company, has a faster onset of absorption/action and a shorter duration of action than insulin lispro due to 2 excipients: treprostinal, a vasodilator, and citrate which enhances vascular permeability.113-115 In clinical studies in people with T1D URLi achieved non-inferior glycemic control (HbA1c) to insulin lispro, but improved postprandial glucose control, in particular after breakfast and lunch. 116 Hypoglycemia rates were similar between URLi and lispro, however, late hypoglycemia more than four hours after the meal were 30% lower with URLi. 116 Likewise, URLi improved postprandial glucose in patients on insulin pumps vs lispro with a significantly lower rate of nocturnal and post-meal hypoglycemia and significantly less time in hypoglycemia. 117 While injection site reactions or pain at the injection site were slightly more frequent with URLi, these events were mostly mild or moderate.116,117 Overall, ultrafast insulins like URLi might be beneficial for patients with frequent postprandial hypoglycemic events. Because of their shorter duration of action, they will also be attractive candidates for advanced hybrid closed loop systems.
Endogenous insulin primarily acts on the liver where it transforms glucose into glycogen which can be released to prevent peripheral hypoglycemia. In contrast, exogenous insulins reach adipose and muscles tissue first, leaving the liver insulin deficient. HDV is a novel disk-shaped insulin delivery system that uses a phospholipid/biotin matrix to target proteins such as insulin to the liver. 118 Figure 9 presents a diagram of an HDV. HDV can be used in vials and in pumps. 119 In a recent open-label trial in 61 people with T1D (mean HbA1c 6.9% after a three-month optimization run-in period with lispro and degludec and CGM), HDV Lispro given over three months showed comparable glycemic control (HbA1c 7.0%), but a significant reduction in Level 2 hypoglycemia, in particular overnight. Insulin doses were comparable between the optimized run-in and the treatment period. 120 The results were in line with previous trials that also indicated a reduction in hypoglycemia with hepatic directed insulin. Phase 3 trials, blinded and with an active comparator in the treatment period, are planned for 2022.
Diagram of a hepatic-directed insulin vesicle. Figure provided by Bruce Bode, MD, Atlanta Diabetes Associates, Atlanta, Georgia, USA.
The only FDA-approved inhaled insulin, Afrezza 121 which was developed by MannKind (Westlake Village, California, USA), has a time-action profile with a very fast onset and a very short duration of action which comes close to the endogenous first phase insulin release after meal intake. Therefore, Afrezza has the potential to improve early postprandial glucose control and at the same time minimize the risk of late postprandial hypoglycemia. Because of its short duration of action a second dose of Afrezza might be necessary for slowly absorbed meals. 122 In a clinical study in patients with T1D on CGM, two-third of patients took adequate post-meal doses of Afrezza. 123 This “compliant” patient group achieved more TIR and less time in hypoglycemia than patients on s.c. insulin aspart without gaining any weight in the four week treatment period. Afrezza also improved early postprandial glucose levels 60 and 90 minutes after a meal without increased hypoglycemia in patients on hybrid-closed loop. 123 Afrezza therefore may allow treatment intensification without increasing the risk of hypoglycemia. Because of its short duration of action, Afrezza is a good option for correction doses and for patients afraid of exercise-induced hypoglycemia or patients that like to snack. While Afrezza leads to small, non-progressive decline in lung function (FEV1), there was no excess of pulmonary safety events in clinical trials.124,125
Insulin icodec is a new basal insulin analogue with a half-life of one week. 126 In a double-blind, double-dummy Phase 2 clinical trial, insulin-naïve patients with T2D were randomized to receive insulin glargine once daily or insulin icodec once weekly for 26 weeks. 127 Patients on icodec achieved numerically lower, but not statistically different HbA1c-values than those on glargine (6.7% vs 6.9%). Hypoglycemia rates were low with both insulins, but level 1 hypoglycemia was more frequent with icodec, whereas level 2 and level 3 hypoglycemia rates were similar, as was the duration of hypoglycemic events. 127 A second trial comparing different titration algorithms for icodec showed higher hypoglycemia rates (levels 2 and 3) with weekly icodec dose changes of 28 U vs changes of only 21 U. 128 A third trial looked at initiation of icodec treatment in patients already on basal insulin. CGM-data showed that the initial increase in fasting glucose levels observed in the first weeks of icodec treatment (before it reaches steady-state) can be prevented by giving a loading dose (twice the usual dose) with the first injection. 129 Again, icodec achieved numerically, but not statistically significantly lower HbA1c than glargine, however, the higher values for TIR with icodec did reach statistical significance. Hypoglycemia rates were comparable in this study. Overall, icodec has the potential to improve treatment acceptance and facilitate T2D management in patients needing basal insulin. 129 The role of once weekly insulin icodec will be further investigated in a Phase 3 program.
Session 8: How Should the Performance of CGM Systems Be Evaluated?
Moderators
Craig Kollman, PhD
Jaeb Center for Health Research, Tampa, Florida, USA
Carlos E. Mendez, MD, FACP
Medical College of Wisconsin, Milwaukee, Wisconsin, USA
CGM Accuracy: A Clinical Perspective
Timothy S. Bailey, MD, FACE, CPI
AMCR Institute, Escondido, California, USA
How accuracy is measured
What are the key areas of interest that contribute to clinically-important accuracy
When better accuracy is needed
A Review of the Proposed Performance Metrics for Continuous Interstitial Glucose Monitoring, Under Development by the Clinical and Laboratory Standards Institute (CLSI)
David Horwitz, MD, PhD
DLH Biomedical Consulting LLC, Las Vegas, Nevada, USA
Standardized nomenclature and definitions
Evaluation of point accuracy, trend accuracy, stability and reliability
Considerations for clinical studies
IFCC WG-CGM: Improving Standardization of Continuous Glucose Monitoring
Guido Freckmann, MD
Institut für Diabetes-Technologie, Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany
The International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) Working Group on Continuous Glucose Monitoring aims at defining:
1. the measurand, consisting of (a) substance, (b) unit, and (c) matrix, as defined by the international vocabulary of metrology,
2. suitable means for establishing the traceability, including the measurement uncertainty, of glucose values obtained by CGM systems to materials and methods of higher metrological order according to ISO 17511,
3. procedures suitable for assessment of the analytical performance of CGM systems, and
4. metrics and corresponding minimum acceptance criteria for the analytical performance of CGM systems.
The DTS Guideline for Continuous Glucose Monitors and Automated Insulin Dosing Systems in the Hospital
Rodolfo J. Galindo, MD, FACE
Emory University School of Medicine, Atlanta, Georgia, USA
DTS convened a multispecialty international panel to standardize and provide recommendations on the use of CGM and automated insulin dosing systems in the hospital:
1. for clinical practice (to use the technology optimally)
2. for research (to improve the safety and effectiveness of the technology)
3. for hospital policies (to build an environment for facilitating use of these devices)
Experts discussed five topics:
1. continuation of home CGM systems after hospitalization
2. initiation of CGM systems in the hospital
3. continuation of AID systems in the hospital
4. logistics and hands-on care of hospitalized patients using CGM systems and AID systems
5. data management of CGM systems and AID systems in the hospital.
The current COVID-19 pandemic created the need for innovative approaches for glycemic monitoring in the hospital.
Experts believe CGM and AID have the potential to overcome current limitations of glycemic monitoring in the hospital and improve patients outcomes, but more research is needed.
How the Security Performance of CGM Systems (And Other Connected Diabetes Devices) Should Be Evaluated
David Kleidermacher, BS
Google, Mountain View, California, USA
Why “measure” performance for security of CGM systems and other connected diabetes devices (CDDs)?
How to measure (and build trust in) security for CGM systems and other CDDs
Current status of performance evaluation program for security of CGM systems and other CDDs
There have been several factors that should be considered to evaluate CGM systems. From a clinical perspective, CGM systems are increasingly becoming a critical tool for the outpatient management of diabetes. The optimal device would offer high accuracy represented both by precision and trueness. Performance may vary by glucose levels with higher values leading to more bias and less precision. Areas affecting accuracy and opportunities for improvement include, Preanalytical: such as pressure on sensor site, 130 X Rays, 131 lipohypertrophy, 132 Analytical: Interfering substances,133,134 end-stage renal disease/dialysis, 135 and Postanalytical: hardware (parakeet, nightrider), software (Nightscout, Looping, tomato), and data analysis (human error).136,137 Accuracy may even be more important for patients at high risk of hypoglycemia such as those with hypoglycemic unawareness.
Several guidelines have been developed regarding CGM systems. Guidelines for “Performance Metrics for Continuous Interstitial Glucose Monitoring,” were developed by the Clinical and Laboratory Standards Institute (CLSI). 138 CLSI is a globally recognized, not for profit standards development document organization, and their work includes guidelines available at the CLSI.org 139 website. The guidelines cover how CGM data should be assessed for accuracy, how CGM systems should be assessed for factors that can decrease accuracy, and how CGM systems should be operated for optimal performance. 138
Another guideline for CGM systems was developed by the IFCC. 140 Despite the global availability of CGM systems, no international evaluation guidelines exist to date. Thus, the IFCC working group focuses on improving standardization of CGM performance measures. 141 Although, the MARD is currently the most common metric used to assess the performance of CGM system, several limitations of using MARD exist affecting traceability.142,143 Notably, there are systematic differences not captured by MARD for example, based on blood sample comparison (venous from factory calibrated vs. capillary). 144 This suggests that, in performance evaluations, some CGM systems may have to be compared with venous blood glucose whereas other may have to be compared with capillary blood glucose, despite using the same set of parameters. Figure 10 presents a diagram of the compartments that surround the CGM sensor’s location in the interstitial fluid, and the calibration differences between each compartment.
Body compartment and calibration differences. Figure provided by Guido Freckmann, MD, Institut für Diabetes-Technologie, Forschungs- und Entwicklungsgesellschaft mbH an der Universität Ulm, Ulm, Germany.
A multispecialty international panel convened early in 2020, organized by DTS to develop practice recommendations on the use of CGM and automated insulin dosing systems in the hospital. 145 Unexpectedly, the COVID-19 pandemic presented and, in response, the FDA produced a statement of “no objection” for the inpatient use of CGM during the COVID-19 crisis.36,146 Hence, CGM systems were being used “off label” to help reduce exposure and preserve personal protection equipment. These new guidelines offer recommendations on the potential use of CGM and AID systems for hospitalized patients taking under consideration the existence of evidence to support its safe use. Panelists recognized the need for additional research to validate the efficacy and safety, and obtain FDA approval for use of these devices in the hospital. 145
Cybersecurity is of utmost importance especially for CDDs. However, the development of cybersecurity strategies must evolve alongside the development of new technologies. Thus, DTS has developed a standard to provide the framework for specifying the security requirements for connected diabetes devices and how to obtain a requisite level of independent assurance that the devices meet those requirements.147,148 Self-attestation of compliance and the auditing by external organizations that can determine and test the level of cybersecurity claimed, are simple steps toward creating confidence within the user and healthcare staff environments.
Session 9: Is Digital Health Discriminatory?
Moderators
Andrew Bremer, MD, PhD
NIDDK/National Institutes of Health, Bethesda, Maryland, USA
Sarah Kim, MD
University of California San Francisco, San Francisco, California, USA
Technology is Not the Silver Bullet
Fazlyn Petersen, PhD
University of Western Cape, Cape Town, South Africa
The importance of considering the context for m-health implementation
Challenges for technology adoption
Alternative approaches to encourage use in low resource settings
Digital Health in the Military
Karl Friedl, PhD, COL(ret)
Senior Research Scientist (SES/ST), US Army Research Institute of Environmental Medicine, Natick, Massachusetts, USA
An efficient way to reach soldiers in remote outposts and veterans in low density rural areas
Potential to reduce health disparities through access to specialized care for chronic disease management
Better care through more efficient virtual house calls in naturalistic settings
A Level Playing Field
Celeste Campos-Castillo, PhD
University of Wisconsin-Milwaukee, Milwaukee, Wisconsin, USA
Parallels between inequities in health and health care and the first-level (inequalities in access) and second-level (inequalities in use) digital divides
Sample of interventions to narrow digital divides
Policies during COVID-19 that widened and narrowed digital divides
Developing Technology for Digital Health
Keesha M. Crosby, MS
Tri-Guard Risk Solutions, LTD, Arlington, Virginia, USA
The largest group with diabetes (Blacks, Latinos, and Native Americans) are not adopting the digital health technology due to socioeconomic constraints, access, and lack of products geared towards them.
The digital health landscape of developing technology is exact opposite of largest user group. Therefore, technology is not fit for use. More than 60% of digital technology is funded and developed by white males. According to a Crunchbase report in 2019, female startups receive less than 2.8% of annual venture investment 149 and whereas black females receive less than 0.02% of annual venture investment. How can there possibly be product market fit given this scenario?
Developing technology for user population without inclusion of representation of that group is discriminatory.
Technology Capacity and Literacy for Urban Poor
Elizabeth Burner, MD, MPH, Msci
University of Southern California, Los Angeles, California, USA
Access to high-speed networks needs to be considered in design
Current technology use will impact uptake; consider training
In South Africa, because of a history of apartheid and legalized discrimination, there continues to be large inequities between white groups and people of color. This inequity extends to the use of mobile technology which is much less accessible to people of color due to high cost and the need to prioritize financial resources to acquiring essential needs like food and water. Furthermore, people with diabetes have additional financial burdens such as cost of medications and testing supplies. There may be a false belief that people of color do not want to access technology or care for their diabetes, but often the obstacles to both are due to financial strains or other barriers that are specific to minority communities (eg, a glucose meter might get stolen to be sold on the street). 150
Therefore, the issue of implementation of mobile health needs to consider this major issue of access, especially in South Africa where people with diabetes tend to live on a pension. In order for mobile health technology to work for people who need it most, internet access needs to be less expensive and catered to individuals, such as the elderly, who are not as “tech-savvy.” If not, mobile health technology might advance in South Africa, but will be helpful only to the wealthy. 150
Issues of disparities in diabetes digital health are also prevalent in the United States. While there may be disparities in the use of digital health technologies, digital health may also reduce disparities. The proposed use of technology to deliver health care remotely dates back to 100 years, however we have been slow to implement technology into health care delivery. More recently, a pioneer of digital health, Ray Dorsey, MD, MBA at University of Rochester has been using video house calls for patients with Parkinson’s disease, providing expertise to patients who live far from a neurologist specializing in movement disorders.151,152 The current COVID-19 pandemic will accelerate the use of video visits, and in some aspects, video appointments made from a patient’s home may be superior to clinic visits because the provider can see patients in their natural environment. Notably, the military has been utilizing telehealth technologies well before the pandemic. One example comes from Ron Poropatich, MD, former president of the American Telemedicine Association, who was involved in providing telebehavioral health care to United States and allied soldiers in remote locations throughout Afghanistan. 153 A near future example is the Army’s proposed use of avatars to query behavioral health concerns in a less judgmental manner than a real human provider might, obtaining better disclosure of their health concerns and getting the assistance they need. 154
What are the downsides of digital health? Spending more time in front of smartphones and other devices contributes to a sedentary lifestyle. Counterbalancing this is the use of technology so that people can exercise in their own home. Additionally, future technologies such as eyeglasses that can keep track of food intake might help reduce unhealthy eating. 155
We need to work on matching our current technologies to enhance diabetes management. The use of CGM systems and hybrid closed loop systems are an important example of this. But we need to also focus on delivering technology to those in greatest need who often have less access to technology, and national leadership around technology innovation is important. We also need to ensure that technology does not create more disparity. An example of this is the discovery by Obermeyer and colleagues of a commercial algorithm used in health systems that was systemically biased against black patients who needed diabetes treatment. The work of Alyson Myers, MD from North Shore University Hospital is a counterexample. She piloted the use of telemedicine to reduce barriers for black and Latinx populations. 156 We all need to increase our sense of urgency around this solvable problem and not rely on a pandemic to push progress forward.
Another important discriminatory aspect of digital health to consider is disparities in access to and use of information and communication technologies, and an examination into how to level the playing field for digital health in diabetes care. A fundamental question about how to level the playing field is: who has access to the internet? Campos-Castillo analyzed the data from the Health Information National Trends Survey aims to assess internet access through means in addition to the traditional home computer. 157 Information such as race, gender, and indicators of socioeconomic status were included in the survey. The study showed that overall, whites had greater access to the internet compared to blacks and Latinos. From 2007 to 2012, there was a significant rise in the proportion of black men accessing the internet. The reason for this cannot be elucidated from the data, but a hypothesis is that this may be because of the higher rate of unemployment in black men and the use of the internet for activities such as job searching. 157 A more recent study she conducted assessed telehealth usage during the COVID-19 pandemic. 158 She showed that overall, non-white groups used telehealth more than whites and after adjusting for co-variates, use of telehealth by blacks was significantly higher than whites. 158 She theorizes that this may be because of a higher impact of COVID-19 in black communities but also because blacks have a higher prevalence of chronic diseases that need regular medical attention, such as diabetes. 158 We need to be aware of the racial and gender differences in telehealth usage to better leverage telehealth to improve health outcomes. Additionally, we need to address the ongoing disparities in access to the internet and consider “lower tech” options like texting that are more accessible and familiar to people of lower socioeconomic status.
Chronic conditions sometimes devolve into emergency situations. Burner et al. have been working on improving diabetes management in the emergency department (ED). This approach is starkly different than other diabetes programs that rely on self-referrals and thus cater to an already engaged audience. It also targets high utilizers and thus contributes to cost containment efforts of an already costly disease.
Patients who wind up getting chronic care in the ED have poor access to health care and low financial resources. There are significant gaps that need to be bridged between urgent care and scheduled, regular care. Additionally, the ED setting is potentially a susceptible moment for behavior change, since many people show up to the ED because of a perceived health crisis. In trying to find a way to deliver chronic disease management in a setting that cannot disrupt workflow in a very busy department like the ED, Burner et al. 159 tested an intervention that delivered text messages twice daily for six months, aimed at improving health behaviors. They found that, compared to the control group, there was a trend toward improved quality of life and self-efficacy. They also found a 20% reduction in ED visits for patients that received the intervention vs. the control group. There was also a significant reduction in HbA1c at six months. At one year follow up, they observed that the Spanish speaking patient’s HbA1c rebounded higher than the English-speaking patients. They are still looking into the reasons why and are investigating the different digital health experiences of these populations.
The use of text vs. apps is different among non-English speaking populations in that low-income individuals may have more concerns about data privacy and low-income households often have to ration data usage. This is especially pertinent in diabetes care where large amounts of data, such as blood glucose values over long periods of time, can be prohibitive. Additional concerns are with language fidelity when translating English into other languages. The Spanish language, as an example, is not the same across countries and translation must be thoughtfully conducted. Furthermore, non-English languages may require more characters to communicate the same message in English, abutting against character constraints of different communication modalities. Finally, if we are to fashion mobile health technologies for low-income, non-English speaking populations, we need to consider the role of training to use new technologies and where, when, and by whom this training should be conducted.
As we consider how Hispanic, black and Native American communities are disproportionately affected by diabetes and the racial differences in technology usage, it is also important to consider the disparities for these groups in leading digital health companies. Unfortunately, digital health companies led by women and racial minorities are far less likely to receive either government funding or venture capital support. Figure 11 presents a bar graph of comparing the number of NIH Small Business Innovation Research Phase 1 grant applications from women-owned small businesses (WOSBs) compared to from non-WOSBs. 160 Only 10% to 12% of digital health startups have a female chief executive officer (CEO) and a similar percentage of venture capital partners are women. 161 Since 2009, black women-led startups have raised $289M in venture/angel funding with most of that raised in 2017.162,163 This is a meager 0.06% of the total $424B in tech venture funding.162,163
NIH SBIR phase 1 applications from WOSBs and non-WOSBs, fiscal years 2005 to 2014. Figure provided by Keesha M. Crosby, MS, Tri-Guard Risk Solutions, LTD, Arlington, Virginia, USA. Reproduced from National Academies Press (USA). 160
Tri-Guard Risk Solutions, started by a black female CEO, Keesha M. Crosby, was funded by the Departments of Energy and Defense. However, other worthy counterparts such as Star Cunningham of 4DHealthware and Iman Abuzeid, MD of INCREDIBLE Health could not raise financial support. In fact, when Abuzeid was attending a meeting to raise her second round of venture capital funding, she was mistaken for the mail delivery person. The consequences these discriminatory practices is that most of health technology is currently being built by white-led companies whereas the need to improve health technology is most pressing in the non-white population.
It is important for us to look at how we can bring racial minorities into the digital health atmosphere and into medical device development, and create products that are centered around the needs of these populations. The marketplace of black consumers is worth a trillion dollars. If we can capture some of that market and give minority-led companies the funding they need as well as incentivize them, then we can grow the digital health adoptions within that marketplace.
Session 10: Does Real Time Diabetes Monitoring Require More Metrics Than Glucose?
Moderators
Avner Gal, MBA, MSCEE, MSc
Iridium Consultancy and Technologies, Ltd., Herzliya, Israel
Barry H. Ginsberg, MD, PhD
Diabetes Technology Consultants, Arlington, Virginia, USA
Smart Insulin Pens and Connected Devices
Hope Warshaw, MMSc, RD, CDCES, BC-ADM
Hope Warshaw Associates, LLC., Asheville, North Carolina, USA
Nearly 3M people in the United States who require insulin use MDIs (~1M with T1D, ~2M with T2D).
Taking insulin using a traditional delivery device is challenging and cognitively burdensome. People want more user-friendly, technology-enabled delivery devices.
A range of smart and/or connected delivery devices are under development or currently available in United States, Europe, and elsewhere. There is significant variation in the capabilities of current and under development devices.
Early evidence demonstrates clinical and mental health benefits of Smart Insulin Pens/Connected devices including decreasing missed and delayed bolus doses because of reminders, improved accuracy of bolus dose timing, improved TIR, less TAR, less TBR.
Smart Insulin Pens/devices with connectivity currently assist the person with diabetes who takes insulin to track glycemia metrics, insulin metrics, carbohydrate consumption, and timing of food intake. Other metrics that could provide valuable are: other nutrient intake, physical activity, personal life schedule parameters and other variables, such as sleep, stress, schedule changes, moods, and so on.
Regarding a person’s choice of insulin delivery device, they can select one that will prioritize their wants, needs and personal goals and recognize these, and therefore, their preferred delivery device at various times through life may change.
Use of Exercise Monitors to Provide Additional Information for Monitoring Diabetes
Dessi Zaharieva, PhD
Stanford University, Stanford, California, USA
Exercise wearable technology may increase adherence to physical activity guidelines in T1D.
Integrating exercise metrics (eg, heart rate) with CGM may aid in detecting glucose fluctuations around exercise.
Incorporating exercise metrics into clinic settings could also improve discussions around physical activity and diabetes management.
Use of Activity Data from Wearable Trackers in T2D Research
Christian Cerrada, PhD
Evidation Health, Inc., San Mateo, California, USA
Activity data from wearable trackers allow researchers to characterize disease burden and identify subgroups of individuals who may benefit from lifestyle change interventions (examples from Evidation's research portfolio).
Characteristics of these subgroups can be leveraged to develop tailored interventions.
Wearable data can additionally be used to monitor progress toward behavior change goals and identify time points for additional intervention.
Multi-Sensor Platforms
Jeffrey Joseph, DO
Thomas Jefferson University, Philadelphia, Pennsylvania, USA
Multi-Sensor Platform that detects the onset of a meal.
Sensors that continuously monitor plasma insulin levels.
Sensors that continuously monitor plasma pH and ketone levels.
Sensors to Prevent, Monitor, and Treat Foot Ulcers
Bijan Najafi, PhD
Baylor College of Medicine, Houston, Texas, USA
Discussing current challenges to deliver care to patients with diabetic foot ulcer (DFU) during the pandemic
Overviewing new innovations for remote patient monitoring and triage those with high risk of DFUs
Discussing new opportunities to empower patients and their caregivers to prevent DFUs
Regarding smart insulin pens and connected devices, of the over seven million people in the United States who take insulin, nearly three million use MDIs or continuous subcutaneous insulin infusion to manage glycemia.164,165 Traditional insulin pen usage continues to increase, as they are both more accurate and easier to use. Most pens, however, do not track the insulin doses.
The ADCES and Sanofi (Paris, France) in a commissioned survey of 700 people who take insulin found that most people with diabetes using insulin log the details of their therapy, but 80 percent would appreciate having a streamlined way to assemble and display all of their diabetes data.97,166 People with diabetes who require insulin therapy often transition between various insulin delivery devices depending on product availability, insurance reimbursement, level of desire and/or need for intensivity of diabetes management. Today's most advanced therapy, hybrid, and advanced hybrid closed loop systems have capabilities that smart insulin pens/connected devices do not currently have. Key details about the six devices on or nearing the USA market were presented.
Two devices are currently available: Clipsulin™ from Diabnext (Paris, France) is a reusable, smart pen cap; it is FDA exempt, CE Marked, and available for purchase now. 167 InPen™ is a durable pen from Medtronic Diabetes 168 (formerly Companion Medical 169 ). The InPen detects priming, has a bolus dose calculator, tracks insulin-on-board and allows the user to send their data to their clinicians. 170 FDA has issued additional clearances including use for younger populations and two meal modes in addition to carbohydrate counting.171,172 It is also CE Marked. 173
Four other devices are in development: Mallya™ from Biocorp (Issoire, France) is a Bluetooth, reusable smart device that clips to the pen top. 174 It is CE Marked 175 and FDA filing is expected this year. The Tempo pen, a modified Lilly Kwikpen®, is being developed with DexCom, Inc.176,177 The Novopen Echo Plus from Novo Nordisk is available in Europe, 178 and is expected to be filed with the FDA in 2021. This device is expected to connect to a smart device. Bigfoot Biomedical (Milpitas, California, USA) is on track to file with the FDA for clearance of their system, Bigfoot Unity Diabetes Management Program, consisting of a cloud-connected pen and a proprietary BGM. 179
Limited quality research suggests clinical and mental health benefits. Preliminary studies from the Novo Echo Plus and InPen show improvements in TIR and frequency of bolusing.
Regarding exercise monitoring for people with diabetes, current adult exercise guidelines suggest at least 150 minutes of mixed physical activity each week, but the vast majority of people with diabetes fall far short of this 180 (probably for some, even worse during the pandemic). Systemic glucose values vary with the type of exercise, being most stable with mixed (aerobic and anaerobic) activities. 180 With exercise, dreaded hypoglycemia is common even with carbohydrate feeding and insulin dose adjustments. CGM and AID systems have helped, but recently a new position paper suggested methods of management of exercise using CGM. 181
There are several metrics that could also be useful when performing exercise. They include: (1) heart rate, (2) energy expenditure, (3) step count, (4) calories burned, (5) elevation, (6) exercise intensity (power), (7) galvanic skin response, (8) lactate, (9) insulin, and (10) ketones.
These first six alone can be captured from most commercial wearable technology devices, but galvanic skin response, lactate, insulin and ketones are more complicated to capture, especially in real-time. Several companies are creating CGM combined with additional metrics that can all be helpful around physical activity.
Sensors that provide data on exercise are especially important to AID systems. In a study examining continuous lactate monitoring in high intensity exercise in adults with T1D, there was a rise in lactate with high intensity exercise and then a drop off after exercise. 182 Commercially available wearable trackers have been utilized as surrogates for exercise in some AID devices with some success. 183 Extreme exercise or exercise with inadequate insulin levels can cause ketosis. 184 A continuous ketone sensor is being developed by PercuSense 185 with clinical studies likely beginning in 2021. In addition to AID, this is obviously important for T1D. Currently, at Stanford, they incorporate CGM data into the pediatric EHR for study by the diabetes care team, and a team is working on getting analyzable physical activity data into the EHR. Figure 12 presents the flow of CGM and physical activity tracker data from the device to the EHR. 186
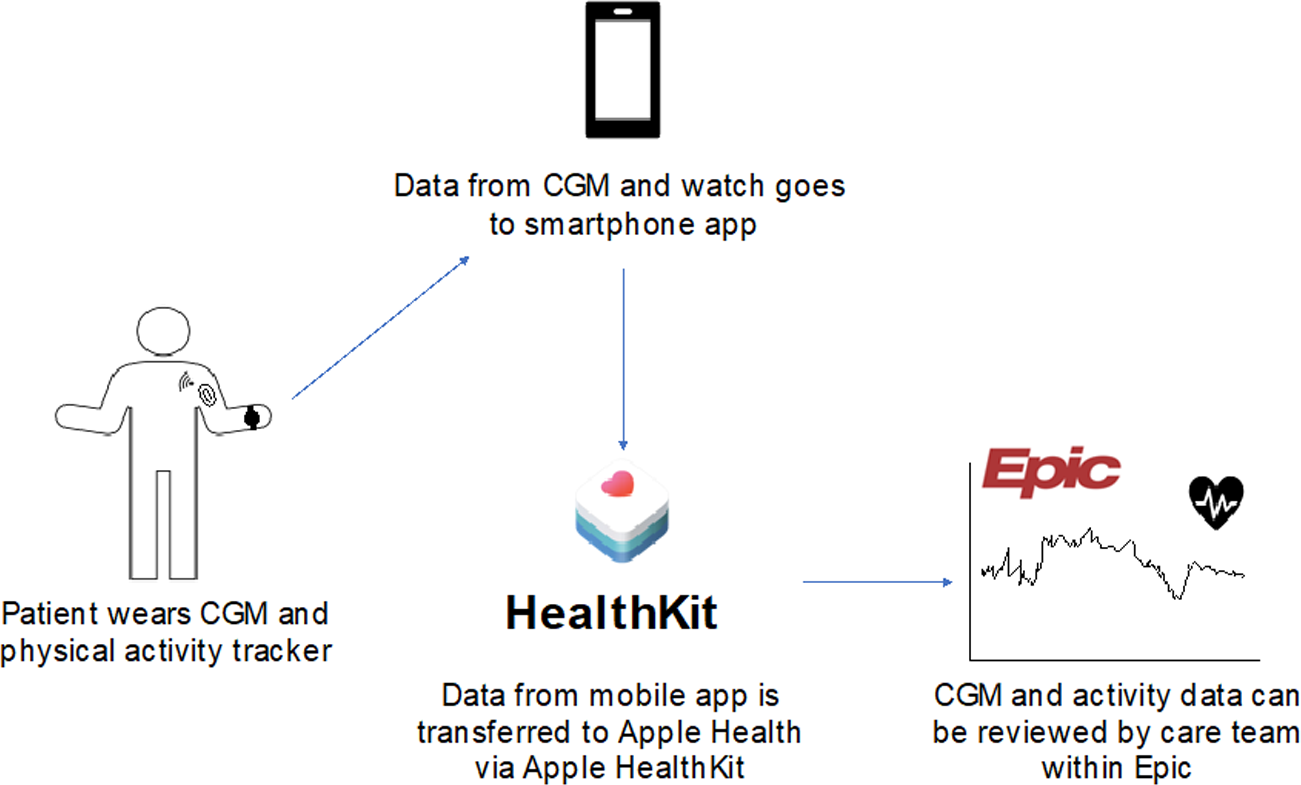
Flow of CGM and physical activity tracker data from a device to the EHR. Figure provided by Dessi Zaharieva, PhD, Stanford University, Stanford, California, USA and adapted from Prahalad et al. 186
The challenges of exercise monitoring include device burden, device accuracy, noise or erroneous values, cost and accessibility of these devices, relevant variables, exercise detection and timing, and, of course, integrating these into AID systems. Many patients already consider the plethora of devices for AID overwhelming, 187 but as the devices get smaller and more integrated, adding exercise monitoring may add more data without more burden.
In addition to insulin and exercise monitoring as metrics for monitoring people with diabetes, activity data has been used in research for people with T2D. Up to 21% of the USA population use a smartwatch regularly for fitness tracking. 188 Using activity data is useful for characterizing, intervening on, and monitoring various behaviors that have been shown to impact blood glucose.
Patients with T2D can be segmented into subgroups of individuals who share similar characteristics and risks related to diabetes management. One set of characteristics that can be used for segmentation purposes are behavior patterns captured from activity trackers. Not only can activity trackers be used to monitor activity and characterize individuals, but they can also be used to provide intervention when necessary, directly within the context of a targeted behavior. So: Monitor, Characterize, Intervene, Repeat.
As one example of characterization, researchers analyzed activity in the context of medication adherence using pharmacy claims data among over 117,000 individuals with T2D. 189 Their analysis showed that across chronic conditions, individuals who tracked any activity (sleep, step, weight, or a food diary) were more likely to be labeled as adherent by about 35%. 189 Further, individuals who are more active based on step counts also had greater odds of being labeled as adherent. 189
So how might you use this information to develop an intervention? This analysis suggests that activity tracker use patterns may be useful for estimating future risk of non-adherence. Intervention among these individuals might include more robust support for medication management, which has downstream impacts for blood glucose control.
Activity data can also be used to characterize the impact of disease on daily life. In a retrospective analysis of 67 individuals identified as having T2D and having dense tracker data, researchers observed lower step counts compared to a baseline period in the two-week period surrounding a flu diagnosis. 190 In addition, they also observed earlier sleep times and more sleep disturbance in the days leading up to the flu among people with diabetes. This kind of insight might be useful as an intervention message delivered to activate individuals to health promoting behaviors, in this case, getting a flu shot. 190
You could also imagine characterizing the relationship between an individual’s activity or diet with their blood glucose in order to deliver personalized coaching and recommendations over the course of a behavior change program. Real time BGM can be supplemented with data from activity trackers or other types of sensors allowing us to situate blood glucose in the context of someone’s lifestyle or behavior pattern. We can characterize segments of people who may need specific types of support to manage their blood glucose, and we can quantify the impact of diabetes on everyday behaviors. This insight can be used to deliver adaptive interventions directly to people, and we can further monitor the impact of such interventions, both in terms of blood glucose and subsequent activity over the long term. This framework could be used to provide long-term lifestyle change and support for diabetes management with the flexibility to intervene where necessary.
Regarding multi-sensor platforms, just as normal glucose control is complicated with a variety of inputs, artificial control would also require a variety of inputs. A CGM sensor may require 20 minutes or more to recognize that a meal is occurring, and if insulin is delivered subcutaneously, there is an additional delay. Complex variables, such as variable insulin absorption, glycemic index of foods and delayed gastric emptying, make this more complex, often leading to poor prandial glucose control and sometimes postprandial hypoglycemia. Thus, matching the insulin activity to the absorption of nutrients is especially important. So, are there possibly sensors that could detect that you’re eating a meal?
There is an experimental device that measures gastroparesis, an electrogastrogram. 191 The signal is low and noisy, but it can detect increasing activity as the stomach empties. 191 A lab is working on an acoustic sensor that can detect swallowing. 192 So, it should be able to detect the meal time and perhaps even the size of the meal.
Many other sensors may be important to improve diabetes care. Insulin absorption has a high interpatient and intrapatient variability. For patients on insulin pumps, there is also a high rate of insulin infusion set failures. Thus, the insulin dose calculators that assume the amount of insulin on board, may be incorrect. 193
So, the typical algorithm in the insulin pump that assumes the amount of insulin absorbed may not be good enough. In one glucose clamp experiment, the PK of insulin absorption from a pump catheter varied significantly over five days. The Tmax was shorted from 110 to 60, and the Cmax increased from 60 to 180. 194
Other measures may be important. An implantable infrared sensor was implanted for years around a blood vessel in experimental animals, and the Joseph lab studied flowing blood in order to measure glucose, lactate ketones and other factors. In humans, they drew venous blood and used the infrared sensor in vitro to measure glucose. During activities of daily living, there was a r 2 of 0.98 compared to the HemoCue (HemoCue AB, Ängelholm, Sweden). This sensor should be capable of measuring lactate and ketones as well. 195
Sensor accuracy can be increased by using multiple sensors, a sensor array. The Joseph lab did a study of six early Medtronic sensors. Placed subcutaneously, they varied significantly, but at least three were consistent and accuracy could be improved by polling the sensors. 196 Medtronic has produced a product called the Hospital Glucose Management System with four electrodes, which they tested in volunteers in the hospital. 197 After calibration, the sensor was extremely accurate.
Regarding foot care for diabetes, unfortunately, every 20 seconds somebody loses a limb because of diabetes. 198 Uncontrolled and prolonged hyperglycemia can damage healthy perfusion to lower extremity as well as the ability to feel pain. These losses, along with abnormal pressure in the foot can lead to ulcers that can lead to a highly prevalent amputation. Amputations are extremely expensive and destroy and shorten lives. The good news is that an estimated 85% of all diabetic amputations are preventable. 199 But the bad news is that we are still losing too many limbs because of diabetes. Technology can help prevent amputations. Recently, a paper in Journal of Diabetes Science and Technology summarized those technologies. 200
The most important sign of diabetic foot disease is foot temperature. We know that the skin temperature is a sign of inflammation. So, if you can measure temperature on a regular basis, then perhaps you can prevent amputations. Frykberg et al. 201 studied a smart mat for home use. It measures localized temperature of the foot and sends the data to the cloud. With high sensitivity and fair specificity, it is a good screening tool and can indicate a potential ulcer more than a month before clinical observation. It is an excellent tool for triaging patients for further study. 201
The Najafi lab group has developed and studied “smart sox” that measure temperature pressure and joint angles. 202 There is a commercial sox technology that continuously measures temperature and can notify the patient of the hottest spot. Of course, this concept is still forming and there needs to be more evidence to show the effectiveness.
Once we know there is a ‘hot spot’ of temperature and pressure, we need to off-load the pressure point, using shoe inserts. This very simple, but very effective step is important and technology can ascertain adherence to the use of the inserts. So, Najafi et al.203,204 are developing a technology to monitor the patient location, adherence to using the off-loading inserts and reporting this to the patient and perhaps a patient portal. Through a patient portal, health care professionals can determine and can engage and re-enforce behaviors that work.
Researchers are also working on "smart wound dressings" that can measure glucose levels and moistness, which is very important for good management. 205 It is still in its infancy, but it is very promising to help patients heal properly and avoid amputations.
Session 11: Can Glucose Monitoring Predict the Future?
Moderators
Thanh Hoang, DO
Walter Reed National Military Medical Center, Bethesda, Maryland, USA
Umesh Masharani, MD
University of California San Francisco, San Francisco, California, USA
CGM-Informed Prediction Tools
Chiara Fabris, PhD
University of Virginia, Charlottesville, Virginia, USA
Clinical optimization problem of T1D control: to reduce HbA1c and risk for complications, while avoiding increased occurrent of hypoglycemia
Role of CGM in predicting hypoglycemia: CGM-derived risk of upcoming hypoglycemia
Role of CGM in predicting complications: CGM-derived (through TIR) estimated HbA1c
Classification of Hypoglycemia: The Hypo-RESOLVE Project
Bastiaan de Galan, MD, PhD
Maastricht University Medical Centre, Maastricht, Netherlands
Radboud University Medical Centre, Nijmegen, Netherlands
There is great variation with respect to the classification of hypoglycemia in diabetes among clinical practice guidelines and position statements.
A recent consensus statement on the classification of hypoglycemia received widespread support, but the underlying evidence is slim.
Hypo-RESOLVE uses a multilevel approach to increase the understanding of hypoglycemia and provide robust evidence to refine and solidify the classification of hypoglycemia
Low Sensor Glucose and Hypoglycemia: Are They the Same? The HypoMETRICS Project
Pratik Choudhary, MBBS, MD, FRCP
University of Leicester, Leicester, United Kingdom
Defining hypoglycemia with CGM
The difference in rates of hypoglycemia detected by CGM and SMBG
Overview of the HypoMETRICS project that aims to define the impact of CGM and patient reported hypoglycemia on people living with diabetes.
Yes, Glucose Monitoring Can Predict the Future
Dan Goldner, PhD
One Drop, New York, New York, USA
Event prediction: two-hour forecasts of blood glucose and overnight predictions of hypoglycemia are highly accurate.
Outcomes prediction: six-month forecasts of 30-day average glucose readings are more accurate when made from CGM data than when made from BGM data.
CGM-based predictions facilitate prevention of adverse events, prioritization of attention, and individually optimized programs of self-care.
Simplifying Diabetes Management Through Glucose Prediction
Pratik Agrawal, MS
Medtronic Diabetes, Northridge, California, USA
Novel algorithms and considerations for glucose prediction solution
How to evaluate the accuracy of predictions
How solutions with glucose prediction can improve diabetes management
Software to Achieve Target Glycemia
Jordan Messler, MD, SFHM, FACP
Glytec, Largo, Florida, USA
The best way to predict the future is when you know what is happening in the present, with real-time data and interventions to identify treatment needs.
The status quo is broken. Confusing paper protocols combined with outdated data are ineffective, unsafe, and costly.
Utilizing insulin software to achieve glycemic targets leads to better outcomes for patients, improved workflows, and cost savings for hospitals and health systems.
CGM data can be used to determine risk of hypoglycemia in the short term. Predictors, namely CGM nadir, sleep and curvature, fed to a logistic regression classifier, can be used to determine risk for upcoming hypoglycemia. Using this model and focusing on a one-hour prediction horizon, the classifier can achieve 93% correct assessments. 59 Figure 13 presents an example of CGM data and the types of short-term and long-term predictions that can be extrapolated from the data.
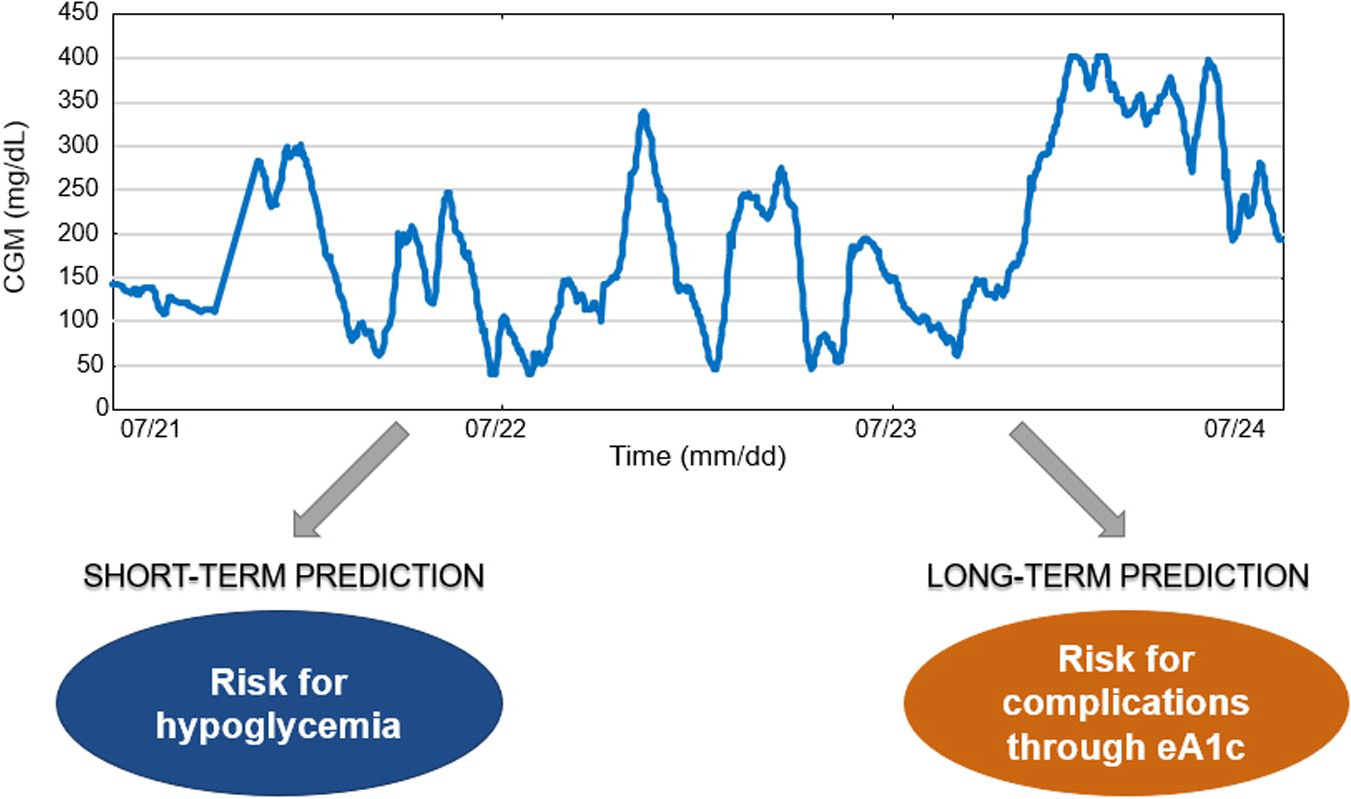
An example of CGM data and the types of short-term and long-term predictions that can be extrapolated from the data. Figure provided by Chiara Fabris, PhD, University of Virginia, Charlottesville, Virginia, USA.
The International Hypoglycemia Study Group has proposed a three-level classification of hypoglycemia: (1) Alert value (Level 1): Blood glucose ≤3.9 mmol/L (70 mg/dL); (2) Clinically important hypoglycemia (Level 2): <3.0 mmol/L (54 mg/dL); and (3) Severe hypoglycemia (Level 3): sufficient cognitive dysfunction to require assistance from another person for recovery. 206 Despite widespread adoption of this classification, data supporting Level 1 and 2 cutoffs are lacking. This is especially true with respect to CGM measurements. The Hypoglycaemia Redefining SOLutions for better liVEs (Hypo-RESOLVE) project aims to collect data on hypoglycemia events from over 100 clinical trials in people with T1D and T2D. The database will be examined for predictors of hypoglycemia and establish glucose thresholds that constitute risk of morbidity and mortality and increase healthcare costs. The significance of continuous glucose sensor detected hypoglycemia will be evaluated. 207
The impact of asymptomatic episodes of CGM hypoglycemia is unclear. With CGM data, one can consider hypoglycemia in a more complex way. There is the question of how rapidly glucose levels are falling, how low are the glucose levels and how long the glucose levels remain below the defined threshold. Hypoglycaemia, Measurement, ThResholds and ImpaCtS (Hypo-METRICS) study aims to recruit 200 patients with T1D with good awareness; 50 patients with T1D and impaired awareness and 350 patients with T2D on insulin. Patients will wear blinded continuous glucose monitors and activity monitors for 10 weeks. The patients will document their sleep quality, mood and hypoglycemia events. The goal will be to provide better definition of clinically relevant low sensor glucose readings. 208
When you pair CGM data with other contextual data that are passively monitored or reported (such as physical activity, sleep, heart rate, food, medications), you can very accurately predict blood glucose levels two hours in advance, and can predict if the glucose is going to be below 70 mg/dL or above 180 mg/dL over the next four hours—or longer when predicting nocturnal hypoglycemia.209,210 Glucose from CGM is better at predicting what will be the average blood glucose level in four to six months, compared to predictions made from finger-stick glucose monitoring. 211 Interestingly, TIR can be predicted even more accurately than average blood glucose. 211 In conclusion, CGM-based predictions facilitate prevention of adverse events, prioritization of attention, and individually optimized programs of self-care.
CGM data can also be used to determine HbA1c levels (and hence risk for complications) in the longer term. Using time in target range (70 to 180 mg/dL) data in a model using population parameters and a subject specific parameter, the correlation between measured laboratory HbA1c and estimated HbA1c was 93%. 59
Physiological models do well in predicting glucose control in the longer term but tend to be inaccurate when predicting what is going to happen to the glucose level in the next couple of hours. Using an AI dynamic estimator to fine tune the parameters within the physiological model, we can improve short term prediction of glucose levels over the next two hours. This enables adjustment for unannounced meals and miscounting of carbohydrates. Personalizing the algorithm by collecting additional data from an individual user, may further improve short term glucose prediction.
Insulin management software integrated with the electronic medical record can be used to recommend insulin doses that adjust to an individual patient’s glucose trends and insulin sensitivity. This may reduce hypoglycemia in the hospitalized patient.
Live Demonstration
Moderators
Rodolfo J. Galindo, MD, FACE
Emory University School of Medicine, Atlanta, Georgia, USA
Eun-Jung Rhee, MD, PhD
Kangbuk Samsung Hospital, Seoul, South Korea
Smart Socks for Diabetic Foot Ulcers
Bijan Najafi, PhD
Baylor College of Medicine, Houston, Texas, USA
The Smart Mat platform, which monitors foot temperature, was able to predict foot ulcer incidents with 37 days lead time on average and 97% sensitivity.
Smart Sox monitors foot temperature, plantar pressure, and big toe motion and was comparable to the gold standard.
New measurements that can improve DFU monitoring include flexible electrochemical sensors to monitors pH, glucose levels (via CGM), and moisture.
Diabetic foot infection is highly prevalent, with an estimated 25% lifetime risk. It is a largely preventable—but devastating—consequence of diabetes. Patients with diabetes often have several risk factors for the development of DFU, including loss of protective sensation due to peripheral arterial disease or neuropathy and abnormal pressures associated with musculoskeletal foot deformities and gait disturbance. Innovative technologies have been developed for (1) triaging patients with an “urgent diabetic foot risk,” (2) provide improved patient foot self-care (preventive care), and (3) remote care delivery.200,203,204 Figure 14 presents ways that the values of thermography can be used to prevent DFU.
Ways that the values of thermography can be used to prevent DFU. Figure provided by Bijan Najafi, PhD, Baylor College of Medicine, Houston, Texas, USA.
A recent prospective multi-center cohort study evaluated the efficacy of a “Smart Mat”: an innovative platform to remotely monitor foot temperature and capture a thermal image of the foot at home, which via a cloud-based artificial-intelligence model was able to predict prospective foot ulcer incidents with 37 days lead time on average and had a sensitivity of 97%. While specificity was poor, this model could help identify high-risk patients for telemedicine or further foot ulcer confirmation. 201 Similarly, Najafi et al. 202 recently published their experience using “Smart Sox”: a continuous monitoring system of foot temperature, plantar pressure, and big toes range of motion. It’s basically an optical fiber based smart textile (also called smart socks), that can simultaneously monitor pressure, temperature, and foot joints’ angles during walking. In the validating study, there was fair to good agreement between the SmartSox and the gold standard. This could also help with telemedicine and provide advice to change shoes, revise shoelaces not being too tight or loose, and so on.
While these are pilot stages, the future is promising for remote monitoring of foot factors that can predispose patients to foot ulcers. In addition to monitoring temperature, pressure, joint angles, and shear forces, integrating new mechanisms to obtain remote clinical information, including flexible electrochemical sensors to monitor pH, glucose levels (via CGM), and moisture, can improve our current gaps for telemedicine and ultimately improve prevention and care of DFUs.
Conclusions
The Diabetes Technology Meeting presented an up-to-date picture of the current state of diabetes technology. The meeting’s presentations covered important aspects of diabetes technology from medical, scientific, engineering, and regulatory perspectives to advance the development and use of better hardware and software for people with diabetes.
Footnotes
Acknowledgements
For contributing key points to this report and reviewing the manuscript, we would like to thank: Pratik Agrawal, MS, David Ahn, MD, Mark Arnold, PhD, Guillermo Arreaza-Rubin, MD, Timothy S. Bailey, MD, FACE, CPI, Richard Bergenstal, MD, Kåre I. Birkeland, MD, Bruce Bode, MD, Bruce Buckingham, MD, Chris Boulton, BSc, Andrew Bremer, MD, PhD, Marc Breton, PhD, Elizabeth Burner, MD, MPH, MSci, Celeste Campos-Castillo, PhD, Eda Cengiz, MD, FAAP, MHS, Christian Cerrada, PhD, Pratik Choudhary, MBBD, MRCP, MD, Faz Chowdhury, PhD, Ali Cinar, PhD, Mark Clements, MD, PhD, Sam Collaudin, PhD, jMBA, Keesha M. Crosby, MS, Ronald Dixon, MD, Frank Doyle, PhD, CPGS, Steven Edelman, MD, Chiara Fabris, PhD, Alexander Fleming, MD, Guido Freckmann, MD, Karl Friedl, PhD, COL(ret), Brian Frier, BSc (Hons), MD, FRCP(Edin), FRCP(Glas), Robert Gabbay, MD, PhD, Avner Gal, MBA, MSCEE, MSc, Bastiaan de Galan, MD, PhD, Wei Gao, PhD, Dan Goldner, PhD, Gerold Grodsky, PhD, Alberto Gutierrez, PhD, Lutz Heinemann, PhD, H. Michael Heise, PhD, Rolf Hinzmann, MD, PhD, Irl Hirsch, MD, Thanh Hoang, DO, David Horwitz, MD, PhD, Roman Hovorka, PhD, FMedSci, Peter G. Jacobs, PhD, Jeffrey Joseph, DO, Francine Kaufman, MD, David Kerr, MBChB, DM, FRCP, FRCPE, Leslie Klaff, MD, PhD, David Kleidermacher, BS, Craig Kollman, PhD, Boris Kovatchev, PhD, Piotr Ladyzynski, PhD, Jordan Messler, MD, SFHM, FACP, Bijan Najafi, PhD, Aaron Neinstein, MD, Anne Peters, MD, FACP, CDE, Fazlyn Petersen, PhD, Andreas Pfützner, MD, PhD, Athena Philis-Tsimikas, MD, Moshe Phillip, MD, Kenneth Quinto, MD, MPH, Mihailo Rebec, PhD, Eun-Jung Rhee, MD, PhD, Mercedes Rigla, MD, PhD, David Rodbard, MD, Julio Rosenstock, MD, Robert Schumm, MBA, Naomi Schwartz, MS, Suzanne Schwartz, MD, MBA, Peter Simpson, MS, Akhil Srinivasan, PhD, Timothy Stenzel, MD, PhD, Andreas Stuhr, MD, MBA, Marc Taub, PhD, Matthew Taylor, CFA, Anders Dyhr Toft, MD, PhD, eMBA, Hubert Vesper, PhD, Hope Warshaw, MMSc, RD, CDCES, BC-ADM, Christopher Wilson, PhD, Howard Wolpert, MD, Yiduo Wu, PhD, and Dessi Zaharieva, PhD. We thank Annamarie Sucher-Jones for her expert editorial assistance.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TS, JZ, BWB, JKR, GC, YP, CEM, SK, BHG, and UM have nothing to disclose. JLS has had research support from the NIH, JDRF, and the Helmsley Charitable Trust and her institution has had research support from Medtronic and Insulet. JLS serves as a consultant to Cecelia Health, Lexicon, Lilly, Insulet, Medtronic, and Sanofi. She also is a member of the advisory board for Bigfoot Biomedical, Cecelia Health, Insulet, Medtronic, and the T1D Fund. JC is a shareholder of Pacific Diabetes Technologies, has received research support from Dexcom, and participates in the advisory board for Insulet. JP has been an advisory board member for BD and Medtronic, and a speaker for Insulet. JE’s efforts were supported by the Food and Drug Administration under award number P50FD006425 for The West Coast Consortium for Technology & Innovation in Pediatrics. The funding source had no involvement in the development of this manuscript or in the decision to submit the paper for publication. The content is solely the responsibility of the authors and does not necessarily represent the official views of the FDA. LHM is a consultant and speaker for Tandem Diabetes, a speaker for Dexcom, and a consultant for Capillary Biomedical. TH’s employer has received research grants from Adocia, Biocon, Eli Lilly, Gan Lee Pharmaceuticals, Mylan, Novo Nordisk and Sanofi. TH has also received speaker honoraria from Novo Nordisk, and is an advisory board member for Novo Nordisk. RJG is supported in part by grants from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) of the National Institute of Health (NIH) under award numbers P30DK111024-04S2 and 5K23DK123384-02. RJG received unrestricted research support (to Emory University) for investigator-initiated studies from Novo Nordisk, Eli Lilly and Dexcom, and consulting fees from Abbott Diabetes Care, Sanofi, Novo Nordisk, Eli Lilly and Valeritas. DCK is a consultant for EOFlow, Fractyl, Lifecare, Novo, Roche, Samsung, and Thirdwayv.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Abbreviations
ACE, alternate controller enabled; ADCES, Association of Diabetes Care & Education Specialists; AGP, ambulatory glucose profile; AI, artificial intelligence; AID, automated insulin delivery; AIMS, AI for Medical Systems Lab; AP, artificial pancreas; BGM, blood glucose monitoring; BGRI, Blood Glucose Risk Index; CDD, connected diabetes device; CE, Conformitè Europëenne; CEO, chief executive officer; CGM, continuous glucose monitoring; CGP, comprehensive glucose pentagon; CLSI, clinical and laboratory standards institute; COVID-19, coronavirus disease 2019; DFU, diabetic foot ulcer; DTS, Diabetes Technology Society; ED, emergency department; EHR, electronic health record; FDA, United States Food and Drug Administration; FNIR, flat, narrow, and in range; GMI, glucose management indicator; GRADE, glycemic risk assessment diabetes equation; HbA1c, hemoglobin A1c; HCP, healthcare professional; HDV, hepatic-directed vesicle insulin; Hypo-METRICS, Hypoglycaemia, Measurement, ThResholds and ImpaCtS; Hypo-RESOLVE, Hypoglycaemia Redefining SOLutions for better liVEs; iAGC, interoperable automated glycemic controller; iCGM, integrated continuous glucose monitoring; IFCC, International Federation of Clinical Chemistry and Laboratory Medicine; IGC, index of glycemic control; INSPIRE, Insulin Dosing Systems: Perceptions, Ideas, Reflects, and Expectations; iPDM, integrated Personalized Diabetes Management; KNN, k-nearest neighbors; MARD, mean absolute relative difference; MDI, multiple dose injection; MGLR, more green, less red; ML, machine learning; PCCP, predetermined change control plan; PD, pharmacodynamics; PGS, personal glycemic state; PK, pharmacokinetics; PMA, premarket approval; RCT, randomized controlled trial; RWD, real world data; RWE, real-world evidence; SAGE, Study on Global AGEing and Adult Health; SMBG, self-monitoring of blood glucose; T1D, type 1 diabetes; T2D, type 2 diabetes; TAR, time above range; TBR, time below range, TIR, time-in-range; URLi, ultra-rapid lispro; USA, United States; WOSB, women-owned small business.
