Abstract
Diabetes Technology Society hosted its annual Diabetes Technology Meeting on November 4 to November 6, 2021. This meeting brought together speakers to discuss various developments within the field of diabetes technology. Meeting topics included blood glucose monitoring, continuous glucose monitoring, novel sensors, direct-to-consumer telehealth, metrics for glycemia, software for diabetes, regulation of diabetes technology, diabetes data science, artificial pancreas, novel insulins, insulin delivery, skin trauma, metabesity, precision diabetes, diversity in diabetes technology, use of diabetes technology in pregnancy, and green diabetes. A live demonstration on a mobile app to monitor diabetic foot wounds was presented.
Introduction
On November 4 to November 6, 2021, Diabetes Technology Society (DTS) gathered healthcare professionals (HCPs), industry representatives, academicians, researchers, and United States (US) regulatory officials for the Diabetes Technology Meeting (DTM). This three-day meeting included two workshops, eleven sessions, and a keynote presentation by the Chief Science and Medical Officer of the American Diabetes Association, all covering current research and emerging topics in diabetes technology. Table 1 presents the agenda for the meeting, with a list of the workshop and session topics. This meeting report summarizes the key points of each speaker’s presentation.
Agenda of the Meeting, With a List of the Workshop and Session Topics.

Workshop A: New Technologies for Glucose Monitoring; Panel 1: Blood Glucose Monitoring
Moderators
Piotr Ladyzynski, PhD, DSc
Nalecz Institute of Biocybernetics and Biomedical Engineering Polish Academy of Sciences, Warsaw, Poland.
John Pickup, MA, BM, DPhil, DSc, FRCPath
King’s College London, London, UK.
Noninvasive Glucose Monitoring
Andreas Thomas, PhD
AGDT (Working group of Diabetes Technology), Germany, Ulm, Germany.
Patients desire noninvasive glucose measurement (NIGM) not only for painless measurement but also for a number of other benefits, such as dispensing with consumables and thus avoiding waste.
Measuring glucose concentration in the mmol/L or mg/dL range is a challenge if it is to be done noninvasively using physical methods. In principle, this is possible: an analysis by DTS showed 28 projects for measurement using optical methods alone.
Developments in NIGM to date have not achieved the necessary accuracy for widespread use in diabetes therapy, but nanotechnology could offer a possibility for NIGM in the future.
Future of Blood Glucose Monitoring
Marcel Gmünder, JD
Roche Diagnostics International AG, Basel, Switzerland.
Blood glucose monitoring remains a strong element in the future of diabetes management as it supports meeting the needs of the majority of persons with diabetes (PWD) across the world.
Roche Diabetes Care (Mannheim, Germany) is continuously leading the development of cutting-edge technologies for blood glucose monitoring and is driving innovation and access. Both need to go hand-in-hand to alleviate the burden of diabetes and support sustainable healthcare around the world.
Roche Diabetes Care drives integration of both their own and partner devices and digital solutions so as to offer holistic treatment options for PWD and HCPs, while utilizing all relevant data to provide custom-made insights and personalized therapy support.
Polypharmacy, Interfering Substances, and Glucose Monitoring Devices
Elizabeth Holt, MD, FACE
LifeScan Global Corporation, Malvern, PA, USA.
EN ISO 19157:2015 and CLSI EP07 specify the evaluation of interference testing for blood glucose monitors (BGMs), but not continuous glucose monitors (CGMs), against single substances one-at-a-time, but testing against combinations of medications is not performed.
Polypharmacy is common in PWD.
Post-marketing surveillance of the OneTouch Verio (LifeScan Global Corporation, Malvern, Pennsylvania, USA) and OneTouch Select Plus test strip platforms show excellent clinical accuracy performance against EN ISO 15197:2015, with no systematic effect from either increasing number of medication classes taken or increasing average number of individual medications.
Innovative Glucose Sensing Principles—Optical Transmission Absorbance System and Open Circuit Potential-Based Sensing System
Koji Sode, PhD
The University of North Carolina at Chapel Hill and North Carolina State University, Chapel Hill, NC, USA
Two innovative and “green” enzymatic glucose sensing principles are introduced, one for BGMs and one for CGMs.
An optical transmission absorbance system provides a highly accurate blood glucose monitoring technology, achieving ±7.1% accuracy in glycemic measurement, with 84% of all measurements within ±5% of the reference values.
An innovative principle for future CGMs is an open-circuit potential (OCP)-based sensing system, which provides a size-independent sensor design by employing an engineered direct electron transfer type enzyme.
NIGM has been a long-standing patient preference, with advantages that include absence of blood sampling, pain, consumables, waste and contamination, and little measurement effort. Several devices have been developed in recent decades, but none have functioned well enough for everyday clinical use. The requirements for effective NIGM include specificity for glucose, a large and linear signal change, and robustness. There are many options for measuring glucose-related physical parameters such as light absorption, light scattering, polarization changes, fluorescence, Raman scattering, photoacoustic spectroscopy, and viscosity; as well as physiological parameters such as heart rate and changes in electrical conductivity or tissue resistance associated with glucose-related electrolyte concentrations. A recent survey by DTS revealed that, despite the many challenges, there are a surprisingly large number of current NIGM projects in progress, but they are unlikely to meet fully the above requirements, such as glucose specificity, a large signal-to-noise ratio (SNR), and clinical robustness. 1 In the future, nanotechnologies may offer some advantages for NIGM, including improved sensitivity and good SNR.
Roche Diabetes Care has a vision for blood glucose monitoring in the coming years, called “Integrated Personalized Diabetes Management.” It enables the patient to master the complexity of diabetes by allowing data collection, integration, and analysis, so as to support effective therapy decisions, while providing integration of both their own and third-party hardware and digital solutions. 2 A central part of the strategy is accurate blood glucose monitoring, which is needed for correct therapy decisions. Poor accuracy leads to missed hypoglycemia, over-bolusing, increased glycemic variability, increased hemoglobin A1c (HbA1c), and less glycemic time-in-range (TIR, 70-180 mg/dL). Although there is increasing use of CGMs in high-income countries, spot blood glucose monitoring will continue to be of major importance in the future, particularly in countries with limited resources, where there are the largest number of PWD and where access to technology is often very limited. A technology which may help with these problems is Accu-Check Sugar View, where a blood glucose value or range can be obtained without a meter by taking a smartphone picture of a special test strip, thereby also allowing digital access to additional app-based resources such as therapy advice. 3
Polypharmacy (five or more medications) is common and increasing—for example, the average number of different medications used by people with type 2 diabetes (T2D) is eight per day and with type 1 diabetes (T1D) is five per day.
4
Known interfering substances for BGMs depend on the enzyme system used: uric acid, galactose, xylose, acetaminophen,
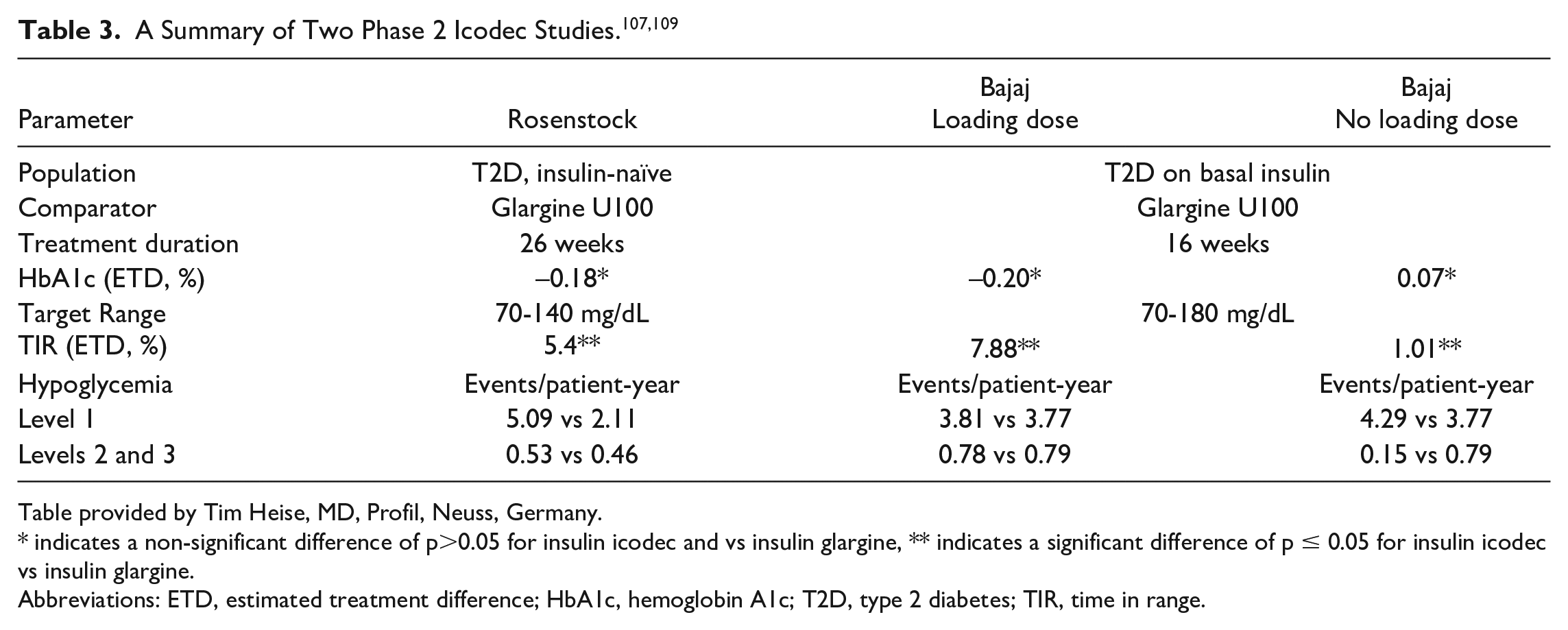
Two innovative glucose sensing principles are described. First, an optical transmission absorbance system for blood glucose monitoring has been developed, in conjunction with Terumo Corporation (Tokyo, Japan), with a strip chemistry based on a new and highly stable flavin adenine dinucleotide (FAD)-dependent glucose dehydrogenase and a tetrazolium salt electron acceptor, which results in strong signal color development at 635 nm and excellent strip storage (Figure 1). A five-wavelength light-emitting diode (LED) array in the detection system allows an algorithm to be used that distinguishes the glucose signal from the blood signal and from cell scattering noise (with little or no hematocrit influence). In a clinical trial, the overall accuracy was within 7.1%. Second, a new system for continuous glucose monitoring has been developed with Arkray Corporation (Kyoto, Japan), called OCP sensing. Unlike most CGMs in use now, which are amperometric and where the signal (current) is proportional to sensor size, OCP sensing has a sensor size-independent signal (voltage change), allowing the creation of a very small sensor. 8 An engineered mutant of a highly stable direct electron transfer type of glucose dehydrogenase enzyme was used for the OCP sensor. Performance was not affected by ascorbic acid or acetaminophen.
The chemical reaction for a new optical transmission absorbance system for blood glucose monitoring. Abbreviation: FADGDH, flavin adenine dinucleotide-dependent glucose dehydrogenase.
Workshop A: New Technologies for Glucose Monitoring; Panel 2: Continuous Glucose Monitoring
Moderators
Thanh Hoang, DO, FACP, FACE
Walter Reed National Military Medical Center, Bethesda, MD, USA
Jennifer Sherr, MD, PhD
Yale University, New Haven, CT, USA
Traceability for BGM and CGM
Guido Freckmann, MD
Institute for Diabetes Technology GmbH, Ulm, Germany
Traceability defined in ISO 17511 is essential for accuracy and comparability.
Traceability for BGMs and laboratory analyzers needs to be improved.
Traceability is a challenge for CGMs and needs to be better defined, especially when CGMs are used for clinical targets.
Abbott Technology
Marc Taub, PhD
Abbott Diabetes Care, Alameda, CA, USA
Abbott’s FreeStyle Libre (Abbott Diabetes Care, Alameda, California, USA) platform of products, which includes the FreeStyle Libre 2 and FreeStyle Libre 3 systems, is used by more than 3.5 million people globally.
A novel model was presented which utilizes sensor glucose readings to determine an individual’s adjusted personalized HbA1c to guide therapy decisions to minimize the risk of both hypoglycemia and long-term hyperglycemia complications.
Early in vivo data on a continuous ketone sensor based on the FreeStyle Libre platform were presented.
Dexcom Technology
Peter Simpson, MS
Dexcom, Inc., San Diego, CA, USA
Dexcom G7 (Dexcom, Inc., San Diego, California, USA) recent clinical trial data highlight the system’s accuracy of 8.7% mean absolute relative difference (MARD) and hypoglycemia accuracy of 6.7 mg/dL mean absolute difference (MAD).
Dexcom is expanding its digital health partnerships, enabled by new tools that provide simple and secure integration with low regulatory effort to the partners.
Dexcom continues to expand its market in T2D through partnerships that leverage continuous glucose monitoring, coaching and population health programs.
WaveForm Technology
Mihailo Rebec, PhD
WaveForm Technologies, Wilsonville, OR, USA
Studies confirmed that US users are able to operate the Cascade CGM (WaveForm Technologies, Wilsonville, Oregon, USA) as effectively as those in Europe.
A reduction in the calibration frequency did not negatively impact the performance of the Cascade CGM.
Additional 15-day studies have not resulted in an increase in skin reactions to the CGM over those observed with 14 days of use.
Medtronic Technology
Robert Mucic, PhD
Medtronic Diabetes, Northridge, CA, USA
Medtronic (Northridge, California, USA) is focused on transforming diabetes care with simpler, smaller, and smarter systems.
Medtronic’s CGM is moving from current technology to the next stages by eliminating fingersticks and simplifying use—providing closed loop and stand-alone CGM therapies.
Preliminary data from the investigational study with an algorithm optimized for the next-generation sensor indicate promising results.
Nemaura Technology
Faz Chowdhury, PhD
Nemaura Medical, Loughborough, UK
A noninvasive sensor has been designed for daily use with a sensor that sits on the surface of the skin and not inside the skin.
A continuous glucose sensor was developed with clinical applications in T2D management.
There are many consumer applications of a noninvasive glucose sensor in diabetes prevention.
Ascensia Technology
Francine Kaufman, MD
Senseonics, Inc., Germantown, MD, USA
Patients on CGMs need access to the most accurate BGM, the CONTOUR Next One (Ascensia Diabetes Care, Parsippany, New Jersey, USA), for confirmation of glucose values, particularly in the low range, and for calibration when required.
The PROMISE Study evaluated two sensors for up to 180 days and showed MARDs of 8.5% and 9.1%, MADs in the hypoglycemia range of <8 to 8.8%-9.4%, event detection rates 93%-99%, and values consistent across the 180-day sensor duration.
The Senseonics product pipeline innovates to extend sensor life, significantly reduce calibrations, allow for intermittent scanning and real-time wear, and use a one-step insertion tool.
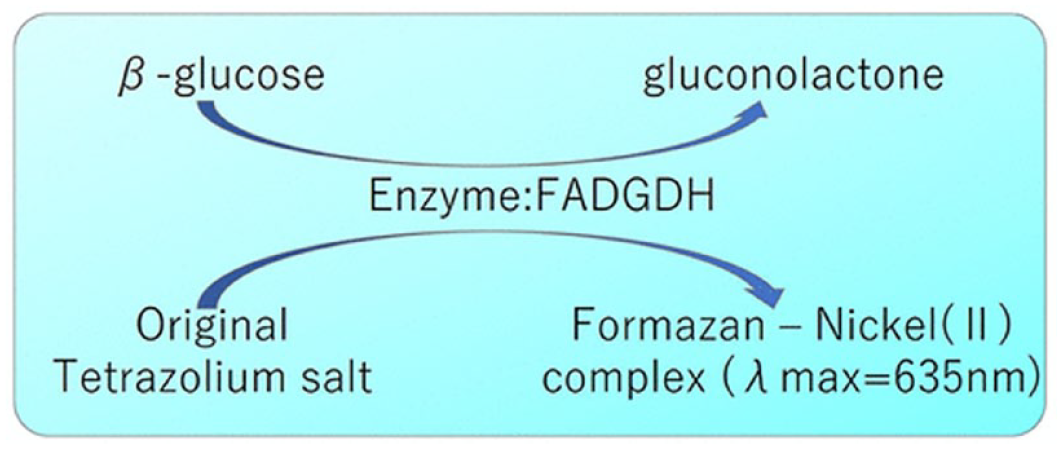
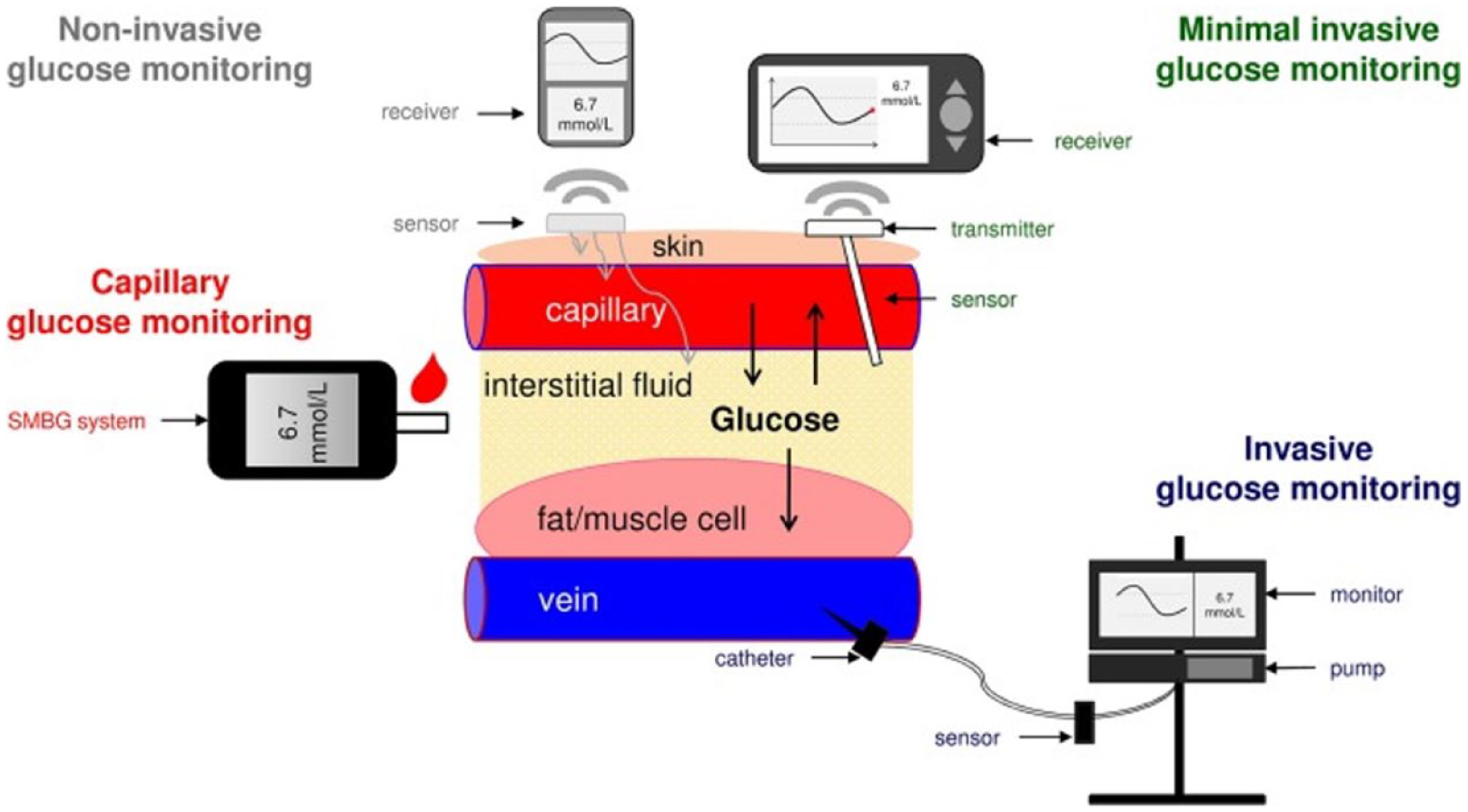
As sensor technology has vastly altered diabetes care, this session focused on updates from six device manufacturers. However, the discussion first began with the concept of traceability. Metrology is the science of measurement, and metrological traceability is defined as the property of measurement whereby the result can be related to a reference through a documented unbroken chain of calibrations. 9 Each part of the chain contributes to the measurement of uncertainty faced. With BGMs, it has been established that even though traceability chains exist, laboratory analyzers and BGMs have systematic differences. 10 Thus, steps have been taken to harmonize and standardize procedures that should overcome these issues. But what about CGMs? While methods should exist for CGM traceability, this is even more difficult because CGMs measure glucose in interstitial fluid, and it is not feasible to obtain reference measures in this compartment as shown in Figures 2 and 3. Thus, it was suggested that for the traceability of CGM measurement, one must consider where the reference measure is being obtained (capillary vs venous blood) and whether an algorithm for compartment correction needs to be considered. 11 This first presentation ended with a call to action to create worldwide standards for CGM traceability and performance evaluation, focusing on the increased role these devices have both in research and clinical settings and use of CGM-derived indices, like TIR, being used to develop treatment targets.
Sampling arrangements of different types of continuous glucose monitors compared with self-monitoring of blood glucose. Interstitial fluid reference measurements are currently not feasible. Abbreviation: SMBG, self-monitoring of blood glucose.
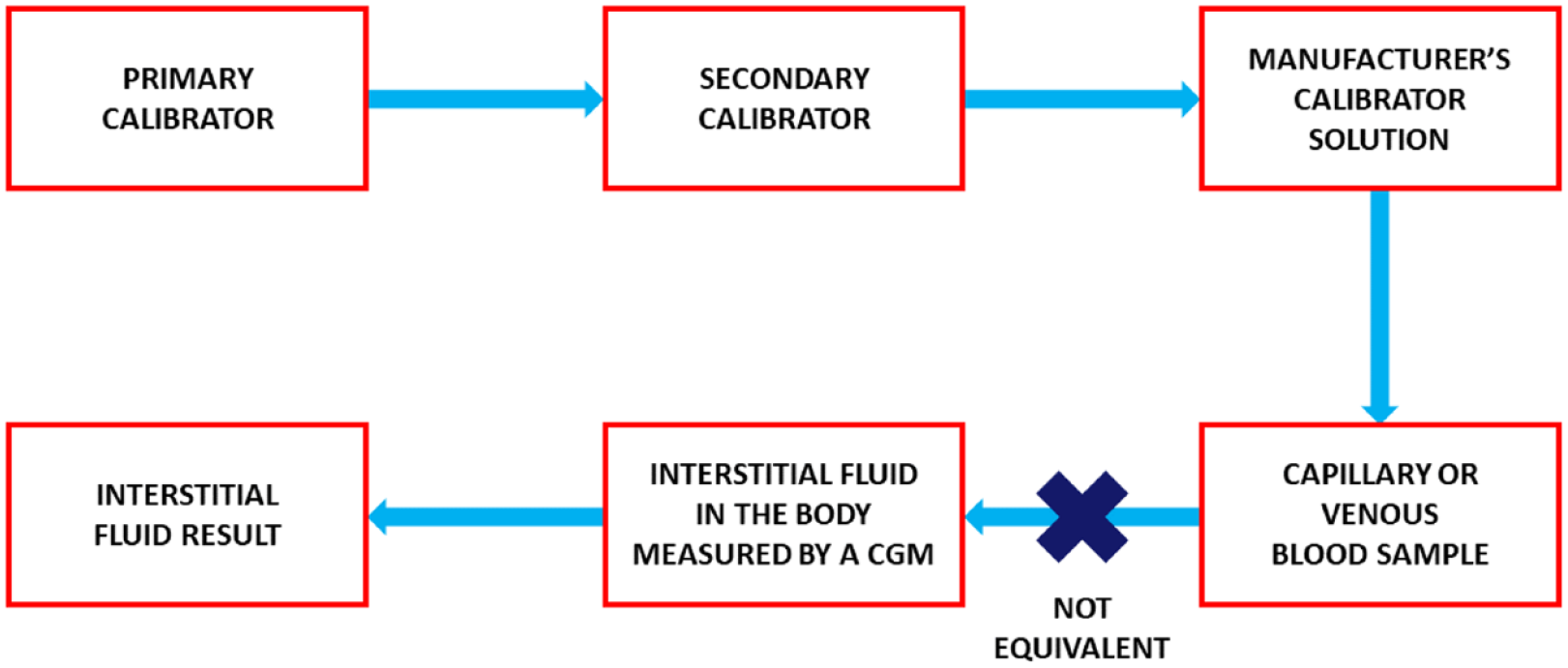
A metrological traceability diagram where the glucose concentration of interstitial fluid measured by a continuous glucose monitor is not necessarily comparable to the glucose concentration of simultaneously obtained capillary or venous blood. 12
Transitioning to discussing individual technologies, the first presented was Abbott’s portfolio. The global impact of this technology was noted as more than 3.5 million PWD are using Libre sensors across 59 countries. In Europe, the Libre 3 has been released, which has the same 14-day duration and accuracy of Libre 2 but has the added benefits of 70% reduction in sensor volume. It is a ready to apply device (whereas Libre 2 requires the user to load the sensor in the applicator), and therefore a reduced environmental footprint. Recognizing that while CGM-based indices, like TIR, are being used more in clinical care, HbA1c remains an important measure. Yet it was highlighted that there is discordance with how HbA1c and mean glucose may relate, which may be alleviated by incorporating red blood cell (RBC) lifespan. While mean RBC lifespan is 105 days, there can be a ±20% variation. 13 Furthermore, RBC glucose uptake is also noted to have ±20% variation. 14 Thus, by modeling for these variables, this new measure has a higher agreement for lab-based HbA1c as compared with the Glucose Management Indicator (GMI) and may be more relevant in helping refine management for PWD. 15 Finally, a recent publication on a novel continuous ketone monitor (CKM) was presented. In a study of 12 participants on low-carbohydrate diets, each of whom wore three sensors, there was concordance between CKM and capillary beta-hydroxybutyrate concentrations using a single retrospective calibration. 16 In total, 82% of CKM values fell within 0.2 mM or 20% of the capillary readings. 16 This work represents the first step for Abbott in the continuous measuring of analytes other than glucose.
The next presentation focused on updates from Dexcom. Since its clinical launch three years ago, the Dexcom G6 has had a number of partner relationships develop. While insurance coverage for CGMs was described as excellent for those on intensive insulin therapy, understanding how to improve coverage for those with T2D who do not require intensive insulin therapy remains problematic. Thus, a real-world study of the Level2 program through United Healthcare (Minneapolis, Minnesota, USA) allowed for assessment of 790 participants in a pilot study, which showed an HbA1c reduction of >1% for those with a baseline HbA1c >8%. In addition, three out of four participants lost weight within three months of starting the program. Additional partnerships that have been developed include Teladoc (Purchase, New York, USA), Livongo (Mountain View, California, USA), Welldoc (Columbia, Maryland, USA), and Onduo (Newton, Massachusetts, USA). With the ongoing COVID-19 pandemic, temporary allowance for CGMs used in hospitalized patients has shown more than 100 hospitals were able to integrate the Dexcom CGM, 24 publications in 2021 showed the accuracy and benefit of Dexcom use in the hospital settings, and the company is working with the United States Food and Drug Administration (FDA) to expand clearance in hospitals beyond this temporary authorization. The presentation culminated with data on the Dexcom G7, which will be more than 60% smaller than the G6 with faster start-up and less of an environmental footprint. 17 The pivotal trial data of 91 adult participants showed an overall MARD of 8.7%, with accuracy noted to be consistent over the beginning, middle, and end of the sensor wear period. The app has been updated to include integrated summary statistics, such as TIR, GMI, and the 24-hour glucose profile, so users will not need to toggle between the Dexcom CGM app and the Clarity app.
Two studies of the Cascade CGM from WaveForm Diabetes were then presented. The CUSP study was a single-center assessment of the CGM to support regulatory approval of the device in the US. In this trial, ten participants were recruited and underwent four 8-hour accuracy assessments of the CGM compared with reference YSI glucose values. The overall average MARD was 11.2%, with the highest MARD of 13.0% noted on the first day of sensor wear. The second study, CCTDCP, assessed the accuracy of the sensor with one calibration performed every two days. The currently commercially available systems require two calibrations on day one and then one calibration daily for the duration of wear. For this trial, 15 participants were recruited, each of whom wore two sensors. They each underwent four 8-hour accuracy assessments. With the hybrid algorithm allowing for a reduced frequency of calibrations, overall MARD was 9.2%. These data have been submitted to modify the frequency of required calibrations to regulators in Europe. Future planned advancements include reduction of calibrations to once weekly, reduced physical footprint of the device, improvement in day one performance, and improved adhesives to continue their goal of minimal skin irritation caused by these devices.
With the goal of creating devices that are simpler, smaller, and smarter, Medtronic offered information on products that are recently approved, ongoing studies, and products for which they are seeking regulatory approval. Discussion was held regarding two recent approvals: FDA approval of the InPen, a connected insulin pen, that allows for integration of real-time sensor data as part of its ecosystem and FDA approval of extended wear insulin infusion sets. In terms of automated insulin delivery (AID), the Medtronic 780G with the Guardian 4 sensor is approved outside the US and is under FDA review. This sensor has changes in the software allowing it to forego the need for calibrations. The synergy sensor builds on this technology but has the benefit of a sensor pre-attached to a disposable transmitter and smaller physical footprint along with easier insertion. A pivotal trial of the synergy sensor involving 200 participants has been conducted. While the findings from this study are not yet available, data regarding sensor performance in hypoglycemia (<70 mg/dL) showed 85% concordance of sensor glucose readings to YSI reference values within ±15 mg/dL.
Nemaura Medical shared an update on their sensor technology, SugarBEAT, which is a transcutaneous sensor that has a reusable transmitter and disposable sensors that measure the interstitial glucose without requiring needle insertion. This system works via electrochemical reactions and reads glucose levels in fluid that is diffused across the skin, which in some situations has led to a mild erythema. Overall MARD for the system was noted to be around 12.0%. Understanding that many people with T2D do not regularly perform fingerstick glucose readings, application of SugarBEAT technology is now planned with a specific focus on this population in the United Kingdom. Ultimately, there are plans for direct-to-consumer (DTC) availability of the device.
The final presentation encompassed the Eversense implanted sensor. The PROMISE study, which evaluated the next-generation Eversense CGM that has an intended lifespan of 180 days, was presented. A total of 181 participants were recruited and each underwent ten clinic visits lasting up to ten hours to assess accuracy of the device. 18 Nearly 25% of the participants had sensors with a specific chemistry modification to reduce oxidation of the glucose-binding indicator chemistry. 18 Notably, while the sensor requires two calibrations per day for the first 21 days, calibration frequency for the duration of sensor wear drops to one per day. 18 The overall MARD across all participants was found to be 9.1%, which dropped to 8.5% in the sensors with modified chemistry, with accuracy metrics being consistent across the 180-day sensor life. 18 Future innovations will include extension of wear out to one year with the Rome sensor, with the benefit of only one calibration per week and the addition of a battery to the internal sensor, which will allow for point in time readings. With these alterations, the Eversense product will be available both as a real-time CGM and as an intermittently scanned sensor.
The past year has seen continued innovation in the field of continuous glucose monitoring. Efforts to improve device accuracy, reduce sensor volume, and ensure the environmental impact of the consumable products is lessened remain areas of active investigation. A number of the presenters have devices under review by regulatory agencies or plan submissions in the next year. This will, no doubt, continue to impact the introduction of sensor therapy for all PWD.
Workshop B: Connected Health; Panel 1: Which Novel Sensors Will Become Established for Diabetes?
Moderators
Eda Cengiz, MD, MHS, FAAP
University of California, San Francisco, San Francisco, CA, USA
Hazhir Teymourian, PhD
University of California, San Diego, San Diego, CA, USA
Smart Pens
Søren Smed Østergaard, MS
Novo Nordisk, Copenhagen, Denmark
With smart pens, people can feel confident speaking about their dosing patterns.
Smart pens will hopefully decrease the prevalence of incomplete paper diaries.
HCPs can retrospectively assess blood glucose and insulin dose data, enabling better titration and establishment of routines.
Wireless Blood Pressure Monitoring
Dessi Zaharieva, PhD
Stanford University, Palo Alto, CA, USA
Remote blood pressure monitors are important digital health devices that can monitor and report blood pressure data.
Lack of accuracy and cybersecurity of current blood pressure monitoring devices as well as user error in proper use of these devices pose challenges to widespread adoption of remote blood pressure monitoring.
The value of these devices will increase by improving accuracy, implementing patient training, and developing new cybersecurity standards.
Apps for Estimating Food Intake
Ricardo Gutierrez-Osuna, PhD
Texas A&M University, College Station, TX, USA
Poor diet is an important contributor to chronic diseases.
Monitoring food intake is a critical problem in dietary research.
Advances in sensing and machine learning (ML) are contributing to technical solutions to diet monitoring.
Foot Sensors
David G. Armstrong, DPM, MD, PhD
University of Southern California, Los Angeles, CA, USA
Every 1.2 seconds, someone develops a diabetic foot ulcer.
Following healing of a diabetic foot ulcer, 40% of people will have a recurrence in one year. Healing is now called “remission.” Our goal is to maximize ulcer-free, hospital-free, and activity-rich days.
Over the past decade, numerous wearable, ambient, and even implantable technologies have been developed and are now being used to help maximize those metrics.
Continuous Ketone Monitoring
Kristin Castorino, DO, BC-ADM
Sansum Diabetes Research Institute, Santa Barbara, CA, USA
Continuous ketone monitoring technology, which measures beta-hydroxybutyrate, is feasible in humans.
Access to beta-hydroxybutyrate-based ketone monitoring should be assured for all PWD who use insulin.
The clinical utility and justification for CKMs have yet to be determined.
Recent advances achieved in monitoring other parameters and biomarkers beyond glucose were discussed in the context of improved diabetes management. The workshop began with a description of smart insulin pens to automatically and passively register dose and time of insulin administration. NovoPen 6 (Novo Nordisk, Bagsværd, Denmark) is an insulin injection device capable of recording and storing the last 800 doses and transferring the information via near-field communication technology to a smartphone app to be viewed by authorized HCPs. In a clinical proof-of-concept study conducted on patients with T1D in Sweden, the introduction of NovoPen 6 showed better outcomes for the patients, including a 21% increase in TIR and 43% fewer missed meal-time insulin injections. 19
With diabetes, there is an increased risk of elevated blood pressure and cardiovascular diseases. Remote home blood pressure monitoring can bring about numerous benefits to the field of personalized healthcare as it saves time and enhances access to care for the patients. Studies have shown that it can effectively help lower blood pressure levels. 20 However, the current technologies of blood pressure monitors are facing three major barriers including (1) limited accuracy, (2) user error in proper operation of the device, and (3) lack of device-specific standards to ensure cybersecurity for wireless monitors, which should be addressed to warrant the wide adoption of at-home blood pressure devices in the future.
Personalized nutrition is an emerging field aimed at preventing and managing diseases by tailoring diet based on the specific requirements of each person. 21 Mobile apps are currently available for diet logging through their massive databases that contain nutritional content of a very large number of foods and meals. 22 In parallel with advances in mobile apps, progress has been made in sensor-based approaches including both wearable physical sensors (e.g., gesture recognition and smart utensils) and chemical sensors (e.g., CGMs and CKMs) to enable a more accurate tracking of eating behaviors. Further advances in wearable sensing technology and its integration with measurements of gut microbiome and ML methods will greatly empower the personalized nutrition programs.
Diabetic foot ulcers and related maladies continue to be among the most common serious diabetes-related sequelae. 23 The incidence of ulcer recurrence for a patient in diabetic foot remission is 40% in the first year, 60% in three years, and at least 65% in five years. 24 In the face of neuropathy or the loss of protective sensation, there are two variables, namely pressure and activity, that lead to diabetic foot ulceration. Studies suggested that variability in activity among individuals with diabetes is linked to higher risk of developing ulceration. 23 Modulating pressure, either externally using protective shoes and insoles or internally through surgery and physical therapy, along with modern wearable activity monitors, has been proven to prevent severe diabetic foot ulcer recurrence and thus to enhance quality of life for PWD.
Continuous ketone monitoring bears significance value primarily as a means to prevent or reverse diabetic ketoacidosis (DKA). Notably, the impressive results of the first evaluation of a wearable CKM in 12 human subjects over a 14-day period were presented. 16 The sensor used wired enzyme electrochemistry technology similar to that in an Abbott CGM, but in connection to the nicotinamide adenine dinucleotide (NAD+)-dependent beta-hydroxybutyrate dehydrogenase enzyme. Figure 4 presents the correlation between sensor results and reference values using a single retrospective calibration. Continued innovations are planned, including studies in intended patient populations of both DKA and intentional ketosis along with the prospective factory calibration to enable real-time CKM.
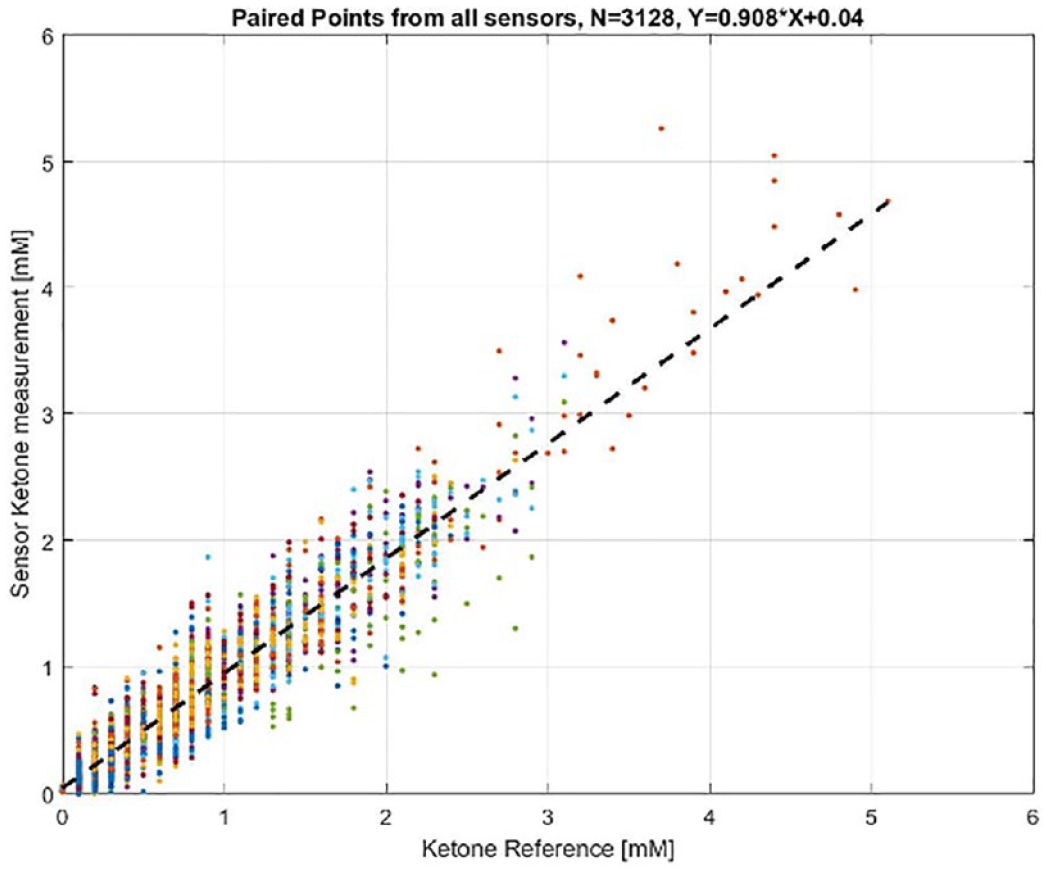
Plot of interstitial fluid ketone values measured by the wearable continuous ketone monitor sensors against capillary ketone strip reference measurements.
Workshop B: Connected Health; Panel 2: Can Direct-to-Consumer (DTC) Telehealth Programs Meet Local Needs?
Moderators
Andjela Drincic, MD, FACP
University of Nebraska Medical Center, Omaha, NE, USA
Barry Ginsberg, MD, PhD
Diabetes Technology Consultants, Arlington, VA, USA
Panel Discussion
Shailendra Bajpai, MBBS, MD
Becton Dickinson Greater Asia, Singapore
The current healthcare system in large parts of the world is constrained by multiple factors including inefficiencies, lack of infrastructure, and gaps in HCP knowledge and skills leading to delayed adoption of standard-of-care and barriers to access.
There is a need to do a gap analysis of the unmet needs across the healthcare value chain and evaluate suitable options through a concerted effort by all the key stakeholders to improve the standard-of-care by deploying lean and agile telehealth models to improve outreach and access to healthcare for this population.
Although there has been enhanced uptake of DTC and telehealth solutions in the developing world, there still remains a good scope of improvement to support consumerization of healthcare and to unburden the stretched healthcare system by addressing remaining challenges in the areas of regulatory, legal, and payer system as well as better adoption by all the key stakeholders.
Ronald Dixon, MD
CareHive, Austin, TX, USA
DTC solutions are improved by front-end data and analytics.
Asynchronous-first telehealth provides value for those consumers willing to engage.
Real-time return on investment provides value to purchasers as consumers engage with telehealth solutions.
Leslie Eiland, MD
University of Nebraska Medical Center, Omaha, NE, USA
Michelle Griffith, MD
Vanderbilt University, Nashville, TN, USA
Telehealth is well suited to diabetes.
Diabetes care is complex and requires a team.
DTC models have limitations in addressing the full spectrum of diabetes care.
Gretchen Zimmermann, RD, CDCES
Vida Health, San Francisco, CA, USA
Virtual care improves healthcare delivery; barriers to accessibility are overcome with DTC models of care.
DTC telehealth can support an integrated, patient-centered care model that treats the whole person by addressing mental health and physical health together, leading to stronger outcomes.
DTC telehealth allows for a continuous care model, individualized treatment plans and long-term behavior change support, enabling greater HbA1c outcomes.
Although the COVID-19 pandemic has been disastrous in many ways, it has improved telehealth adoption and spurred communications development. Initially, the use of telehealth was limited to healthcare centers providing care for rural communities, but the pandemic expanded the population of telehealth users and prompted rapid innovation in healthcare delivery. Perhaps most importantly, an example of this innovation is DTC telehealth. This session discussed telehealth as a whole and DTC telehealth, covering both US and international perspectives. Ambulatory or outpatient telehealth visits are structured in three main ways. The first is a clinic-to-clinic model or facility-based model where a patient presents to a remote clinic and may be aided by a telepresenter. The visit can be enhanced by peripheral examination tools like remote stethoscopes, and lab results can be incorporated as well. The second are direct-to-patient and DTC models where the patient, typically not at a healthcare facility, will use their own device to connect with an HCP. The third is the kiosk model, where large healthcare systems will place kiosks in areas such as pharmacies that provide patients with an access point to healthcare without needing their own technology.
In the direct-to-patient model, the patient is often virtually connecting with their own HCP, who would be available for in-person care as well if desired. In the DTC model, there is an independent entity, usually a company, directly providing a service to the patient with no option for in-person care. DTC telehealth is bringing competitive pressure to traditional healthcare, as it evolves from focusing on acute care to encompassing more boutique services of chronic care and health maintenance.
Telehealth is well suited for care of PWD for several reasons. Diabetes is becoming increasingly prevalent and is about 17% more prevalent in rural areas than urban ones. 25 However, 62% of rural counties lack diabetes self-management education and support services, so telehealth would help more PWD in these areas access appropriate healthcare services. 25 Diabetes specialists are also scarce in rural areas. 26 Also, many diabetes technologies now allow for cloud-based data sharing, so HCPs are able to access the medical history of PWD during telehealth visits. In these aspects, DTC telehealth is able to support traditional healthcare. There still exist limitations for DTC telehealth for diabetes care. HCPs that are able to understand limitations provided by the local environment, such as the availability of food, forms of exercise, or equipment, can provide personalized care. This quality of care may be difficult if PWD are not seeing the same HCP at each telehealth visit and if the HCP is not located in the same city or even state as the PWD. There is certainly a place for DTC telehealth in diabetes education and technology management, but there exist limitations to be improved upon.
Healthcare hybridization refers to a hybrid combination of what we predict and what we fail to anticipate. The future is a nonlinear hybrid in which patterns and trends coalesce in ways we can never fully anticipate. The hybridization of healthcare refers to simplification of access. The concept of hybridization applies to many aspects of healthcare delivery as shown in Figure 5. Healthcare can be provided either synchronously or asynchronously. Asynchronous-first telehealth means the establishment of contact with a patient through text or access to a set of questions to understand their health issue before arranging a synchronous interaction with an HCP. With a network of different healthcare institutions and technologies in use, clinical navigation services can help a person connect with their HCP or locate the specific clinic or pharmacy that they need to visit. These services can also all occur over a spectrum from fully in-person care to fully virtual care via telehealth. Telehealth is able to cover a broad range of healthcare needs from acute care to post-acute care to chronic care.

Hybrid healthcare promotes simplified access to healthcare. Abbreviations: ER, emergency room; PCP, primary care physician.
Telehealth is best used with data informing the clinical needs of the population to be cared for and measurement of the impact of the intervention on this population. Data analytics can help tailor the best solution to impact a particular population based on their specific healthcare needs. The effective use of data can also help with the telehealth reimbursement model by facilitating delivery of both synchronous and asynchronous telehealth to help with at-risk entities and comprehensive coding. This will allow patients to stay engaged or become re-engaged with healthcare visits to benefit their own health and facilitate payment for missing diagnostic codes. CareHive 360 is an intervention solution that focuses on digital engagement through an asynchronous-first telehealth model. 27 Patients will first be engaged through text and then escalated to a navigator that will act as triage that will guide people to the correct service. The clinical information gathered will be sent to the patient’s primary care physician and will be used to better manage their chronic disease. This solution focuses on patient experience, navigator experience, and the back-end agent engine. Data analytics at the back-end quantifies the impact of the intervention and allows for continuous improvement and predictive capacities for real-time return on investment.
Traditional brick and mortar healthcare settings can have barriers to accessibility such as HCP availability, the ability to integrate mental health and physical health support, literacy challenges, and social determinants of health. DTC telehealth can improve healthcare delivery by meeting patients where they are, within their schedule, and without the commute. At Vida Health, each person’s healthcare journey is tailored to their needs. Based on their healthcare information and health goals, people are matched with a care team and program that suits them. As part of the integrated care model, the care team will often involve various HCPs such as physicians, pharmacists, dieticians, certified diabetes educators, and therapists. Also, instead of using only a didactic approach, Vida Health uses motivational interviewing and understands where patients are in their healthcare journey while having patients put in the work to do tasks like track their blood sugar or their weight. 28 This engagement with HCPs and the Vida Health app was shown to help patients reduce their HbA1c levels by 60% compared with those who were not as engaged. 29 Effective DTC telehealth should improve three areas in particular with diabetes: nutrition, blood glucose self-monitoring, and medication adherence. In addition, diabetes never stands alone. People with T2D are two times more likely to develop depression, and people with depression are 60% more likely to develop T2D. 30 A DTC telehealth model that can integrate mental and physical healthcare can sometimes more easily address these issues than in a brick and mortar setting. DTC telehealth solutions can also easily integrate into existing health benefit ecosystems through using data to coordinate care and help patients navigate the healthcare system and make necessary referrals.
Developing countries face particular challenges such as limited healthcare infrastructure (facilities, specialty care, outreach), rapid lifestyle changes (higher birth rate, aging population, urbanization, changing family patterns), an increase in chronic diseases, and limited per capita healthcare expenditures. There is also less emphasis on preventative care, causing resources to be wasted in management of chronic diseases and other comorbidities downstream. Many patients need to pay out-of-pocket, so affordability is another barrier that can cause other issues like poor medication adherence in an attempt to save on costs. Healthcare is evolving in developing countries, with some countries adopting universal healthcare to try to focus on population health. There has also been the rapid consumerization of health segments such as the growth of over-the-counter medications and the prescription model that allows for more choices for consumers. Data is also relatively affordable with supporting technology infrastructure that is helping to reduce the digital divide. There has also been rapid e-commerce and marketplace evolution and adoption, which allows for DTC telehealth opportunities. Many healthcare companies are engaging consumers through social media, which also provides a lot of relevant data. These developments increase the availability and accessibility of digital health solutions in developing countries as cost-effective and time-saving solutions. In addition, there is the trend of “uberization” (on-demand service delivery through direct contact between a customer and supplier by way of electronic or mobile communication) of certain pockets of healthcare such as E-pharmacies that decrease the cost of these services. 31 Especially during the COVID-19 pandemic, there has been enhanced regulatory framework for telehealth that improves payer adoption. 32 There has been an increase in digital engagement on multiple telehealth platforms, again highlighting the opportunities for DTC telehealth in these emerging and developing markets.
Although there still are unmet needs such as covering specialty care, embodying the art of medicine (empathy, compassion, trust, etc that can be achieved through an in-person interaction), and other policy/payer considerations, DTC telehealth has the potential to be a reliable, effective, and safe solution at a reasonable cost for patients. It would allow for an offloading from the current healthcare infrastructure, thereby increasing the efficiency in the system. DTC telehealth would also improve healthcare outreach, especially for remote and inaccessible areas. Overall, it would enhance the patient’s experience, empowerment, decision-making, and opportunities for goal-based behavior changes.
Keynote Presentation: Technology and the ADA—Pushing for Adoption
Robert Gabbay, MD, PhD
American Diabetes Association, Arlington, VA, USA
Despite significant advances in technology for diabetes, adoption remains a challenge.
American Diabetes Association (ADA) has been working to improve adoption among the four key domains: clinicians, patients, regulatory agencies, and payers.
In the future, ADA will be focused on simplifying requirements for insurance coverage, expanding durable medical equipment (DME) coverage for apps and all CGMs, and reinstating automatic insurance coverage rule for breakthrough technology.
The value of diabetes technology in improving patient health outcomes is already well recognized among many endocrinologists and diabetes educators. Diabetes clinicians appreciate technologies such as CGMs for their accurate and comprehensive monitoring of blood glucose measurement trends over time. However, limited insurance coverage or lack thereof and high costs continue to be major barriers for many PWD trying to access diabetes technologies. The fast pace of clinical care, workflow challenges, and short appointment times can make it difficult for HCPs to educate and train their patients to effectively use technologies.
In addition, inequities in technology access also limit their widespread adoption. For young adults with T1D, insulin pump and CGM use were lowest among Blacks, intermediate among Hispanics, and highest among Whites. These disparities could not be solely explained by socioeconomic status. 33 Within the T1D Exchange registry, the lowest socioeconomic status correlated with the lowest use of technology and the highest levels of HbA1c. 34 DKA rates were higher across all racial and ethnic groups that did not have access to technology, particularly CGMs. 35 With all the advancements in diabetes technology over the past two decades, the biggest challenge is no longer innovation but adoption. To overcome barriers to the adoption of diabetes technologies, ADA has been working to address four main domains: (1) clinicians, (2) patients, (3) regulatory agencies, and (4) payers (Figure 6).
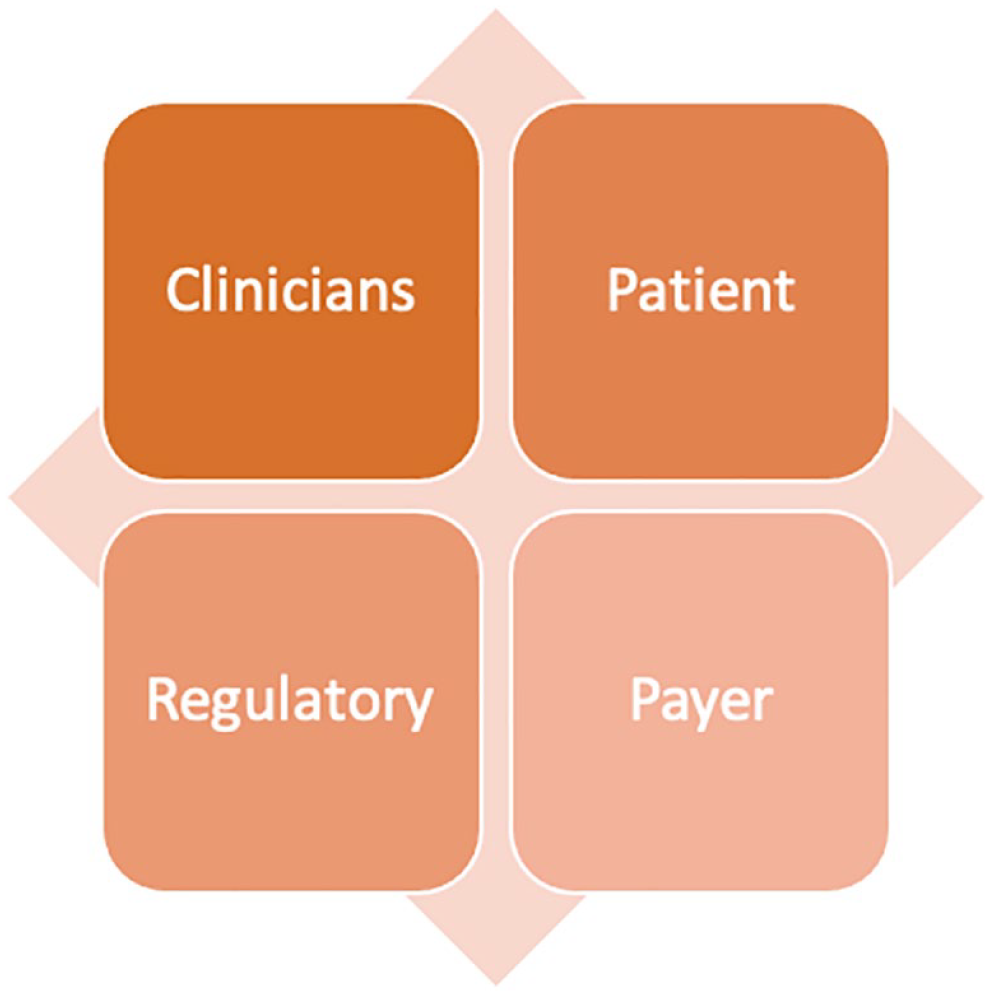
The four domains to address as part of the roadmap to diabetes technology adoption.
A qualitative study of clinicians conducted by ADA showed that while TIR is generally understood by endocrinologists and certified diabetes care and education specialists, it was not common knowledge for primary care professionals, who provide care for 90% of PWD in the US. Following the study, ADA launched three major efforts. The first is a nationwide effort to encourage using and monitoring blood glucose levels beyond HbA1c using TIR to better diabetes management. 36 The second aims to help HCPs operationalize new technologies through a team-based approach to care. 37 The implementation of technology into the clinical workflow would necessitate more time during appointments, particularly for primary care professionals. The increased volume of data from CGMs can be overwhelming and lead to device burnout. By configuring the workflow and delegating new roles to their team, an HCP can reduce the burden of implementing the use of diabetes technology. The third is a free online professional education portal maintained by ADA. 38 The website is globally accessible to both HCPs and patients anywhere in the world.
ADA also conducted a qualitative study on PWD, finding that while many PWD did not understand TIR (how TIR relates to HbA1c or how TIR can be used to promote better long-term health outcomes), they would be willing to adopt technology in hopes of it reducing their disease burden. HCPs will have a crucial role in educating PWD on TIR and its benefits to diabetes management, particularly for changes to insulin doses, carbohydrate intake, and exercise. ADA maintains an online consumer guide of available diabetes products with information that PWD may need when purchasing or using drugs and devices. However, coverage and cost barriers would still need to be overcome to expand access to technology.
On the regulatory level, ADA has been leading discussions with the Center for Devices and Radiological Health (CDRH), which oversees the premarket approval of all medical devices, and the Center for Drug Evaluation and Research (CDER), which regulates all over-the-counter and prescription drugs. Although CDRH pivoted to the approval of COVID-19 tests during the pandemic, ADA has been working to help and is optimistic that CDRH can soon refocus on the many diabetes devices that have been waiting in the pipeline. ADA has also discussed with CDER about including TIR data into clinical trials and on package inserts to signal to HCP the importance of TIR. In addition, regulatory agencies have begun to recognize the value of real-world evidence (RWE) in providing important data, including economic data, that cannot effectively be collected through randomized controlled trials. The ADA Advocacy Group has formed a Technology Action Coalition to promote these efforts on both the regulatory level and the payer level.
On the payer level, there is considerable variation in the use of technology by PWD with various payers. For example, CGM use is two to five times lower for Medicaid than for commercial plans among similar age groups. 39 There is also considerable state-level variation across payers. Payer coverage has the potential to expand technology access to even more PWD, so addressing payer challenges will be crucial. Payers often have a short-term mindset, wanting to see an immediate return on investment. This is not always possible in the case of diabetes because complications can take time to develop. Although payers require a strong business case for coverage, studies do not often focus on economic issues. This may be resolved as more studies begin to embrace RWE, increasingly the available economic data. There is also a need to standardize and simplify DME coverage.
ADA has been successful in working with payers to overcome challenges to adoption. At the beginning of the COVID-19 pandemic, ADA facilitated CGM coverage changes to allow virtual visits to count as acceptable visits. For Medicare, PWD need regular visits to qualify for CGM coverage. ADA also successfully advocated for in-hospital use of CGM and eliminated the four-times-a-day fingerstick glucose testing requirement for CGM coverage. However, more work needs to be done. ADA’s partnership with Centers for Medicare & Medicaid Services (CMS) is focused on three future goals: (1) eliminating the requirement that PWD must measure C-peptide and be on three or more injections, (2) expanding DME coverage for all apps and all CGMs, and (3) establishing an automatic rule for breakthrough coverage, such that if the FDA approves of a novel technology, then CMS will automatically provide insurance coverage for it.
Session 1: Best Metrics for Glycemia Using CGM
Moderators
Will Cefalu, MD
NIDDK/NIH, Bethesda, MD, USA
Claudio Cobelli, PhD
University of Padova, Padova, Italy
Clinical Importance of Glycemic Variability
David Rodbard, MD
Biomedical Informatics Consultants LLC, Potomac, MD, USA
The Coefficient of Variation (%CV) is the most useful and practical metric to measure and control glycemic variability. When %CV exceeds 20%, there is a progressive linear or even exponential increase in risk of hypoglycemia below 70 mg/dL and below 54 mg/dL.
The frequency distribution for glucose is asymmetrical with positive skewing (to the right), making it more difficult to measure variability. The distribution for the logarithm of glucose is more nearly symmetrical and Gaussian. %CV is best because %CV is roughly proportional to the standard deviation of log glucose.
The display of a simplified distribution for categories of glucose (glucose ranges) enables one to evaluate variability for any day, time of day, day of the week, or over longitudinal time.
A longer period of CGM recording is needed for measuring glycemic variability than metrics such as TIR or mean glucose. Ideally one would have four weeks of CGM data to achieve a relative error of ±5% in %CV.
Time in Range Is Predictive of Diabetic Vascular Complications
Roy Beck, MD, PhD
Jaeb Center for Health Research, Tampa, FL, USA
TIR largely reflects time in hyperglycemia and is strongly correlated with HbA1c after adjusting for individual differences in glycation.
TIR computed from quarterly 7-point blood glucose testing in the Diabetes Control and Complications Trial is strongly associated with the development or progression of retinopathy and microalbuminuria, the results of which have been supported by several recent studies that have demonstrated the association between CGM-measured TIR and diabetic complications.
TIR should be considered to be a clinically meaningful endpoint for clinical trials.
The New Composite Dysglycemia Index for Assessing CGMs
David Klonoff, MD, FACP, FRCP (Edin), Fellow AIMBE
Mills-Peninsula Medical Center, San Mateo, CA, USA
The Glycemia Risk Index (GRI) is a single number that expresses the quality of glycemia in a CGM tracing that answers the two questions: (1) How is this patient doing? and (2) If the patient is not doing well, then what is needed?
The GRI is a composite score that represents the mathematicized opinions about the quality of glycemia in a CGM tracing per crowdsourced information from 330 clinicians who use CGM.
The GRI is expressed as a single number percentile score from 0 to 100 (where 0 is the best and 100 is the worst) based on the magnitude of dysglycemia present, which is a weighted combination of time spent in Ambulatory Glucose Profile (AGP)-defined zones of mild and severe hypoglycemia and mild and severe hyperglycemia.
By summarizing a 14-day tracing as a single point on a grid, multiple tracings for one patient or multiple tracings for a population can all be presented in a single figure, allowing progress over time to be easily visualized.
Using Time in Range to Optimize and Personalize Diabetes Management
Richard Bergenstal, MD
International Diabetes Center, Minneapolis, MN, USA
CGM has expanded the goals of glucose control, and the AGP report helps organize the data for effective action (more green, less red; flat narrow and in-range).
Glycemic-independent factors (cardiovascular disease/heart failure/chronic kidney disease) and diabetes distress (quality of life, reducing burden) need to be considered and could be added to the AGP report.
Innovations for effective therapy personalization and tracking are needed, for example, integrating CGM directly into the electronic health record (EHR) or generating an AGP risk score like the GRI may minimize therapeutic inertia.
CGM has revolutionized diabetes treatment: the ability to continuously measure glucose concentration is a unique situation in the field of chronic diseases. The amount of data available by CGM is tremendous, and there is a need to translate this rich database into actionable strategies for an optimal diabetes therapy. The session moves along this direction trying to provide insight into the best metrics for glycemia using CGM. Four investigators in the field were chosen to provide their perspective: Drs. David Rodbard, Roy Beck, David Klonoff, and Richard Bergenstal. Each of them provided some key points listed below on how to use the data-rich information provided by CGM.
An interesting new metric was also presented, the GRI (Glycemia Risk Index), which is a single number percentile score from 0 to 100 (where 0 is the best and 100 is the worst) based on the magnitude of dysglycemia present as hypoglycemia risk and hyperglycemia risk. The score is a weighted combination of time spent in four AGP-defined zones of mild and severe hypoglycemia and mild and severe hyperglycemia. 40 This new metric should answer two key clinical questions: (1) How is this patient doing? and (2) If the patient is not doing well, then what is needed? The future validation of GRI in larger outpatient cohorts is expected to set the stage for this index to enter our clinical practice in the near future as well as describe the quality of glycemia in population studies and outcome studies.
Additional insights included (1) %CV is the most useful and practical metric to measure and control glycemic variability; (2) the frequencies of interstitial fluid glucose at various times of day are asymmetrical and skewed to the right (Figure 7); (3) TIR largely reflects time in hyperglycemia, is strongly correlated with HbA1c after adjusting for individual differences in glycation, and should be considered to be a clinically meaningful endpoint for clinical trials 42 ; and (4) glycemic independent factors (cardiovascular disease/heart failure/chronic kidney disease) and diabetes distress (quality of life, reducing burden) need to be considered and could be added to the AGP report. The AGP report helps organize the data for effective action with a goal of more TIR (the green zone) and less time below range (the red zone). 43
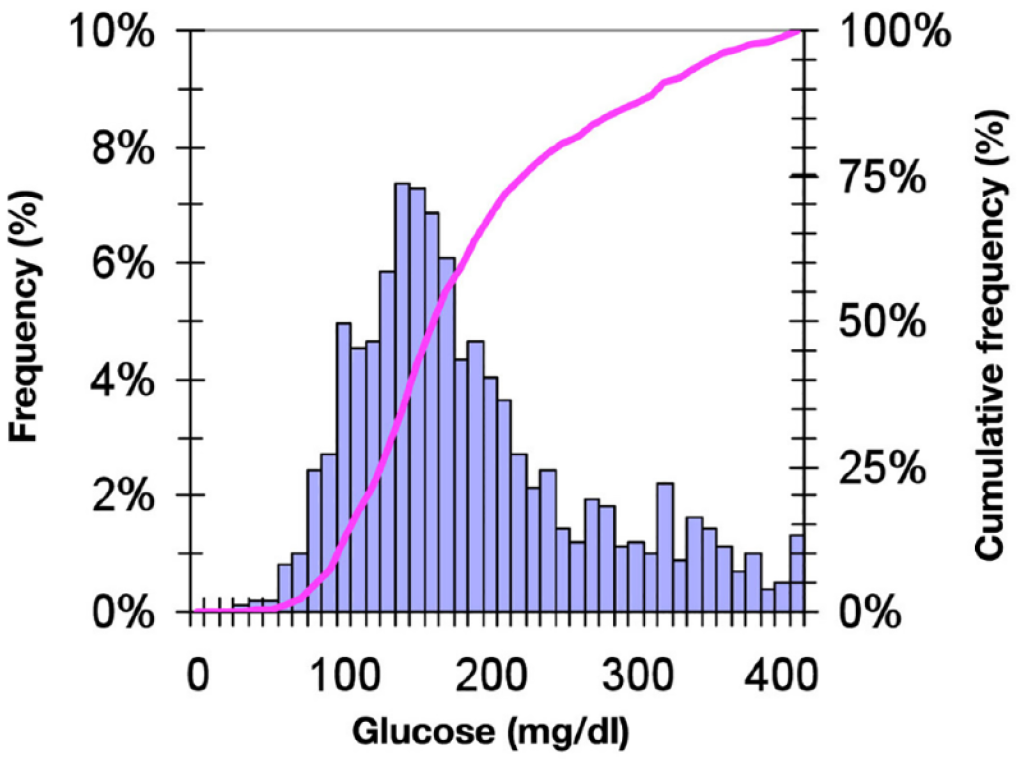
A glucose frequency distribution with a histogram for all times of day (blue bar chart), which is asymmetrical and skewed to the right, and the corresponding cumulative frequency distribution (pink curve).
Session 2: Software for Diabetes
Moderators
David Ahn, MD
Mary & Dick Allen Diabetes Center at Hoag, Newport Beach, CA, USA
Guillermo Umpierrez, MD
Emory University, Atlanta, GA, USA
Insulin Dosing Software
Moshe Phillip, MD
National Center for Childhood Diabetes
Schneider Children’s Medical Center of Israel, Petah Tikva, Israel
Metabolic outcomes of PWD have not improved despite the introduction of new advances in insulin delivery and glucose monitoring.
Decision support systems are needed to cope with the increasing amount of health data being gathered by diabetes devices.
Clinical studies have shown that artificial intelligence (AI)-driven decision support systems are not inferior to physician management at highly regarded academic hospitals.
Hurdles to Bringing Digital Health Technologies to the Market
Chris Boulton, BSc
Sanofi, Bridgewater, NJ, USA
There is a large opportunity to boost the adoption of digital therapeutics, particularly post-COVID-19.
Many challenges remain to the adoption of diabetes software, including the negative stigma of technology, health disparities due to cost, and privacy concerns.
The path to commercialization still remains intriguing yet uncertain, with different product combinations, stakeholders, pricing, and promotional models.
Getting the Most Out of the EHR
Juan Espinoza, MD
Children’s Hospital Los Angeles, USC, Los Angeles, CA, USA
Technology plays a critical role in the management of diabetes, and these tools capture large amounts of patient data that can be used to guide and personalize treatment.
HCPs need streamlined, integrated EHR workflows to efficiently leverage the vast amount of patient data available.
Data standards are urgently needed in all aspects of diabetes data management to support effective dissemination and interoperability.
The session started with describing the process of creating and validating a software platform intended to help standardize clinical recommendations for dosing insulin. The DreaMed Advisor platform (DreaMed Diabetes, Ltd., Petah Tikva, Israel) makes clinician-facing insulin dosing suggestions and has received FDA clearance for use with T1D and T2D, for PWD who utilize any combination of multiple daily injections, continuous subcutaneous insulin infusion (CSII), BGMs, and CGMs.
As an iterative model for how to demonstrate clinical efficacy for novel digital therapeutics, the earliest DreaMed Advisor study was an expert validation study, showing that their platform made similar dosing recommendations as clinician experts in a small subset of 15 patients.44,45 The next progression was to do larger feasibility studies in several trials showing that Advisor suggestions resulted in noninferior clinical outcomes when compared with an expert-driven control group. Now, the DreaMed Advisor platform is going through larger proof-of-concept studies to determine whether its recommendations can be safely made directly to patients, without requiring approval from an HCP. If successful, the final step would be to demonstrate the algorithm’s noninferiority in a large-scale, multicenter randomized control trial.
Next, the session addressed the overall landscape for digital therapeutics from a broader perspective. The upside for diabetes software is that diabetes represents the largest share (19%) of the digital therapeutics market for treatment-related applications in 2020, but there are significant outstanding challenges to widespread adoption such as regulatory hurdles, the healthcare sector’s resistance to change, and the need for low-cost solutions that are accessible to diverse populations. 46
A key takeaway related to privacy and security was that the fast-evolving world of consumer technology raises many questions: (1) how do we deal with the increasing amounts of personal health-related data being generated, both from social media and wearable sensors? and (2) how do we differentiate between medical and nonmedical data as consumer technology companies such as Amazon, Apple, Google, Meta, and Samsung enter the digital health market?
The session then covered the vital role that EHR integration plays in using diabetes software. With the extensive amount of quantifiable data that PWD gather from both consumer and medical devices, diabetes would appear to be ideally suited for EHRs that serve as the crucial hub for clinical management. Unfortunately, the current state is plagued by a clear gap in interoperability and a distinct lack of functional integration across these devices. As a roadmap to follow, six levels of data integration ranging from low fidelity (i.e., manually entered health data as text in a physician progress note) to high fidelity (i.e., continuous health data as discrete, structured data) were presented. An in-depth Diabetes Technology Integration Framework (Figure 8) shows the key considerations that need to be addressed from beginning (data sourcing) to middle (data exchange and storage) to end (governance). 47
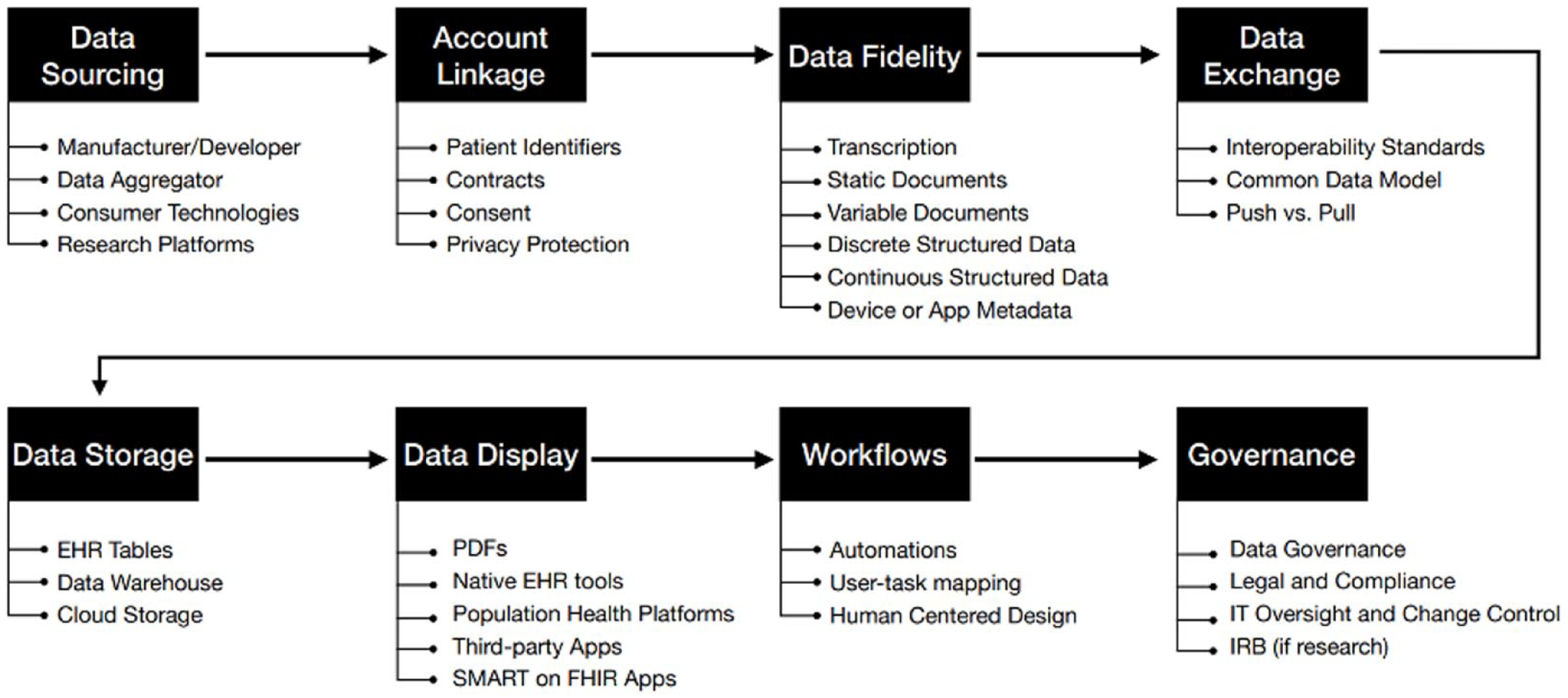
A diabetes technology integration framework consisting of eight concepts for integration of CGM data into the EHR. Abbreviations: CGM, continuous glucose monitor; EHR, electronic health record; FHIR, Fast Healthcare Interoperability Resources; IRB, Institutional Review Board; IT, information technology; SMART, Substitutable Medical Applications, Reusable Technologies.
Although the session included distinct perspectives on the topic of diabetes software and digital therapeutics, the overall message was clear. To succeed, diabetes software must provide clinical outcomes, develop a strong business case, and integrate seamlessly with EHRs.
Session 3: Regulation of Diabetes Technology
Moderators
Alexander Fleming, MD
Kinexum, Harpers Ferry, WV, USA
Alberto Gutierrez, PhD
NDA Partners, LLC, Bethesda, MD, USA
Diabetes Hardware and Software
Naomi Schwartz, MS
FDA, Silver Spring, MD, USA
An overview of the current landscape of various diabetes devices and the interoperable ecosystem including various glucose testing devices and mobile apps allowing people to track their CGM values, insulin pump types, AID systems, drug dose calculators, insulin therapy adjustment software, and smart pens and adapters.
A discussion on the FDA CDRH total product lifecycle approach and how that translates into use of RWE and real-world data (RWD).
How post-market data can give the FDA and medical device manufacturers an overarching insight into potential problems and features that can be incorporated into subsequent device versions and/or new devices.
Medical Device Cybersecurity
Kevin Fu, PhD
FDA, Silver Spring, MD, USA
Cybersecurity and medical sensor data integrity must be designed into devices and into the total device product lifecycle rather than considered after device release and implementation.
Cybersecurity is a vital component of safety, particularly when it has clinical implications.
The FDA-recognized consensus standard, AAMI TIR57, discusses categorization of various types of computer security risks in medical devices.
Privacy of Diabetes Devices
Alice Leiter, JD
EHealth Initiative, Washington, DC, USA
The proliferation of digital health tools and mobile technologies provides immense benefit to consumers and patients desiring to be more involved in their own health and healthcare; however, the advantages are accompanied with disadvantages.
The current legal and regulatory regime governing health apps lag far behind innovation and data collection and storage in digital platforms are under-protected.
The Health Insurance Portability and Accountability Act of 1996 (HIPAA) protects health data held by covered entities—HCPs and payers—but in most cases, not data held by a health app developer. As a result, in addition to posing cybersecurity risks, diabetes apps can pose significant privacy risks to users.
There are three classifications of medical devices established by the FDA, and the device class assignment is an integral component of the type of premarketing application required for FDA clearance to the market. 48 Each regulated medical device is assigned to one of three regulatory classes based on the level of control necessary to assure both device safety and effectiveness. 48 Class I includes low-risk devices such as bandages and nonelectric wheelchairs, Class II includes moderate-risk devices such as fingerstick blood glucose meters and insulin dose calculators, and Class III includes high-risk devices such as implanted cardiac pacemakers. 48
With the increased prevalence of interoperable devices, such as smart pen-cap adapters, mobile apps and CGMs, there are several devices communicating with each other. 49 This increased inter-device communication requires secure data transfer and handling. At present, the FDA considers interoperability agreements between the different medical device manufacturers in the context of each medical device’s design to evaluate the data transfer security. In the future, standardization may support a more consistent approach to secure data sharing.
There are opportunities to leverage information beyond conventional clinical trial data. 50 The traditional regulatory pathway includes design, conducting studies and analyzing results, and bringing a device to market. However, there are occasional medical device reports where the device does not function as intended for a particular patient subset. The FDA considers this valuable RWD as part of the total product life cycle approach. Additionally, RWD can be used by manufacturers to inform clinical decision making and device innovation. This process is shown in Figure 9. To emphasize the value and use of RWE and RWD, the FDA Reauthorization Act includes a new commitment to the use of RWE to support pre- and postmarket decision, and the FDA issued a guidance on use of RWE in medical device review.51,52 The current status of simulations is not adequate to replace clinical evidence. The FDA predicts that software as a medical device (SaMD) will become increasingly prevalent beyond 2021; however, it is important that data used to support the regulatory review of medical devices including SaMD be generated in a way that can be validated, whether it is data generated from robust clinical studies or from clinical studies augmented with simulation. Manufacturers may also leverage RWE from other countries or from previous iterations of the device to support FDA’s decision-making.
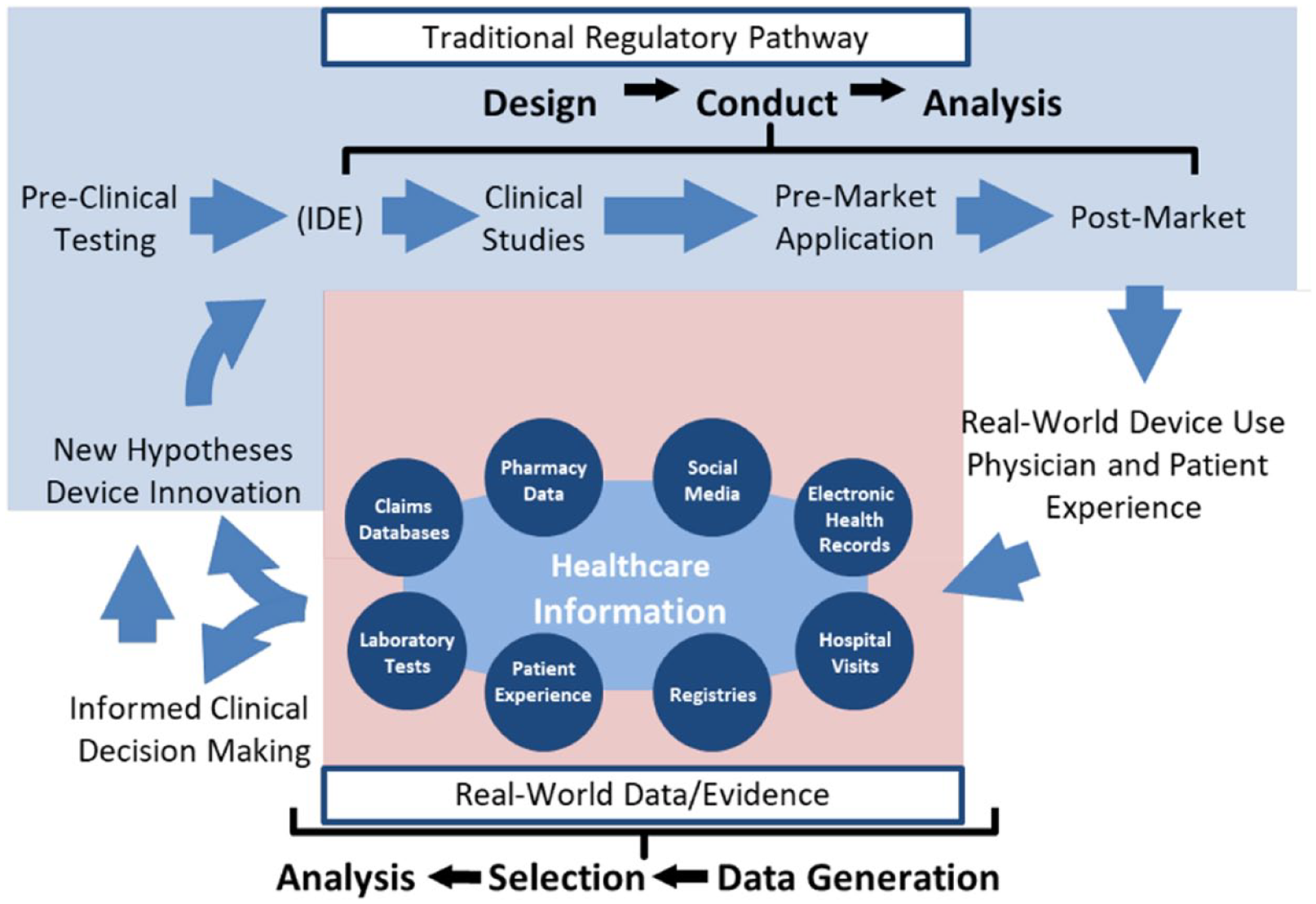
The total product lifecycle for medical devices. Abbreviation: IDE, Investigational Device Exemption.
There are two important things to focus on for medical technology: (1) wide-scale availability to deliver patient care and (2) integrity of data and data flow. Cybersecurity vulnerabilities are prevalent and affect the ability to deliver patient care, so it is important to have medical devices designed to cope with and to avoid becoming unsafe or ineffective due to cybersecurity vulnerabilities, including ransomware. Cybersecurity concerns alone can prevent a device from reaching the US market, so to ensure a medical device can reach the market, it is beneficial to have reasonable cybersecurity designs built in.
At the FDA, there are premarket clearance and approval processes. The FDA has cybersecurity guidance documents that apply to medical devices both pre- and post-market: (1) the premarket guidance provides recommendations for designing cybersecurity into the devices as a manufacturer prepares for FDA review and (2) the postmarket guidance provides recommendations for managing cybersecurity and coordinating effective communication after a device has been marketed and a cybersecurity problem arises.53,54 Both documents are publicly available and can be utilized by various stakeholder groups, including manufacturers, clinicians, and patients to emphasize the importance of cybersecurity for medical devices. Under the Healthcare and Public Health Sector Coordinating Council, the Medical Device and Health IT Joint Security Plan provides a framework for medical device manufacturers and healthcare delivery organizations to enhance product cybersecurity. In 2016, the FDA formally recognized AAMI TIR57 “Principles for medical device security—Risk management” as a cybersecurity standard for medical devices where it discusses how to categorize various types of computer security risks on medical devices. 55 The FDA is also heavily involved in the International Medical Device Regulators Forum where they help increase harmonization to regulations across various borders.
This past year, the Medical Device Innovation Consortium and FDA kicked off a large threat modeling initiative, which teaches manufacturers how to apply science to integrating cybersecurity during the design process to make their devices more secure by design. Manufacturers need to move from making claims such as “We have never been attacked” and “We use an obscure programming language” to developing threat models which represent the totality of information that affects the security of a device. A more appropriate threat model includes one that relies less on “gut judgment assessments” and takes a risk-management based approach instead.
The Health Insurance Portability and Accountability Act of 1996 (HIPAA) application to new-age digital technologies and mobile health apps is increasingly difficult to implement and enforce, especially with the push by various administrators to increase the amount of data that patients have access to in order to increase data flow from the healthcare system to patients through various applications.56,57 However, once data leaves the healthcare system or leaves the medical device itself and is transferred into the hands of the individual via their web browser, mobile, or wearable device, HIPAA no longer protects the data. 58 With increased access and interoperability, the data loses all of its HIPAA protections once it is held by an app developer to then be transferred to the individual. It must be noted that mobile health apps contain largely sensitive data that would otherwise possess robust protections if it were to reside within a traditional healthcare system, but outside of such a system possesses very minimal protection.
There are a few laws that are applicable to mobile technologies and medical devices. Section V of the Federal Trade Commission (FTC) Act of 1914 broadly prohibits deceptive or unfair acts of practice often communicated through a company’s terms of service where companies are required to protect consumer privacy and ensure that their notice of privacy practices are true, followed, and not misleading. 59 In the case of violation or data breach, the FTC can take action. Although regulation of health data does exist outside of the healthcare system, it is not as robust as it should be which results in significant privacy risks.60-64
Notices of privacy practices have been shown to be inadequate as they poorly demonstrate to users how their data will be used.65-67 For instance, terms of service tend to be densely written, lengthy, and often ignored by an individual.
HIPAA does not apply to de-identified data; however, it must be realized that if enough data is collected, digital data included, then the data can be easily identified and linked to other data elements.68,69 Congress and US Health and Human Services are aware of a need for new comprehensive data privacy legislation, although federal progress is minimal and slow. In the meantime, Europe passed and implemented the General Data Protection Regulation (GDPR) in 2018 and several states also passed legislation to protect consumer privacy including California with the California Consumer Protection Act (CCPA) and Virginia with the Virginia Consumer Protection Act.70-72 Both the GDPR and CCPA standardize data protection law across companies and give greater individual control over how personal information is used given the increasingly digital economy, and both apply far more obligations on companies with respect to how they protect individual data and how they inform individuals of how that data will be used.70-72
Session 4: Diabetes Data Science
Moderators
Guillermo Arreaza-Rubin, MD
NIDDK/NIH, Bethesda, MD, USA
Riccardo Bellazzi, PhD
University of Pavia, Pavia, Italy
Diabetes Data Science
Boris Kovatchev, PhD
University of Virginia, Charlottesville, VA, USA
Discussion of the following aspects of data science as it relates to diabetes, with most emphasis on biosensing, data farming, and analytics:
Data Acquisition and Biosensing—an Ecosystem of Diabetes Devices;
Data Engineering—Creating Interfaces and Mechanisms for the Flow and Access of Information;
Data Farming—The process of using computational experiments to “grow” data, which can then be analyzed to obtain insight into complex systems;
Data Analytics—Metrics, ML, Algorithms and AI;
Data Visualization—Making Information Available to Those Who Need It;
Data Ethics—Doing the Right Thing with Data: a framework defining the moral obligations of gathering, protecting, and using personally identifiable information and how it affects individuals.
Artificial Intelligence for Diabetes
Martina Vettoretti, PhD
University of Padova, Padova, Italy
AI techniques have the potential to advance diabetes care.
Applications of AI techniques include to enhance the management of diabetes, to predict its complications early, and to support its diagnosis and prevention.
There are still some challenges to be addressed (data availability, interpretability, generalization, integration, and safety) and future research efforts (structured data collection, AI techniques, adaptation/learning, integration to decision-making, and definition of new regulations) are needed for AI-enabled medical devices.
Machine Learning for Diabetes
Josep Vehi, PhD
University of Girona, Girona, Spain
ML has been intensively used for glucose and adverse event prediction. However, data-driven models have the same limitations as the data on which they are based.
Synthetic data can be used to overcome some of these limitations. However, current simulators based on physiological models are limited, as they do not cover the full range of human physiology and also lack information on patients’ behavior.
Because of their ability to approximate complex probability distributions and then generate new samples from the learned distribution, deep generative models (DGMs) may be a reasonable tool for synthetic data generation.
Since the 1980s, researchers have proposed a variety of approaches based on data science and AI to deal with diabetes management. However, it is only over the last decade that technology has made a decisive step to enable data science and AI to become part of deployed products and to support day-by-day patient management.
The first presentation discussed some key elements of diabetes data science. The first component is represented by data acquisition and biosensing. Nowadays, ecosystems of diabetes devices provide a large flow of information that can be conveniently exploited to support diabetes management. To do so, data engineering is needed to create interfaces and mechanisms to allow for the flow and access of information. Data engineering is currently applied to summarize and integrate information into EHRs. A complement to obtaining data from patients is represented by generating data with mathematical models, or data farming (the process of using computational experiments to grow data), which can then be analyzed to obtain insights into complex systems. For example, data farming allows for in silico trials to generate and optimize treatment options. One of the most important topics of diabetes data science is represented by data analytics, which is a wide field that includes a plethora of methods ranging from statistics to ML. One fundamental component of analytical approaches is the computation of suitable metrics to assess outcomes, like blood glucose control that can be evaluated in terms of TIR.73,74 Then, data-driven methods can be used to extract suitable and representative patterns from the data. For example, 24-hour “basic” motifs can be found in a large series of CGM data so that one can classify any other 24-hour blood glucose profile as belonging to these clusters for the purpose of data compression and encryption, pattern recognition and predictions, decision support, and closed-loop control. 75 Moreover, analytics and algorithms are now components of devices for closed-loop control available on the market, and more sophisticated analytical solutions will likely be developed. Another crucial element of diabetes data science is represented by data visualization, that is, the pictorial representation of information made available to those who need it. 73 The combination of data metrics, data analytics, and data visualization allows for the depiction of patients’ metabolic control trajectories. For example, in a diabetes optimization plane, where hypoglycemia and hyperglycemia risks are on the x- and y-axis (Figure 10), an outcome metric like TIR colors the plane area. Finally, data ethics or doing the right things with the data should be noted; this aspect deals with the moral obligations of gathering, protecting, and using personally identifiable information and how it affects individuals. 76
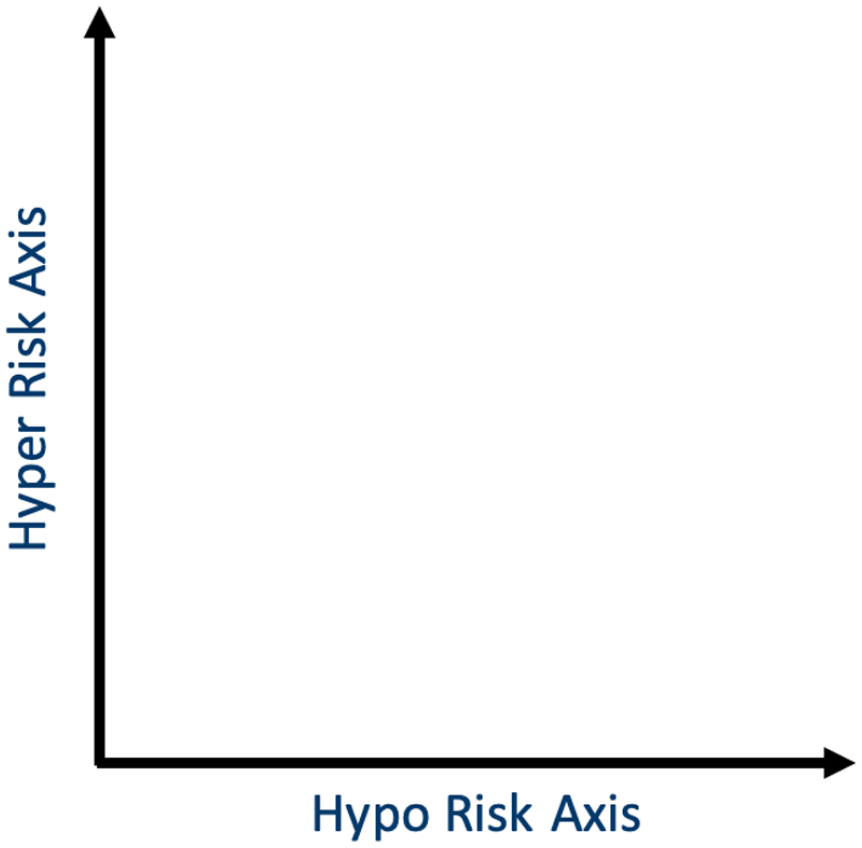
The diabetes optimization plane for continuous glucose monitoring data is defined by two principal dimensions or components, hypoglycemia and hyperglycemia risk, which explain around 90% of the variance of diabetes data.
The session then discussed some recent applications of AI in diabetes. AI is seen as the branch of computer science dealing with the simulation of intelligent behaviors in computers. It has been applied in many research projects dealing with healthcare and medicine for more than 40 years. As diabetes is a data-intensive and knowledge-intensive area, AI seems a natural technological instrument to support patient care. An important area of AI application is the enhancement of T1D management. As T1D is a complex optimization problem with many factors involved, there is an opportunity for AI to leverage patient-generated data to provide personalized decision support such as optimal insulin dosing. For example, case-based reasoning and instance-based learning have been used to adjust the carbohydrate-to-insulin ratio and correction factors based on the data collected by patients.77,78 Linear and nonlinear models based on ML methods have been applied to adapt the standard formula for bolus calculation based on several variables that can be collected in a home monitoring context, including with the use of CGMs.79,80 Furthermore, a lot of attention has been devoted to applying AI approaches for predicting future blood glucose levels or events related to hypoglycemia. This work has been boosted by the availability of time series and open-source data sets, such as one provided by the Ohio University. 81 Another area of AI approaches is represented by early prediction of diabetic micro- and macrovascular complications in people with both T1D and T2D. Nonsupervised methods have been used to identify clusters of T2D patients, with a similarity-based stratification approach to the characterization of this type of subject. 82 ML has been used to predict microvascular complications of T2D, exploiting EHR data 83 and to forecast them with administrative data only. 84 Finally, another area where AI methods can be used is to identify subjects at risk of developing diabetes and allow a timely diagnosis. ML models have been applied to distinguish between healthy subjects and those with prediabetes based on CGM-derived glycemic variability indices. 85 Dynamic probabilistic models have been also studied to describe and predict diabetes onset relying only on common risk factors, such as obesity, lifestyle, or hypertension. 86 This latter approach provides an explainable model that seems particularly interesting in a safety critical context as medicine. In summary, AI provides great opportunities for diabetes diagnosis, treatment, and prognosis, but there are open issues: (1) ML methods need large and curated datasets, which are not always available; (2) some recent AI models are difficult to interpret; (3) a data-driven model can poorly generalize when new data from different clinical contexts are collected; (4) safety must be ensured; and (5) integration with human intelligence and clinical workflow is essential. Answers to all those issues will be topics for future research, including explainable AI, data governance and stewardship approaches, and new regulatory settings.
The last presentation covered novel data generation methods. The need for powerful data generation strategies is demonstrated by the current limitations in the performance of data-driven approaches. For example, while CGM data have proven to be a great source of information to build ML models for short-term blood glucose prediction 87 and to predict short-term adverse events, more information is needed for medium to long term prediction. Meals, insulin, and physical activity seem crucial variables to be analyzed and collected, as shown in studies to forecast nocturnal hypoglycemia. 88 Moreover, in hypoglycemia prediction, the actual performance of ML tools seems adequate only for patients at high risk. In low-risk contexts, the false-positive rate is too high. 89 Finally, data-driven models have the same limitations as the data on which they are based: it is difficult to obtain quality data sets for training personalized models, many data are unbalanced in case of rare events, large data collections can be lengthy, costly, and cumbersome. For all these reasons, synthetic data can help. There are currently a lot of new approaches that allow for generation of synthetic data, including in diabetes. Conventional simulators based on physiological models lack information about some patient behaviors such as alcohol consumption, eating habits, or disturbances (such as physical activity, illness, medications, or depression). On the contrary, data-driven function approximators are a promising set of methods to learn from data complex probability distribution, resorting, for example, to DGMs. The three most popular DGMs are normalizing flows, variational autoencoders, and generative adversarial networks (GANs). 90 At the 2021 DTM, an abstract by Mujahid et al. 91 showed that a conditional GAN can be used to generate blood glucose time series with highly similar characteristics compared with the original (real) training data. When this data generation model is available, synthetic data can be used to improve accuracy of ML algorithms to predict hypoglycemia for low-risk patients and to support the achievement of several other difficult prediction goals.
Session 5: Artificial Pancreas: Scientific Advances
Moderators
Wayne Bequette, PhD
Rensselaer Polytechnic Institute, Troy, NY, USA
Peter Jacobs, PhD
Oregon Health and Science University, Portland, OR, USA
Use of an Artificial Pancreas During Stress Disturbances
Frank Doyle, PhD, CPGS
Harvard University John A. Paulson School of Engineering & Applied Sciences, Cambridge, MA, USA
Artificial pancreas systems have not been designed to contend with psychological and physiological stress.
Prior studies have noted psychological stress can cause an increase in catecholamine and cortisol levels, which can impair insulin sensitivity and cause changes in glucose variability.
An artificial pancreas platform using zone model predictive control (MPC) has been developed to run on the Harvard University interoperable Artificial Pancreas System to improve closed-loop response under stress disturbances.
Using an Online Disturbance Rejection and Anticipation System to Reduce Hyperglycemia in a Fully Closed-Loop Artificial Pancreas System
Marc Breton, PhD
University of Virginia, Charlottesville, VA, USA
Fully closed loop (FCL) is achievable for T1D.
Anticipation of regular behaviors seems to improve glycemic control.
Early supervised studies must be confirmed in a less controlled environment.
Incorporating Prior Information in Adaptive Model Predictive Control for Multivariable Artificial Pancreas Systems
Ali Cinar, PhD
Illinois Institute of Technology, Chicago, IL, USA
The impact of glycemic disturbances such as meals, physical activity, psychologic stress, and sleep characteristics cannot be mitigated by fully-automated AID systems by relying exclusively on continuous glucose monitoring and insulin dosing information.
Wearable devices provide valuable data (accelerometer, blood volume pulse/heart rate, galvanic skin response, skin temperature) that can indicate the characteristics of physical activity, stress, and sleep states in real time.
Signal processing and ML techniques can extract valuable personalized patterns of behavior and real-time capture of the user’s state from wearable device data to provide proactive (feedforward) control to complement feedback control relying on CGM and insulin information.
There were several common themes on the current state of the art in AID, including MPC, disturbance (stress, exercise, eating) rejection, and the use of prior and/or feedforward information. It should be noted that there were often debates on the relative merits of MPC, proportional-integral-derivative, and other control strategies at prior DTMs,92,93 but the three presentations in this session solidly supported the use of MPC.
It was noted that meals and exercise disturbances have received the most attention, without much emphasis on psychological or pharmacological stress disturbances. Bevier et al. 94 conducted a study in which they administered prednisone to ten subjects and found that average glucose increased from 110 to 149.2 mg/dL, with significantly more time in hyperglycemia and an average increase in total daily insulin dose of 69%. Thus, there was a significant reduction in insulin sensitivity.
The next study discussed correlated psychological stress with glucose variability, based on open-loop results presented by Gonder-Frederick et al., 95 with a discussion of future closed-loop applications. The study found that naturally occurring daily stressors can be associated with increased glucose instability and hypoglycemia, as well as decreased carbohydrate consumption.
The next studies involved closed-loop system using an AID system based on zone MPC with insulin-on-board constraints. Just before the COVID-19 pandemic, there were studies by Pinsker et al. 96 of two subjects with the background stress of COVID-19. The AID system improved TIR for both subjects, while reducing the time in hypoglycemia.
Preliminary closed-loop results were presented by Kaur et al., 97 based on a randomized crossover trial of AID versus sensor-augmented pump, involving 14 adults with T1D under both psychological and pharmacological stress. The AID system resulted in increased TIR and decreased hypoglycemia and hyperglycemia. It was also clear that the AID system shifted the burden in making micro-adjustments from the “human-in-the-loop” to the closed-loop algorithm.
The concept of FCL versus hybrid closed loop (HCL) was clarified, noting that the FCL system has no meal announcement, carbohydrate amounts, or exercise mode. That is, there should be no interaction between the patient and the automated system. Because of current limitations to insulin analogues and pharmacodynamics, FCL will result in large postprandial excursions. A “bolus priming” module can be added to MPC to react to glycemic disturbances that look like meals more efficiently. It is a simple system that looks at about 30 minutes of CGM data and uses a logistic regression classifier based on ML. The result is a probability that an event that looks like a meal has occurred. The higher probability that a meal has occurred allows a larger magnitude of insulin to be given. The results of a feasibility study involved a “mini-camp” with a strict COVID-19 testing protocol, in a hotel admission with 18 adolescents with T1D. Four days were repeated for each individual, with an unannounced meal on the second day. Garcia-Tirado et al. 98 present results comparing the new controller (“RocketAP”) with the previous controller (“USS-Virginia”). Particularly during the six hours following the unannounced meal, the RocketAP outperformed the USS-Virginia, with a reduced time-above-range.
Another clinical study focused on the effect of physical activity by enrolling 15 adults and comparing two HCL controllers, a MPC and a modified MPC called APEX to anticipate and detect unannounced exercise. The APEX system reduced hypoglycemic episodes overall, during exercise, and in the four hours following exercise. An ongoing study involves adults and compares three control strategies: HCL, FCL, and anticipatory FCL. Preliminary results for the first 18 subjects indicate that HCL is best, followed by anticipatory FCL, then FCL. Similar multiple model approaches to handle uncertainty and anticipation were presented in simulation studies by Cameron et al., 99 in-patient studies by Cameron et al., 100 and in closely supervised outpatient (hotel) studies by Cameron et al. 101 and Forlenza et al. 102 using a multiple model probabilistic predictive control strategy.
The next presentation was based on a series of papers by Sevil et al.103-105 and Askari et al. 106 and was focused on incorporating prior information to improve the prediction of future blood glucose concentrations. Meals, exercise, sleep, and stress become more challenging to account for when some of these events occur simultaneously. A plot of an individual from the Tidepool data set showed that there is substantial variability in the probability of an event occurring throughout the day. The approach makes use of historical data to detect trends in behavior and uses this information to predict expected disturbances (Figure 11). This method requires personalized models and the need to understand the various changes in patterns over many periods of time: day, week, month, year. Based on a simulation over 21 days, there was significant improvement as the system learned the behavior. There is a limitation to using only CGM information for predictions, so the use of additional sensor information is important. Historical data should be complemented with current information. The Empatica E4 wristband is used to classify and estimate energy expenditure. Furthermore, psychological stress can be detected with and without concurrent physical activity. Understanding that the current detected stress will affect future glucose concentrations enables the MPC law to make better insulin infusion decisions.
The current trend of data (current day’s evolving pattern) can be compared with historical data to find the most similar scenarios for the realization of unknown and unmodeled disturbances.
In a multivariable artificial pancreas, personalized adaptive multivariable models with recursive updating are used to improve the accuracy of glucose predictions. The adaptive MPC with regularized partial least squared (rPLS) is robust to disturbances caused by unannounced meals and physical activities even with missing glucose data. In a simulation-based study of 20 virtual subjects, the proposed MPC with rPLS had improved TIR and reduced hypoglycemia compared with MPC based on autoregressive moving average.
Session 6A: Patient Panel
Moderators
Bruce Buckingham, MD
Stanford University, Palo Alto, CA, USA
Laura Gandrud Pickett, MD
International Diabetes Center, Minneapolis, MN, USA
Patient Panel (Insulet, Diabeloop, Tandem, Medtronic)
Four panelists shared their experience using different AID systems: Omnipod 5 (Insulet Corporation, Acton, Massachusetts, USA), Diabeloop (Roche pump/Dexcom CGM/Diabeloop DBLG1 algorithm; Diabeloop, Grenoble, France), Control-IQ (Tandem Diabetes Care, San Diego, California, USA), and Medtronic MiniMed 780G. All experiences were universally positive. The Omnipod 5 user most appreciated reduction of overnight lows, which was noted as “life changing” since the user lives alone, and rated the system at 8.5 out of 10. The Diabeloop user noted that the system was less work than prior systems used, provided more “peace of mind,” and allowed the user to sleep better. The Diabeloop system was rated at 9 out of 10. The Control-IQ user was happy with how the system had simplified life and rated the system at 11 out of 10. The MiniMed 780G user enjoyed the accuracy of the Guardian 4 sensor and significant reduction in blood glucose calibrations. They called the system a “game changer” and rated it at 9 out of 10.
In terms of future improvements, most panelists look forward to smart phone integration for system use. The Omnipod 5 user would like to be able to wear the same pump for longer, have a smart phone replace the dedicated device called the personal diabetes manager, and for the algorithm to correct hyperglycemia more aggressively. In the future, they do not want to be constrained to wearing the Omnipod 5 and CGM on the same plane of their body for optimal function. The Diabeloop user would like smart phone integration and smart watch integration for exercise. They hope that in the future, the systems can handle strenuous exercise more easily and would be willing to use a fitness tracker to aid in this. The Control-IQ user would like to have pump control from their phone. The MiniMed 780G user would like an instant start for the transmitter and coupling a disposable transmitter with a sensor for 15 days of wear.
Everyone was excited about FCL systems in the future, even if they would be more expensive (recognizing the need for more affordable insulin for all). The Diabeloop user would not sacrifice TIR below 80% or more than 1% hypoglycemia to use an FCL system. They also would not be interested in a dual hormone pump if it required two infusion sets. However, they would be interested in co-infusion of insulin and amylin. All panelists would be willing to use other devices such as fitness trackers, with shared data, if needed for optimal AID function.
Session 6B: Artificial Pancreas: Comparison of Products
Moderators
Ananda Basu, MBBS, MD, FRCP (UK)
University of Virginia, Charlottesville, VA, USA
Jeffrey Joseph, DO
Thomas Jefferson University, Philadelphia, PA, USA
Features of Insulet Omnipod 5
Sue Brown, MD
University of Virginia Health, Charlottesville, VA, USA
Wearable tubeless insulin-filled Pod communicates directly with CGM.
MPC algorithm is built into the Pod—AID can continue without the handheld controller nearby.
Customizable glucose targets from 110 to 150 mg/dL, in 10 mg/dL increments, adjustable by time of day is the main method to modify an AID system.
Features of Medtronic FLAIR
Elvira Isganaitis, MD, MPH
Joslin Diabetes Center Harvard University, Boston, MA, USA
The Medtronic Advanced Hybrid Closed-Loop remains an investigational device in North America.
Based on published data, the system holds promise for improving:
HbA1c
TIR
Usability—higher likelihood of staying in closed loop than with MiniMed 670G
Features of Tandem Control-IQ
Paul Wadwa, MD
University of Colorado Anschutz Medical Campus, Aurora, CO, USA
Control-IQ is commercially available for use in PWD age six years and older requiring insulin.
Control-IQ is approved by the FDA and Health Canada and is Conformité Européenne (CE) Marked.
The system requires the user to pre-bolus for meals and use carbohydrate counting.
Studies and RWE show high rates of staying in closed loop.
Features of Beta Bionics iLet Bionic Pancreas
Steven Russell, MD, PhD
Massachusetts General Hospital Harvard University, Boston, MA, USA
The iLet bionic pancreas is initialized using only the user’s body weight. No information about the previous insulin regimen is used. The system is designed to automatically and continuously adapt insulin dosing.
Meal announcements to the iLet bionic pancreas do not involve carbohydrate counting. Meals are announced by type (breakfast, lunch, or dinner) and amount of carbohydrates compared with other meals of that type (less, usual, or more). The system is designed to give 75% of the predicted meal insulin requirement as a “meal priming dose” and to automatically and continuously adapt this amount.
All correction doses are automated. The only adjustment that can be made is the glucose target, which can be set to lower, usual, or higher. A schedule of two different targets may be set.
Features of CamAPS FX
Roman Hovorka, PhD, FMedSci
University of Cambridge, Cambridge, UK
CamAPS FX is an interoperable hybrid closed loop app CE-marked for ages one year and up including pregnancy (not FDA approved).
CamAPS FX is uniquely adaptive, independent of pump settings, to accommodate ever-present changes in insulin needs.
Key personalization features include time-segmented personal glucose target (80-200 mg/dL), “Ease-off,” and “Boost” modes.
An AID system, in the past referred to as artificial pancreas, is now a reality with more systems becoming commercially available. The latest available models of AID systems are shown in Table 2. CGMs and insulin pumps are communicating via an algorithm for optimized automatic modulation of insulin delivery. These HCL systems share many features, although each of them has individual proprietary algorithms and hardware features that have nuances which are unique and should be taken into consideration when PWD are choosing a system. It is important to note that currently, the FDA-approved systems are not FCL and require the user to self-initiate food boluses.
Latest Available Model of Every AID System.
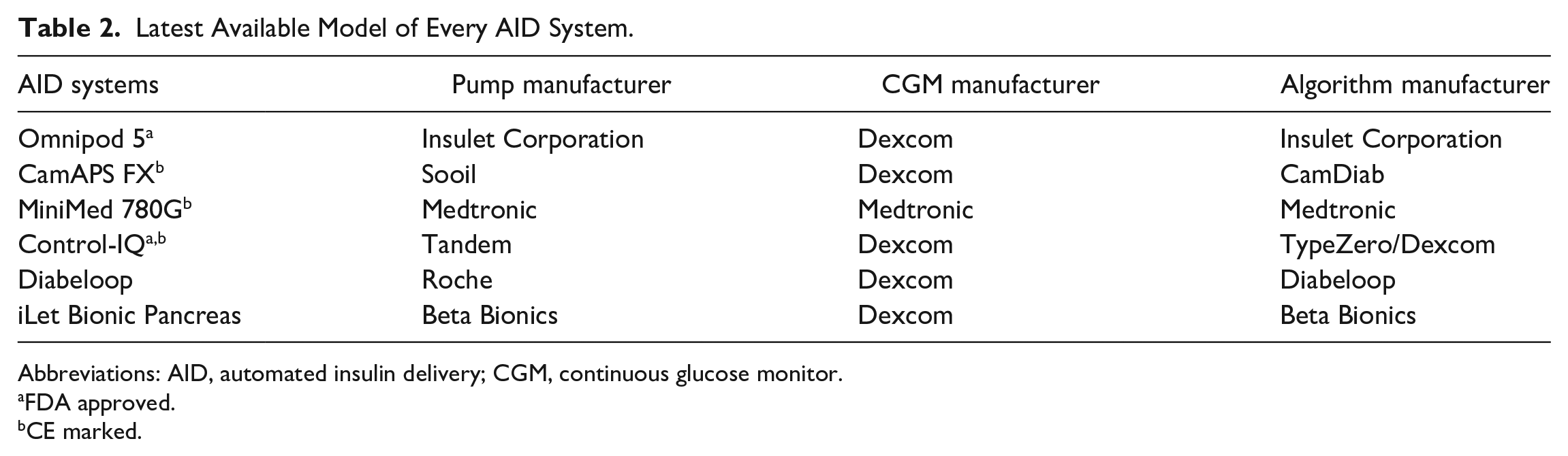
Abbreviations: AID, automated insulin delivery; CGM, continuous glucose monitor.
FDA approved.
CE marked.
The other similarities that many of the interoperable HCL systems (either approved or under review by the FDA) share are that they utilize the Dexcom G6 CGM to gather glucose data. They infuse insulin via micro-boluses every five minutes, have an auto and manual mode, and offer a mobile app that assists with data management and sharing. Currently, the ability to bolus insulin via a mobile phone is not available apart from the CamAPS FX app.
The unique algorithms, while mostly using an MPC foundation, offer several different features. The degree of “tight control” is largely influenced by what the glucose target can be set at and the baseline glucose control. The options include different settings, that is, sleep or exercise and have customizable targets that can be programmed to different amounts up to eight times a day.
The pumps, other than the iLet Bionic Pancreas, require preset basal rates, insulin sensitivity factor, carbohydrate ratios, and insulin duration times, but depending on the system, the settings may not affect the learned algorithm and insulin delivery. The importance of pump setting is when the connection between the CGM and insulin pump is lost, and therefore must return to manual mode to continue insulin delivery. The iLet Bionic Pancreas is the only one of the five systems presented that does not return to preset delivery settings because there are no presets. If the connection is lost, then it will continue with the last “learned” settings.
For any HCL system to be successful, training and troubleshooting strategies need to be learned and utilized by the user. In current times, new offerings of training are now virtual as well as in person. Much like with self-driving cars, the user needs to retain the basic safety skills of CSII in times when they need to revert to manual mode.
Session 7: Novel Insulins
Moderators
Gerold Grodsky, PhD
University of California, San Francisco, San Francisco, CA, USA
Elias K. Spanakis, MD
University of Maryland, Baltimore, MD, USA
Gold Student Research Award Winner: Predicting Postprandial Glucose Excursions With Nutrient Content Using an Interpretable Random Forest Augmented by a Digital Twin ODE Model
Rahul Narayan, MS
Oregon Health & Science University, Portland, OR, USA
The research explored how knowledge of macronutrient intake (carbohydrates, fats, proteins) as well as fiber, water, and caffeine may improve the prediction accuracy of the postprandial glucose response in people with T1D using a random forest augmented by an ordinary differential equation (ODE) physical model of metabolism (digital twin).
Data used for model development was from 25 adults with T1D monitored for 28 days using CGMs, insulin pumps/pens, and exercise fitness trackers. A custom smartphone app was used to collect food intake data and nutrient content was estimated by nutritionists from photos of food consumed in four days per week (977 meals). A random forest model augmented by an ODE digital twin was trained to predict incremental area under the glucose curve (iAUC) three hours following meals; 89% of meals were used for training and 11% were used for validation. The effects of macronutrient content and other predictor variables were assessed using Shapley coefficients.
Shapley analysis revealed that carbohydrates, caffeine, and alcohol all tended to increase post-prandial iAUC, whereas larger amounts of fats, proteins, and fiber decreased iAUC; the impact was dependent on meal size. Root-mean-squared-error decreased by 1.4% when including protein and fat as predictors. The ODE model alone explained 6% of iAUC variance while random forest alone explained 25% of the variance and the combined ODE and random forest explained 30% of the variance.
Including macronutrient inputs to postprandial glucose prediction algorithms improves prediction accuracy. A combination of data-driven machine learning and ODE-based physical models offers promise for improving prediction accuracy.
New Insulins
Tim Heise, MD
Profil, Neuss, Germany
Two once-weekly insulins, insulin icodec (Novo Nordisk, Bagsværd, Denmark) and basal insulin Fc (BIF; Eli Lilly, Indianapolis, Indiana, USA), are currently in clinical development and show pharmacokinetic and pharmacodynamics properties suitable for once-weekly dosing.
Clinical studies show good tolerability of both once-weekly insulins. There was a trend toward improved glycemic control (HbA1c) with icodec and significantly lower rates of documented hypoglycemia with BIF.
New rapid-acting insulins in development include AT247 (Arecor, Saffron Walden, United Kingdom), which showed a faster onset and short duration of action than Fiasp (Novo Nordisk, Bagsværd, Denmark) in a glucose clamp trial, and ultrafast-absorbing insulin lispro (UFAL) (Surf Bio, Palo Alto, California, USA), a monomeric insulin stabilized through polymer excipients, that showed faster onset and shorter duration of exposure in mini-pigs.
Smart Glucose-Responsive Insulins
Michael Weiss, MD, PhD, MBA
Indiana University School of Medicine, Indianapolis, IN, USA
Structural mechanisms informing how insulin binds to and triggers its receptor define a structural switch.
This switch may be regulated by an exogenous ligand such as a monosaccharide, provided that appropriate sensors are linked to insulin.
A prototype of glucose-responsive insulin has been synthesized and validated in vitro and in rat clamp studies.
Update on New Glucagon Products
Anne Peters, MD
Keck School of Medicine of USC, Los Angeles, CA, USA
Glycemic variability can be worsened by using only one hormone (insulin).
Newer formulations of glucagon are much easier to administer than previous versions.
Novel uses for glucagon may improve outcomes for patients.
Heat Stability of Various Insulin Types
Leonardo Scapozza, PhD
University of Geneva, Geneva, Switzerland
Insulin is stable at oscillating temperature during the four weeks of use.
Many challenges still confront HCPs and patients trying to manage diabetes in low-resource settings.
The ability to use insulin without cold chain during the period of use in hot climates removes at least one significant barrier.
A hundred years after the discovery of the first insulin, a life-saving medication for patients with diabetes, novel insulins are under development. Two of them are once-weekly formulations, including insulin icodec and BIF. Initiation of icodec insulin can be done by administering a double dose at the start of therapy, which is able to achieve steady state almost immediately. 107 Titration can be achieved by adjusting insulin dosing by 28 units per week. 108 Insulin icodec has been tested in two different phase 2 studies (Table 3) and showed a trend toward reduction of HbA1c.107,109 In one of these studies, a higher incidence of level 1 (and not level 2 or 3) hypoglycemia was found, 109 a finding that was not confirmed in other studies. Duration of hypoglycemic events was not increased with icodec insulin. 108 Another basal insulin is BIF. In an early phase 2 study, BIF subjects had a lesser reduction of HbA1c and lower rates of hypoglycemia compared with subjects receiving degludec, which might partially be due to different titration targets. UFAL 110 and AT247 111 are promising prandial insulins, which have faster and shorter durations of action compared with available prandial insulins.
Table provided by Tim Heise, MD, Profil, Neuss, Germany.
indicates a non-significant difference of p>0.05 for insulin icodec and vs insulin glargine, ** indicates a significant difference of p ≤ 0.05 for insulin icodec vs insulin glargine.
Abbreviations: ETD, estimated treatment difference; HbA1c, hemoglobin A1c; T2D, type 2 diabetes; TIR, time in range.
Novel insulins and especially “smart” glucose-responsive insulins represent an exciting field for patients with diabetes. Unimolecular glucose-responsive insulins represent one of the different classes of glucose-responsive insulins. 112 The concept of the glucose-responsive insulin design is that glucose will unlock and activate insulin while insulin will remain inactive in the absence of glucose. 113 Structural mechanisms informing how insulin binds to and triggers its receptor define a structural switch, 114 a switch that can be regulated by a monosaccharide. In vitro studies with HepG2 cells examining glucose or fructose as the activating monosaccharide showed a progressive autophosphorylation of the insulin receptor with increasing doses of glucose or fructose and an enhanced effect of glucose compared with fructose. In vivo insulin clamp studies, which were performed in normal rats, showed a glucose-dependent marked enhancement of insulin action in hyperglycemic conditions.
Using insulin alone can lead to hypoglycemia and worsening glucose variability, an increasingly recognized marker of dysglycemia. 115 Glucagon can not only treat but can also prevent hypoglycemia, thus reducing glucose variability. Compared with the first generation of glucagon emergency kits that required reconstitution, newer second-generation formulations such as nasal glucagon, premixed/prefilled glucagon autoinjector, 116 and the first glucagon analogue, dasiglucagon (Zealand Pharma, Søborg, Denmark) 117 autoinjector are available. Compared with users of glucagon emergency kits, users of glucagon autoinjectors were more likely to be successful at administrating glucagon. 116 In addition to treating hypoglycemia, mini-dose glucagon has been found to be more effective than insulin reduction for preventing exercise-induced hypoglycemia in patients with T1D. 118 Future directions can be glucagon solutions that will be utilized with insulin in bi-hormonal pumps or “glucose sensing glucagon” formulations which will be released in response to low glucose values in a similar concept as the glucose-responsive insulins.
Developing new insulins represents a significant topic; however, ensuring that available insulin types are stable during heat or high-tropical temperatures is also important. A study was performed which evaluated seven commercially available insulins in different storage conditions. 119 Insulin was analyzed by multiple methods: (1) high-performance liquid chromatography (HPLC), which is used for insulin quantification and potency determination; (2) circular dichroism, which is used to detect changes in 3D structures; (3) readout insulin receptors and Akt phosphorylation, which is used for bioactivity testing; and (4) visual inspection, which represents a simple and easily performed method. Using HPLC for insulin stored under oscillating conditions at 25°C to 37°C for four weeks showed stability and conformity to pharmacopeia guidelines. The 3D structure of the tested insulin confirmed that insulin monomer conformation did not undergo significant modifications. Similarly, insulin bioactivity was not significantly altered. Insulin formulation under other oscillating temperature settings of 17°C to 45°C also retained its pharmacopeia properties during four weeks of testing.
Session 8: Insulin Delivery
Moderators
Umesh Masharani, MD
University of California, San Francisco, San Francisco, CA, USA
Anant Nigam, MD, FRCP
Nigam Diabetes Centre, Jaipur, India
Novel Needles
Teresa Oliveria, PharmD
BD, Franklin Lakes, NJ, USA
The primary focus of needle innovation is to ensure reliable delivery of insulin to the subcutaneous tissue for predictable insulin absorption.
Innovations in cannula design focus on needle length and gauge, needle lumen, and needle hub design.
Along with advances in technology, proper insulin injection technique and the prevention of lipohypertrophy are essential to obtain the full benefits of insulin therapy, whether injecting with a pen, syringe, or CSII.
Novel Insulin Delivery Systems
Jeffrey Joseph, DO
Thomas Jefferson University, Philadelphia, PA, USA
Insertion of an insulin infusion set needle/cannula into the subcutaneous tissue damages cells and connective tissue and initiates an acute inflammatory/immune/foreign body response. The inflammatory process may be enhanced by the infusion of rapid acting insulin, its excipients, and fibrils/precipitates.
The inflammatory tissue adjacent to the cannula may become dense, thick, and continuous over wear-time, forming a mechanical barrier that inhibits the diffusion of insulin into healthy capillary and lymph vessels. The delivery of insulin into the subcutaneous tissue via a commercial pump may be variable because of the variable compliance of the adjacent layer of inflammatory tissue.
Insulin molecules may be denatured/degraded within the inflamed tissue and lymph nodes by protease enzymes, free radicals, and the acidic environment.
Novel Infusion Sets
Robert Vigersky, MD
Medtronic Diabetes, Northridge, CA, USA
Research into the mechanism by which traditional infusion sets fail after two to three days has led to the development by Medtronic of an extended wear infusion set capable of lasting up to seven days in most PWD using insulin pump therapy.
Longer-wear infusion sets successfully address a previously unmet need for pump users, reduce the burden of diabetes management, and reduce insulin and plastic waste.
Medtronic’s extended infusion set can be successfully used with rapid-acting insulins (insulin lispro and insulin aspart) as well as ultra-rapid-acting insulin (faster-acting insulin aspart or Fiasp).
Forget Diabetes: A Novel Automated Insulin Delivery System
Andreas Pfützner, MD, PhD
Pfützner Science and Health Institute, Mainz, Germany
The Forget Diabetes European Union (EU) Grant project aims to develop a unique intraperitoneal artificial pancreas device.
The pump will be supplied with insulin via orally administered containers and by an innovative micromechanical filling mechanism.
An osmotic pressure–based intraperitoneal sensor will feed the dosing algorithm with the necessary glucose information.
Smartwatch- and Smartphone-Mediated Cell-Based Diabetes Control
Maysam Mansouri, PhD
ETH Zürich, D-BSSE, Basel, Switzerland
GlowControl is a mammalian engineered cell which can sense green light emitted from a smartwatch and produce glucagon-like polypeptide-1 (GLP1) in response. This system was used for treatment of T2D upon green light illumination.
iβ-cell is a light-inducible designer cell with fast kinetics that releases insulin in response to light irradiated from a smartphone flashlight.
Optogenetically engineered cells can be the foundation of next-generation precision medicine tools.
The next generation of insulin pen needles will be 34-gauge (as thin as a human hair) and 4 mm long. They appear to perform as well as 8 mm long pen needles in terms of insulin leakage. There are technical challenges in making the needles so thin. First, the needles have to be engineered to resist breakage. Second, to make sure that the injection force remains acceptable, the needle wall must be made thinner, allowing for expansion of the lumen diameter.120,121 Figure 12 shows the different types of needle wall thickness. The hub of the needle is also contoured so that the insertion force is dispersed over a wider surface area of the skin when compared with the standard cylindrical hub. 122
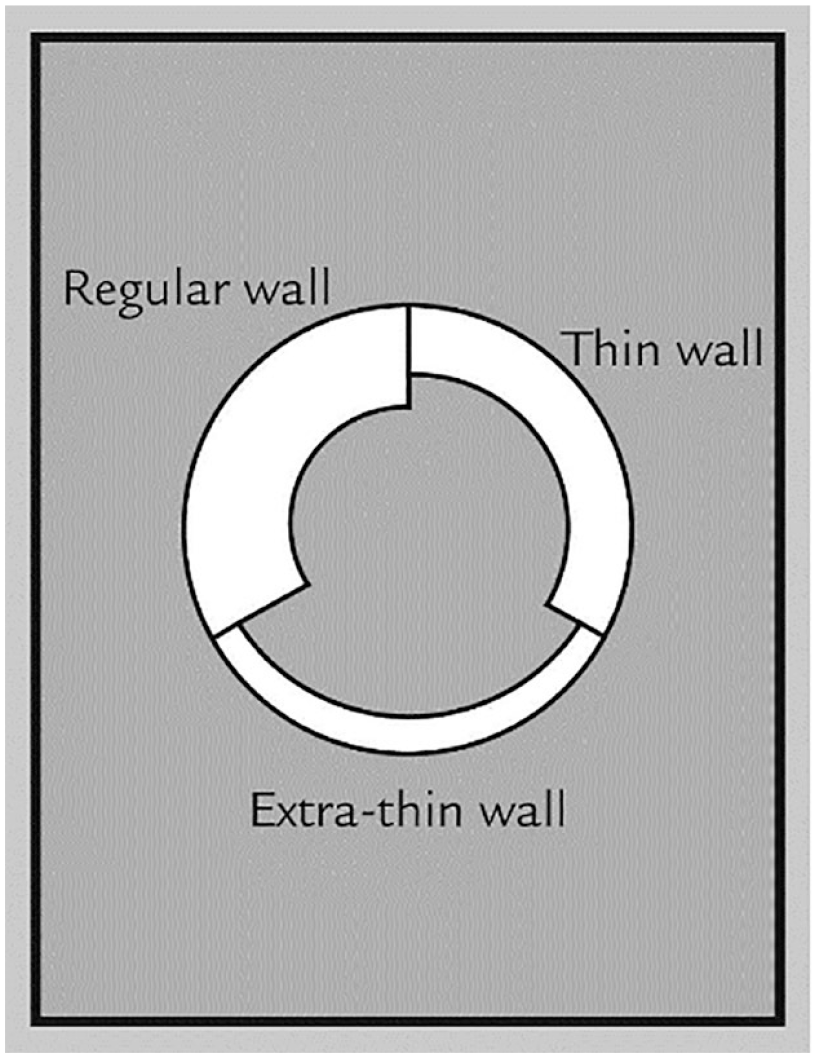
Comparison of different insulin pen needle wall thicknesses.
Neutrophils and macrophages migrate to the site of the insulin infusion cannula because of tissue damage. In porcine models of human skin, inflammatory tissue at the cannula lumen interferes with insulin delivery within two days of insertion. Micro-computed tomographic (CT) images can demonstrate what percentage of the insulin volume is delivered and how this percentage decreases over time. Insulin infusion sets that cause less tissue damage and less inflammation are in development. In one of these infusion sets, the cannula is made of a soft flexible polymer that is less likely to cause injury. It also has multiple orifices (distal and proximal) so that insulin delivery is not impaired if the most distal orifice gets occluded. These types of novel infusion sets can be worn for up to five to seven days.
The Medtronic extended infusion set has been designed for extended wear up to seven days. Innovations with this system include a new connector cap that improves insulin stability, new tubing that improves preservative retention, and new adhesives to keep the infusion set in place for a prolonged period. 123 One additional advantage of having prolonged wear is reduced insulin wastage and plastic wastage compared with more frequent infusion set changes with standard infusion sets. Importantly, these modifications fulfill an unmet need and thereby reduce patient burden.
An EU-funded research program has been established to develop a fully automatic intra-abdominal pump that will deliver insulin and glucagon into the peritoneum. 124 Features of the pump include reservoirs for insulin and glucagon, inductive wireless charging, an integrated nonenzymatic glucose sensor, and the capability to refill insulin and glucagon reservoirs from capsules of hormones swallowed by the patient. A number of the technologies for this project are already in development.
The nonenzymatic osmotic pressure glucose sensor uses the competitive and reversible binding of glucose versus dextran to concanavalin A. The system measures the osmotic pressure difference arising between a reagent chamber (containing concanavalin A and dextran) and a diffusion chamber in direct contact with interstitial fluid. Changes in tissue glucose will proportionally alter the osmotic pressure within the reagent chamber, and the information is then translated into an accurate glucose reading.
To refill the insulin and glucagon reservoirs in the pump, the patient will swallow magnetic capsules containing insulin or glucagon. The capsules will travel down the small bowel and position themselves adjacent to the pump reservoirs. Fine needles will puncture the bowel wall and transfer the insulin or glucagon cargo to the appropriate reservoirs.
Mammalian cells can be engineered so that they can produce and release hormones on demand. The concept is to engineer cells with a green light–inducible gene switch linked to a specific gene such as insulin or GLP1. 125 The engineered cells can then be implanted subcutaneously and insulin or GLP1 production and release can be controlled by the green LED of a smart watch.
Session 9: Skin Trauma Due to Diabetes Devices
Moderators
Halis K. Akturk, MD
University of Colorado Anschutz Medical Campus, Barbara Davis Center for Diabetes, Aurora, CO, USA
Mohammed Al-Sofiani, MBBS, MSc
King Saud University, Riyadh, Saudi Arabia
Bruising
Lutz Heinemann, PhD
Science Consulting in Diabetes GmbH, Kaarst, Germany
Bruising has been an underreported problem with injections in diabetes therapy.
Bruising may have a relevant impact on the performance of technologies used for diabetes therapy.
While visible bruising is a cosmetic issue (which is disturbing for patients), it might be the bruising in the subcutaneous tissue that is of clinical relevance with respect to glucose control.
Allergic Reactions to Adhesives
Jannet Svensson, MD, PhD
Department of Pediatrics and Adolescent Medicine, Herlev and Gentofte Hospital, Herlev, Denmark
Acrylates are the most common allergens in diabetes devices and adhesives used for these devices.
Many patients are thought to have nonallergic eczema probably because not all chemicals released from infusions sets and sensors are known.
Nonspecific eczemas seem to not be resolving over time in most affected patients.
Preserving Skin Integrity With Wearable Diabetes Devices
Cari Berget, RN, MPH, CDCES
University of Colorado Anschutz Medical Campus, Barbara Davis Center for Diabetes, Aurora, CO, USA
The most common skin problem for PWD wearing diabetes devices is contact irritation of the skin from adhesives used in CGMs.
To help preserve skin integrity and prevent skin irritation, place devices in areas with enough fat to pinch up and rotate insertion sites, clean the insertion site with soap and water and dry thoroughly, use a barrier technique to reduce exposure of skin to the device, and remove tapes slowly, with a low angle to reduce skin injury.
Clean the skin to remove adhesive residues after removal and apply lotions and anti-itch creams to intact skin and antimicrobial creams to wounds to promote skin healing.
Advances in Adhesives
Naunihal Virdi, MD
Abbott Diabetes Care, Alameda, CA, USA
The goals of adhesion include robust adhesion, breathability, removal without skin trauma, minimization of adverse events during wear, and no interference with medical device functionality.
Numerous factors can lead to adverse events because of the device adhesive.
Device improvements and proper education can be used to address poor adhesion and adverse events patients may encounter.
Bruising due to diabetes device placements and injections frequently occurs in PWD. The number of blood vessels at the application or injection site, the state of vasoactivity, the size of blood vessels ruptured by the penetration of injectables, the amount of blood extravasated, the depth of bleeding, the age and weight of the patient, and the amount of blood clotting factors are some of the main determinants of bruising due to diabetes devices and injections. A recent study of 790 PWD using insulin injections showed that bruising occurs most commonly in females, older adults, and those with a higher body mass index. 126 Whether subcutaneous bleeding may affect the accuracy of CGM systems remains largely unknown. However, the higher MARD level of CGMs during the first 24 hours may be related to subcutaneous bleeding. Further research should study whether bruising has any correlation with lipohypertrophy and any effect on insulin absorption.
Skin irritation, contact dermatitis, and eczema are common dermatologic problems encountered by PWD with the use of diabetes devices. Itchiness and redness are the most frequent complaints. In a recent study, nonspecific eczema was the most reported dermatologic complication in children and adults using diabetes devices.127,128 Follow-up studies with the same population showed that majority of the patients had still had eczema five months later that significantly decreased their quality of life.129,130 Isobornyl acrylate, abitol, and colophonium were the most common allergens.131,132 A patch test, similar to the one shown in Figure 13, may be necessary to identify the irritant. Complete labeling of ingredients may help to prevent recurrent eczema. Thin hydrocolloid or silicone dressing may help to ease the irritation.
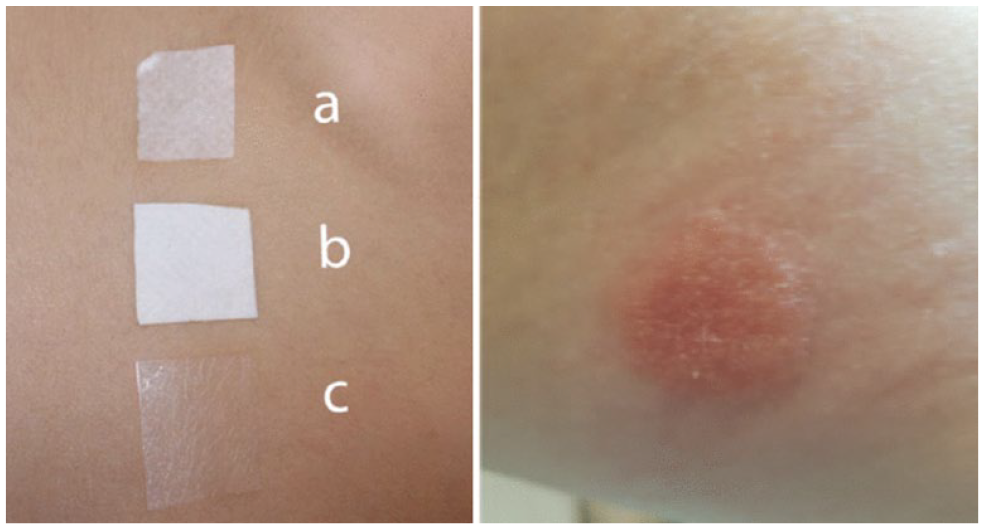
Left: a patch test with (a) continuous glucose monitor adhesive that sticks to the skin, (b) double layer of adhesive, and (c) thin-layer adhesive that binds continuous glucose monitor to the adhesive. Right: a positive skin reaction from the patch test.
Preserving skin integrity is fundamental to continue diabetes technology use. Proper device placement is the first step to prevent dermatologic problems. 134 Placing devices in healthy skin with enough subcutaneous tissue, avoiding areas with bending, and rotating sites with each insertion is recommended. 134 Cleansing the skin with alcohol-free wipes and using liquid or physical barriers may help prevent skin irritation. 134 Removing the tape slowly and at a low angle, using topicals to unstick the adhesive, and using lotions to promote skin healing will help the removal process and prevent further skin damage. 134
Besides dermatitis and irritation, adhesives can also cause skin stripping, tearing, and tension injuries. In addition, the prolonged accumulation of moisture under the adhesives lead to increased permeability and infection susceptibility. Adhesive strength, pliability, breathability, hydrophobicity, and duration of wear should be considered in selecting adhesives to reduce skin trauma. 135 Using a hydrophilic layer to allow moisture to evaporate, holes within the adhesive layer to allow moisture egress, and silicone-based taping can facilitate water vapor transmission.
Session 10: Metabesity: Preventing Metabolic Deterioration and Chronic Disease
Moderators
Brian Frier, BSc (Hons), MD, FRCP (Edin), FRCP (Glas)
University of Edinburgh, Edinburgh, Scotland
John S. Melish, MD
University of Hawaii, Honolulu, HI, USA
Overview of Metabesity
Alexander Fleming, MD
Kinexum Services LLC, Harpers Ferry, WV, USA
Geroscience (the study of aging biology) has demonstrated a promise of various interventions for slowing the aging process and the onset of multiple chronic diseases, including diabetes. The major geroscience objective is to increase healthspan—the period of lifespan free of chronic diseases and disabilities.
Metabesity is the constellation of chronic diseases (diabetes, nonalcoholic steatohepatitis, cardiovascular diseases, and neurodegenerative diseases), cancer, and the aging process itself, all which share metabolic roots and therefore can be targeted collectively.
Diabetes technologies such as CGMs, digital apps, ketone meters, activity monitors, and other wearable diagnostics are now being used for disease prevention as first and relatively easy steps in targeting metabesity.
Repurposing Diabetes Sensors for Other Metabolic Diseases
Mark Clements, MD, PhD, CPI, FAAP
Children’s Mercy Kansas City, Kansas City, MO, USA
Normative data for CGMs in healthy individuals without diabetes have been collected.
Data from CGMs can be used to predict worsening metabolic disease states and to monitor response to noninsulin medications among those with obesity.
Data from CGMs can be used to provide actionable insights related to food choices, food architecture, and physical activity in individuals with obesity and metabolic disease.
Use of CGM for Identifying Glucotypes
Michael Snyder, PhD
Stanford University, Stanford, CA, USA
CGMs can be used to measure glucose dysfunction in healthy, prediabetic people.
People can be classified into glucotypes.
Different people show a significant rise in glycemia following different foods.
Metabesity is a new term that is intended to individualize the comprehensive approach to chronic disease by understanding and prevention, rather than focusing on the treatment of those diseases when they become overt, progressive, and life-limiting. This is particularly relevant to diabetes, which is expressed in different groups, and ultimately in individuals, depending on their genetic predisposition, social circumstances, lifestyle, gut microbiome, and environmental conditions. A more complete understanding of all elements affecting an individual’s health is effectively the ultimate expression of precision medicine. This currently looks at identifying multiple factors that affect individual health and well-being (Figure 14). This process includes disease risk identification and prevention of the commonly associated morbidities which affect people with T1D and T2D such as hypertension and hyperlipidemia, disease duration, age, and obesity. Metabesity is the multidisciplinary study of preventive medicine and the use of technology for the preservation of well-being and function as people age. Scientists from a multiplicity of disciplines are now meeting regularly to further define and study the predispositions to ill health and to extend healthy, satisfying, and productive life. Long-term study of drugs such as metformin, exercise, low carbohydrate diets, and application of technologies such as CGMs should be undertaken with longevity and disease-free aging as the desired and hoped for outcomes.
Metabesity is the constellation of chronic diseases and cancer, which all share metabolic root causes and can therefore be targeted collectively. Abbreviation: NASH, non-alcoholic steatohepatitis.
As a part of understanding disease predisposition and treatment, technology is increasingly being used to monitor physiologic function and perhaps understand and predict progression of disease processes, which eventually appear as overt diabetes. Such devices should be studied and should be helpful in disease amelioration and/or prevention. Currently, there is evidence that devices such as glucose monitors might be useful in identifying the early changes associated with developing prediabetes as well as monitoring those with the condition. Individuals currently identified as “normal” have been shown to have a range of responses to mealtime challenges depending on age, sex, weight, and current fasting glucose. Do these different responses to meal composition impact long-term health? Could CGM use in normal individuals be used for earlier prediction of the risk and progression of glucose intolerance toward overt diabetes? Could it predict longevity and disease process development in those who currently have normal definitions of health and well-being? What is the impact of continuous glucose monitoring in diseases such as hypertension? What is the impact in normal individuals of currently suggested diets, such as the low-carbohydrate diet, and the prevention and treatment of T2D? A variety of reviewed papers studying normal individuals demonstrated differences in glucose variability, which might have an impact on health over the period of a lifetime and be useful in disease amelioration and prevention. Repurposing sensors for diabetes to study other chronic diseases such as hypertension and hyperlipidemia may be useful in terms of identification, treatment, and outcomes.
“Glucotyping” is the use of CGM profiles to show differences between normal individuals that might predispose them to different degrees of glucose intolerance. The data and the pilot study presented reviewed the use of CGMs in metabolic profiling of 53 individuals with normal glucose tolerance by current criteria. This demonstrated marked variability in dietary responses to meals of varying composition, although the greatest elevations in blood glucose occurred with high-glycemic-index foods such as cornflakes and milk. However, marked variations in glucose excursions and variability were seen even with high-protein meals. Spectral analysis was used to assess periodic CGM tracings for variability. High degrees of glucose variability were observed, in some cases similar to those with overt diabetes.
In addition to continuous glucose monitoring, an example was given of an individual followed with dense fasting glucose and HbA1c profiling - this individual acquired unexpected T2D, and early detection enabled temporary amelioration with diet and exercise, and the subsequent rational selection of treatment with the addition of oral medication appropriate for that individual’s metabolic circumstances. DNA methylation changes occurring with a viral illness were associated with the onset of glucose intolerance. A second viral illness was associated with a further deterioration of glucose tolerance and the need for medication rather than diet and exercise alone. Thus, increased DNA methylation occurred at the same time as these viral exposures and the progression of diabetes, an interesting association which was felt to be of likely etiologic significance and possibly wider applicability if studied. Additional metabolic sensors were broadly described along with their potential use in the early identification of metabolic illness, cancer, and infectious disease.
Session 11: Hot Topics
Moderators
Sarah Kim, MD
University of California, San Francisco, San Francisco, CA, USA
Kayo Waki, MD, PhD, MPH
The University of Tokyo, Tokyo, Japan
Precision Diabetes
Louis Philipson, MD, PhD
University of Chicago, Chicago, IL, USA.
Precision diabetes medicine (PDM) is an emerging approach for disease treatment and prevention that accounts for individual variability in genes, environment, and lifestyle for each person.
“Precision” means exact and accurate, whereas “personalized” means meeting individual requirements.
Clustering of T2D into subgroups illustrates aspects of the PDM concepts.
CGM in Type 2 Diabetes Not on Insulin
Eugene Wright, MD
Charlotte Area Health Education Center, Charlotte, NC, USA
For the past ten years, numerous studies have demonstrated the glycemic benefits of CGM use in individuals with T1D and intensively treated T2D, with reductions in healthcare resource utilization and costs.
There is a growing body of evidence suggesting that CGM use confers similar benefits in PWD who are treated with less-intensive therapies. Moreover, behavioral interventions that included use of CGM have been shown to improve dietary habits, increase physical activity, reduce body weight and cardiometabolic risk, and enhance problem-solving skills.
While the benefits of CGM in people with T1D and T2D, who are intensive insulin using patients, are well established, the use of CGM beyond these patients is becoming established with data on: Glycemic control and HbA1c reduction; Behavior modification to improve glycemic control and diet and activity; Enhanced problem-solving skills for glycemic control.
Diversity in Diabetes Technology
Katharine Barnard-Kelly, PhD
BHR Ltd, Portsmouth, Hampshire, UK
It is clear that inequalities have not reduced in any meaningful way. These inequalities are exacerbated by structural, societal, and individual biases.
Confusion exists surrounding what diabetes technology actually is (e.g., devices, telemedicine). This enables “cherry picking” of easier issues to discuss or address.
Greater efforts are needed to ensure inclusivity in research, decision-making bodies, and healthcare as a whole.
Using Diabetes Technology in Pregnancy
Lynn Yee, MD, MPH
Northwestern University Feinberg School of Medicine, Chicago, IL, USA
Pregnancy is a period of enhanced learning requirements, amplified health behavior change demands, intensified healthcare engagement, and tighter glycemic control requirements. The intensity of the perinatal period is a burden on patients, and the appropriate use of evidence-based technology can improve perinatal outcomes.
CGMs and insulin pumps are now standard of care for pregnant individuals with T1D and are increasingly available to people with T2D or gestational diabetes. Findings of the CONCEPTT trial showed that the use of CGM during pregnancy in patients with T1D was associated with better maternal glycemic control and improved neonatal health outcomes.
Electronic patient portal use, smartphone applications for health education and support, telemedicine for improved access to perinatal diabetes specialists, and glucose monitors with direct input of data to clinician portals are all emerging or standard technologies that may enhance perinatal outcomes in the setting of diabetes.
Green Diabetes
Lutz Heinemann, PhD
Science Consulting in Diabetes GmbH, Kaarst, Germany
Diabetes sustainability and waste management is a complex topic.
Changes need to be made at all levels: patients, hospitals, practices, storage, and so on.
Sustainability should be part of all considerations about how to design new products and how to pack them.
This session included technologies that aim to ease the burden of managing diabetes and reduce healthcare disparities.
There exists a global interest in PDM, which ventures to move away from a “one-size-fits-all” approach to biomarker-driven diabetes treatment and prevention. Although T2D is a heterogeneous disease, it is therapeutically addressed with the assumption that it is a uniform disease. This approach inevitably leads to “trial and error” when it comes to medication and lifestyle recommendations. PDM recognizes that T2D varies in several characteristics including but not limited to the nature and degree of beta cell failure, insulin resistance, dyslipidemia, and body composition. 136 Using these characteristics, PWD can be placed in specific “clusters” or subtypes. 137 The idea with PDM is to predict, a priori, what therapies and methods of monitoring would be most beneficial to each subtype based on its unique characteristics.
There is also increasing evidence that CGM use may be beneficial in PWD who are not on intensive insulin regimens. A database study evaluated HbA1c levels prior to and six months after being prescribed a flash CGM. There was a 1.5% drop in HbA1c overall and 1.1% drop among those who were not on insulin. 138 CGM use may improve glucose levels by providing real-time, detailed feedback on diet, exercise, and medications.
Technology use could inadvertently increase healthcare disparities because access to technologies such as smartphones and Internet and even technology literacy is higher in wealthier populations. This structural inequality, also known as the “digital divide,” may exacerbate health-related challenges already faced by vulnerable populations.
Different technologies can aid in glycemic control during pregnancy (Figure 15). Gestational diabetes is a unique condition in which women must quickly learn to execute intense glycemic control for a relatively short but critical period. In addition, health learning behaviors and social determinants of health impact the course of pregnancy and may further impact glycemic control during pregnancy. CGM use in people with T1D has been shown to improve glycemic control (without an increase in hypoglycemia) and reduce both obstetric and fetal complications. 139 Although the data supporting insulin pump use has been mixed, there is promise in leveraging AID systems. An early, small study showed improved overnight glucose levels with CGM sensor augmented insulin pump delivery among pregnant women. Telemedicine and mobile health, particularly in the face of the COVID-19 pandemic, are additional technologies that would reduce in-person visit burden.140,141
Technology uses in pregnancy. Abbreviation: EMR, Electronic Medical Record.
The session concluded with a discussion about the impact of diabetes treatment on the environment, a timely topic that was first addressed on an international platform at the 2021 Green Diabetes Summit. 142 The treatment of diabetes with injectable medications, fingerstick glucose testing devices, CGMs, and insulin pumps generates waste. 143 An environmentally responsible approach to diabetes treatment should be the concern of not only HCPs and patients but also device manufactures, pharmaceutical companies, and government regulatory agencies. It will require collaboration across these stakeholders as well as financial commitment to make diabetes care environmentally sustainable. 144
Live Demonstration
Moderators
Gu Eon Kang, PhD
The University of Texas at Dallas, Richardson, TX, USA
Dorian Liepmann, PhD
University of California, Berkeley, Berkeley, CA, USA
A Mobile App for Foot Selfies to Monitor Diabetic Foot Wounds
Mark Swerdlow, MS
University of Southern California, Los Angeles, CA, USA
There exists an important unmet need to help patients with diabetic foot disease easily screen their feet at home. Current recommended solutions of asking someone else to examine their feet or using a hand mirror are insufficient.
The Foot Selfie System is a novel, low-cost, smartphone-based tool that achieves this goal. In a small-scale study, we showed that patients found it easy-to-use and felt that it gave them more control over managing their disease; it permitted physicians to make clinical-management decisions on a remote basis, especially important during the pandemic.
Combining the system with a sensitive machine learning algorithm and advanced imaging technologies will likely further increase its utility by reducing the time and cost associated with screening images and helping to identify problem areas early.
Diabetic foot ulceration is a leading cause of disability and mortality. A recent study suggests the five-year mortality for diabetic foot ulceration of 30.5% is comparable to that of cancer at 31.0%. Despite the devastating consequences of diabetic foot ulceration, studies have shown that early detection of ulcerations is key to improving clinical outcomes and quality of life for the affected individuals; however, early detection of ulcerations is a challenge because many of these patients also suffer from diabetic peripheral neuropathy, which makes them unaware of an ulcer on their feet.
To address this challenge, a simple, low-cost, smartphone-based system, called “Foot Selfie” was developed. The Foot Selfie system is composed of (1) an apparatus on which a patient can place the plantar surface of the foot, (2) a smartphone, and (3) a smartphone app that is voice activated (Figure 16). It enables patients to photograph their foot on their own by using their voice and then to upload the photos to a remote server. Using the Foot Selfie system, a proof-of-concept feasibility study in 15 patients recruited from the University of Southern California Keck Limb Preservation Clinic was performed. All 15 patients were followed for at least one month, and 10 of the 15 patients were followed for six months. The overall adherence rate was 73% at one month and 80% at six months. 145 Based on a survey, the Foot Selfie system was recognized as easy-to-use, useful, empowering, and preferable to participants’ previous methods of ulceration monitoring.
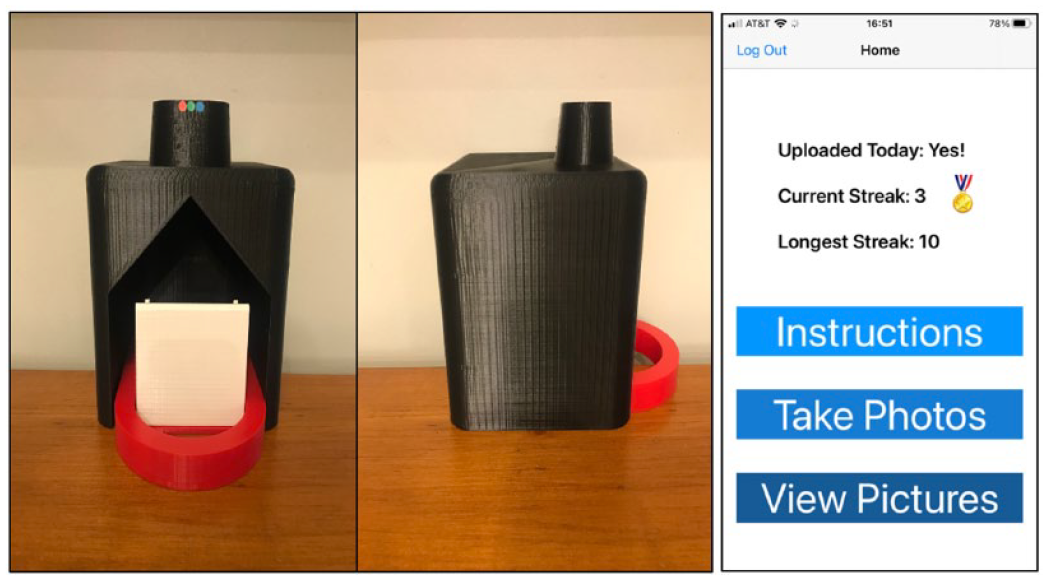
Apparatus and app for taking a Foot Selfie.
The Foot Selfie system is at the pilot stage, but it showed a promise as a monitoring tool for diabetic foot ulceration. In addition to monitoring other risk factors for diabetic foot ulcerations such as temperature and pressure, the Foot Selfie system can help address current challenges to prevent and early detect diabetic foot ulcerations.
Conclusions
The Diabetes Technology Meeting presented various perspectives on the current state of diabetes technology. The meeting’s presentations examined the development of diabetes technology through medical, scientific, regulatory, and engineering lenses.
Footnotes
Acknowledgements
For contributing key points to this report and reviewing the manuscript, we would like to thank Mohammed Al-Sofiani, MBBS, MSc; David G. Armstrong, DPM, MD, PhD; Guillermo Arreaza-Rubin, MD; Shailendra Bajpai, MBBS, MD; Katharine Barnard-Kelly, PhD; Ananda Basu, MBBS, MD, FRCP (UK); Roy Beck, MD, PhD; Cari Berget, RN, MPH, CDCES; Richard Bergenstal, MD; Chris Boulton, BSc; Marc Breton, PhD; Sue Brown, MD; Bruce Buckingham, MD; Kristin Castorino, DO, BC-ADM; Will Cefalu, MD; Eda Cengiz, MD, MHS, FAAP; Faz Chowdhury, PhD; Ali Cinar, PhD; Mark Clements, MD, PhD, CPI, FAAP; Ronald Dixon, MD; Francis J. Doyle III, PhD, CPGS; Andjela Drincic, MD, FACP; Leslie Eiland, MD; Juan Espinoza, MD; Alexander Fleming, MD; Guido Freckmann, MD; Brian Frier, BSc (Hons), MD, FRCP (Edin), FRCP (Glas); Kevin Fu, PhD; Robert Gabbay, MD, PhD; Marcel Gmünder, JD; Michelle Griffith, MD; Gerold Grodsky, PhD; Alberto Gutierrez, PhD; Ricardo Gutierrez-Osuna, PhD; Lutz Heinemann, PhD; Tim Heise, MD; Thanh Hoang, DO, FACP, FACE; Elizabeth Holt, MD, FACE; Roman Hovorka, PhD, FMedSci; Elvira Isganaitis, MD, MPH; Peter Jacobs, PhD; Jeffrey Joseph, DO; Francine Kaufman, MD; Boris Kovatchev, PhD; Piotr Ladyzynski, PhD, DSc; Alice Leiter, JD; Dorian Liepmann, PhD; Maysam Mansouri, PhD; Robert Mucic, PhD; Rahul Narayan, MS; Anant Nigam, MD, FRCP; Teresa Oliveria, PharmD; Søren S. Østergaard, MS; Anne Peters, MD; Andreas Pfützner, MD, PhD; Louis Philipson, MD, PhD; Moshe Phillip, MD; Mihailo Rebec, PhD; David Rodbard, MD; Steven J. Russell, MD, PhD; Leonardo Scapozza, PhD; Naomi Schwartz, MS; Peter Simpson, MEng; Michael Snyder, PhD; Koji Sode, PhD; Jannet Svensson, MD, PhD; Mark Swerdlow, MS; Marc Taub, PhD; Andreas Thomas, PhD; Guillermo Umpierrez, MD; Josep Vehi, PhD; Martina Vettoretti, PhD; Robert Vigersky, MD; Naunihal Virdi, MD; Paul Wadwa, MD; Kayo Waki, MD, PhD, MPH; Michael Weiss, MD, PhD, MBA; Eugene Wright, MD; Lynn Yee, MD, MPH; Dessi Zaharieva, PhD; and Gretchen Zimmermann, RD, CDCES. We thank Annamarie Sucher-Jones for her expert editorial assistance.
Abbreviations
ADA, American Diabetes Association; AGP, Ambulatory Glucose Profile; AI, artificial intelligence; AID, automated insulin delivery; BGM, blood glucose monitor; BIF, basal insulin Fc; CCPA, California Consumer Protection Act; CDER, Center for Drug Evaluation and Research; CDRH, Center for Devices and Radiological Health; CGM, continuous glucose monitor; CKM, continuous ketone monitor; CMS, Centers for Medicare & Medicaid Services; COVID-19, coronavirus disease 2019; CSII, continuous subcutaneous insulin infusion; CT, computed tomography; DGM, deep generative model; DKA, diabetic ketoacidosis; DME, durable medical equipment; DTC, direct-to-consumer; DTM, Diabetes Technology Meeting; DTS, Diabetes Technology Society; EHR, electronic health record; EU, European Union; FAD, flavin adenine dinucleotide; FCL, fully closed loop; FDA, United States Food and Drug Administration; FTC, Federal Trade Commission; GAN, generative adversarial network; GDPR, General Data Protection Regulation; GLP1, glucagon-like polypeptide-1; GMI, Glucose Management Indicator; GRI, Glycemia Risk Index; HbA1c, hemoglobin A1c; HCL, hybrid closed loop; HCP, healthcare professional; HIPAA, Health Insurance Portability and Accountability Act of 1996; HPLC, high-performance liquid chromatography; iAUC, incremental area under the glucose curve; LED, light-emitting diode; MAD, mean absolute difference; MARD, mean absolute relative difference; ML, machine learning; MPC, model predictive control; NAD+, nicotinamide adenine dinucleotide; NIGM, noninvasive glucose measurement; OCP, open-circuit potential; ODE, ordinary differential equation; %[percent]CV, coefficient of variation; PDM, precision diabetes medicine; PMS, post-market surveillance; PWD, persons with diabetes; RBC, red blood cell; rPLS, regularized partial least squared; RWE, real-world evidence; SaMD, software as a medical device; SNR, signal-to-noise ratio; T1D, type 1 diabetes; T2D, type 2 diabetes; TIR, time in range; UFAL, ultrafast-absorbing insulin lispro; US, United States.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NYX is a consultant to Abbott Diabetes Care. KTN is a consultant to Abbott Diabetes Care. JP reports consultancy fees from Medtronic and BD. JLS has conducted clinical trials for Eli Lilly, Insulet, and Medtronic and has received in-kind support for research studies from Dexcom and Medtronic. She has consulted for Eli Lilly, Lexicon, Medtronic, and Sanofi. She has been a member of advisory boards for Bigfoot Biomedical, Cecelia Health, Eli Lilly, Insulet, the T1D Fund, and Vertex. EC is an advisory board member and consultant for Novo Nordisk, Adocia, Eli Lilly, and Arecor. DA has received speaker’s honoraria from Lilly Diabetes, Novo, Xeris Pharmaceuticals, and Zealand Pharma. He has received consulting fees from Mannkind, Ascensia Diabetes Care, Roche Diagnostics, and Senseonics. RB is shareholder of Biomeris s.r.l, a university spin-off dealing with clinical research informatics. Moreover, he has received consulting fees from Merck Healthcare to support the design of decision support tools. EKS reports that this work was supported in part by a VA MERIT award from the U.S. Department of Veterans Affairs Clinical Sciences Research and Development Service (1I01CX001825). He has received unrestricted research support from Dexcom (to the Baltimore VA Medical Center and to the University of Maryland) for the conduction of clinical trials. UM reports research funding from Clementia Pharmaceutical and is an advisor for Ryse Health. HKA received research support through University of Colorado from Dexcom, Senseonics, Medtronic, Eli Lilly, Mannkind. DCK is a consultant to AI Health, Dexcom, Eli Lilly, EOFlow, Integrity, Lifecare, Medtronic, Novo, Roche Diagnostics, Rockley Photonics, and Thirdwayv. AYD, HT, BHG, CC, LGP, LP, RB, BWB, JSM, SK, and GEK have nothing relevant to disclose.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.