Abstract
Background:
Glucagon-like peptide-1 (GLP-1) receptor agonists are a relatively recent addition to the treatment options for type 2 diabetes mellitus (T2DM) and are administered using prefilled pen devices.
Method:
In this open-label task and interview-based pilot study, 3 GLP-1 receptor agonist pen devices—exenatide (Byetta®, Bristol-Myers Squibb/AstraZeneca), liraglutide (Victoza®, Novo Nordisk), and lixisenatide (Lyxumia®, Sanofi-Aventis)—were comparatively assessed in a randomized order in 30 participants with T2DM for ease of use, using a series of key performance measures (time taken to complete a series of tasks, number of user errors [successful performance], and user satisfaction rating). Linear and logistic regression analysis was conducted for the lixisenatide and liraglutide pens versus the exenatide pen. Participants’ mean age was 60 years; 27% and 20% of the participants had visual impairments and reduced manual dexterity, respectively.
Results:
Tasks were completed faster (
Conclusions:
Lixisenatide and liraglutide pens are associated with higher user satisfaction compared with the exenatide pen. In addition, the lixisenatide pen is faster and results in fewer errors than its comparator (exenatide). The lixisenatide pen may therefore be a suitable choice for patients with T2DM, including older and pen device-naïve patients, and those with visual impairments and reduced manual dexterity.
Keywords
Glucagon-like peptide-1 (GLP-1) receptor agonists are a relatively new therapeutic option for the treatment of type 2 diabetes mellitus (T2DM). These agents are generally well tolerated and provide glycemic control and other clinical benefits.1,2 Aside from their efficacy in reducing glycated hemoglobin via effects on both fasting plasma glucose and postprandial plasma glucose, GLP-1 receptor agonists allow patients to achieve glycemic targets, with beneficial effects on weight being reported.1,3-6 One of the key benefits of GLP-1 receptor agonists is that they stimulate insulin secretion and inhibit glucagon secretion in a glucose-dependent manner, and therefore carry a limited risk of hypoglycemia.3,7 The GLP-1 receptor agonists currently available are exenatide (Byetta®, Bristol-Myers Squibb/AstraZeneca), administered as a twice-daily injection; 8 liraglutide (Victoza®, Novo Nordisk), administered as a once-daily injection; 9 exenatide (Bydureon®, Bristol-Myers Squibb/AstraZeneca), administered as a once-weekly injection; and lixisenatide (Lyxumia®, Sanofi-Aventis), a once-daily prandial GLP-1 receptor agonist. 10 Twice-daily exenatide as well as once-daily liraglutide and once-daily lixisenatide are available as ready-to-use prefilled pen devices for subcutaneous injection. Exenatide is available in 2 different fixed-dose pens: a 5 µg pen and a 10 µg pen, with each prefilled pen designed to deliver a premeasured dose. Exenatide should be initiated at 5 µg per dose administered twice-daily for at least 1 month to improve tolerability; the dose can then be increased to 10 µg twice-daily to further improve glycemic control. 11 Liraglutide is a multidose pen that delivers doses of 0.6 mg, 1.2 mg or 1.8 mg. Liraglutide should be initiated at 0.6 mg per day for 1 week; after 1 week the dose can then be increased to 1.2 mg, and if this does not result in acceptable glycemic control, the dose can be increased to 1.8 mg. 12 Lixisenatide is available in 2 different fixed-dose pens: a 10 µg pen and a 20 µg pen, with each prefilled pen designed to deliver a premeasured dose. Lixisenatide is initiated at 10 µg once-daily for 14 days, with a fixed maintenance dose of 20 µg once-daily commencing on Day 15. 13
Since their introduction in the 1980s, pen devices have been used to administer insulin and are preferred by many patients over traditional administration with a vial and syringe.14-16 Studies demonstrate that pen devices can increase treatment satisfaction and quality of life (QoL) in patients with T2DM,15-18 and provide more accurate and precise delivery of medication doses compared with a vial and syringe, both of which can help improve glycemic control and long-term outcomes.19-21 Ease of use is a particularly important feature of a pen device. In a recent questionnaire-based survey of more than 1000 patients with T2DM, conducted to evaluate which features of a medication injector device were most important for overall user satisfaction, ease of use was found to be the single most important feature. 22 Among patients with diabetes, there is a high prevalence of visual impairment due to diabetes-associated retinopathy and maculopathy. 23 Reduced manual dexterity, such as limited joint mobility syndrome, is often seen in these patients, and may limit finger or hand strength and the ability to inject medication without assistance.19,24-26 In such patients, the ease with which a pen device can be used without error is of particular importance. In addition, many patients with diabetes are older and may require more time to learn the various functions of a pen device, and ease of use is therefore also important in this population. Research studies on pen devices that include patients with manual and visual impairments, especially with devices that deliver GLP-1 receptor agonists, however, are limited.
Pen device technology is continuously evolving, and such devices are becoming easier to use, more convenient, more flexible, safer, and more socially acceptable.16,19,27,28 Until recently, pen devices had only been used to administer insulin. However, with the introduction of the injectable GLP-1 receptor agonists that are also available in prefilled pen devices, ease of use and satisfaction with device use will become even more important. Pen devices can have varying designs and features, which can impact on dosing accuracy and the ability of patients to easily use the devices. These include the ability to hear and feel clicks (tactile and auditory feedback), easy dialing and delivery of a dose, ease of performing the safety test and cartridge replacement, and overall ease of use. The primary objective of this study was to comparatively assess GLP-1 receptor agonist pen devices—the once-daily lixisenatide pen, with the once-daily liraglutide pen and the twice-daily exenatide pen—for ease of use using 3 important practical aspects.
Methods
Study Design
This was an open-label, task, and interview-based pilot study, conducted in Frankfurt, Germany, and London, United Kingdom. Key performance measurements that were used to assess the 3 pen devices were the time taken to complete a series of tasks, the number of user errors (successful performance), and user satisfaction ratings for each device. This study was not approved by the institutional review board. Following a risk assessment, it was identified that the study involved little or no risk to the patients recruited as the study made use of simulated tasks, and therefore did not require the participant to self-inject, and a moderator was present at all times to observe any potential needle stick injuries.
Participants
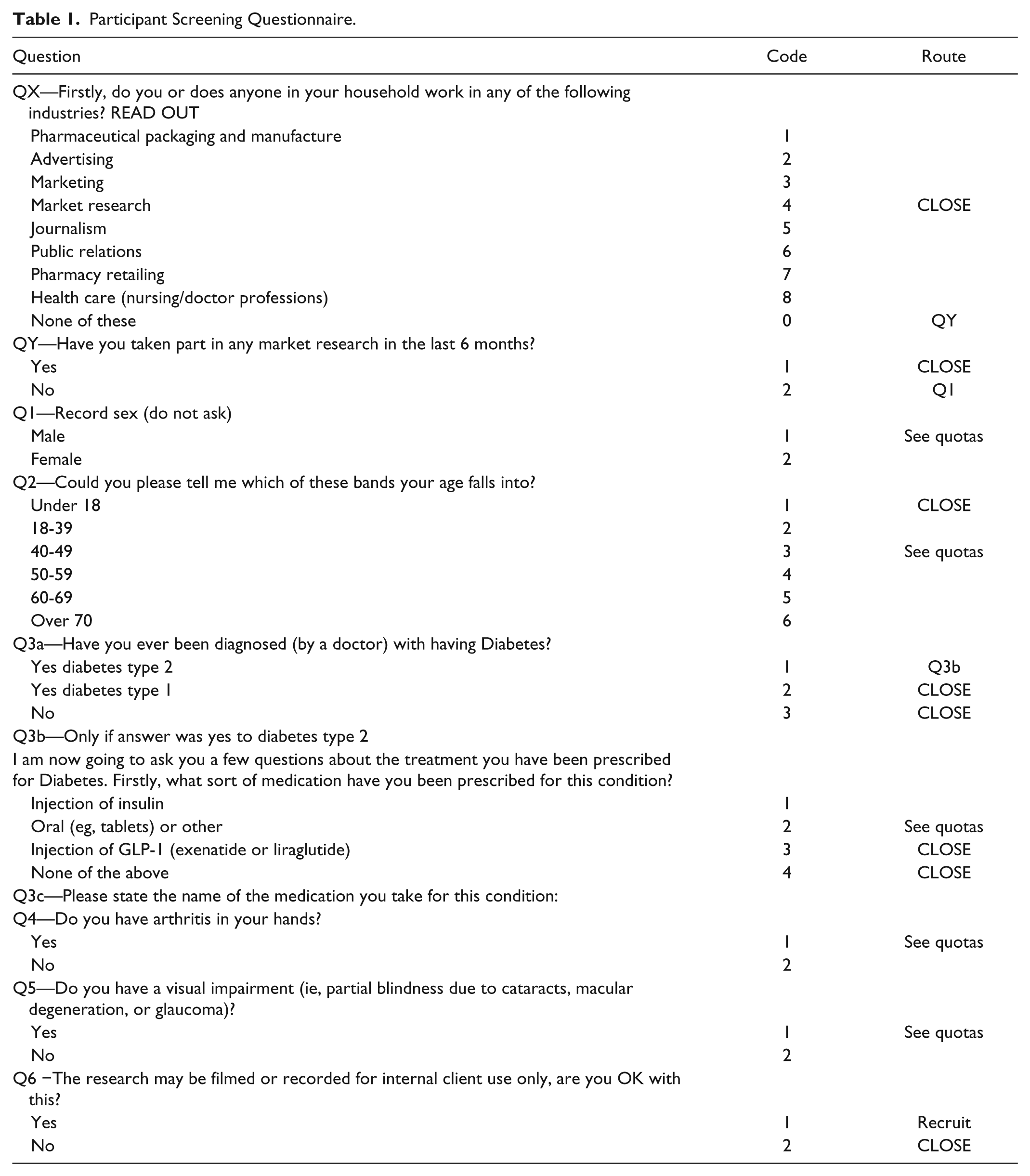
A specialist recruitment agency (WorldOne HealthCare Research) recruited participants for this study using a detailed recruitment screener document that clearly specified the participant profile for recruitment into the study and included a series of screening questions (Table 1). Research was conducted at a nonclinical research unit. Overall, 30 participants from Germany (n = 15) and the United Kingdom (n = 15) with T2DM were enrolled using a stratified sampling method, and all enrolled participants completed the study. Participants were GLP-1 receptor agonist-naïve subjects ≥18 years of age with T2DM for >1 year. The study aimed to enroll a total of 60% of participants >60 years old, 25% of participants had to have a visual impairment (self-reported) and 25% a form of manual dexterity impairment (self-reported), and an equal number of males and females. Two-thirds of participants had to be pen device-naïve.
Participant Screening Questionnaire.
Assessments
Assessments were conducted in January 2012 at outpatient clinics by independent moderators from a research agency (DCA Design International, UK; 22030425 and WorldOne, Germany) in line with the study protocol. Each assessment took 90 minutes to complete. To ensure consistency, task assessment was conducted by 1 moderator after study completion using video data and data gathered from study observations (real time via a note taker). The order in which participants tested each device was randomized using a 3 × 3 Latin squares design to avoid bias from user learning. Participants received the manufacturer’s country-specific patient instruction leaflet for each device (prospective leaflet for lixisenatide pen device). Patients were not given formal training on the use of the pen devices. All participants were asked to consecutively demonstrate the use of each of the 3 pens by injecting them into a simulator pad. To measure successful performance, participants carried out a series of tasks with each pen: priming the pen, administering a first dose into a simulator injection pad (without priming), administering a second dose into a simulator injection pad (without priming), determining the remaining dose level in the pen, and identifying when the pen was empty. The number of participants who completed each task without error (successful performance) and the time taken to complete tasks were recorded in real time by direct observation by a researcher (DCA Design International) using a digital stopwatch (Tables 2 and 3).
Summary of User-Based Trial and Time Taken to Complete Tasks.
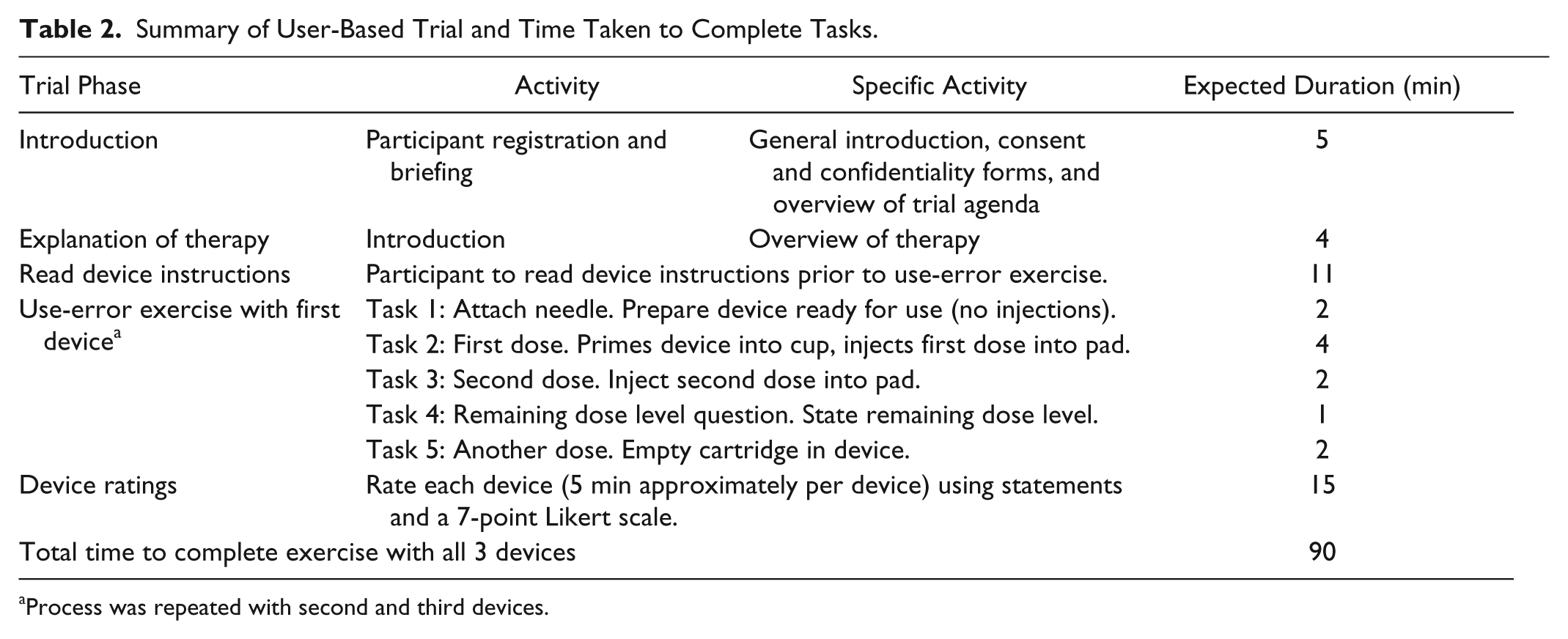
Process was repeated with second and third devices.
Use-Error Exercise.
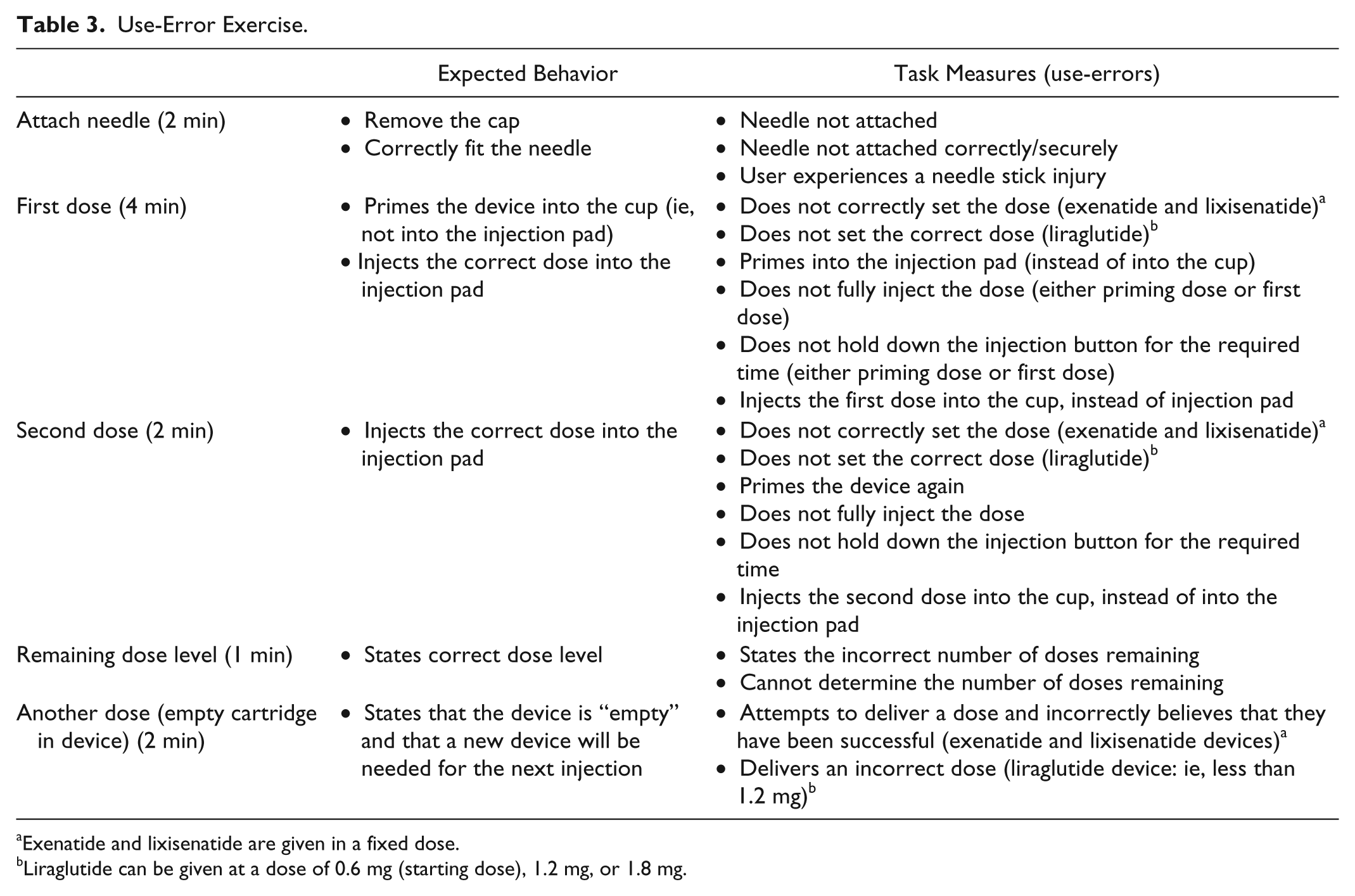
Exenatide and lixisenatide are given in a fixed dose.
Liraglutide can be given at a dose of 0.6 mg (starting dose), 1.2 mg, or 1.8 mg.
Participants were asked to provide their own assessment of the overall usability of the 3 pen devices based on a number of questions using a 7-point Likert-type scale (1 =
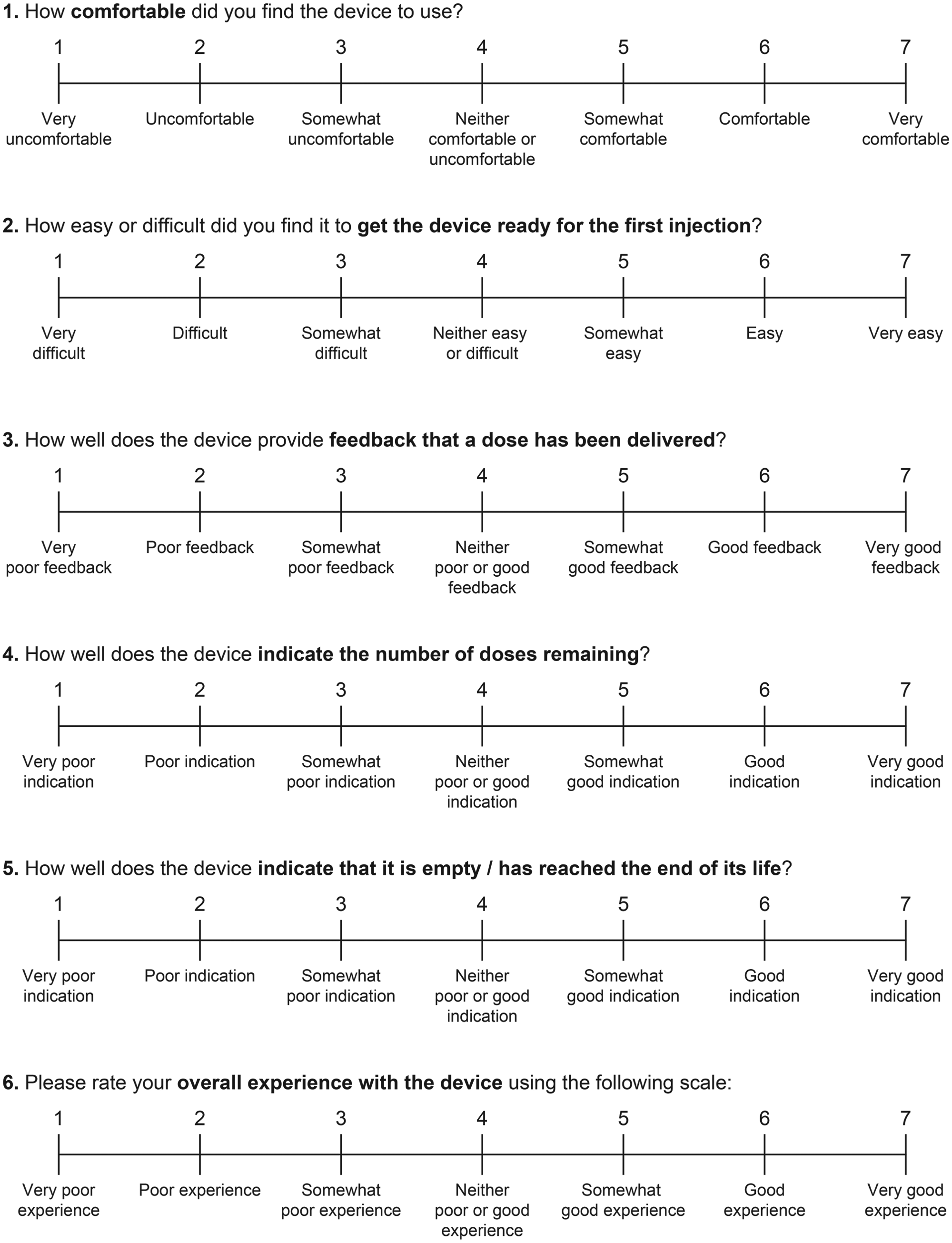
Participant assessment of the overall usability of the 3 pen devices based on questions using a 7-point scale.
Statistical Analyses
Statistical analyses were conducted on the 3 main outcome variables: time taken to complete the tasks, successful completion of the tasks, and user satisfaction ratings. The lixisenatide pen and the liraglutide pen were compared with the exenatide pen (reference pen).
Time taken to complete the tasks was derived using random effects linear regression of the total time (seconds) taken to complete all 4 tasks, and represents the difference in time for the comparator pens and the reference pen. For successful performance, a binary variable for overall success was derived, taking an overall score of at least 3 out of 4 successful tasks (administration of first and second doses, determining the remaining dose level in the pen and identifying an empty pen) as overall success. Odds ratios (ORs) were derived using random effects logistical regression. For the user satisfaction rating, random effects linear regression was conducted using the sum of the scores (42 points) for all 6 questions, and refers to the difference in score between the comparator pens and reference pen. The intraclass correlation coefficient and the Akaike information criterion were determined for the random effects logistic regression model.
Results
Patient Characteristics
A total of 19 (63%) participants self-reported that they had never before used an injection device. Nine patients (30%) had experience with injecting insulin. In total, 8 participants (26%) had a visual impairment (partial blindness due to cataracts, macular degeneration, or glaucoma) and 6 participants (20%) had impairment in manual dexterity (arthritis in the hands). Impairments were also self-reported by the participants (Table 4)
Patient Characteristics.
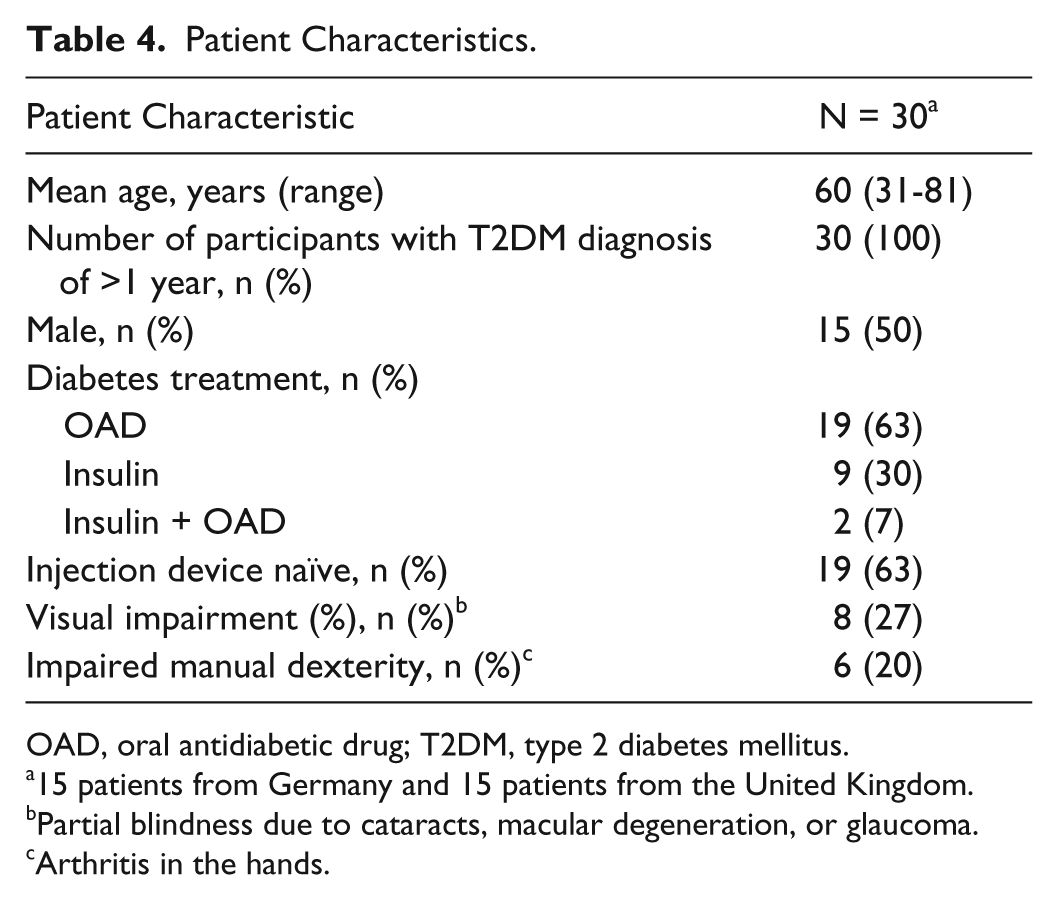
OAD, oral antidiabetic drug; T2DM, type 2 diabetes mellitus.
15 patients from Germany and 15 patients from the United Kingdom.
Partial blindness due to cataracts, macular degeneration, or glaucoma.
Arthritis in the hands.
Successful Performance
In administering a first dose, a second dose, and assessing the remaining dose, a high proportion of participants (83%) were able to complete all 3 tasks without error using the lixisenatide pen. Lower proportions of patients were able to complete these tasks without error with the exenatide and liraglutide pens (43%, 77%, 0%, and 60%, 57%, 10%, for the exenatide and liraglutide pens, respectively; Figure 2). No participants were able to assess a remaining dose without error with the exenatide pen, and 3 participants were able to do so with the liraglutide pen. Almost all participants were able to identify an empty device with all the pens (97% of participants for all 3 pens). Regression analysis of successful performance (using successful completion of at least 3 out of 4 tasks) produced an OR of 11.27 for the lixisenatide pen versus the exenatide pen (
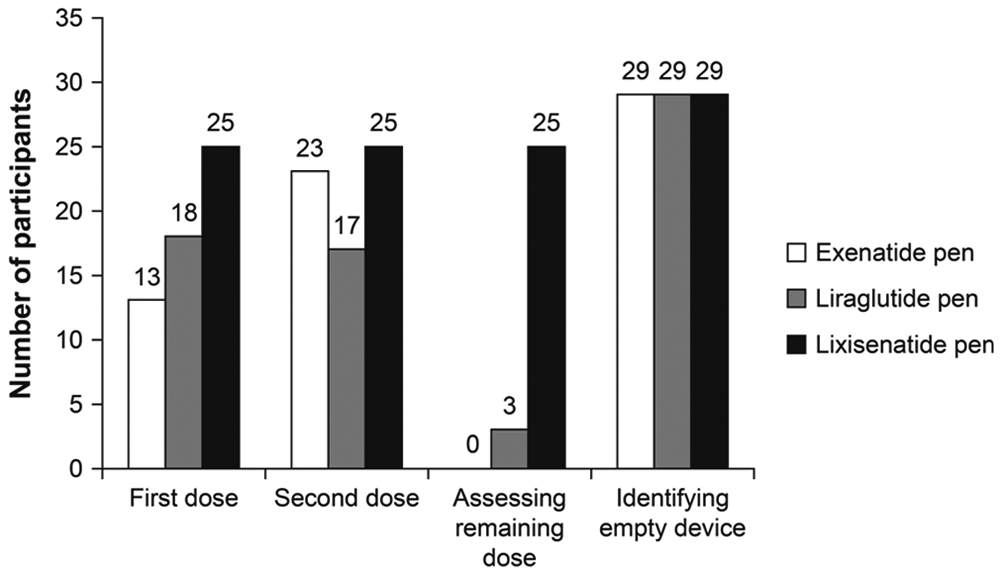
Total number of participants who completed the tasks error free (n = 30 participants).
Regression Analysis for Performance, Task Time, and Overall User Satisfaction of Lixisenatide and Liraglutide Pens Versus the Exenatide Pen.
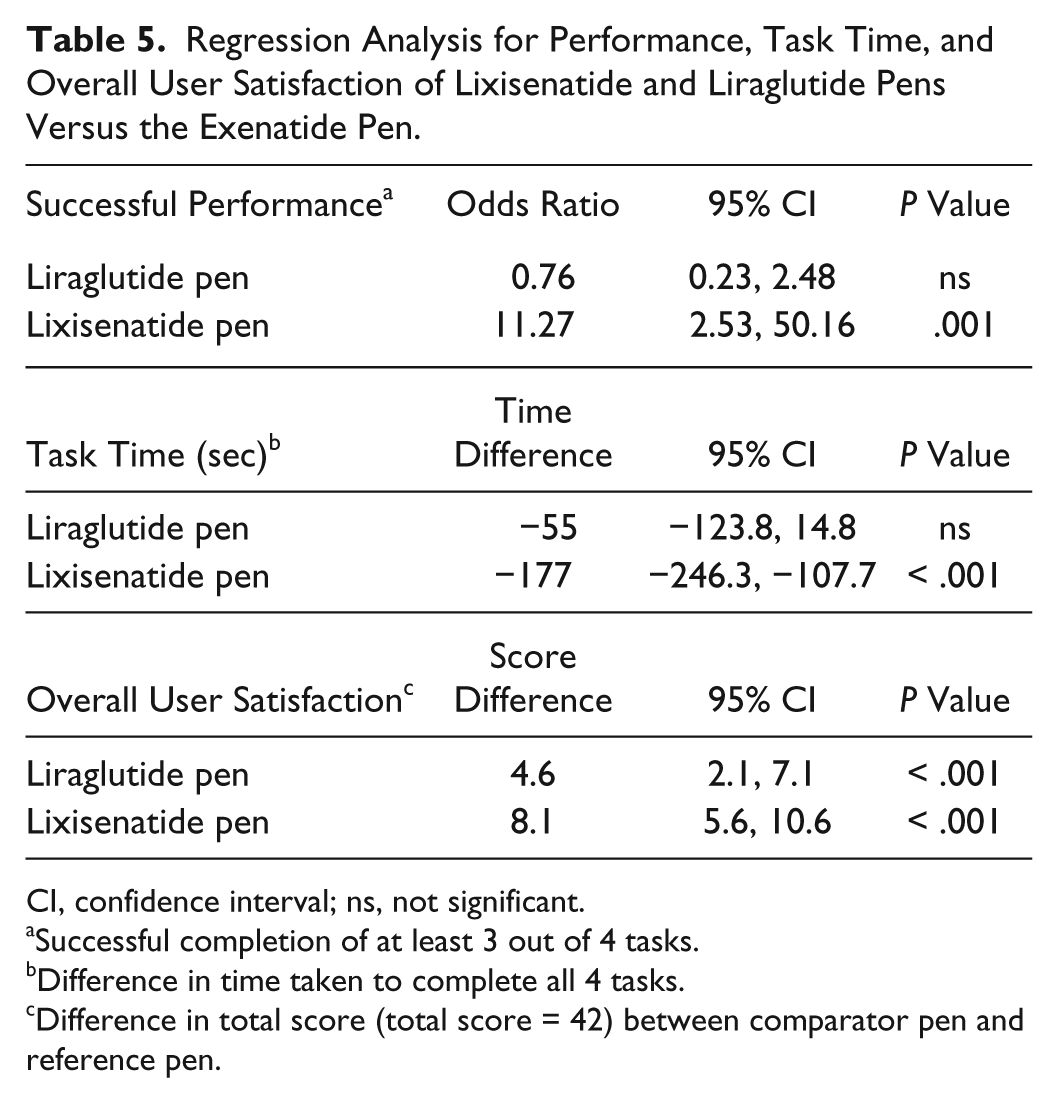
CI, confidence interval; ns, not significant.
Successful completion of at least 3 out of 4 tasks.
Difference in time taken to complete all 4 tasks.
Difference in total score (total score = 42) between comparator pen and reference pen.
Task Time
Time taken to complete the first task (administering a first dose) was considerably higher for the exenatide pen than for the other 2 pens (3.39 vs 2.18 minutes and 2.20 minutes for the exenatide, liraglutide, and lixisenatide pens, respectively; Figure 3). Participants also took longer to complete the remaining tasks (administering the second dose, assessing the remaining dose, and identifying an empty device) with the exenatide pen than with the lixisenatide and liraglutide pens. With these tasks, participants took a shorter time with the lixisenatide pen than with the other 2 pens (Figure 3). Time taken to complete all 4 tasks was significantly lower for the lixisenatide pen versus the exenatide pen (time difference: –177 seconds;
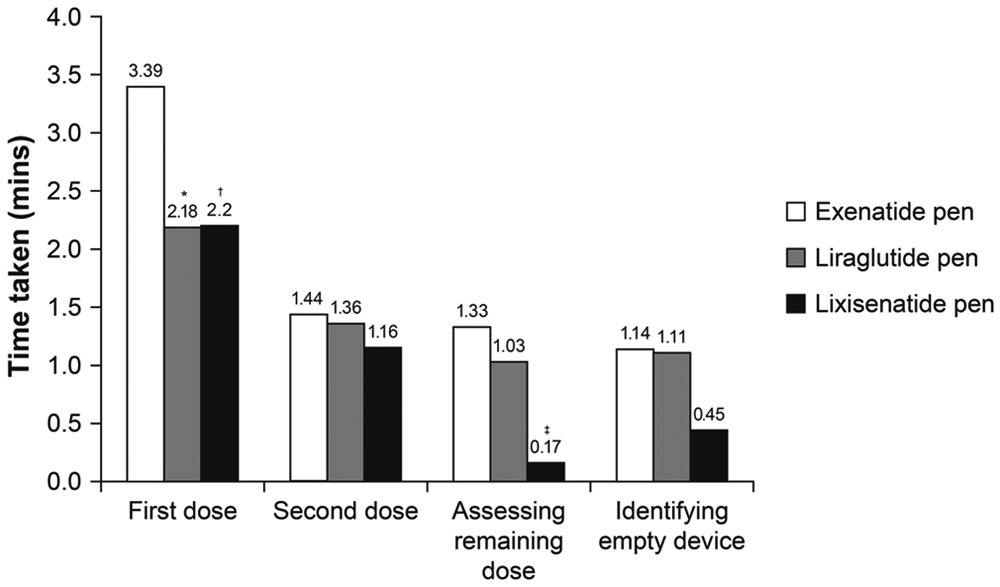
Time taken to complete each task (n = 30 participants). *
Overall User Satisfaction
For delivered dose feedback, indication of the remaining dose and indication of an empty device, the lixisenatide pen was given a higher satisfaction rating by the participants compared with the rating given for the liraglutide and exenatide pens (Figure 4). For device comfort and priming the pen, the lixisenatide and liraglutide pens were rated equally high versus exenatide. For overall user experience, participants rated the lixisenatide pen higher than the other 2 pens (scores of 5.8, 4.4, and 5.3 for lixisenatide, exenatide, and liraglutide pens, respectively; Figure 4). Overall user satisfaction score was significantly higher for the lixisenatide pen (score difference 8.1) and the liraglutide pen (score difference 4.6) versus the exenatide pen (
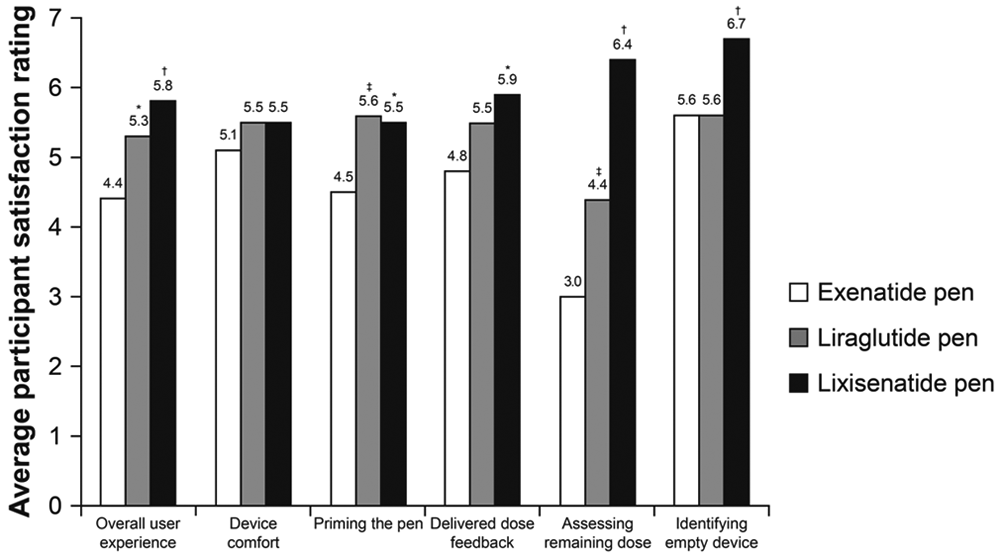
User satisfaction rating of the 3 pen devices (n = 30 participants). *
Discussion
Owing to their many advantages over traditional syringes and needles, pen devices have become common for the administration of insulin, and are also being used for administration of the GLP-1 receptor agonists, lixisenatide, exenatide, and liraglutide. Studies of insulin pen devices have shown that accuracy, ease of use, and patient satisfaction are among the most important factors for achieving glycemic control and patient compliance, and thus long-term beneficial outcomes.18,21,22 Real-world studies are a necessary and important aspect of pen device development, and provide assurance to patients and health care professionals regarding the usability of the device in a real-world situation. In this pilot study in participants with T2DM, we comparatively assessed 3 GLP-1 receptor agonist pen devices for ease of use, using time taken to use the device, successful performance, and user satisfaction.
In this study, more than half the study population was pen device-naïve, and none of the participants received any formal training with the use of pen devices. This is an important aspect of the present study, as it demonstrates that accurate use of a pen device can be enhanced through well-designed instructions and simple and intuitive device design that may remove some of the need for costly and time-consuming training by health care providers. However, in this study no comparisons were made between device-naïve and pen experienced patients in terms of overall ease of use.
Participants enrolled in this study had a mean age of 60 years, and 27% and 20% had visual impairments and reduced manual dexterity, respectively. Visual impairments and reduced manual dexterity are frequently found in people with T2DM, especially among the more elderly population, and it is important that pen devices can be accurately and easily used by this population. Studies that include participants with impaired vision and reduced manual dexterity are limited, but recent research with insulin pen devices showed that visual impairments and reduced manual dexterity in patients with T2DM can influence patient preference. 24
Pen devices that can be used with accuracy are important tools that contribute to safety in diabetes treatment. In this study, participants made fewer errors with the lixisenatide pen device than with the other 2 pen devices, with significantly higher successful performance versus the exenatide pen (
Although intended to be a pilot study, this study has some limitations that should be considered when interpreting the results. The study was open-label, and therefore carries an intrinsic risk of bias. In addition, user feedback with a scale rating may not be the most comprehensive means of evaluation: verbal discussion or comparative rating scales may be more effective. The sample size was small, which may have been a determining factor in the large confidence intervals observed in this trial. In addition, the small sample size did not permit subgroup analyses based on country, insulin-naïve and -experienced users, and visual and dexterity disabilities to be performed. However, this was a pilot study and a large scale study is planned in the future. Although this study included pen device-naïve patients, and those with visual impairments and reduced manual dexterity, no multivariate analyses were performed to assess whether these subgroups had differences in the outcomes assessed and to ascertain which device is the most beneficial for those with these impairments. The fact that visual impairments and reduced manual dexterity were self-reported may also be regarded as a limitation of this study. However, a full study would be designed to screen for such impairments through prestudy assessments. Finally, although not a study limitation, it would be of interest to assess whether twice-daily dosing with exenatide influenced ease of use and to compare the 3 pen devices with exenatide extended-release injection.
Conclusions
Use of the lixisenatide or the liraglutide pen was found to be associated with higher user satisfaction compared with the exenatide pen. In addition, the lixisenatide pen was faster to use and resulted in fewer user errors than its comparator (the exenatide pen), and may be a suitable choice for patients with T2DM, including older and pen device-naïve patients, and those with visual impairments or reduced manual dexterity. Larger-scale, prospective, randomized assessments of these 3 devices involving larger numbers of participants and across a wider range of countries should be conducted.
Footnotes
Acknowledgements
Medical writing assistance was provided by Frances Gambling, Medicus International, London, UK, and was supported by Sanofi-Aventis.
Abbreviations
CI, confidence interval; GLP-1, glucagon-like peptide-1; NS, not significant; OAD, oral antidiabetic drug; OR, odds ratio; QoL, quality of life; T2DM, type 2 diabetes mellitus.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Udo Stauder is an employee of Sanofi-Aventis. Hina Elton is an employee of DCA Design International, which received funding from Sanofi-Aventis. Alfred Penfornis has received fees for consultancy, advisory boards, speaking, travel, or accommodation from Lilly, Novo-Nordisk, and Sanofi-Aventis. Steve Edelman has received consultation fees from Animas Corporation, LifeScan, Inc, DexCom, and Sanofi-Aventis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This pilot study was funded by Sanofi-Aventis (Germany).
