Abstract
Objectives:
To examine changes in device use and glycemic outcomes for pregnant women from the T1D Exchange Clinic Registry between the years 2010-2013 and 2016-2018.
Methods:
Participant-reported device use and glycemic outcomes were compared for women aged 16-40 years who were pregnant at the time of survey completion, comparing 2010-2013 (cohort 1) and 2016-2018 (cohort 2). Hemoglobin A1c results within 30 days prior to survey completion were obtained from medical records.
Results:
There were 208 pregnant women out of 5,236 eligible participants completing the questionnaire in cohort 1 and 47 pregnant women out of 2,818 eligible participants completing the questionaire in cohort 2. Continuous glucose monitor (CGM) use while pregnant trended upward among cohort 2 (70% vs 37%, P = .02), while reported continuous subcutaneous insulin infusion (CSII) use while pregnant declined (76% vs 64%, P = .04). HbA1c levels trended downward (6.8% cohort 1 vs 6.5% cohort 2, P = .07).
Conclusions:
Self-reported CGM use while pregnant increased over the studied intervals whereas CSII use decreased. Additional evaluation of device use and the potential benefits for T1D pregnancies is needed.
Introduction
Tight glycemic control in pregnancies complicated by type 1 diabetes reduces the risk of adverse fetal and maternal outcomes. 1 The glucose targets for pregnancy recommended by the American Diabetes Association for women with preexisting diabetes are a glycated hemoglobin A1c (HbA1c) level of <42 mmol/mol (<6%) and glucose targets for fasting, one-hour, and two-hour postprandial of <5.2 mmol/L (<94 mg/dL), <7.8 mmol/L (<141 mg/dL), and <6.7 mmol/L (<121), presuming these targets can be obtained without severe hypoglycemia. 1 Diabetes technologies provide both patients and their health care team additional tools to help achieve these glycemic targets.2,3 Results from predominantly small trials evaluating continuous subcutaneous insulin infusion (CSII) therapy and continuous glucose monitors (CGM) during pregnancy for women with type 1 diabetes have yielded mixed results in terms of both use and efficacy of the devices.2-7 Most published studies are retrospective analyses completed outside of the United States. Over the last 20 years, very few publications have been prospective or reported the frequency of technology use or its potential impact on glycemic control for pregnant women in the United States.7,8 The T1D Exchange clinic registry provides a unique opportunity to capture prospective data on changes in device adoption and blood glucose control over time in a real-world, large cohort of pregnant women with type 1 diabetes. We assessed differences in use of CSII and CGM therapies and changes in glycemic control in pregnant women who participated in the T1D Exchange clinic registry over two time intervals, from 2010-2013 to 2016-2018.
Methods
The T1D Exchange Clinic Network includes over 80 US-based pediatric and adult endocrinology practices providing specialized diabetes care that have enrolled over 35 000 individuals with type 1 diabetes. Written informed consent was obtained from adult participants and parents/guardians of minor participants. Minor participants provide written assent, according to Institutional Review Board requirements and conforming to the Declaration of Helsinki. Details on Clinic Registry participant eligibility criteria, the informed consent process, and data collection have been published previously.9,10 This report includes data from 5236 females between 16 and 40 years old who completed the pregnancy and childbirth portions of the registry participant questionnaire either at enrollment or year 1 administered between 2010 and 2013, and 2818 female clinic registry participants between 16 and 40 years old who completed the questionnaire between 2016 and 2018, for a year 5 data collection.
Sex, race/ethnicity, insurance status, pregnancy status, self-monitoring blood glucose (SMBG), and occurrence of severe hypoglycemia (SH; defined as seizure or loss of consciousness) within the prior three months also were collected on the participant questionnaires. Information about age, duration of diabetes, anthropomorphic measures including BMI, height, and weight, CSII and CGM use at most recent clinic exam, and HbA1c concentrations obtained as part of usual care were collected from medical records. In addition, CSII and CGM device use specifically during pregnancy was obtained through self-report in the pregnancy and childbirth portions of the questionnaires. To ensure HbA1c measurements reported were collected during gestation for participants, HbA1c data were restricted to within 30 days prior to questionnaire completion.
Statistical Methods
Calculation of frequency of pregnancy in both time cohorts (2010-2013 and 2016-2018) used the full analysis cohort; remaining analyses included only participants who indicated they were pregnant at the time of questionnaire completion (n = 208 in 2010-13; n = 47 in 2016-18). Multivariable linear regressions were performed to assess the association between HbA1c and time cohort and the association between binary outcomes (achieving HbA1c ≤45/≤48 mmol/mol [≤6.0%/≤6.5%], participant-reported use of CSII while pregnant, participant-reported use of CGM while pregnant, participant-reported occurrence of SH) and time cohort.
Results are expressed as means (M) ± standard deviations (SD) for normally distributed variables or medians (interquartile range [IQR]) for non-normally distributed variables. To account for possible confounding, the following covariates were assessed for association with each outcome through bivariate analysis and selection models: age, race/ethnicity, duration of diabetes, SMBG, insurance status, CSII use at time of most recent clinic exam (clinic-reported), CGM use at time of most recent clinic exam (clinic-reported), and clinic site. If an association with an outcome was present, the covariate was included in the model for that outcome.
Data analyses were performed using SAS 9.4 (SAS Institute, Cary, NC). In view of multiple comparisons, only P values <.01 were considered suggestive of a true association. All P values were two-sided.
Results
Among the 5236 eligible participants in cohort 1 who completed the questionnaire in 2010-2013, 208 (4.0%) indicated they were pregnant at the time of questionnaire completion (enrollment or year 1 questionnaires); among the 2818 eligible participants in 2016-2018 in cohort 2, 47 (1.7%) indicated they were pregnant at the time of questionnaire completion (year 5 questionnaire).
Table 1 provides clinical characteristics of cohort 1 (n = 208) and cohort 2 (n = 47). In both cohorts, the majority of participants were non-Hispanic white (cohort 1 = 86% vs cohort 2 = 85%), mean age was similar (29 ± 6 years vs 30 ± 6 years), with most respondents indicating they had private insurance coverage (76% vs 80%). Cohort 1 had a median diabetes duration of 15 years (range 9-22), while in cohort 2 median disease duration was 17 years (range 13-23) years. Reported SMBG frequency was higher in cohort 1 than in cohort 2, a finding that held true whether or not participants reported using a CGM.
Participant Characteristics.
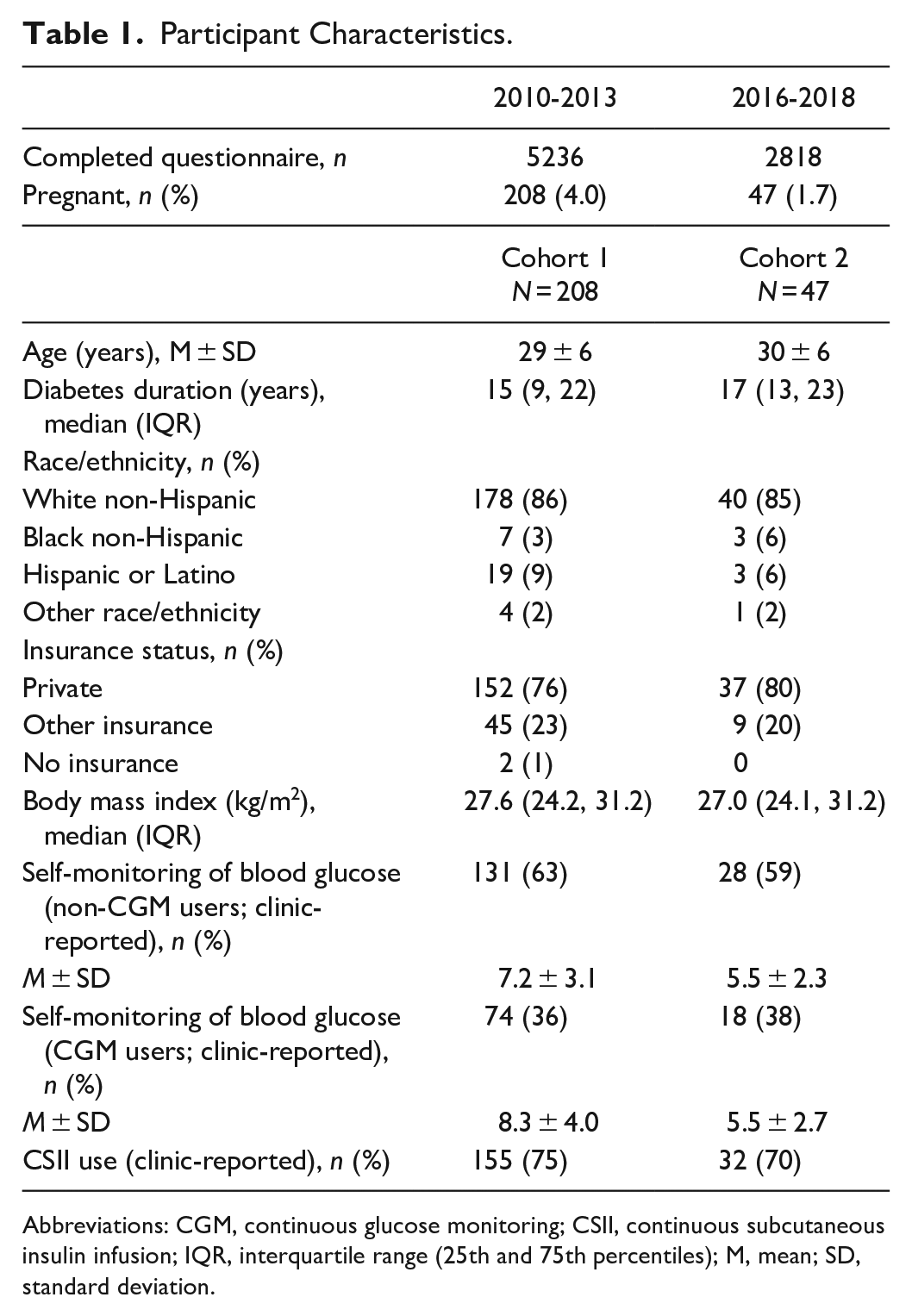
Abbreviations: CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; IQR, interquartile range (25th and 75th percentiles); M, mean; SD, standard deviation.
There was a trend for decrease in HbA1c in pregnant women between cohort 1 and cohort 2 (51 ± 10.9 mmol/mol [6.8% ± 1.2%] vs 48 ± 10.9 mmol/mol [6.5% ± 1.0%]; P = .07 adjusted for age, race/ethnicity, SMBG, and CGM use; Table 2). The proportions of participants achieving an HbA1c goal of ≤42 mmol/mol (≤6.0%) were 41 (28%) for cohort 1 and 12 (39%) for cohort 2 (adjusted P = .04), while those achieving the HbA1c goal ≤48 mmol/mol (≤6.5%) were 79 (54%) for cohort 1 and 17 (55%) for cohort 2 (adjusted P = .54). Participant-reported use of CGM while pregnant increased 1.9-fold between cohort 1 and cohort 2 (37% vs 70%, P = .02 adjusted for age, diabetes duration, race/ethnicity, insurance status, CSII use, and SMBG). Conversely, participant-reported use of CSII while pregnant decreased from 76% to 62% between cohort 1 and cohort 2 (P = .04 adjusted for age, diabetes duration, SMBG, and CGM use). There was no association between occurrence of SH and time cohort (P = .38 adjusted for duration of diabetes, race/ethnicity, SMBG, and insurance status).
Association Between Maternal Glycemic Outcomes and Time Cohort (2010-2013 vs 2016-2018) for Self-Reported Device Use.
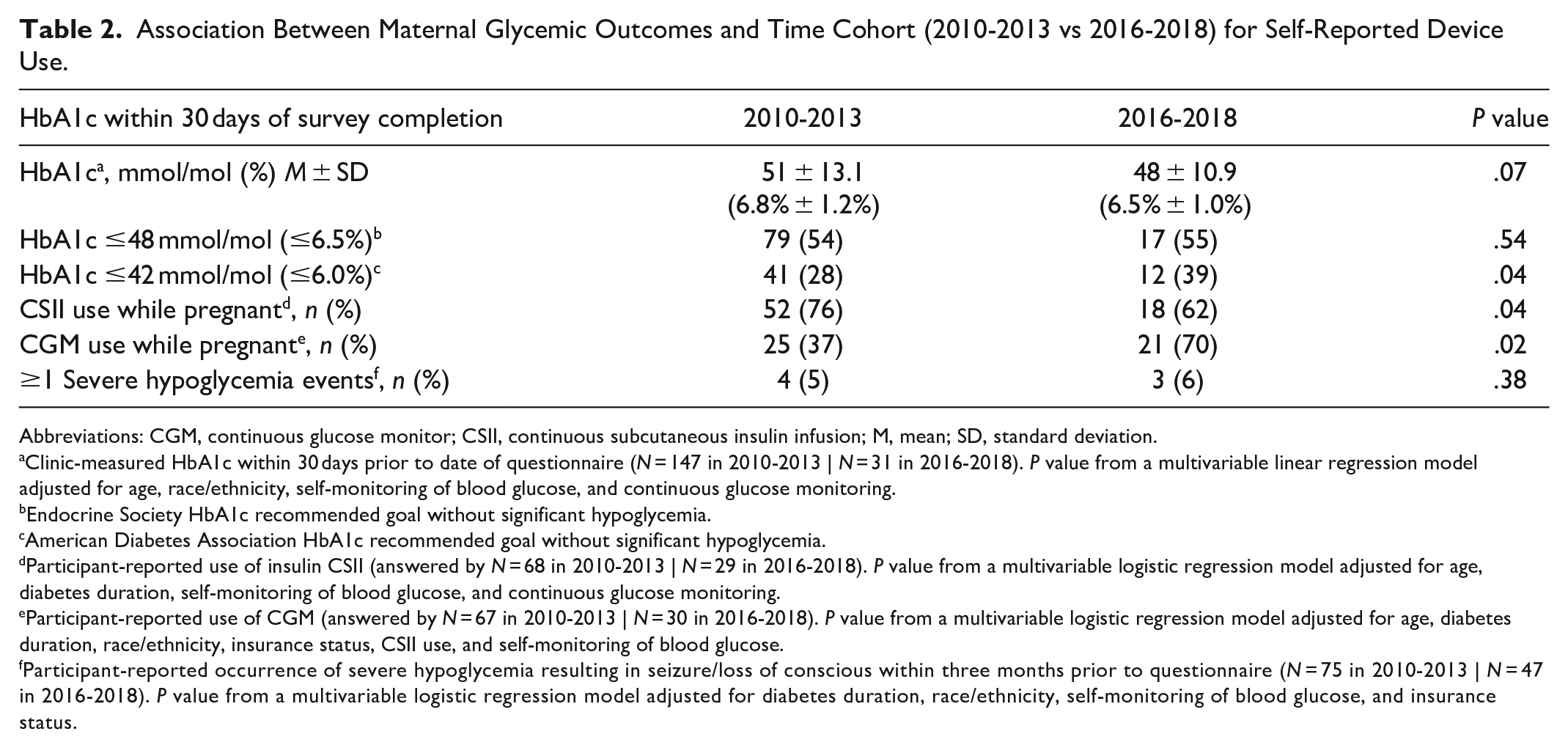
Abbreviations: CGM, continuous glucose monitor; CSII, continuous subcutaneous insulin infusion; M, mean; SD, standard deviation.
Clinic-measured HbA1c within 30 days prior to date of questionnaire (N = 147 in 2010-2013 | N = 31 in 2016-2018). P value from a multivariable linear regression model adjusted for age, race/ethnicity, self-monitoring of blood glucose, and continuous glucose monitoring.
Endocrine Society HbA1c recommended goal without significant hypoglycemia.
American Diabetes Association HbA1c recommended goal without significant hypoglycemia.
Participant-reported use of insulin CSII (answered by N = 68 in 2010-2013 | N = 29 in 2016-2018). P value from a multivariable logistic regression model adjusted for age, diabetes duration, self-monitoring of blood glucose, and continuous glucose monitoring.
Participant-reported use of CGM (answered by N = 67 in 2010-2013 | N = 30 in 2016-2018). P value from a multivariable logistic regression model adjusted for age, diabetes duration, race/ethnicity, insurance status, CSII use, and self-monitoring of blood glucose.
Participant-reported occurrence of severe hypoglycemia resulting in seizure/loss of conscious within three months prior to questionnaire (N = 75 in 2010-2013 | N = 47 in 2016-2018). P value from a multivariable logistic regression model adjusted for diabetes duration, race/ethnicity, self-monitoring of blood glucose, and insurance status.
Discussion
With the strict glycemic targets recommended during pregnancy, knowing how women with type 1 diabetes in the real world (outside the confines of strict clinical research protocols) leverage advanced technologies to manage their diabetes is important to understand. Our findings provide insights regarding the use of CSII and CGM therapies over time for pregnant women with type 1 diabetes in the United States. While there was a trend for decreased use of CSII during pregnancy over the two time periods studied, this mirrors stable penetrance of pump therapy found in the larger T1D Exchange clinic registry, which equates to roughly 60% of participants at both initiation of the registry and the 5-year follow up data. 10 Conversely, use of CGM was increased between cohort 1 and cohort 2 in the present analysis. Indeed, reports of CGM use in these pregnant cohorts exceeds that described in the larger registry data set, where CGM use was reported in 7% of the participants in 2010-2012 and 30% in the 2016-2018 data. 10 Though the etiology of these changes is unclear, the trend in improvement in HbA1c could be related to increased CGM incorporation into diabetes management, as well as other concurrent factors. Polsky et al 8 reported in 2018 on data from the T1D Exchange clinic registry, comparing the use of both CSII and CGM therapies in women who were recently pregnant (defined as women pregnant between 2010 and 2013) with those who had ever been pregnant (defined as a history of previous pregnancy or miscarriage, but not pregnant at the time of enrollment) and with women who had never been pregnant. In that analysis, frequency of diabetes technology use increased in recently pregnant women, both when examining use of CSII as well as frequency of CGM utilization. CSII and CGM use during pregnancy were both independently higher in recently pregnant women compared with ever pregnant women in the 2010-2013 cohort. Our previous data analyzed use of technologies prior to 2013 and showed that those who were recently pregnant had the highest rates of use of both technologies compared to ever- and never-pregnant women. Interestingly, our present data examining pregnant women in cohort 2 do not show a continued increase in pump wear during pregnancy over time. The reasons for the apparent decreased CSII use for cohort 2 in our current findings are unclear, especially given that CSII use outside of pregnancy has been shown to lower HbA1c results as well as decreased hypoglycemia compared to treatment with multiple daily injections (MDIs). 11 The physiologic changes throughout pregnancy leading to changes in insulin sensitivity12,13 would suggest that CSII use would be advantageous for pregnant women with type 1 diabetes. 14
Much of the literature regarding the benefits of CSII therapy compared with MDI therapy in a pregnancy has been largely reliant on retrospective observational case series with baseline differences between CSII and MDI users. 14 This type of analysis can lead to a risk of bias when making conclusions. Earlier publications reported results from small, randomized studies of varying quality using outdated insulin pumps from which only limited conclusions can be drawn. 15 Women using CSII in these studies typically tended to be older, have a longer duration of diabetes, and have a higher frequency of receiving preconception counseling than women on MDI, making comparisons between the groups challenging.3-6 More recent trials have shown conflicting reports regarding benefits of CSII therapy during pregnancy. One of the largest cohort studies of pregnant women with diabetes was performed by Kallas-Koeman et al in Western Canada. In that retrospective analysis, 133 patients using CSII as their method of insulin delivery were compared to 218 patients treated with MDI therapy and there were significantly lower HbA1c levels throughout pregnancy for those using CSII therapy. Yet, rates of macrosomia were also higher in the infants of the women on CSII. 16 Abell et al reported data from Australia reviewing 40 pregnancies managed with CSII compared with 127 pregnancies managed with MDI, where the two groups achieved similar glycemic control and no differences in pregnancy outcomes were demonstrated. 5 Most recently, a secondary analysis of the Continuous Glucose Monitoring in Pregnant Women with Type 1 Diabetes Trial (CONCEPTT) evaluated patient outcomes based on insulin delivery modality, where choice of therapy was made by both patients and their providers rather than being determined by the protocol. Feig reported lower HbA1cs for MDI users at 34 weeks’ gestation (48 ± 7.5 mmol/mol [6.54% ± 0.69%] for CSII vs 46 ± 6.3 mmol/mol [6.37% ± 0.58%] for MDI adjusted P = .001), but increased hypoglycemia (defined as time spent <3.5 mmol/L [<63 mg/dL] on CGM). Newborns of CSII users were more likely to have hypoglycemia at delivery as well as admissions of greater than 24 hours to the neonatal intensive care unit (NICU). 6 However, details about the appropriate use of the insulin pumps, pump education, and methods of utilizing or titrating pump or MDI therapy were not available in CONCEPTT, limiting the ability to properly evaluate comparisons between the MDI and CSII users.6,17 Hauffe et al recently published data for an observational retrospective cohort of 399 pregnant women with type 1 diabetes who delivered between January 2010 and May 2017 after using either MDI or CSII therapies for diabetes management while receiving care in three tertiary Berlin centers specializing in pregnancies complicated by diabetes. Both groups had similar HbA1c results throughout pregnancy; however, CSII users had increased maternal weight gain and an increased number of large-for-gestational age babies (birth weight >90% percentile for gestational age) 18 compared with those on MDI. The authors attributed this difference in offspring size to maternal weight gain since HbA1c levels were similar in both groups. 19 However, a greater number of patients who smoked were in the MDI group vs CSII subjects (P = .01), which may have also contributed to these findings.20,21 With conflicting results complicated by possible maternal factors, prospective, randomized trials with newer insulin pump models are needed to fully elucidate the benefits and drawbacks of CSII use in pregnant women with type 1 diabetes.
For over a decade, the evidence regarding the clinical benefits of CGM therapy has been clear for those with type 1 diabetes. 22 Our data show a trend toward increased CGM uptake in this US population of pregnant women with type 1 diabetes, at a rate that is exponentially greater than what has been seen in the larger T1D Exchange clinic registry. Earlier publications revealed conflicting data for CGM benefits during pregnancy,2,23-25 with discordant results emerging from more recent trials, the GLUCOMOMS and CONCEPTT trials. In the GLUCOMOMS trial conducted in the Netherlands, women with type 1 diabetes, type 2 diabetes, or gestational diabetes requiring insulin were randomized to intermittent CGM for five-seven days every six weeks versus standard of care. With over 300 participants at 20 clinical sites who were equally distributed amongst the three forms of diabetes studied, the authors found no significant difference in macrosomia or HbA1C between the two groups and concluded that use of intermittent retrospective CGM as a treatment strategy did not translate into improved pregnancy outcomes. 25 The CONCEPTT trial, the most recent, large randomized prospective study evaluating real-time CGM used continuously, reported improved fetal outcomes as well as modest improvements in HbA1C for CGM users with T1D when compared with women not using CGM in pregnancy. 2 Importantly, the differences in maternal glycemic control and gestational outcomes between these trials may be a result of the frequency of CGM use (intermittent vs constant).
Though we did not inquire about the reasons for CGM use, it is possible that the increased uptake of CGM in our cohort is due to increased accuracy, longer device wearability, increased patient confidence, changes in insurance coverage for CGM use overall, and/or increased health care provider confidence regarding the benefits of this technology. With the findings of the CONCEPTT trial, currently available CGM with longer duration of wear, non-adjunctive indications (though these are not currently approved for pregnancy), and factory calibrations of present-day CGMs, we anticipate increased adoption rates of CGM use for women with type 1 diabetes during pregnancy.
Although our data describe trends in technology use and glycemic control, caution should be used in interpreting these results. Since our report is observational in nature, we are unable to confirm that the trend toward lower HbA1c levels can be attributed to increased CGM use. Furthermore, we relied on self-reported data on the use of these technologies during pregnancy for which some questions were left unanswered by a subset of the subjects and several subjects did not have HbA1c levels available within 30 days of survey completion. Future studies of technology use in the real-world, clinical-care setting of pregnant women with type 1 diabetes could leverage data downloads to verify device use and the frequency with which the technology is employed. Finally, with <2% of eligible participants reporting pregnancies in the second cohort of our study, the small sample size may limit our findings. Yet, inclusion of pregnant women from a number of different clinical centers with data collected in a structured manner is a strength of the study. Additionally, development and implementation of surveys to gather data on device use in pregnancy at the registry onset with repeat data collection between 2016 and 2018 allowed us the opportunity to explore changes in device use between these two time periods.
Technology use in the treatment of type 1 diabetes in pregnancy including CGM, CSII, and potentially the use of hybrid closed loop systems may help the management of this challenging condition.26,27 The potential impact of advanced diabetes technology use by pregnant women with type 1 diabetes remains unclear. Further research is needed to comprehensively evaluate the benefits and barriers to adoption of these technologies as well as ways to implement the most effective methods for pregnant women with type 1 diabetes to use these devices to help them achieve the best outcomes during pregnancy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported through the Leona M. and Harry B. Helmsley Charitable Trust
