Abstract
Automated insulin delivery (AID) systems mimic an artificial pancreas via a predictive algorithm integrated with continuous glucose monitoring (CGM) and an insulin pump, thereby providing AID. Outside of pregnancy, AID has led to a paradigm shift in the management of people with type 1 diabetes (T1D), leading to improvements in glycemic control with lower risk for hypoglycemia and improved quality of life. As the use of AID in clinical practice is increasing, the number of women of reproductive age becoming pregnant while using AID is also expected to increase. The requirement for lower glucose targets than outside of pregnancy and for frequent adjustments of insulin doses during pregnancy may impact the effectiveness and safety of AID when using algorithms for non-pregnant populations with T1D. Currently, the CamAPS® FX is the only AID approved for use in pregnancy. A recent randomized controlled trial (RCT) with CamAPS® FX demonstrated a 10% increase in time in range in a pregnant population with T1D and a baseline glycated hemoglobin (HbA1c) ≥ 48 mmol/mol (6.5%). Off-label use of AID not approved for pregnancy are currently also being evaluated in ongoing RCTs. More evidence is needed on the impact of AID on maternal and neonatal outcomes. We review the current evidence on the use of AID in pregnancy and provide an overview of the completed and ongoing RCTs evaluating AID in pregnancy. In addition, we discuss the advantages and challenges of the use of current AID in pregnancy and future directions for research.
Introduction
Pregnant women with type 1 diabetes (T1D) have an increased risk for maternal and neonatal complications, such as congenital malformations, preeclampsia, preterm delivery, and perinatal mortality as well as the need for prolonged hospital admissions after delivery and need for neonatal intensive care compared with the background population. 1 Several population-based cohort studies have shown that the use of technical devices, such as continuous glucose monitoring (CGM) systems and insulin pumps have not significantly improved pregnancy outcomes over time.2,3 It is often challenging for women with T1D to achieve and maintain strict glycemic control throughout pregnancy due to the metabolic changes, such as the increased risk of hypoglycemia in early pregnancy,4,5 and the increasing insulin resistance with delayed insulin absorption later in pregnancy. 6
Continuous glucose monitoring is increasingly being used in women with T1D, especially following the results of the “Continuous Glucose Monitoring in Pregnant Women with Type 1 Diabetes” (CONCEPTT) trial, 7 and CGM is routinely offered during pregnancy in some countries, including the United Kingdom, Denmark, Belgium, and Sweden.8-14 International consensus on CGM targets recommends to spend > 70% of time in range in pregnancy (TIRp: 3.5-7.8 mmol/L or 63-140 mg/dL) and < 4% of time below range in pregnancy (TBRp: < 3.5 mmol/L or 63 mg/dL). 15 However, pregnant women with T1D often do not achieve TIRp > 70% until the final weeks of pregnancy, if at all.8,9,16-18
Automated insulin delivery (AID) systems mimic an artificial pancreas via a predictive algorithm that can be hosted on a smartphone that integrates CGM and insulin pump data and thereby provide automated glucose-responsive insulin delivery between meals and overnight with manually triggered premeal doses. Some AID also provide automated hyperglycemia correction boluses.19,20 Outside of pregnancy, the use of AID has led to a paradigm shift in the management of T1D, with a 10% higher time in range (TIR; 3.9-10.0 mmol/L or 70-180 mg/dL), with low hypoglycemia risk and improved quality of life compared with stand-alone insulin pumps.21-23 As the use of AID in clinical practice is increasing, the number of women of reproductive age becoming pregnant while using AID is also expected to increase. The requirement for a lower glucose target than outside of pregnancy and the need for frequent adjustments of insulin doses in women with T1D during pregnancy may impact the effectiveness and safety of AID when using algorithms for non-pregnant populations. 24 Clinicians and pregnant women are therefore faced with the dilemma whether to continue with the AID in pregnancy or to switch back to stand-alone insulin pumps, sensor-augmented pump (SAP) therapy, or multiple daily injections (MDIs). However, women are often hesitant to cease the use of AID during pregnancy. 25 For this narrative review, we review the current evidence on the use of AID in pregnancy and provide an overview of the completed and ongoing randomized controlled trials (RCTs) evaluating AID in pregnancy. In addition, we discuss the advantages and challenges of current AID for use in pregnancy and future directions for research.
AID Approved for Use in Pregnancy
The first feasibility studies evaluating AID with an algorithm developed for use in T1D pregnancy were performed more than ten years ago, confirming their safety in the second and third trimesters in small studies (n ≤ 12) (Table 1).26,27
Results of the Smaller AID Studies in Pregnant Women With Type 1 Diabetes.
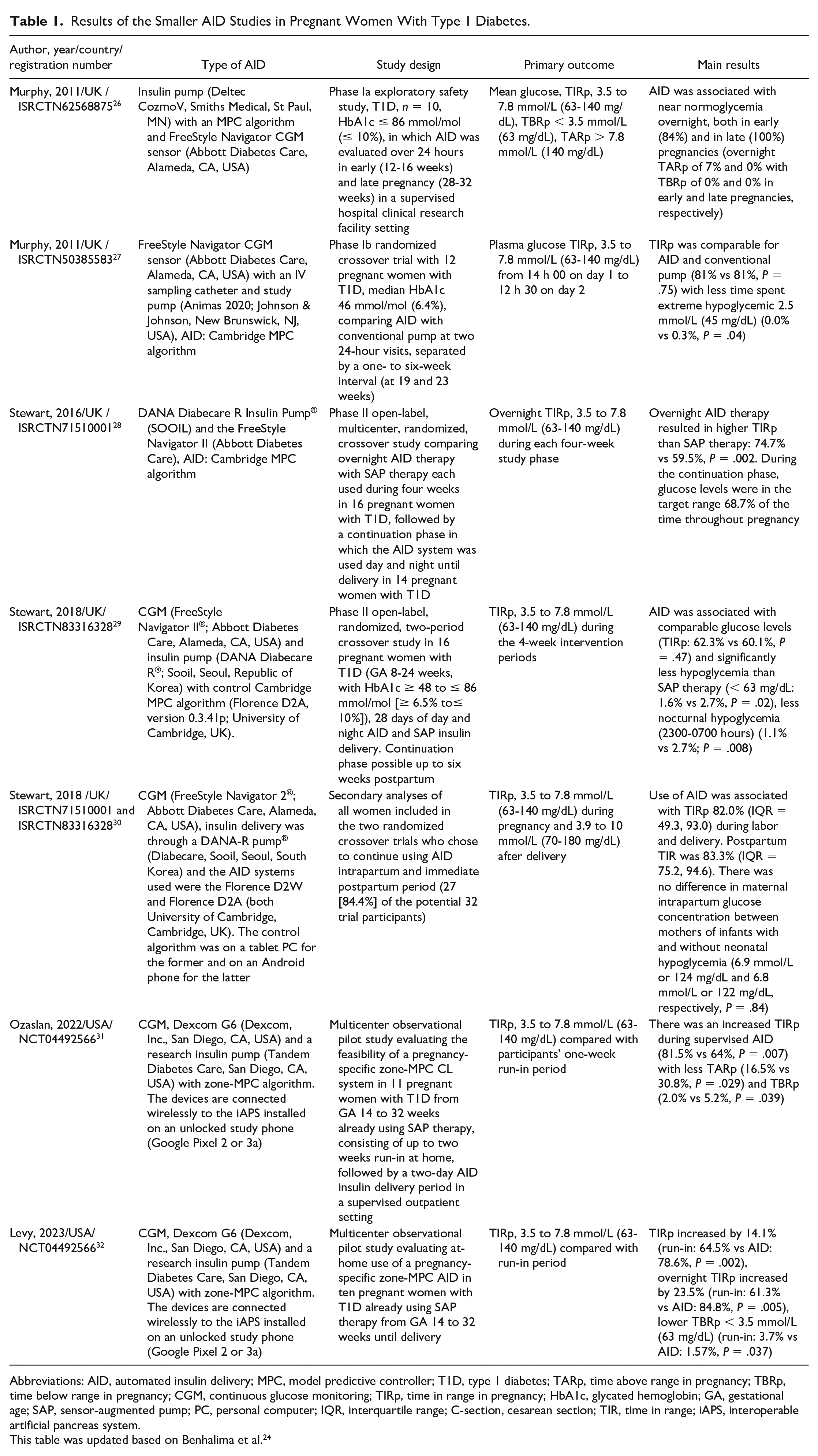
Abbreviations: AID, automated insulin delivery; MPC, model predictive controller; T1D, type 1 diabetes; TARp, time above range in pregnancy; TBRp, time below range in pregnancy; CGM, continuous glucose monitoring; TIRp, time in range in pregnancy; HbA1c, glycated hemoglobin; GA, gestational age; SAP, sensor-augmented pump; PC, personal computer; IQR, interquartile range; C-section, cesarean section; TIR, time in range; iAPS, interoperable artificial pancreas system.
This table was updated based on Benhalima et al. 24
Two crossover RCTs with this algorithm, confirmed proof of concept of the CamAPS® FX AID (CamDiab Ltd. Cambridge, UK) in pregnancy (Table 1) with improvement of TIRp overnight and less hypoglycemia when using the AID compared with SAP therapy. These RCTs had, however, only small sample sizes (16 women) and limited duration of AID use of four weeks.28,29 A secondary analysis of these two crossover RCTs (including the continuation phase) reported a mean TIR of 82.0% during delivery and 83.3% postpartum. 30 The AID performed well, both in vaginal and cesarean section deliveries without hypoglycemia. 30 In general, women expressed satisfaction with improved glucose control and increased peace of mind, and they also reported concerns about CGM accuracy and burden of maintenance requirements. 33 Most women became more positive with the technology throughout pregnancy. 34 However, the insulin pump used in these crossover RCTs was less sophisticated than the current commercially available insulin pumps and the algorithm was housed on a tablet, which might have led to more device burden. 24
Based on these pilot RCTs,28,29 the CamAPS® FX AID has been approved for use in pregnancy in the United Kingdom, Europe, and Australia, and to date, this is the only AID approved for use in pregnancy (Table 2). The CamAPS® has been available since 2020, and is the first interoperable AID android app. It is compatible with different insulin pumps: YpsoPump, with mylife loop (Ypsomed AG, Burgdorf, Switzerland), DANA Diabecare RS and DANA-IIs insulin pumps (SOOIL Development Gyeonggi-do, Korea) with the Dexcom G6/G7 (Dexcom, San Diego, CA, USA) and Free Style Libre 3 sensors (Abbott Inc., Abbott Park, IL, USA).35,36 Both rapid and ultra-rapid insulin analogues can be used. 37
Overview of the Different Commercially Available AID Systems.
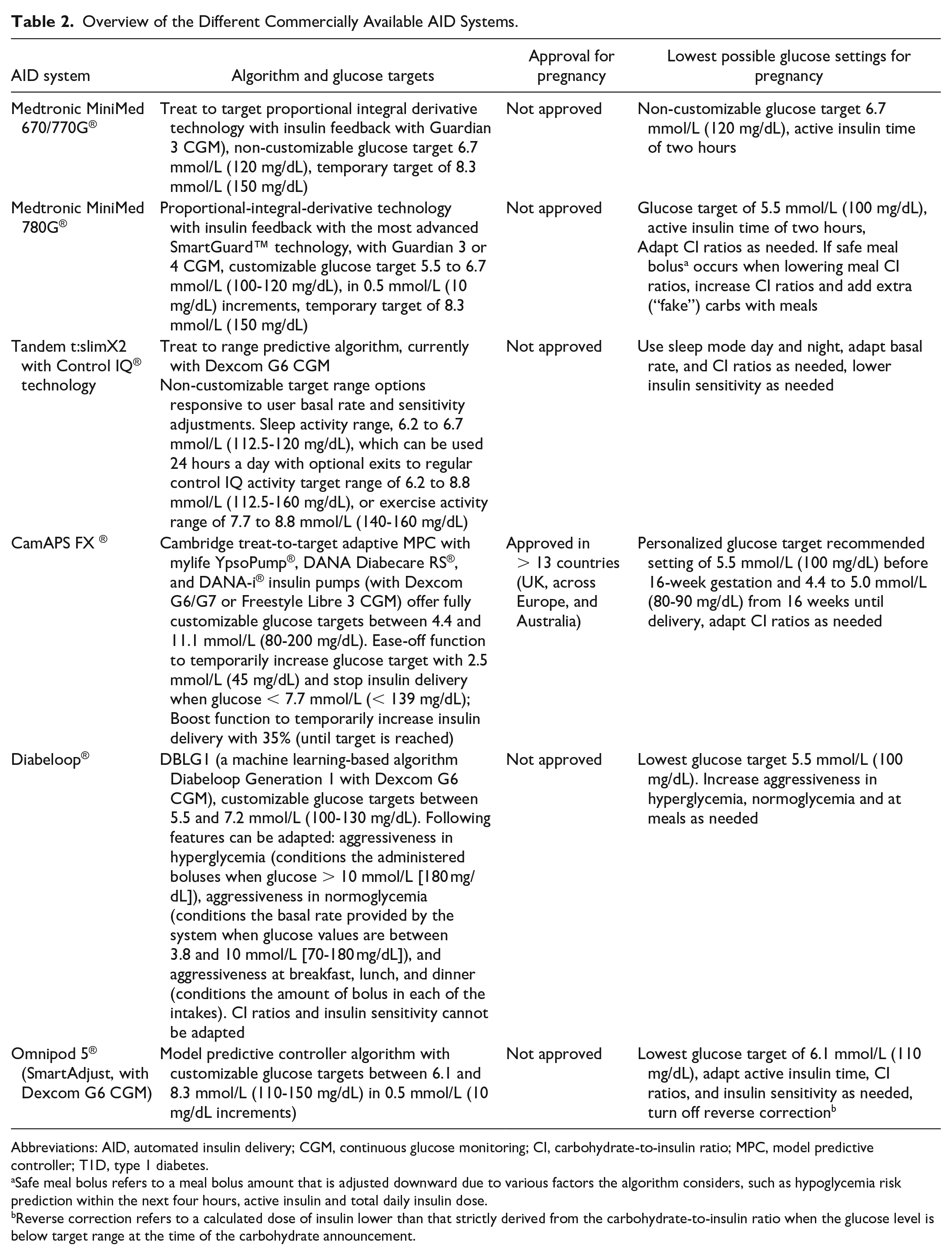
Abbreviations: AID, automated insulin delivery; CGM, continuous glucose monitoring; CI, carbohydrate-to-insulin ratio; MPC, model predictive controller; T1D, type 1 diabetes.
Safe meal bolus refers to a meal bolus amount that is adjusted downward due to various factors the algorithm considers, such as hypoglycemia risk prediction within the next four hours, active insulin and total daily insulin dose.
Reverse correction refers to a calculated dose of insulin lower than that strictly derived from the carbohydrate-to-insulin ratio when the glucose level is below target range at the time of the carbohydrate announcement.
The CamAPS® FX AID contains a treat-to-target adaptive model predictive controller (MPC) algorithm. The algorithm is initialized using total daily insulin dose and bodyweight. Insulin sensitivity and active insulin time are automatically calculated and adjusted over time by the algorithm. Adaptive learning is incorporated with regards to total daily insulin requirements, diurnal variations, and meal patterns. It also contains an optional exercise mode (Ease-off function), which temporarily raises the glucose target and suspends algorithm-directed insulin delivery if sensor glucose is lower than 7·0 mmol/L or 140 mg/dL. In addition, this AID contains a Boost function to intensify algorithm-driven insulin delivery by approximately 35% when glucose is above target level. 38
Based on the experiences with the pilot RCTs,28,29 this AID was updated since 2018. Glucose measurements from CGM can now be used to inform user-initiated premeal boluses. 39 In addition, the glucose target can be intensified or relaxed. In contrast to other available commercial AID, the CamAPS® FX offers fully customizable glucose targets between 4.4 mmol/L or 80 mg/dL and 11.1 mmol/L or 200 mg/dL. During pregnancy, targets are typically set at 5.5 mmol/L or 100 mg/dL during the first trimester and 4.4 to 5.0 mmol/L or 80 to 90 mg/dL thereafter (Table 2). 39
Other Pilot Studies in Pregnancy
A consortium from the United States designed a zone-MPC algorithm specifically customized for use in pregnancy, allowing for lower glycemic targets during the day (4.4-6.0 mmol/L or 80-110 mg/dL) and during the night (4.4-5.5 mmol/L or 80-100 mg/dL) 40 compared with most commercially available AID. The zone-MPC algorithm runs on an interoperable artificial pancreas system, which consists of a CGM, an insulin pump (Tandem research pump), and an unlocked smartphone. 41 The optimal insulin delivery plan is updated every five minutes based on the glucose data from the CGM. Meals need to be announced to the system by the user, and meal boluses are calculated based on the participants’ prescribed bolus settings (ie, carbohydrate ratio and insulin sensitivity factor) and user-estimated carbohydrate intake. Two small observational studies in women using this algorithm showed a significant increase in TIRp of 14% to 17% (Table 1).31,32 The question remains whether AID with a customized pregnancy algorithm should preferably be used, as illustrated by the promising results of these small pilot studies.31,32 Since this algorithm allows for lower glycemic targets compared with most other commercially available AID, this might be particularly useful for the stricter glycemic targets needed during pregnancy. However, larger and longer-duration trials are warranted.
Off-Label Use During Pregnancy
The MiniMed 670G® and the next version 770G® (Medtronic Inc., Northridge, CA, USA) were the first commercially available AID. The limitation is that their algorithms target an average glucose of 6.7 mmol/L or 120 mg/dL, which is not customizable to lower glucose levels and is higher than the recommended target for use in pregnancy 42 (Table 2). A few case reports and case series reported improvement in TIR with no pregnancy complications nor other safety issues, in pregnant women with T1D switching to this AID during pregnancy (often starting in auto mode later on in pregnancy).43-45 Although glycemic control improved, the TIRp was often not reached.43-45
Several next generation AID, such as the MiniMed 780G®, Tandem t:slim X2 Control IQ®, Diabeloop® and Omnipod 5® systems received approval for use in non-pregnant people in 2020, but are not approved for use in pregnancy.46-48 These AIDs allow for a lower glucose target, which might potentially lead to improved glycemia compared with the MiniMed 670G/770G® systems (Table 2). The MiniMed 780G® uses an algorithm that automatically adapts the basal rate and also provides automated hyperglycemia correction boluses. This type of AID might be particularly useful for women who prefer an AID without the need for much intervention. In contrast, with the Tandem t:slim X2 with control IQ®, the basal rate and insulin sensitivity can be adapted, which might be a good option for women who prefer an AID that is less automated and allows for more personal intervention. 24 A case report was presented on the use of the Diabeloop® (Grenoble, France) in a pregnant T1D patient with highly unstable glycemic control and severe hypoglycemia unawareness. 49 Both daytime and nighttime periods in target range were steadily improved, with a mean TIRp of 82.1% for the daytime and 77.1% for the nighttime from 13 weeks until delivery. The Omnipod 5® is the first patch pump that is available as an AID. However, there are currently no data on the use of the Omnipod 5® in pregnancy. Recent case series on the use of the MiniMed 780G® and Tandem t:slim X2 control IQ® in pregnancy show, respectively, that women spent more time in range and had lower glucose variability compared with their previous pregnancy using SAP therapy, 50 and that control IQ technology could be safely used in pregnancy with increased TIRp and less diabetes management burden. 51 In contrast, a recent report of six pregnant women with a baseline HbA1c of 6.6% using the MiniMed 780G® throughout pregnancy, indicated challenges in achieving the desired glycemic targets for pregnancy in most women. 52 Differences between these case series, could be related to differences in baseline characteristics, such as glycemic control at the off-set of pregnancy. It could be expected that AID can more easily increase time spent in target glucose range in women starting with a higher baseline HbA1c in early pregnancy, compared with women with already tight glycemic control on SAP therapy. Data from RCTs are therefore needed to evaluate whether AID can provide additional benefit for glycemic control compared with SAP therapy.
Data on do-it-yourself artificial pancreas systems are limited to case reports in selected women with T1D and strong diabetes self-management skills, showing that these systems might be used during pregnancy with improvements in quality of life.53-56 Insulin pump settings should be adapted similarly to that of conventional insulin pumps to ensure safety in case of loss of AID. However, since these systems are unregulated with no formal technical support, their use is not recommended during pregnancy.
RCTs on AID During Pregnancy or Postpartum
Three open-label RCTs have recently been completed39,57,58 and one is still ongoing, 59 to determine the clinical efficacy and safety of AID in T1D pregnancy. These studies are powered for maternal glycemic outcomes, with the primary outcome TIRp of 3.5 to 7.8 mmol/L or 63 to 140 mg/dL. Secondary outcomes include several pregnancy and neonatal outcomes, adverse events (including severe hypoglycemia and diabetic ketoacidosis), and patient-reported outcomes (including treatment satisfaction and quality of life). 24 In addition, one small RCT evaluates AID in early postpartum 60 (Table 3).
The Larger (Ongoing) AID Studies in Pregnant Women With Type 1 Diabetes.
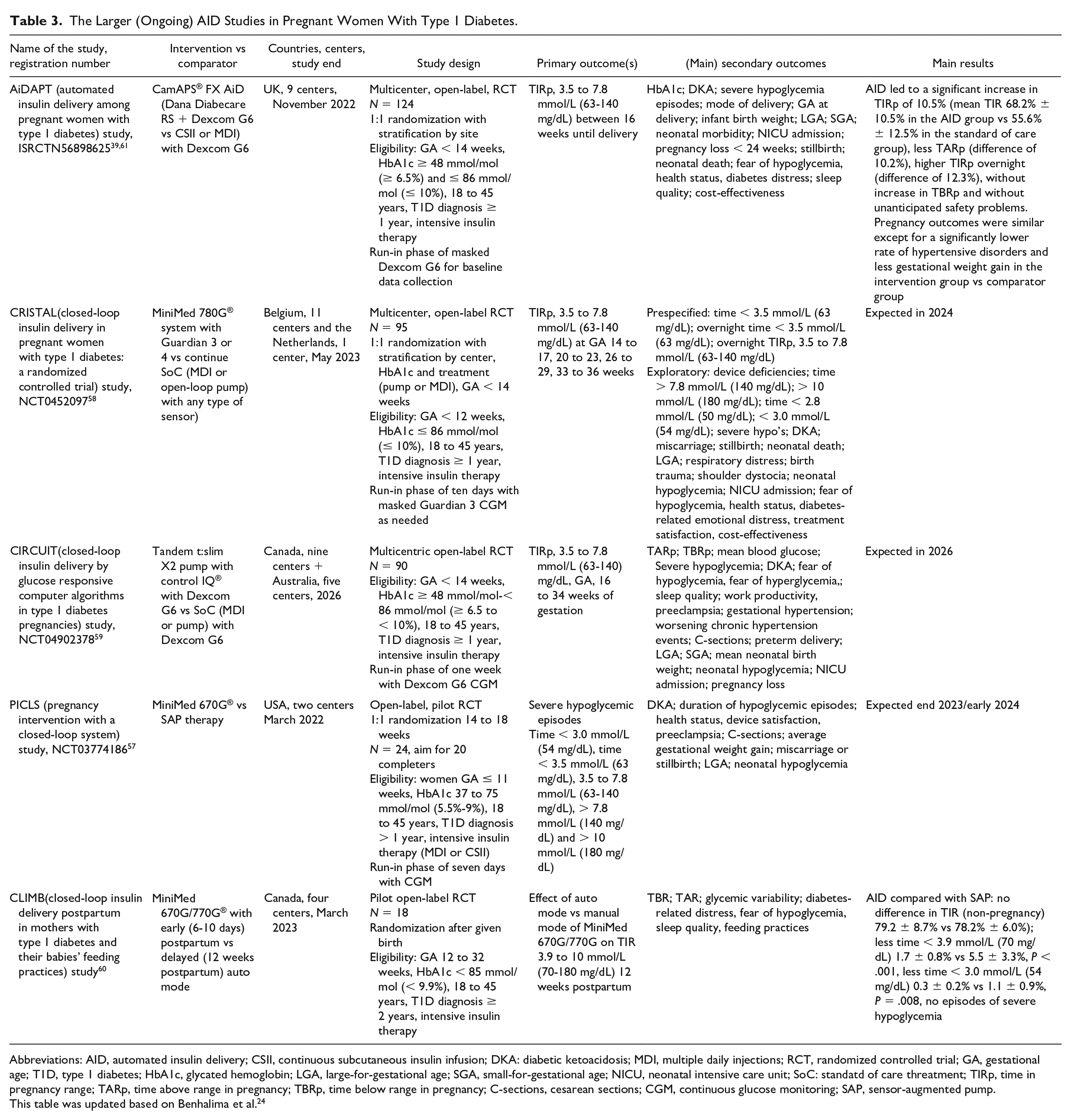
Abbreviations: AID, automated insulin delivery; CSII, continuous subcutaneous insulin infusion; DKA: diabetic ketoacidosis; MDI, multiple daily injections; RCT, randomized controlled trial; GA, gestational age; T1D, type 1 diabetes; HbA1c, glycated hemoglobin; LGA, large-for-gestational age; SGA, small-for-gestational age; NICU, neonatal intensive care unit; SoC: standatd of care threatment; TIRp, time in pregnancy range; TARp, time above range in pregnancy; TBRp, time below range in pregnancy; C-sections, cesarean sections; CGM, continuous glucose monitoring; SAP, sensor-augmented pump.
This table was updated based on Benhalima et al. 24
The AiDAPT trial investigated the updated CamAPS® with Dexcom G6 compared with standard of care in 124 women with an HbA1c between 48 and 86 mmol/mol (6.5%-10%). 61 The AID led to a significant increase in TIRp of 10.5% without increased TBRp. 39 Pregnancy outcomes were similar except for a significantly lower rate of hypertensive disorders and less gestational weight gain in the AID group. 39 The study was not powered for pregnancy outcomes and women were randomized at about 11 weeks, thus missing the crucial period of organogenesis. Interviews with women using the AID in pregnancy, highlight that it can facilitate positive pregnancy experiences, with less fear of complications and better sleep quality. Women also indicated the need for intensive support during early pregnancy to develop confidence in the AID technology. 62
The CRISTAL study, including 95 women, compared the MiniMed 780G® system and Guardian 3 or 4 sensor with routine care. The study allows for the use of all types of CGM in line with routine care and with no lower HbA1c limit for inclusion, thus enabling the evaluation of women with tight glycemic control in early pregnancy. The study is completed and results are expected in 2024. 58 The CIRCUIT study, including 90 women, compares the Tandem t:slim X2 pump with control IQ® and Dexcom G6 vs routine care with Dexcom G6 sensor 59 (Table 3). Results are expected in 2026.
The PICLS (pregnancy intervention with a closed-loop system) study is a pilot RCT on 24 women with T1D investigating the MiniMed 670G® compared with SAP therapy in pregnancy. 57 The study was completed in 2023 but is not yet published.
The CLIMB (closed-loop insulin delivery postpartum in mothers with type 1 diabetes and their babies’ feeding practices) study was a pilot RCT followed by an observational cohort study in 18 women, of the MiniMed 670G/770G® system with early (6-10 days postpartum) versus delayed (12 weeks postpartum) use of the auto mode. 60 Participants in the AID group had less hypoglycemia compared with the SAP group (Table 3). 60 These findings are reassuring for the use of AID postpartum. However, the AID was used after the first week postpartum. Studies exploring the safety of AID in the first week after delivery are needed.
The RCTs on AID in pregnant women with T1D will report on whether AID can improve glycemic management. Several studies will also investigate the cost-effectiveness of AID in women with T1D during pregnancy.58,61
Advantages and Challenges of Current AID in Pregnancy
Although many AID are not specifically designed for use in pregnancy, these systems may also offer several advantages, such as less glycemic variability, better TIRp overnight, less risk for hypoglycemia, and improved treatment satisfaction. 24 Due to the magnitude in increase in TIRp, it can be anticipated that if the CamAPS® AID is started at preconception or very early in pregnancy, this will probably also lead to reduced rates of other complications, such as congenital anomalies and large-for-gestational age (LGA) infants in women with higher baseline HbA1c.
The challenges of most commercially available AID are that the glucose target cannot be lowered < 5.5 mmol/L or 100 mg/dL (Table 2). The glycemic target might therefore not be strict enough for pregnancy (Figure 1). In addition, the algorithms designed for non-pregnant populations, might not be sufficiently flexible to adapt fast enough to the increased insulin need later in pregnancy. When using AID off-label in pregnancy, it is important to provide counseling on the potential risks and benefits to women, and offer an assisted AID approach. 25 The lowest target glucose available for the AID should be used (Table 2). Until now, studies have introduced women to AID in early pregnancy 39,57-59 or postpartum, 60 and none initiated AID before pregnancy. Some women may be reluctant to initiate new diabetes technology in pregnancy, suggesting that study participants might be highly selected.

Challenges and advantages of available automated insulin delivery (AID) systems for use in pregnancy. This Figure was updated based on Benhalima et al. 24 RCT, randomized controlled trial.
To facilitate the real-world adoption of AID in pregnant women with T1D, presenting AID to pregnant women (or preferably before pregnancy) and healthcare professionals as a pillar of a three-party collaboration may help to promote optimal use. 63 It is also important to develop mentorship services to upskill and support inexperienced staff attending pregnant women using AID. 64 However, for broad implementation of AID in pregnancy, improved equity in access to AID worldwide is needed. Based on the AiDAPT trial, the “National Institute for Health and Care Excellence” guidance (under review) recommends that AID should be offered to all pregnant persons with T1D. 39
Future Research
To have strong evidence on the impact of AID on pregnancy outcomes, a large RCT would be needed powered for maternal and neonatal outcomes with initiation of the AID before pregnancy throughout pregnancy and delivery, and with follow-up postpartum to also evaluate the impact of breastfeeding. However, it would be very challenging to perform such a large study as the number of women of reproductive age already using AID before pregnancy is increasing and due to the advantages associated with AID, this will potentially limit the willingness for recruitment.
As none of the current RCTs are powered for pregnancy outcomes (Table 3), a meta-analysis is needed to evaluate the impact of AID use on obstetric and neonatal outcomes. In addition, this meta-analysis should also include participant-reported outcomes to determine the psychosocial impact of AID use in T1D during pregnancy. 24 However, existing standardized questionnaires do not adequately capture important participant-reported outcomes, such as sleep quality, fear of hypoglycemia and hyperglycemia during pregnancy and fear of hypoglycemia postpartum. 65
More research is also needed to determine whether the current proposed consensus glucose targets for use in pregnancy are adequate or if the glucose targets should be adjusted as AID become more broadly available for use in T1D during pregnancy. 15 When using AID in pregnancy, TIRp might be more easily achieved, allowing for a lower upper target of 6.6 mmol/L or 120 mg/dL instead of 7.8 mmol/L or 140 mg/dL. Until now, there is no consensus on an appropriate target for mean glucose in pregnancy, even though this metric might help to further improve pregnancy outcomes as recent studies in normal glucose tolerant women have demonstrated that mean glycemia is around 5.4 mmol/L or 98 mg/dL throughout pregnancy.66,67
More research is also needed on the timing of the bolus when using AID in pregnancy. Due to the automatic adaptation of the basal rate, bolus should be given a minimum of 10 to 15 minutes before meals, and there may be less of a need to administer the bolus up to 30 to 45 minutes before meals in late pregnancy, as recommended when using MDI or insulin pumps. 6 The response should be individualized based on each woman and system used. 24
Conclusion
Glycemic management in T1D during pregnancy remains challenging with risk of pregnancy complications. The CamAPS® FX is the first AID approved for use in pregnancy, demonstrating a 10% increase in TIRp in a population with a baseline HbA1c ≥ 48 mmol/mol (6.5%). Off-label use of AID not approved for pregnancy, are currently also being evaluated in RCTs. More research is needed on the impact of AID on maternal and neonatal outcomes as well as during delivery and postpartum. This will help to guide clinicians and women with T1D on the use of AID during pregnancy.
Footnotes
Abbreviations
AID, automated insulin delivery; CL, closed-loop; MDI, multiple daily injections; T1D, type 1 diabetes; RCT, randomized controlled trial; SAP therapy, sensor-augmented pump therapy; MPC, model predictive controller; CGM, continuous glucose monitoring; HbA1c, glycated hemoglobin; TIRp, time in range in pregnancy; TBRp, time below range in pregnancy; TARp, time above range in pregnancy
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KB received an unrestricted grant and study devices from Medtronic for the investigator-initiated CRISTAL study. KB received study devices from Dexcom for the investigator-initiated GLORIA study. KB received study medication from Novo Nordisk A/S for the investigator-initiated SERENA study. KB received speaker fee from Novo Nordisk, AstraZeneca and Medtronic. KaatB has no disclosures. LR received a grant from Novo Nordisk A/S for an investigator sponsored study. Grant number: U1111-1209-6358 and participated in the European Association for the Study of Diabetes annual meeting 2023 as an invite by Novo Nordisk A/S. JJ has been a lecturer/member of the scientific advisory board in the following companies: Abbott, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Medtronic, Nordic InfuCare, NovoNordisk A/S, and Sanofi. KB received a fundamental clinical investigatorship from FWO Flanders (1800220N).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
