Abstract
Background:
Perioperative diabetes patients are often treated with sliding-scale insulin, despite a lack of evidence to support therapeutic effectiveness. We introduced an automated subcutaneous insulin algorithm (SQIA) to improve glycemic control in these patients while maintaining the simplicity of a q4 hour adjustable sliding-scale insulin order set.
Methods:
In this pilot study, we implemented a fully programmed, self-adjusting SQIA as part of a structured order set in the electronic medical record for adult patients who are nil per os, or on continuous enteral tube feedings or total parenteral nutrition. The nurse only enters the current glucose in the Medication Administration Record, and then the calculated dose is shown. The new dose is based on previous dose, and current and previous glucoses. The SQIA titrates the glucose to 120-180 mg/dL. For this pilot, this order set was utilized for complex perioperative oncologic patients.
Results:
The median duration on the SQIA was 58 hours. Glucoses at titration initiation were highest at 206 ± 63 mg/dL, and came down to 156 ± 29 mg/dL by 72 hours. The majority of measured glucoses (66.8%, n = 647) were maintained between 80 and 180 mg/dL. There were no glucoses lower than 60 mg/dL, and only 0.3% (n = 3) were below 70 mg/dL. There was a low rate of errors (1%).
Conclusions:
A simple automated SQIA can be used to titrate insulin to meet the changing metabolic requirements of individuals perioperatively and maintain glucose within the target range for these hospitalized patients.
Introduction
Hyperglycemia in the perioperative period has been associated with multiple adverse outcomes including infection, reoperative interventions, and death.1-4 The most compelling evidence that hyperglycemia worsens outcomes comes from cardiac surgery and critically ill patients admitted to surgical intensive care units (ICUs). In these populations, improved glycemic control reduces multiorgan failure, infections, and mortality.5-7 Limited data also suggest that improved glycemic control reduces perioperative complications in the general surgery population, 8 and as well as multiple other surgical populations.3,9,10
Despite these known complications, glucose-lowering therapy is often overlooked and inadequate. The reason for this is multifactorial, including fear of hypoglycemia, use of sliding-scale coverage only, and dynamic fluctuations in insulin requirements.1,11 General anesthesia and surgery trigger a neurohormonal stress response with release of inflammatory cytokines and counterregulatory hormones such as glucagon, cortisol, and catecholamines. Even in the absence of underlying diabetes, metabolic derangements as a result of this stress response can lead to hyperglycemia.12-16 Additional factors complicating glycemic control in postoperative patients are frequent interruptions in meal schedules, altered nutritional intake, emesis, and hyperalimentation.
There are many efforts being made to improve inpatient glucose control.17-23 Guidelines for achieving glycemic control in perioperative patients exist, but there remain a lack of details and accessibility to physicians who do not specialize in diabetes care. The American Diabetes Association recommends basal insulin, or basal plus correctional insulin for noncritically ill hospitalized patients who are restricted from oral intake. Patients receiving total parenteral nutrition (TPN) or enteral tube feedings (TFs) may receive short-acting nutritional insulin plus correction insulin. Achieving appropriate glucose control may be delayed as timely adjustments in insulin doses are rarely made to titrate to the needs of the individual patient. In addition, the insulin content of TPN can only practically be adjusted every few days and is fixed over the course of the intravenous (IV) bag.
One promising approach to improve glycemic control is integration of real-time glucose data with electronic prescribing to create insulin-treatment algorithms that titrate insulin doses based on glucose trends. IV insulin algorithms are routinely used in the ICU or perioperative setting, where nurses adjust insulin infusion rates every 1-2 hours based on glucose levels. Prior studies have shown that simple subcutaneous (SQ) insulin algorithms used in the ICU or perioperative setting can have adequate glycemic control as compared to the labor-intensive IV insulin algorithms in patients who are nil per os (NPO).24,25 These algorithms can be applied to other hospitalized populations, particularly in patients who are receiving calories in a controlled and continuous fashion, such as those who are NPO or receiving continuous enteral or parenteral nutrition.
In this pilot study, we implemented a fully programmed, self-adjusting SQ insulin algorithm (SQIA) as part of a structured order set in the electronic medical record (EMR) for adult patients who are NPO or on continuous TPN or TFs. Our goal was to determine if this approach can improve glycemic control in this population and reduce rates of hypoglycemia and potentially complications during hospital stays.
Methods
Subcutaneous Insulin Protocol Design and Implementation
This algorithm is a variation on a simple protocol that was first described by Pezzarossa et al in 1988, 24 and then modified as part of a Quality Improvement (QI) project at the University of California, San Francisco (UCSF) using a shorter-acting insulin analog, aspart (NovoLog, Novo Nordisk Pharmaceuticals Inc.). 25 Based on our experience with the algorithm during the QI project, a new multidisciplinary team composed of endocrinologists, pharmacists, nurses, and programmers was formed. The SQIA underwent further refinement and details are shown in Supplemental Figure S1. To simplify use of the SQIA, the algorithm was fully programmed into a calculator in the EMR (Epic Systems) and embedded in the Medication Administration Record (MAR). When a physician checks off the box in the order set for the SQIA to be used, they are only required to enter an initial starting insulin dose. There are suggestions given based on the patient’s previous insulin dosing. When it is time for insulin to be given, the nurse does not see the algorithm but simply goes to the MAR, enters the current glucose level, and then the new calculated insulin dose is shown (Figure 1). If the nurse or provider wants to review the algorithm, it is available via a hyperlink within the MAR. As detailed in Supplemental Table S1, the new dose is based on the previous dose, and current and previous glucose levels. The SQIA is designed to titrate the glucose to 120-180 mg/dL for patients who are NPO, or on TPN or TFs. No new orders are required even if insulin is added to TPN, or changes made in TF rates. In the event of a significant change in the patient’s status, for example a significant addition of insulin to the TPN, the physician has the option to go to the active orders and modify the next insulin dose. The subsequent insulin dose will be based on this new entry. If new diet orders were placed, the nurses’ MAR indicates that the ordering physician needs to be contacted to place new, appropriate insulin orders. If enteral feedings are interrupted, there are standing orders to initiate an IV glucose infusion. Prior to implementation, extensive real-time testing with mock patients was performed to assure that the calculator produced the correct new insulin dose.
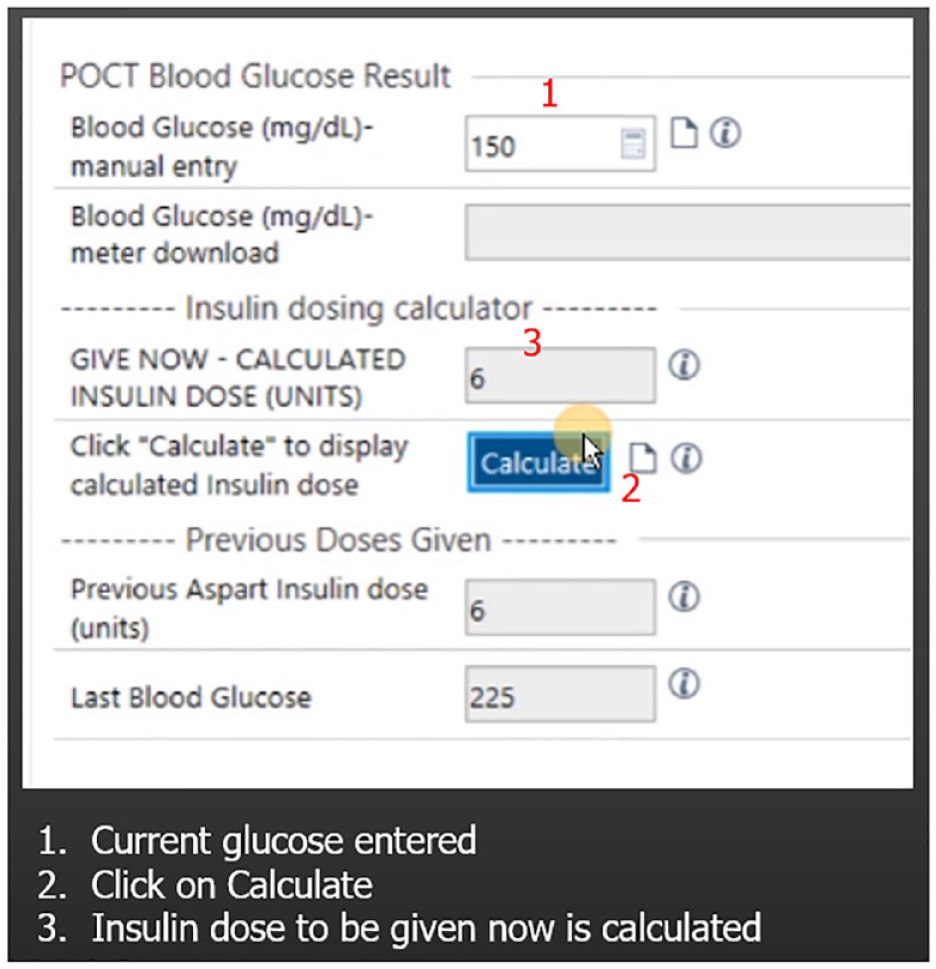
Medication Administration Record subcutaneous insulin algorithm calculator. Example of SQIA calculator in Medication Administration Record. Previous glucose 225 mg/dL with 6 units aspart insulin given. Current glucose is 150 mg/dL. As glucose in range (120-180 mg/dL), the same dose of insulin given 4 hours ago is now to be delivered. For details of algorithm see Supplemental Table S1. For more details on the calculator see Supplemental Figure S1.
The SQIA order set was introduced on the two surgical units and the surgical intensive care unit (SICU) at the UCSF Mission Bay Campus in June 2019. Surgical services there include pancreatic and colorectal oncology, otolaryngology oncology, urology oncology, gynecology oncology, and plastic and oral and maxillofacial surgery. Specific online training (<10 minutes in length) was available for nurses, physicians, and pharmacists. Although taking the training was highly encouraged, it was not mandatory. In-person training sessions were provided to unit nursing managers and tip sheets were given to the nursing staff.
Standard protocol was followed with all admitted patients having any previously administered oral antidiabetic agents and/or noninsulin injectable hypoglycemic agents discontinued. In addition, all insulins, with the exception of insulin glargine, in those on the pilot algorithm were discontinued. In our institution, insulin glargine is the formulary basal insulin. Levemir is not used, and neutral protamine Hagedorn is rarely used. Patients could be on basal insulin if ordered by the physician, but it was not mandatory. Hypoglycemic events were treated per standard protocol.
Multiple patient safety committees reviewed all this preliminary work and the data collection was approved by the Institutional Review Board at the UCSF.
Subjects
All non-obstetric patients admitted to the UCSF Mission Bay Campus who were NPO or on continuous TPN or continuous enteral feedings were eligible for the SQIA. For adults, only surgical patients are located at that campus with two Surgical Units and the SICU. At the time of this data analysis, a total of 68 patients had the SQIA ordered, but 22 required no insulin or were on the protocol for <12 hours and were not included in final analysis. Of those 46 patients, two were excluded because they did not undergo any procedure. Forty-four patients were included in the final analysis.
Glucose Monitoring
Glucose measurements were made using a point-of-care glucose meter (ACCU-CHEK Inform [Roche Diagnostics]). Blood samples obtained were either capillary (from finger sticks) or arterial (from arterial lines), if an arterial line was available.
Data Monitoring
A daily report showed all patients who were on the SQIA. One of the physicians would then review all patients on the SQIA to assure the protocol was being followed and the calculated values were correct and that the SQIA was only being ordered for patients who were NPO, or on continuous TPN or enteral feedings.
Results
Baseline Characteristics
Baseline characteristics for these patients are described in Table 1. Mean age was 66.5 ± 12.1 years. The majority of patients were admitted for cancer-related issue (75%), and all 44 patients underwent some invasive procedure. The majority of procedures were intra-abdominal (75%), and seven (16%) were Whipple procedures. Head and neck surgeries were second most common (18%). Twenty-four (55%) patients had pre-existing diabetes and were on some antiglycemic therapy prior to admission. Nine (20%) were on insulin only, 12 (27%) were on non-insulin agents only, and 3 (7%) were on a combination of insulin and non-insulin agents. Twenty-three of these patients were presumed to have type 2 diabetes, and one patient had immune checkpoint inhibitor-induced diabetes mellitus. Fifteen (34%) patients received at least one dose of high-dose glucocorticoids while on the SQIA, defined by greater than 20 mg prednisone steroid equivalent.
Baseline Characteristics of Patients Treated with the Subcutaneous Insulin Algorithm.

Glucose Results
Table 2 shows nutrition order, insulin dosing, and glucose levels for patients on the SQIA. The average duration on the SQ protocol was 88 hours (median duration 58 hours; range 16-404 hours), yielding a total of 983 glucose checks. The average glucose was 166 ± 51 mg/dL. Glucoses at titration initiation were highest at 206 ± 63 mg/dL, and decreased to 156 ± 29 mg/dL by 72 hours on the SQIA. There were no glucoses lower than 60 mg/dL, and only 0.3% (n = 3) were below 70 mg/dL. The majority of measured glucoses (66.8%, n = 647) were maintained between 80 and 180 mg/dL. Although 2.1% (n = 18) of measured glucoses were above 300 mg/dL, these were accounted for by only 6 of the 44 patients, and 2 of the patients accounted for 61% (n = 11) of these values. Seventy-two percent (n = 13) of glucose levels above 300 mg/dL occurred within 8 hours of SQIA initiation or missed doses of insulin.
Diet, Insulin Doses, and Glucose Levels for Patients on the SQIA.
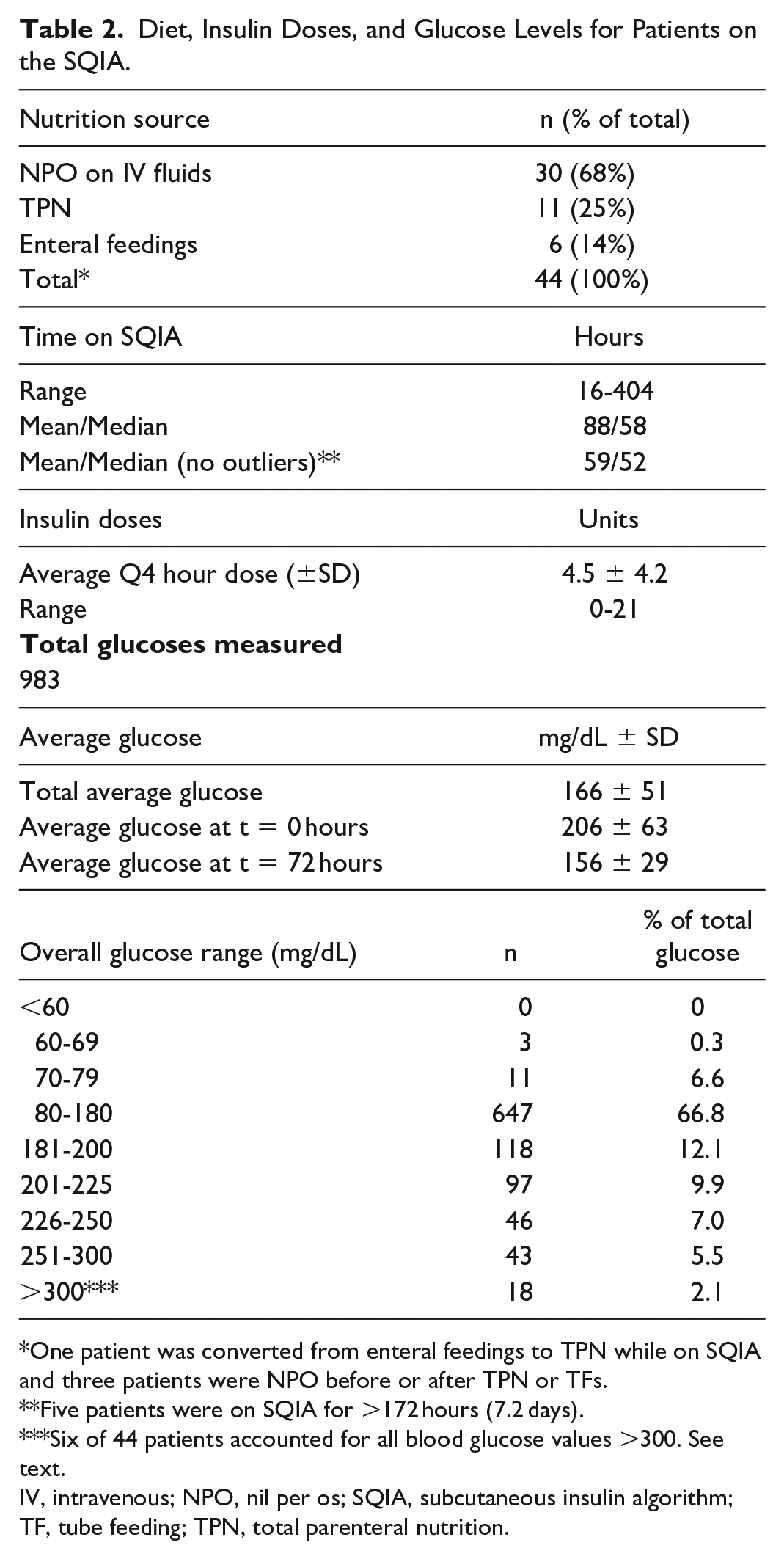
One patient was converted from enteral feedings to TPN while on SQIA and three patients were NPO before or after TPN or TFs.
Five patients were on SQIA for >172 hours (7.2 days).
Six of 44 patients accounted for all blood glucose values >300. See text.
IV, intravenous; NPO, nil per os; SQIA, subcutaneous insulin algorithm; TF, tube feeding; TPN, total parenteral nutrition.
Figure 2 demonstrates individual patient glucose and insulin data while on the SQIA. Specific examples include a patient previously without diabetes who is NPO after a Whipple procedure (Figure 2(a)); a patient with type 2 diabetes on continuous TPN with subsequent addition of insulin to TPN and basal insulin (Figure 2(b)); a patient previously without diabetes on continuous tube feeds (Figure 2(c)); a patient with type 2 diabetes receiving tube feeds at increasing rates and with addition of basal insulin (Figure 2(d)); a patient with type 2 diabetes who is NPO but receives high doses of dexamethasone (Figure 2(e)); and a patient with immune checkpoint inhibitor-induced diabetes who is NPO with a stress dose glucocorticoid taper (Figure 2(f)).
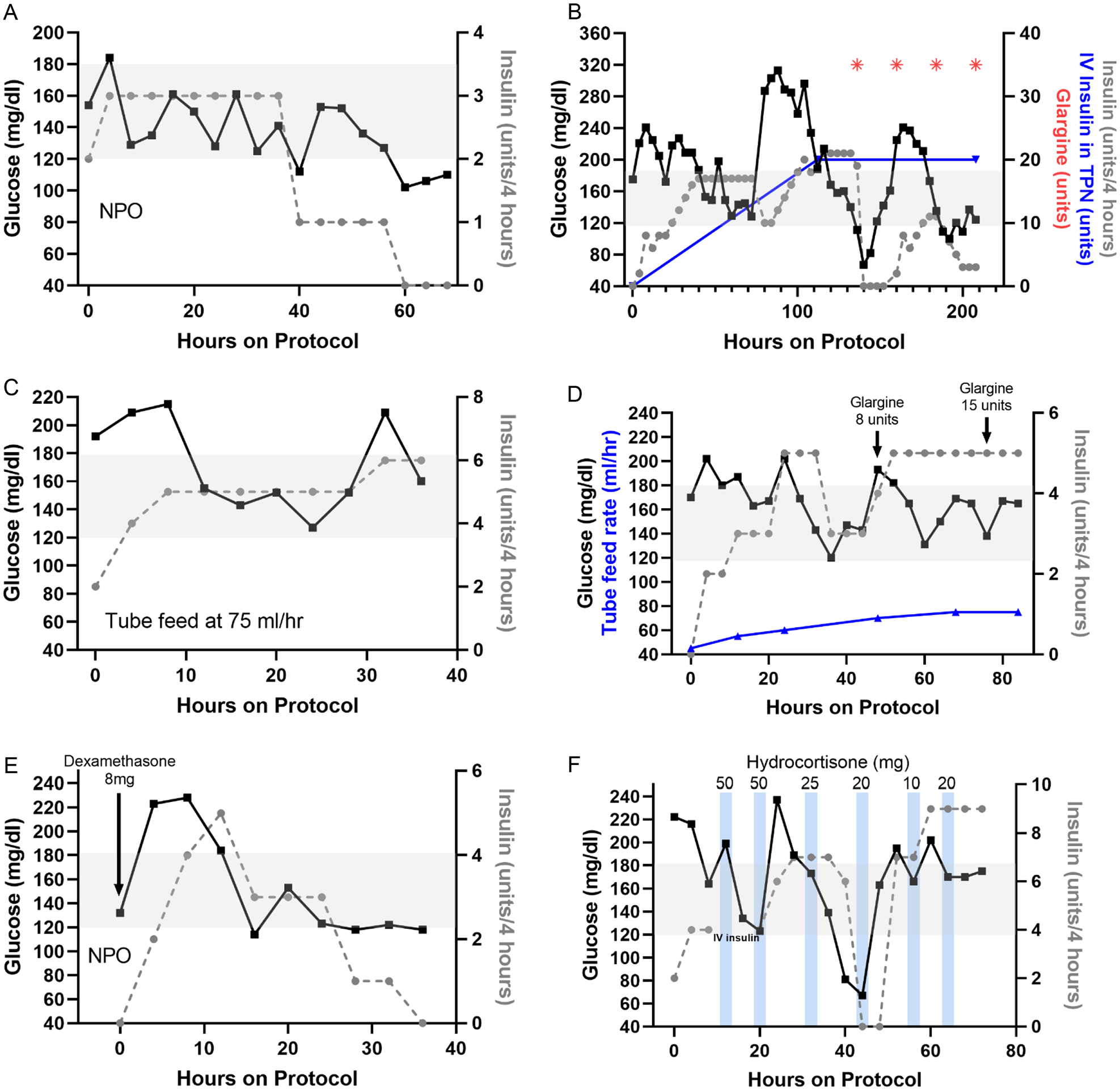
Individual patient data while on SQIA. (a-f) Glucose levels and insulin doses are indicated by black solid lines and gray dotted lines, respectively. Gray shading indicates SQIA glucose target range. (a) A patient without diabetes who is NPO after a Whipple procedure. (b) A patient with type 2 diabetes on continuous TPN with subsequent addition of 20 units of regular insulin to TPN (indicated by blue line) and addition of 35 units of glargine (indicated by red asterisk). (c) A patient previously without diabetes on continuous tube feeds. (d) A patient with type 2 diabetes on increasing rates of tube feeds (indicated by blue solid line) and with addition of basal insulin. (e) A patient with type 2 diabetes who is NPO but receives 8 mg of dexamethasone at initiation of SQIA, as well as 10 mg several hours prior. (f) A patient with immune checkpoint inhibitor-induced diabetes who is NPO and receiving a stress-dose steroid taper. Timing of hydrocortisone indicated by blue bars, and dose is above the bars.
Six of the eleven patients receiving continuous TPN never had insulin added to TPN, but two of these had basal insulin added on top of the SQIA. Four patients had insulin added to TPN later, and only one patient’s blood sugar dropped to 67 mg/dL, but he also had a significant dose of basal insulin started around the same time (see Figure 2(b)). One patient was started on TPN with insulin added to the IV bag from the beginning.
Four patients had basal insulin started with TPN. One patient on tube feeds had basal insulin added, and this patient’s glucose and insulin trend is shown in Figure 2(d), and his glucose was well controlled.
Errors
There were 10 protocol administration errors out of 983 total potential insulin injections (rate of 1%; Table 3). Six (60%) of these were errors in administration of the correct dose of insulin, and four of those (67%) were resolved by improving and simplifying the nursing MAR interface, as well as by making adjustments to the actual algorithm. No patient was harmed as a result of these errors.
Errors Made Using the Subcutaneous Insulin Algorithm in Pilot Units.*
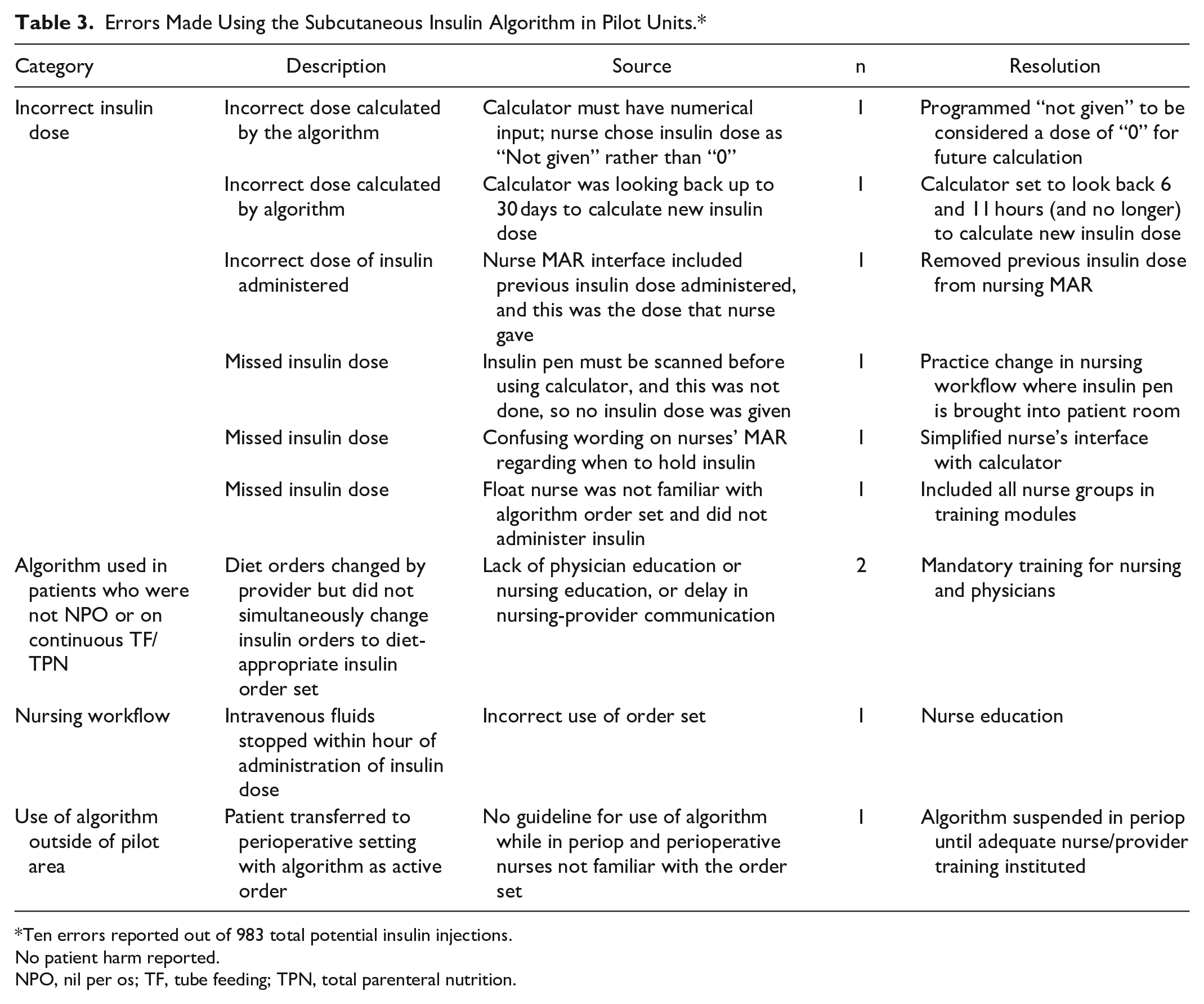
Ten errors reported out of 983 total potential insulin injections.
No patient harm reported.
NPO, nil per os; TF, tube feeding; TPN, total parenteral nutrition.
Discussion
As published in 2012, we previously demonstrated the feasibility of implementing a paper-based self-titrating SQIA and that it maintains the majority of glucose levels within the target range of 80-180 mg/dL. 25 In that study, where we compared the SQIA to use of an IV insulin infusion, the SQIA resulted in similar glucose levels, but only required nursing intervention every 4 hours instead of every hour. As we prepared to launch this paper-based protocol into use throughout the medical center, inpatient use of EPIC began. To simplify use for the nursing staff, we were asked to program into EPIC the algorithm as a calculator. Unfortunately, that functionality was not possible at that time and the project was put on hold. With changes in technology, we were able to revisit this project in 2016. As a result of weekly meetings of our new multidisciplinary group, refinements were made to the previous algorithm and the orders were moved from being paper-based to a fully programmed calculator located in the nurses’ MAR. The “calculated insulin dose” was based on the current and past glucose levels and the previous insulin dose. Thus, no insulin dosing calculations were required by the nursing staff, and there was a very low rate of errors in medication administration. This SQIA maintained the majority of glucoses within the target range with very low rates of hypoglycemia and was effective in surgical patients with and without diabetes.
As we have previously highlighted, 25 inpatient diabetes management using sliding-scale insulin, where a fixed dose of insulin is given based on the current glucose level, can lead to a “rollercoaster” effect on glucose levels. When the glucose is at goal, no insulin is given; then the glucose increases and insulin is given, lowering the glucose, and the cycle begins again. With an algorithm, whether used with IV insulin infusions or with SQ insulin, such as in this study, the insulin dose is titrated over time to the specific requirements of the patient. This method of insulin delivery mimics the physiologic response to glycemia and eliminates the rollercoaster effect. Indeed, the average insulin dose to maintain glucose levels within our target range was 4.5 units every 4 hours, but the range was 0-21 units every 4 hours, reflecting how different the requirements are in each person, how insulin requirements vary over time, and why self-adjusting algorithms are necessary. With the algorithm in place, glucoses were maintained at goal even with administration of high-dose glucocorticoids, changes in enteral feeding rates, the addition of insulin to TPN, and addition or changes made in basal insulin.
For purposes of our pilot data presentation, we excluded patients who had this automated algorithm ordered, but their glucose level did not rise high enough so that insulin dosing was required. Generally, glucose averages for hospitalized patients are based on all patients who have glucoses above 120 mg/dL. Including patients not requiring insulin would have led to lower average glucoses but would have not reflected the impact of the algorithm.
There are some limitations to this work. This was designed as a pilot program at our institution, and is not a randomized study. With adoption of this programed SQIA for all adult inpatients at UCSF, further studies will be needed to determine outcome benefits. There may be patient populations we have not yet encountered for whom this automated SQIA will not be appropriate. Finally, as with any IV or SQ protocol, as the clinical situation changes with alterations in enteral feedings or IV glucose rates or use of glucocorticoids, overriding the protocol orders and resetting the SQ insulin dose may still be required.
Conclusion
A straightforward fully automated SQIA titrates insulin doses to the individual patient’s needs and achieves glycemic goals for hospitalized patients. It is effective for patients who are NPO and those receiving continuous calories via TPN or TFs. In addition, this SQIA can be fully integrated into the EMR, such that a calculator embedded within the nurses MAR requires only input of current glucose level.
Supplemental Material
Supplemental_Tables_PDF – Supplemental material for Automated Self-Adjusting Subcutaneous Insulin Algorithm for Patients NPO or on TPN or Enteral Feedings
Supplemental material, Supplemental_Tables_PDF for Automated Self-Adjusting Subcutaneous Insulin Algorithm for Patients NPO or on TPN or Enteral Feedings by Sophie Patzek, Heidemarie W. MacMaster, Esther Rov-Ikpah, Craig San Luis, Craig Johnson, Venkateswarlu Juttukonda and Robert J. Rushakoff in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
We acknowledge the additional assistance of Mekhala Patwardhan (initial programming), Amy Kuwata, MSN, RN-BC, CNS (planning, testing), and the nursing staff, pharmacists, and physicians at the UCSF Mission Bay Campus.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
