Abstract
Background:
Regular moderate-to-vigorous-intensity physical activity results in health benefits in people with diabetes. No study has observed the moderate-to-vigorous-intensity physical activity typically performed by people with diabetes-related foot ulcers (DFU) in their everyday free-living environments. We observed the bouts, and accumulated time, spent doing moderate-to-vigorous-intensity physical activity in cases with DFU compared with diabetes-related peripheral neuropathy (DPN) and diabetes (DM) controls over a one-week period.
Methods:
This was a secondary analysis of a cross-sectional case-control study. Participants wore a multisensor device for >5 days (>22 hours per day). Primary outcomes included the number, duration (minutes) and intensity (metabolic equivalent tasks [METs]) of bouts of moderate-to-vigorous-intensity physical activity (defined as at least >3 METs for >10 consecutive minutes). Secondary outcomes included the total accumulated times spent doing moderate-to-vigorous-intensity physical activity (>3 METs) and doing sedentary-intensity activity (<1.5 METs). DFU subgroups with minor amputations and nonremovable offloading devices were also analyzed.
Results:
Overall, 15 DM, 23 DPN, and 27 DFU participants were included. All groups recorded similar low median daily numbers (0.33, 0.29, 0.25 numbers, respectively), duration (15, 17, 14 minutes), and intensity of daily bouts of moderate-to-vigorous-intensity physical activity (4.1, 4.3, 3.9 METs) (all, P > .1). Median accumulated daily time spent doing moderate-to-vigorous-intensity physical activity was also similar (40, 37, 36 minutes; P > .8). Those with DFU had more mean accumulated daily time spent doing sedentary-intensity activity (796 minutes) compared to DPN (720 minutes; P < .05), but not compared to DM (728 minutes; P < .08). DFU subgroups with minor amputations had more median accumulated daily time spent doing moderate-to-vigorous-intensity physical activity (66, 28 minutes; P < .05) and less mean time doing sedentary-intensity activity (745, 837; P < .05) than those without amputations.
Conclusions:
People with DFU performed similar low numbers of daily bouts of moderate-to-vigorous-intensity physical activity to controls, but spend more time doing sedentary-intensity activities. Interventions that gradually increase the moderate-to-vigorous-intensity physical activity in people with DFU should be investigated.
Keywords
Diabetes-related complications are rapidly becoming one of the leading causes of global morbidity, mortality and health care cost burdens.1-3 Diabetes-related foot ulcers (DFU) are now very much a leading cause of these large diabetes-related burdens.4-6 Foot ulcers typically develop in people with the diabetes-related peripheral neuropathy (DPN).4,7 Thus, to reduce diabetes-related burdens more needs to be done to understand, prevent, delay and manage these diabetes-related foot complications.
Performing regular moderate-to-vigorous-intensity physical activity has been reported to prevent or delay the onset of diabetes and other diabetes-related complications.8,9 Specific health benefits gained from performing regular moderate-to-vigorous-intensity physical activity in people with diabetes includes improved insulin sensitivity, 10 improved endothelial function, 11 lowered glycated HbA1c,12-14 reduced body mass index, 12 reduced systolic blood pressure, 15 and reduced risk of complications.8,9,15
To achieve these health benefits international guidelines recommend adults need to perform an accumulated 30 minutes of exercise on at least five days of each week.8,16 They define exercise as being a bout of at least 10 consecutive minutes of any moderate-to-vigorous-intensity physical activity (defined as >3 metabolic equivalent tasks [METs]). One MET is defined as approximately 1 kcal/kg/hour and includes activities such as sleeping, lying quietly or meditating. 17 Activities achieving levels of moderate-intensity physical activity (3-6 METs) typically include brisk walking, swimming leisurely, or cycling leisurely,8,16,17 whereas activities achieving vigorous-intensity physical activity (>6 METs) include jogging, cycling fast, or playing a game of football. 17 To help achieve these moderate-to-vigorous-intensity levels of physical activity guidelines also recommend that people with diabetes should decrease their sedentary activity. Sedentary activities (<1.5 METs) typically includes lying, sitting, eating or standing quietly. 17 Therefore, people with diabetes should be aiming to perform at least one bout of moderate-to-vigorous-intensity physical activity on most days, and reduce their daily sedentary activity, if they are to achieve optimal health benefits.8,16
Many studies have now begun to investigate if people with type 2 diabetes are achieving these physical activity recommendations.18-22 However, to our knowledge none have investigated this in people with DFU. Therefore, the primary aim of this study was to investigate the bouts of moderate-to-vigorous-intensity physical activity performed by people with DFU and to compare it to those with DPN without DFU history (DPN) and those with diabetes without DPN or DFU (DM) in their everyday free-living environments. Secondary aims were to investigate the accumulated daily time spent doing any moderate-to-vigorous-intensity physical activity and any sedentary-intensity activity. Last, we also investigated these outcomes in DFU subgroups with and without minor amputations, and with and without nonremovable offloading devices.
Methods
Study Design
This study was a secondary analysis of data collected in an original cross-sectional case-control study that investigated the activity of people with DFU, DPN, or DM in their free-living environment over a one-week period. 23 The original study recruited participants from three diabetes-related foot outpatient clinics in Brisbane, Australia. 23 Findings from the original study on the outcomes of average daily steps, energy expenditure (kJ), lying and sleeping durations have previously been published. 23 Ethical approval was granted for this study by the Human Research Ethics Committees (HREC) of the Prince Charles Hospital (No. HREC/17/QPCH/13) and Queensland University of Technology (No. 1700000228), Australia. Written informed consent was voluntarily obtained from all individual participants. The design and methodology have been described in detail elsewhere, 23 and will be summarized below.
Participants
All participants were adults with type 2 diabetes mellitus. Participants were grouped into those with: (1) DPN and a current DFU (DFU group), (2) DPN and no DFU history (DPN group), and (3) no DPN or DFU history (DM group). 23 The DFU group was further subdivided into those (1) with a history of minor amputation(s) compared with those without any amputation history and (2) those wearing a nonremovable knee-high offloading device compared with those wearing another offloading device. 23 DPN was defined as an inability to detect a 10-gram monofilament on at least two of three sites on the plantar forefoot.7,24 DFU was defined as an active full thickness wound on the plantar aspect of the foot.7,24 Minor amputation was defined as a previous amputation below the ankle.7,24,25 Nonremovable knee-high offloading devices were defined as either a total contact cast or removable knee-high walker made irremovable.25,26 Other offloading devices were defined as either removable ankle-high devices (such as a postoperative shoe), therapeutic footwear, or felted foam applied in appropriate footwear.25,26
The exclusion criteria for the original study were designed to avoid conditions that may confound the impact of DPN and DFU on typical daily activity and sleep patterns and included those with (1) peripheral arterial disease (defined as a toe pressure of <70 mmHg), (2) major amputation (previous amputation above the ankle), (3) mobility impairment (inability to walk without an aide), (4) a DFU on the contralateral foot, (5) painful DPN, (6) sleep apnea, or (7) those deemed to be cognitively unable or unwilling to perform the study procedures by the researchers.23,24
Sample Size
Sample size was calculated for the original study based on previously reported mean daily steps in people with DM,27,28 DPN,27,29,30 and DFU,27,29 a power of 80% and an alpha of .05. 23 These calculations suggested 25 participants were required in each group to determine a statistical difference for daily steps which is considered a surrogate marker of daily activity.23,24
Participant Characteristics
Participant’s characteristics were recorded using the Queensland High Risk Foot Form (QHRFF)23,25 and included demographics (age, gender, body mass index [BMI]), diabetes history (diabetes duration, HbA1c), comorbidity history (hypertension, dyslipidemia, cardiovascular disease, chronic kidney disease), foot complications (foot deformity, DPN, DFU, and amputation history), and treatment (offloading devices).23,25
Procedure
A SenseWear armband (Model MF-SW; BodyMedia Inc, Pittsburgh, PA, USA) was used to measure physical activity in all participants. 23 All participants were instructed to wear the armband on their mid upper arm continuously for one week at all times except when engaging in water-based activities such as showering or swimming. 23 No further advice was provided as the study aim was to observe typical daily activity of participants in their normal free-living everyday environment. 23 At least five days of data from wearing the device for at least 22 hours on each of those days, with at least one of those days being a weekend day, was required to be eligible for the participant’s data to be included, as this period has been previously validated to be representative of typical daily activity.31-34 Days in which a participant wore the device for less than 22 hours were excluded.
The armband consists of multiple sensors such as tri-axial accelerometer (measures motion), skin temperature (measures armband use), galvanic skin response (measures skin impedance), and heat flux (measures the rate of heat dissipation).23,35,36 The armband uses validated proprietary algorithms to transform data captured from the multiple sensors along with demographic characteristics to detect and record wear time (compliance with wearing the device) and calculate activity parameters such as METs (mean error <5%), steps (mean error <9%), physical activity intensity and duration (mean error <5%), lying and sleep duration (mean error <10%), and estimated energy expenditure (mean error <5%).23,35-37 The use of the armband has been validated in comparisons with gold standard criterion measures for a range of daily physical activity parameters, including METs.31,32,34-36,38,39
Outcome Measurement
The primary outcomes of interest for this particular study were the number of bouts of moderate-to-vigorous-intensity physical activity (defined as activity recording an intensity of >3 METs per minute for >10 consecutive minutes),8,16 and the average duration (minutes per bout) and intensity (METs per bout) of these bouts. Secondary outcomes included the overall accumulated time spent doing any moderate-to-vigorous-intensity physical activity (total of any nonsleeping minutes recording intensity >3 METs per minute) and the overall accumulated time spent doing any sedentary-intensity activity time (total of any nonsleeping minutes recording intensity <1.5 METs per minute) and intensity (METs per minutes).8,16 The data from the five or more eligible days were averaged to determine daily outcomes.
Statistical Analysis
Data were analyzed using SPSS 23.0 for Windows (IBM Corp, Armonk, NY, USA). For categorical variables, proportions were reported and Person’s chi-squared or Fisher’s exact test used to test differences between three groups and two groups respectively. For continuous variables, Kolmogorov-Smirnov tests were used to assess normality of distribution. For those with normal distributions, means and standard deviations (±SD) were reported and ANOVA with Fisher’s least significant difference were used to test differences between three groups and Student’s t-tests between two groups. For those nonnormal distributions, median, and interquartile ranges (IQR) were reported and Kruskal-Wallis and Mann-Whitney U-tests were used to test differences between three and two groups respectively. A significance of P < .05 was used throughout. In addition, for those outcomes with P < .2 and normal distributions we estimated effect sizes (standardized mean differences) between pairs of groups using Cohen’s d. 40 The degree of the effect size difference was graded based on Cohen’s d values as <0.1 trivial difference, 0.1-0.2 small difference, 0.2-0.6 medium difference, 0.6-1.2 large difference, and >1.2 very large difference. 41
Results
Seventy-seven participants were recruited in the original cross-sectional study, including 23 DM, 24 DPN, and 30 DFU. Five (6.5%) participants were excluded from the original study (four DM and one DPN) as their armband failed to collect any data. A further seven (9.1%) participants also had to be excluded from this analysis (four DM and three DFU) as the armband failed to collect all continuous minute-by-minute physical activity data over the five days (>22 hours per day) to determine the primary outcome of bouts. Therefore, 65 participants were included in this secondary analysis, including 15 DM, 23 DPN, and 27 DFU.
Table 1 displays participant characteristics and outcomes of interest for the three groups. Most characteristics were similar between groups (P > .05), except for those with DPN being older (DPN 68 ± 9, DM 59 ± 11 and DFU 56 ± 11 years; P = .001) and those with DFU having more foot deformities (DM 0%, DPN 16.7%, DFU 83.3%; P < .001). All groups recorded similar low median [IQR] numbers of daily bouts of moderate-to-vigorous-intensity physical activity (DM 0.33 [0-0.67], DPN 0.29 [0-1.33], DFU 0.25 [0-0.8] numbers), median durations per these bouts (DM 15 [12-17], DPN 17 [14-21], DFU 14 [11-18] minutes) and median intensity per bout (DM 4.1 [4.0-4.4], DPN 4.3 [3.8-4.5], DFU 3.9 [3.6-4.2] METs) (all P > .1). All groups also recorded similar median accumulated daily time spent doing moderate-to-vigorous-intensity physical activity (DM 40 [21-65], DPN 37 [19-76], DFU 36 [17-64] minutes per day; P > .8). The DFU group recorded significantly more mean accumulated daily time spent doing sedentary-intensity activity than the DPN group (DFU 796 ± 120, DPN 720 ± 140 minutes per day; P < .05; d = 0.58), but not significantly more than the DM group (DFU 796 ± 120, DM 728 ± 123; P < .08; d = 0.56). The mean intensity during this sedentary activity time was significantly higher for the DFU group compared to the DPN group (DFU 0.95 ± 0.10, DPN 0.90 ± 0.07 METs per minute; P < .05; d = 0.58) and compared to the DM group (DFU 0.95 ± 0.10, DM 0.89 ± 0.09 METs per minute, P < .05; d = 0.63).
Participant Baseline Characteristics and Outcomes of Interest for Each Group Stated in Mean ± SD, Median [IQR], or Number (%).
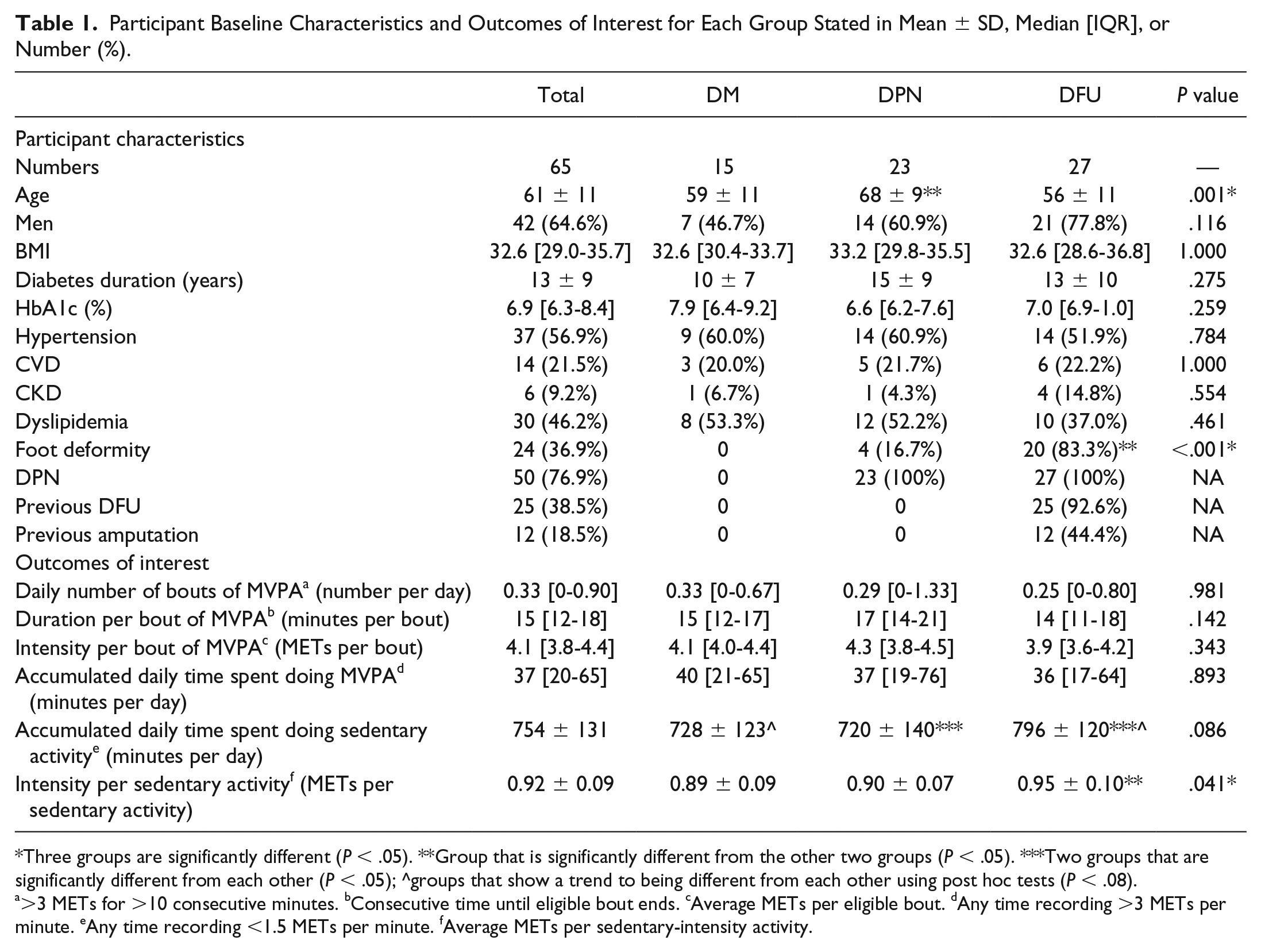
Three groups are significantly different (P < .05). **Group that is significantly different from the other two groups (P < .05). ***Two groups that are significantly different from each other (P < .05); ^groups that show a trend to being different from each other using post hoc tests (P < .08).
>3 METs for >10 consecutive minutes. bConsecutive time until eligible bout ends. cAverage METs per eligible bout. dAny time recording >3 METs per minute. eAny time recording <1.5 METs per minute. fAverage METs per sedentary-intensity activity.
Table 2 displays the participant characteristics and outcomes of interest for the subgroups of those 27 with DFU, including 12 (44%) with minor amputation(s) and 15 (56%) without a minor amputation, and 7 (26%) wearing a nonremovable offloading device and 20 (74%) wearing other offloading devices. Participants with minor amputation(s) compared to those without any amputation had more, albeit nonsignificant, median daily bouts of moderate-to-vigorous-intensity physical activity (0.58 [0.17-1.89], 0.20 [0-0.63]; P < .08), significantly higher median intensity during their bouts (4.2 [3.9-4.3], 3.7 [3.5-4.0] METs; P < .05), significantly more median accumulated daily time spent doing moderate-to-vigorous-intensity physical activity time (66 [24-109], 28 [12-42] minutes per day; P < .05), significantly less mean accumulated daily time spent doing sedentary-intensity activity (745 ± 125, 837 ± 103 minutes per day; P < .05; d = 0.80), and significantly more mean intensity during this sedentary time (1.00 ± 0.11, 0.92 ± 0.07; P < .05; d = 0.87). There were no significant differences between those who wore nonremovable knee-high devices and those wearing other offloading devices in any of their baseline characteristics (P > .15) or outcomes of interests (P > .25).
Participant Baseline Characteristics and Outcomes of Interest for Each DFU Subgroup Stated in Mean ± SD, Median [IQR], or Number (%).
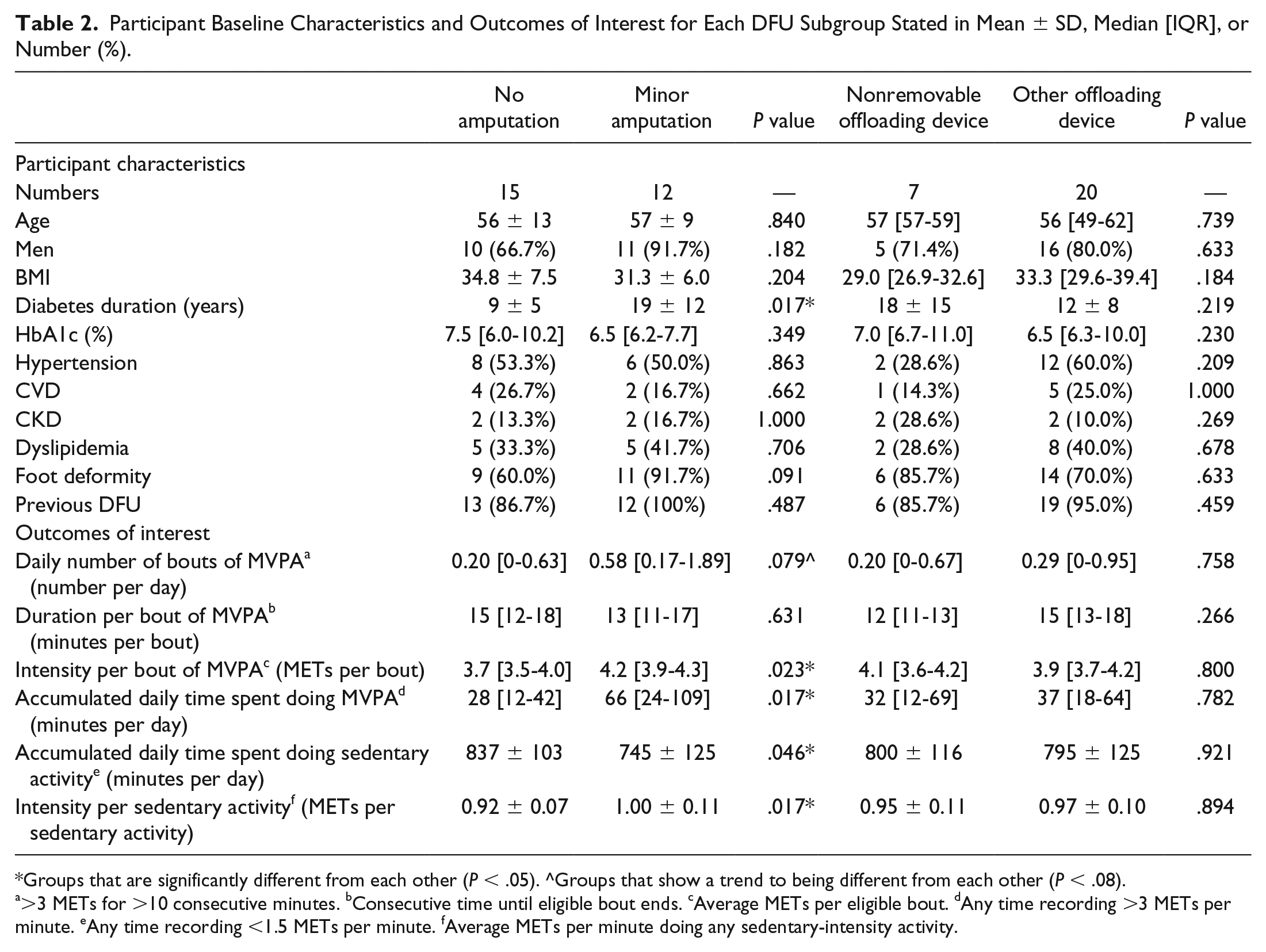
Groups that are significantly different from each other (P < .05). ^Groups that show a trend to being different from each other (P < .08).
>3 METs for >10 consecutive minutes. bConsecutive time until eligible bout ends. cAverage METs per eligible bout. dAny time recording >3 METs per minute. eAny time recording <1.5 METs per minute. fAverage METs per minute doing any sedentary-intensity activity.
Discussion
This is the first study to observe the bouts and accumulated time people with DFU spent performing moderate-to-vigorous-intensity physical activity in their everyday environment. We observed people with DFU performed similar low numbers of daily bouts and accumulated time doing moderate-to-vigorous-intensity physical activity to those with DPN or DM. However, people with DFU spent more daily time doing activity of sedentary intensity. In DFU subgroups, those with minor amputations spent more time performing moderate-to-vigorous-intensity physical activity and less time sedentary than those without minor amputations.
Our findings initially suggest all three groups met international guideline recommendations for performing 30 minutes of exercise per day, with all groups achieving over 30 minutes of accumulated moderate-to-vigorous-intensity physical activity on average per day.8,16 However, as this recommendation defines exercise as a bout of at least 10 consecutive minutes of moderate-to-vigorous-intensity physical activity, our findings actually show that no group met these exercise recommendations as none performed at least one of these bouts per day to be defined as exercise.8,16 We observed that people with diabetes, DPN, or DFU rarely perform a 10-minute bout of moderate-to-vigorous-intensity physical activity, and thus rarely achieve any of the health benefits that regular exercise provides.8,16 In people with established diabetes, these findings may indicate that the presence of DPN or DFU does not impact on moderate-intensity physical activity levels. However, all groups had low levels of moderate-to-vigorous-intensity physical activity indicating that people with DM, DPN, and DFU might not be willing or able to sustain a minimum of 10 consecutive minutes of moderate-intensity physical activity. Further studies are required to investigate first the ability of people with DPN and DFU to perform regular bouts of moderate-to-vigorous-intensity physical activity (exercise), and second the impact that achieving these recommended regular bouts have on their general health as well as their foot health, such as foot ulcer development, prevention and healing.26,42,43
We also observed that all groups were highly sedentary, spending at least 12 waking hours each day (>720 minutes) performing activities that are akin to sitting quietly. Further, our findings showed that the DFU group spent an additional hour each day sedentary compared to controls. A simple explanation for this may be that the DFU group slept less and had an extra hour of waking time than the other groups; however, our original cross-sectional study found no differences in daily sleep time between these same groups or DFU subgroups. 23 Previous studies have reported that foot complications such as chronic foot pain or foot deformity resulted in reductions in physical activity. 44 This may partly explain our findings as the DFU participants had significantly more foot deformities than controls; however, they did not perform any less physical activity than the other groups. Another explanation may be that treating clinicians often recommend that those with DFU reduce their ambulatory activity to reduce plantar pressure and in turn facilitate improved DFU healing. 42 In our study we provided no advice to participants that should have biased their everyday physical activity, although it cannot be discounted that previous advice may have impacted. Regardless, our findings do suggest that those with DFU are not performing any alternative bouts of moderate-to-vigorous-intensity physical activity such as upper body exercises and this may be further negatively impacting on their general health.
An interesting finding from this study that supports those from other studies was that those with DFU had a higher average intensity of sedentary activity. This sounds contradictory, but indicates that people with DFU require more energy or higher intensity to perform the same (sedentary) activities as controls. The original cross-sectional study reported that these same participants with DFU generally walked less but expended more overall energy to do this than controls. 23 Hypotheses for the original study’s findings included that DFU patients may (1) perform other nonambulatory activity that were not accounted for by simply measuring walking activity, (2) have had to ambulate in heavier offloading devices, (3) have more comorbidities and are generally sicker, which in turn requires more energy to function, (4) require more energy to heal their foot ulcer than those without ulcers, and (5) have a more inefficient gait that is compensating for the DFU itself, foot deformity, minor amputations (as indicated by our subgroup results), postural instability or soft tissue rigidity.23,45,46 Our findings in this study seem to suggest we may be able to discount the hypotheses of (1) performing other nonambulatory physical activity as we measured all activity in this study and found no differences, (2) ambulating in heavier offloading devices as the majority of people in the DFU group wore removable ankle-high devices (such as postoperative shoes) which can be considered similar in weight to footwear worn by those in the DPN and DFU group, and the small DFU subgroup wearing nonremovable offloading devices showed no differences to the removable ankle-high devices DFU subgroup, and (3) being generally sicker with more comorbidities as there were no differences in comorbidities between groups. Although we are unable to interpret if the differences in mean sedentary activity intensity between the DFU group and other groups is clinically important, this difference was reported to be statistically different with a moderate-large effect size, and thus any clinical difference cannot be discounted until future studies can confirm. Therefore, this again all suggests that the most likely hypotheses for this increase in average sedentary-intensity activity is either from the increased energy required to heal the foot ulcer or from a more energy inefficient compensatory gait in those with DFU compared with controls. However, future research is still required to verify these hypotheses and investigate if there is any general health impact of such increases in sedentary activity intensity.
Finally, our study observed that those with DFU and minor amputations recorded more accumulated daily time doing moderate-to-vigorous-intensity physical activity, less accumulated time doing sedentary-intensity activity and a trend toward higher numbers and intensity of bouts moderate-to-vigorous-intensity physical activity than those without minor amputations. Many studies have investigated the impact of major amputations on a range of physical activity parameters and have overwhelming found negative impacts;47-49 however, our results suggest this may not be the case for minor amputations. To our knowledge only one other study has investigated the impact on physical activity parameters of minor amputations in people with DFU and also found no negative impact on physical activity. 29 This suggests minor amputations might not impact on physical activity and supports reports suggesting that minor amputations should be viewed as a positive prophylactic procedure with limited negative impact on general health that may prevent major amputations that have significant detrimental impacts on general health. 50
Strengths and Limitations
There were several strengths to this study. First, this study excluded participants with conditions that have been shown to limit physical activity and would have biased findings, such as peripheral arterial disease, mobility impairment and painful DPN. Second, participant characteristics were generally well-matched between groups, except for age and foot deformity. However, it is already well known that people with DFU have much higher prevalence of foot deformities than controls. Regarding age, the DFU group was younger than the DPN group and, if anything, could have been expected to be more active, not less active as found. While we could have adjusted for age in our analysis we considered against this due to the small numbers in each group presenting a significant risk of being underpowered for such an adjustment, and the most likely possibility that this would have increased the effect rather than decreased the effect of our findings and thus not impacted our overall findings. Last, participant characteristics and physical activity were both recorded using tools and devices respectively found to be reliable and valid to collect such variables.25,31-33,35,38
There were also several limitations to this study. First, while we recruited and retained the group numbers required by our original sample size calculations (n = 25) for the DFU group (n = 27), this wasn’t quite the case for the DPN group (n = 23) and especially not the case for the DM group (n = 15). Thus, this study was underpowered when comparing against the DM group in particular; however, our use of two “control” groups (DPN and DM groups) and the reported effect sizes give some context of the potential differences in physical activity outcomes for cases with DFU compared to control groups. 24 Second, our comparison of DFU subgroups resulted in some small subgroups and potentially underpowered findings, particularly for the nonremovable knee-high offloading device group with seven participants. While the significance and effect size findings of these subgroups provide some direction they should be viewed with caution until future adequately powered studies can confirm these findings. Third, due to these small subgroups we lacked the power to be able to investigate combined subgroups of those with minor amputations wearing nonremovable knee-high offloading devices compared to other offloading devices for example. Such further analyses in larger groups may have shed light on the reasons for differences seen in the DFU subgroup with minor amputations in particular. Fourth, although the other offloading device subgroup consisted mainly of similar types of removable ankle-high devices or footwear, unlike the nonremovable offloading device group this was not a homogenous offloading device group. Future studies should investigate the impact of specific offloading device types on daily activity to determine their specific impact on daily activity and offloading. Fifth, while we captured and reported nearly all demographic and comorbidity variables recommended for collection by diabetic foot studies, we did not capture educational, socioeconomic or employment status 24 which may have also had some impacts on daily activity. Sixth, although the SenseWear armband has been demonstrated to be valid and reliable for recording physical activity parameters in different populations with chronic conditions,31-33,35,38,39 it has yet to be validated in a DFU population. Seventh, the international exercise guideline recommendations used as the basis for this study were for 18- to 65-year-old adults and may not readily apply to our older population.8,16 However, our DFU population in particular had a mean age of 56 years, so most of these participants would have still been in the age range for these recommendations. Eighth, our definition of a bout was very tight8,16 and did not account for any long moderate-to-vigorous-intensity physical activities that may have been broken up by a minute or two of <3 METs or for any long bout durations (such as a 10-minute bout being counted the same way as a 60-minute bout). However, this was the same for all groups and the duration and intensity of bouts were found to also be low along with the number. Last, this study was not controlled to represent a particular phase of DFU healing, duration or offloading treatment duration. A previous study identified that activity may change in people with DFU over time, 51 and thus future longitudinal studies are recommended to observe if physical activity changes do occur over time in these populations.
Conclusion
We found that people with DFU, DPN or DM did not meet the international guideline recommendations for regular exercise (bouts of moderate-to-vigorous-intensity physical activity) that have been found to achieve general health benefits. We also found all groups were highly sedentary, and those with DFU more so. Interestingly, we observed that people with DFU and minor amputations were more physically active and less sedentary compared to those with DFU without a minor amputation. More work is now needed to investigate interventions that may increase exercise of moderate-to-vigorous-intensity physical activity, and reduce sedentary-intensity activity, in people with DPN and DFU to determine their effects on general and foot health.
Footnotes
Acknowledgements
We gratefully acknowledge the help and support of the staff and patients at the Community Diabetes Service and Department of Podiatry, Metro North Hospital and Health Service, Australia. We would also like to thank the Allied Health Research Collaborative from The Prince Charles Hospital for data storage and availability.
Abbreviations
ANOVA, analysis of variance; BMI, body mass index; CKD, chronic kidney disease; CVD, cardiovascular disease; DFU, diabetes-related foot ulcer; DM, diabetes mellitus; DPN, diabetes-related peripheral neuropathy; HbA1c, glycosylated hemoglobin; HREC, human research ethics committee; IQR, interquartile range; LSD, least significance difference; MET, metabolic equivalent task; MVPA, moderate-to-vigorous-intensity physical activity; PA, physical activity; QHRFF, Queensland High Risk Foot Form; SD, standard deviation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was originally supported by a research grant from the Prince Charles Hospital Foundation. The funder had no involvement in the study design, data collection, data analysis, manuscript preparation, or publication decisions. Furthermore, no funding was provided to carry out the secondary analyses for this particular study.
