Abstract
Background:
The prevalence of smoking and diabetes is increasing in many developing countries. The aim of this study was to investigate the association of smoking with inadequate glycemic control and glycemic variability with continuous glucose monitoring (CGM) data in people with type 1 diabetes.
Methods:
Forty-nine smokers and 320 nonsmokers were obtained from the Novo Nordisk Onset 5 trial. After 16 weeks of treatment with continuous subcutaneous insulin infusion, risk of not achieving glycemic target and glycemic variability from six CGM measures was investigated. Analyzes were carried out with logistic regression models (glycemic target) and general linear models (glycemic variability). Finally, CGM median profiles were examined for the identification of daily glucose excursions.
Results:
A 4.7-fold (95% confidence interval: 1.5-15.4) increased risk of not achieving glycemic target was observed for smokers compared with nonsmokers. Increased time in hyperglycemia, decreased time in range, increased time in hypoglycemia (very low interstitial glucose), and increased fluctuation were observed for smokers compared with nonsmokers from CGM measures. CGM measures of coefficient of variation and time in hypoglycemia were not statistically significantly different. Examination of CGM median profiles revealed that risk of morning hypoglycemia is increased for smokers.
Conclusions:
In conclusion, smoking is associated with inadequate glycemic control and increased glycemic variability for people with type 1 diabetes with especially risk of morning hypoglycemia. It is important for clinicians to know that if the patient has type 1 diabetes and is smoking, a preemptive action to treat high glycated hemoglobin levels should not necessarily be treatment intensification due to the risk of hypoglycemia.
Introduction
Globally, tobacco smoking trends have been estimated by the World Health Organization to decline from 2000 to -2025 by 1.1 billion smokers. 1 Unfortunately, the prevalence is increasing in many developing countries. 1 Also the prevalence of diabetes is rising rapidly in these middle- and low-income countries. 2 Not only is smoking a risk factor for developing diabetes, smoking also severely affects glycemic management after onset of diabetes. 3 Increased insulin resistance has been observed in smokers with or without diabetes, which leads to inadequate glycemic control. 4 In a cross-sectional study of 292 adults with type 1 diabetes by Melin et al, 5 smoking (any amount of tobacco during the last year) was found to be associated with a three-fold increased risk of inadequate glycemic control as defined by glycated hemoglobin (HbA1c) above 70.5 mmol/mol (8.6%). In another observational study by Peng et al 6 of more than 25 000 people with diabetes, the risk of inadequate glycemic control defined as HbA1c equal to or above 53 mmol/mol (7.0%) was 1.5-fold higher for current smokers compared with nonsmokers. These studies suggest an association between smoking and risk of not achieving glycemic target. However, the effect of smoking on glycemic variability has been less investigated. In an observational study by Hirai et al 7 of 537 people with type 1 diabetes, smoking was associated with a 2.5-fold increase in risk of severe hypoglycemia, but more thorough investigations of glycemic variability are very sparse. In treating type 2 diabetes, assisting individuals adopt healthier lifestyles, includes a focus on smoking cessation, but the target for glycemic control is still based on HbA1c. 8 Moreover, the use of continuous glucose monitoring (CGM) is rare for type 2 disease, which limits insight in glycemic variability. On the other hand, CGM devices are frequently used by people with type 1 diabetes, which enables the study of smoking and glycemic variability. Accuracy of CGM devices has improved significantly in the last decade, and today’s devices have reached an accuracy level, where investigation of daily glucose excursions as a consequence of smoking would be possible. 9
The aim of this study was to investigate the association of smoking with inadequate glycemic control and glycemic variability with CGM data in people with type 1 diabetes.
Methods
Clinical Trial Data
Data were obtained retrospectively from a recent clinical trial from Novo Nordisk A/S investigating efficacy and safety of fast-acting insulin aspart compared with insulin aspart in a 16-week treatment period in 472 adults from Europe and North America with type 1 diabetes using continuous subcutaneous insulin infusion. Two inclusion criteria of interest were HbA1c 53-75 mmol/mol (7.0%-9.0%) and use of the same Medtronic pump (Minimed 530G [551/751], Paradigm Veo [554/754], Paradigm Revel [523/723], and Paradigm [522/722]) for continuous subcutaneous insulin infusion in a basal-bolus regimen with a rapid acting insulin analog for at least six months prior to screening and willing to stay on the same pump model throughout the trial. 10 Prior to the end of trial, the participants wore a blinded Dexcom G4 Platinum (Dexcom, San Diego, CA) CGM device for two weeks. Demography and smoking status data were collected at screening, and smoking was defined as smoking at least one cigarette, cigar, or pipe daily (excluding e-cigarettes). Previous smokers were excluded in this study making smoking status a dichotomous variable of “Smoker” and “Nonsmoker.” Laboratory measurements were derived from blood samples drawn at randomization (baseline) and at end of trial. People without CGM measurements, demographic data, or an HbA1c measurement at baseline/end of trial were not included in this study. During the treatment period, hypoglycemic episodes were recorded by the participants according to guidelines by the American Diabetes Association 11 at the time of trial design. The definition was as follows 12 : (1) Severe hypoglycemia: An episode requiring assistance of another person to actively administer carbohydrate, glucagon, or take other corrective actions. Blood glucose concentrations may not be available during an event, but neurological recovery following the return of blood glucose to normal is considered sufficient evidence that the event was induced by a low blood glucose concentration, (2) Asymptomatic hypoglycemia: An episode not accompanied by typical symptoms of hypoglycemia, but with a measured blood glucose concentration ≤70 mg/dL (3.9 mmol/L), (3) Documented symptomatic hypoglycemia: An episode during which typical symptoms of hypoglycemia are accompanied by a measured blood glucose concentration ≤70 mg/dL (3.9 mmol/L), (4) Pseudo-hypoglycemia: An episode during which the person with diabetes reports any of the typical symptoms of hypoglycemia with a measured blood glucose concentration >70 mg/dL (3.9 mmol/L) but approaching that level, and (5) Probable symptomatic hypoglycemia: An episode during which symptoms of hypoglycemia are not accompanied by a blood glucose determination but that was presumably caused by a blood glucose concentration ≤70 mg/dL (3.9 mmol/L).
Statistical Analyses
Characteristics of people at baseline included in this study are presented as numbers, means, and standard deviations or percentages of participants. T-tests, chi-square, or Mann-Whitney U tests will be used to present statistical differences in subject characteristics. Achievement of glycemic target after 16 weeks of treatment was defined as an HbA1c at the end of trial of <53 mmol/mol (7%). Logistic regression models were constructed to analyze unadjusted and adjusted effect of smoking on risk of not achieving glycemic target. The adjusted model included age, gender, body mass index (BMI), diabetes duration, HbA1c at baseline, and insulin use at baseline as covariates and factors. Reported hypoglycemic episodes during the 16 weeks of treatment were summarized descriptively and are presented according to American Diabetes Association’s categories 11 for smokers vs nonsmokers as number of episodes and number of episodes per 100 person years of exposure.
To further analyze glycemic variability, six measures from the CGM data were calculated as follows: (1) Percentage of time spent in hyperglycemia, interstitial glucose (IG) > 180 mg/dL (10 mmol/L), (2) percentage of time spent in range, 70 mg/dL (3.9 mmol/L) ≤ IG < 180 mg/dL (10 mmol/L), (3) percentage of time spent in hypoglycemia, IG ≤ 70 mg/dL (3.9 mmol/L), (4) percentage of time spent in hypoglycemia, IG ≤ 45 mg/dL (2.5 mmol/L), (5) coefficient of variation (CV), and (6) fluctuation, calculated as the area between the average CGM value and the CGM values above and below divided by the CGM duration. Only periods without gaps in CGM data contributed to the calculation. The effect of smoking on each of the six measures was analyzed with general linear models, and the P-values from the models are illustrated in the results. Each model was adjusted for age, gender, BMI, diabetes duration, HbA1c at baseline, and insulin use at baseline.
Finally, the daily CGM profiles for smokers and nonsmokers are illustrated. The profiles were created by calculating the median of all CGM profiles of all participants stratified by smoking status in five-minute intervals throughout the day, that is, 00:00, 00:05, 00:10, 00:15, and so on. Each CGM value was shifted to the nearest five-minute timepoint. Percentages of readings in hypoglycemia are illustrated in 60-minute intervals in the same plot using the second y-axis. Moreover, average time for bolus insulin infusion for smokers and nonsmokers was found for intervals of interest and is presented in the results section. The bolus insulin infusions were found from continuous subcutaneous insulin infusion data.
Results
One hundred and three of the 472 participants were excluded due to being previous smokers. Of the remaining 369 participants, 13% (49) are smokers. In Table 1, the characteristics of the 369 people in this study are shown. Nonsmokers had a statistically significantly higher BMI at baseline than smokers. Otherwise, characteristics of the people in the two groups were similar. In Table 2, the effect of smoking on risk of not achieving glycemic target is shown. From the adjusted estimate, the risk of not achieving glycemic target is 4.7-fold increased for smokers. Table 3 shows number and rate of hypoglycemic episodes after 16 weeks of treatment. The rate of severe hypoglycemia is almost double for smokers. Furthermore, the rate of documented symptomatic and asymptomatic episodes is higher for smokers, whereas the rate of probably symptomatic and pseudo-hypoglycemia is lower for smokers. In Figure 1, the CGM measures of time spent in hyperglycemia, time spent in range, time spent in hypoglycemia, and measures of variation are shown for smokers versus nonsmokers after 16 weeks of treatment. Smokers spent more time in hyperglycemia and less time in range. Time below 70 mg/dL (3.9 mmol/L) was not different for smokers compared with nonsmokers, but time below 45 mg/dL (2.5 mmol/L) was statistically significantly higher for smokers compared with nonsmokers. Smokers’ IG fluctuated more, but the CV was not statistically significantly different. Figure 2 illustrates daily median CGM profiles and interquartile ranges for smokers versus nonsmokers. Furthermore, it illustrates the daily percentage of readings below 45 mg/dL (2.5 mmol/L). For the median profiles, smokers in general have a higher IG level except during the evening, and the interquartile range is slightly wider as a result of more glycemic variation. From the percentage of time in hypoglycemia, smokers tend to spend more time in hypoglycemia especially in the morning (around 9 AM). From insulin pump data, the average time for bolus insulin infusion for smokers and nonsmokers during mornings (6-10 AM) was 08:04 and 08:21, respectively.
Characteristics at Baseline of People Included in this Study.
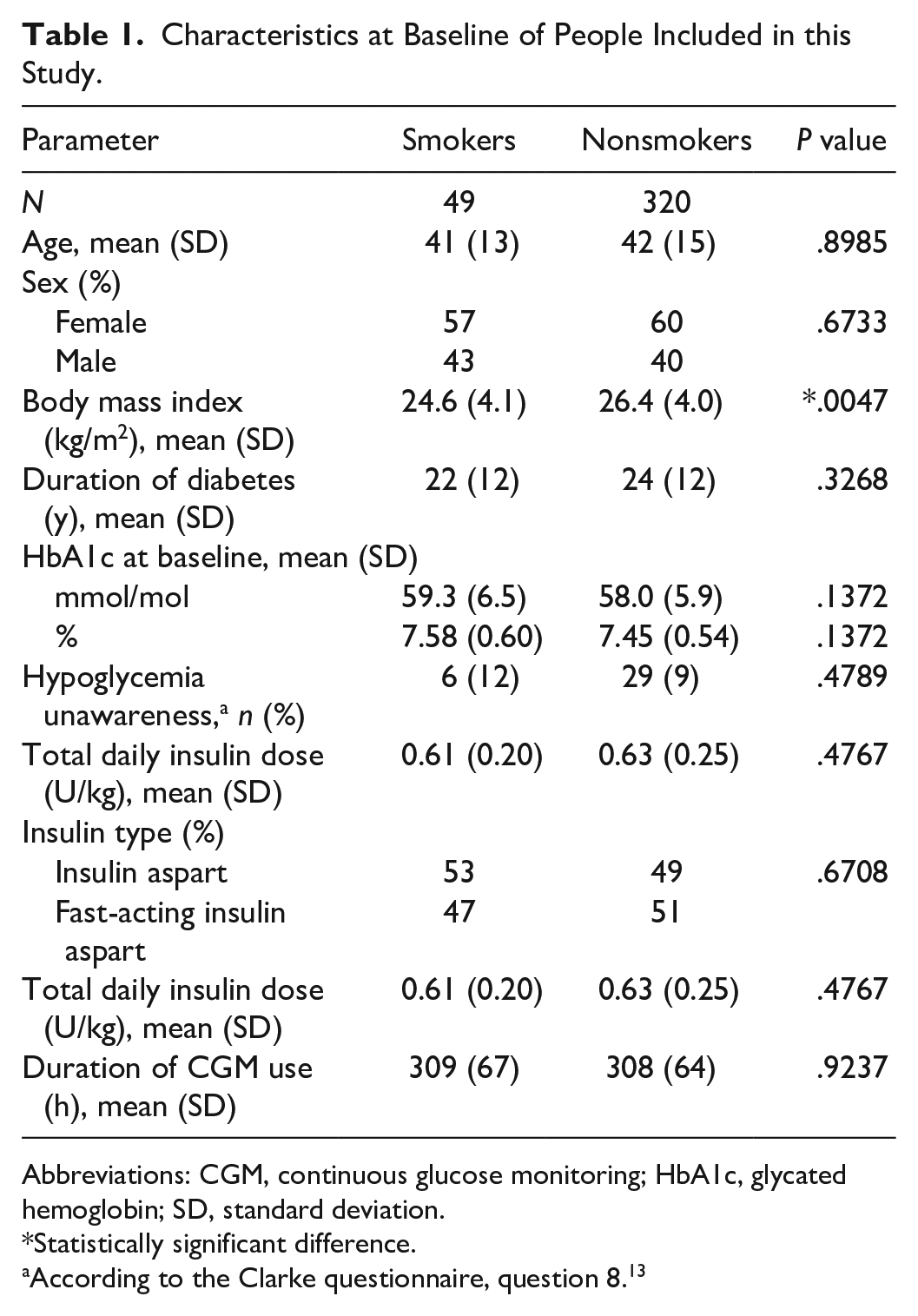
Abbreviations: CGM, continuous glucose monitoring; HbA1c, glycated hemoglobin; SD, standard deviation.
Statistically significant difference.
According to the Clarke questionnaire, question 8. 13
The Estimated Effect of Smoking on Risk of Not Achieving Glycemic Target.
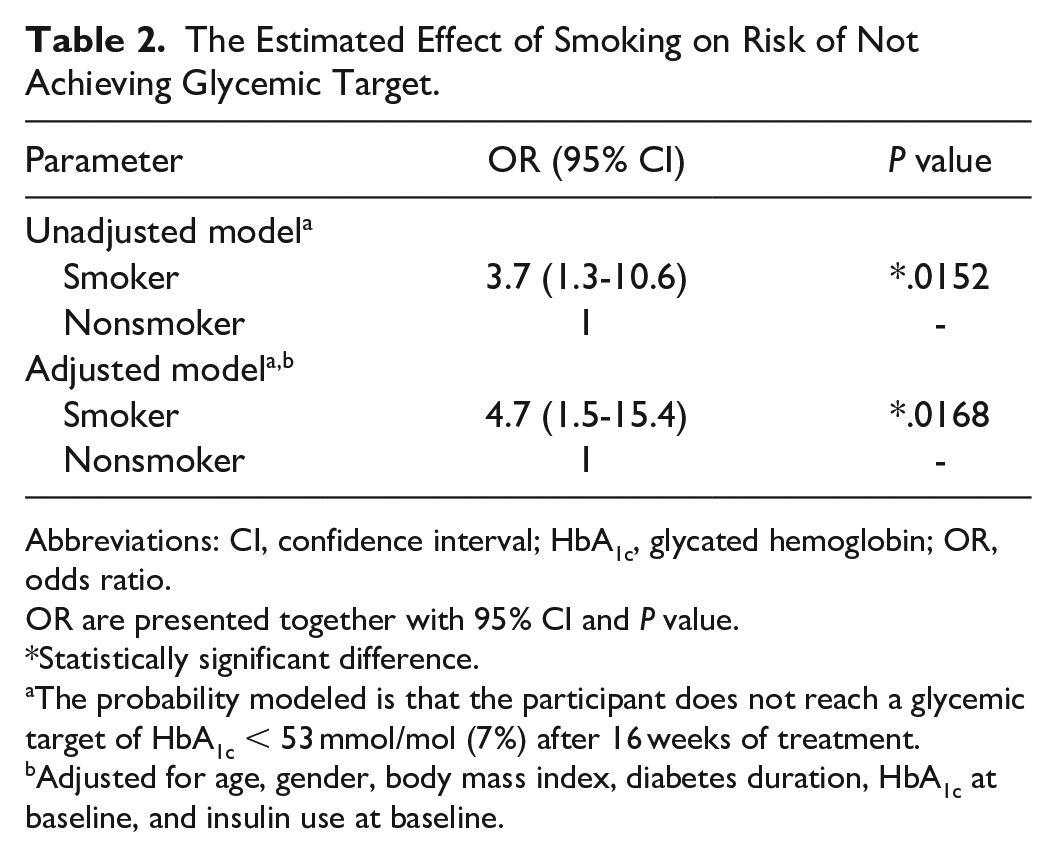
Abbreviations: CI, confidence interval; HbA1c, glycated hemoglobin; OR, odds ratio.
OR are presented together with 95% CI and P value.
Statistically significant difference.
The probability modeled is that the participant does not reach a glycemic target of HbA1c < 53 mmol/mol (7%) after 16 weeks of treatment.
Adjusted for age, gender, body mass index, diabetes duration, HbA1c at baseline, and insulin use at baseline.
Number and Rate of Hypoglycemic Episodes After 16 Weeks of Treatment.
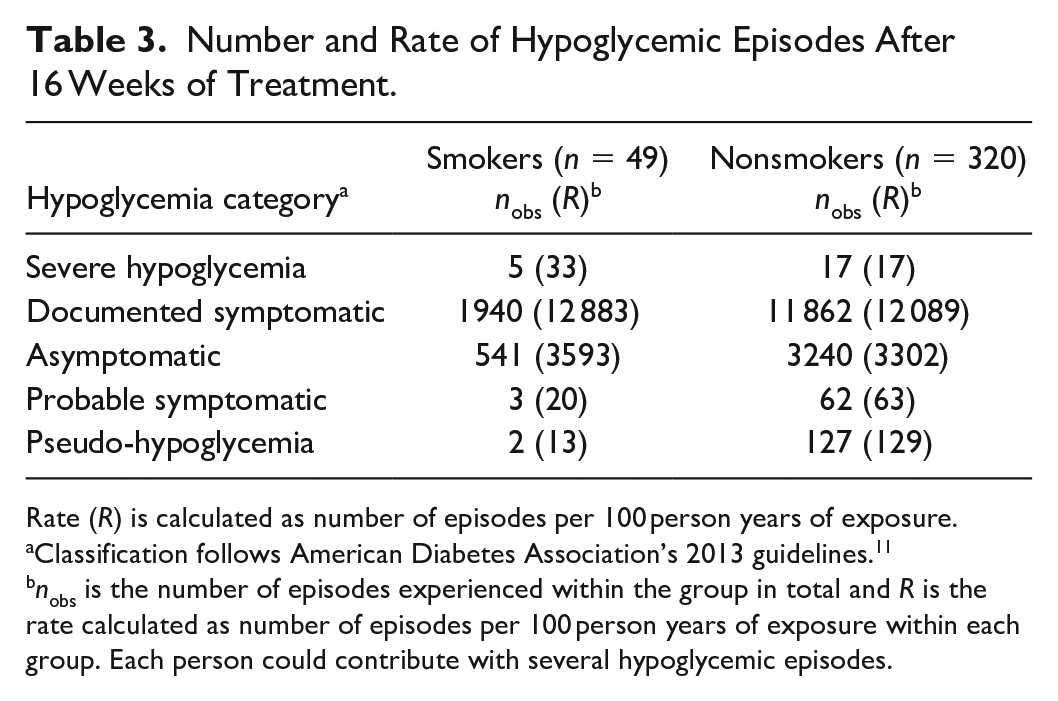
Rate (R) is calculated as number of episodes per 100 person years of exposure.
Classification follows American Diabetes Association’s 2013 guidelines. 11
nobs is the number of episodes experienced within the group in total and R is the rate calculated as number of episodes per 100 person years of exposure within each group. Each person could contribute with several hypoglycemic episodes.
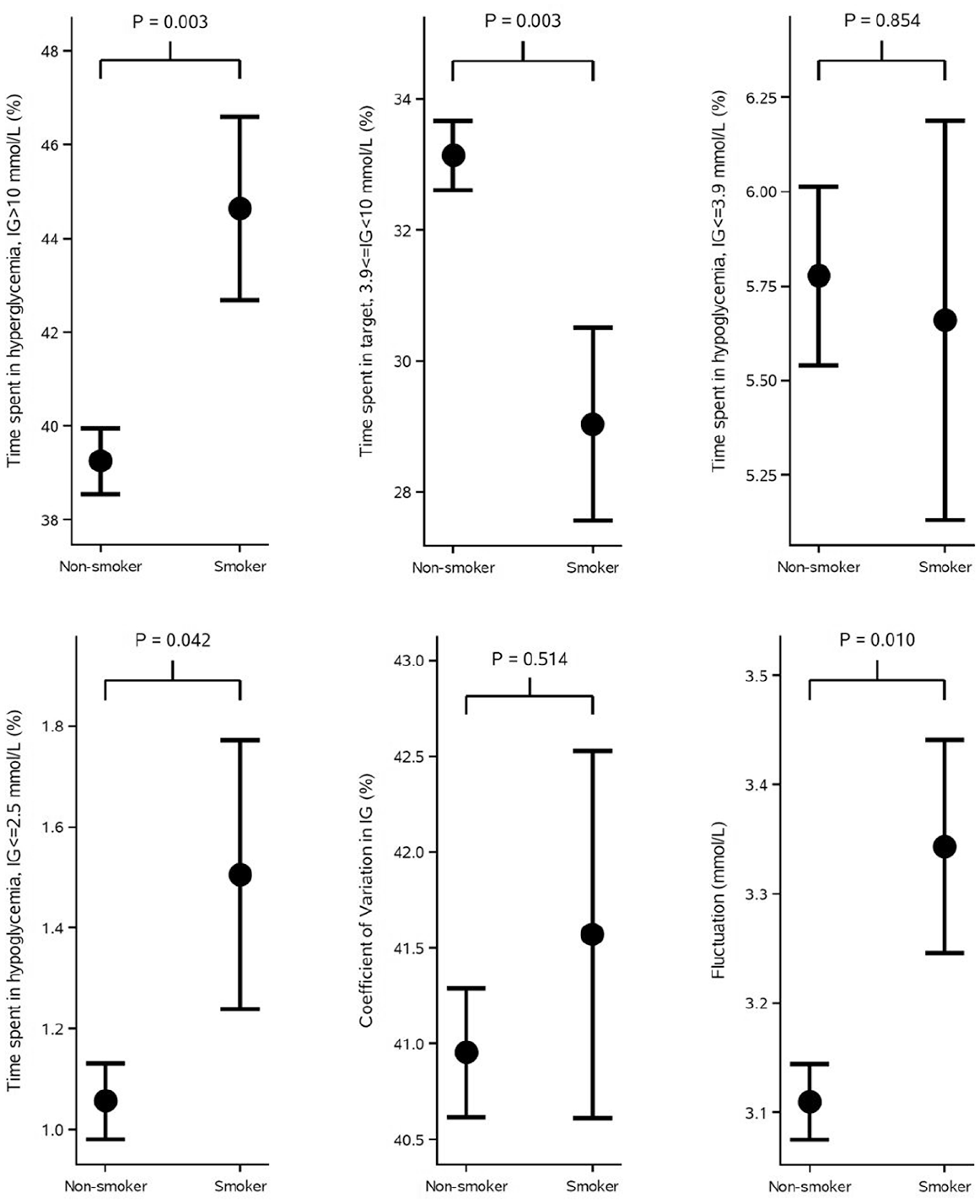
Measures of glycemic variability at the end of treatment period shown as mean and standard error of the mean. Time spent in hyperglycemia, time spent in range, time spent in hypoglycemia, and measures of variation are shown for smokers vs nonsmokers. The P values are from general linear models analyzing the effect of smoking on each of the measures. The models were adjusted for age, gender, body mass index, diabetes duration, HbA1c at baseline, and insulin use at baseline. HbA1c, glycated hemoglobin.
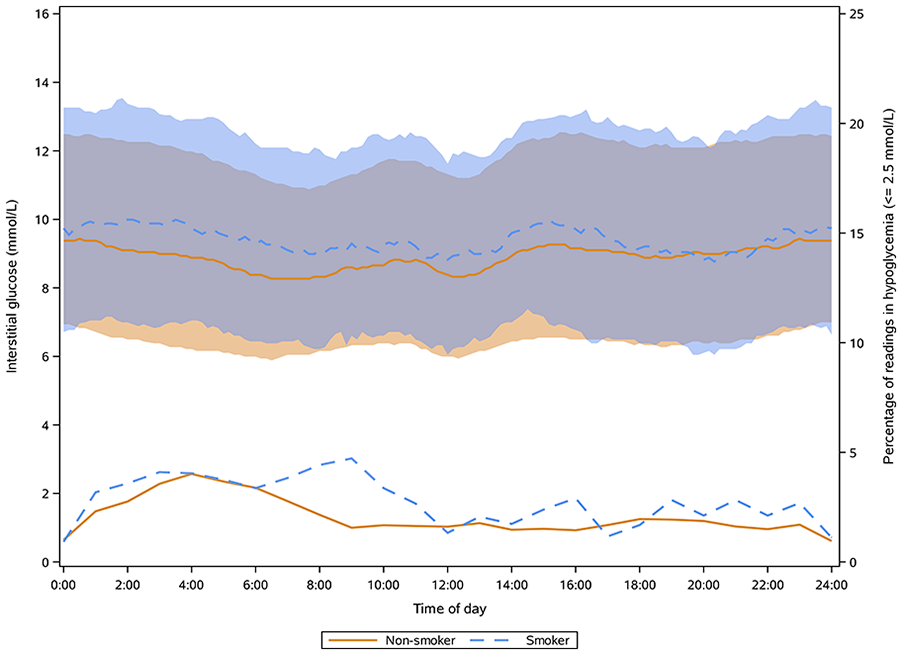
The upper two curves are the median CGM values across all days of all participants and the bands are the interquartile ranges. The lower two curves are the percentage of readings in hypoglycemia defined as IG ≤ 45 mg/dL (2.5 mmol/L) and they are associated with the second y axis. CGM, continuous glucose monitoring; IG, interstitial glucose.
Discussion
This study found that smoking is associated with inadequate glycemic control and increased glycemic variability for people with type 1 diabetes. Smokers had increased time in hyperglycemia, decreased time in range, increased time below very low glucose levels, and increased fluctuation in IG (not confirmed by CV though) after 16 weeks of continuous subcutaneous insulin infusion of either fast-acting insulin aspart or insulin aspart. Descriptive statistics underpinned the increased risk of hypoglycemia with an almost doubled rate of severe hypoglycemic episodes for smokers compared with nonsmokers after 16 weeks of treatment. Daily CGM profiles showed that the excessive time in hypoglycemia among smokers is primarily driven by post-prandial excursions during the morning.
The 4.7-fold increased risk of not achieving glycemic target found in this study is similar to the 4.2-fold increased risk found in the cross-sectional study by Melin et al 5 investigating smoking among people with type 1 diabetes. The lower 1.5-fold risk found in the study by Peng et al 6 may be attributed to that they investigated smoking among people with type 2 diabetes. In an observational study of 537 people with type 1 diabetes, Hirai et al 7 found that smokers had an adjusted 2.6-fold greater risk of severe hypoglycemia compared with nonsmokers, which is in line with the doubled rate of severe hypoglycemia for smokers in this study. The reason for the increased risk of not achieving target might be due to the mechanisms triggered by smoking. Targher et al 4 showed in an insulin clamp study of 28 smokers and 12 nonsmokers with type 2 diabetes that total glucose disposal was markedly reduced for the smokers, leading to the conclusion that cigarette smoking aggravate insulin resistance in people with type 2 diabetes. However, this is not a likely explanation in this study as the insulin use among smokers was lower than nonsmokers.
The increase in glycemic variability for smokers compared with nonsmokers from the CGM measure of fluctuation is in line with the other reported excursions. However, the increased glycemic variability was not confirmed from the CGM measure of CV. Although CV has become a standard CGM measure for assessing glycemic variability, it has some limitations; for example, it does not capture frequency of oscillation in IG and it assumes a normal distribution of IG values. A right skew of CGM values observed in case of more values in the lower glucose range than in the upper will not lead to substantial changes in CV as compared to fluctuation (based on our calculation), which may explain the difference in the two measures in this study.
From the baseline characteristics of the participants in this study, no statistically significant differences existed, except for BMI. The increased weight among nonsmokers compared with smokers is confirmed in literature. 14 Lower BMI is both associated with increased glycemic variability and increased risk of severe hypoglycemia15,16; however, the estimated effects of smoking on CGM measures of glycemic variability were in our study adjusted for BMI and the skewness is thus minimized. The cause behind the observed risk of morning hypoglycemia among smokers is thus difficult to identify, and it would require additional background information about the participants, which is not available. For example, Lohse et al 17 demonstrated that heavy smoking is associated with an unhealthy lifestyle including less consumption of fruit and vegetables and high alcohol intake, and, especially, the latter is important for the risk of hypoglycemia. The risk of hypoglycemia is increased 12-16 hours after alcohol consumption due to depletion of hepatic glycogen stores and reduced hepatic insulin sensitivity. 18 Even moderate consumption of alcohol in the evening increases the risk of post-prandial hypoglycemia after breakfast the following day. 19 If alcohol intake during the evening was higher in the group of smokers compared with nonsmokers in this study, the subsequent increased risk of morning hypoglycemia could be explained by this phenomenon. Moreover, smoking is associated with depression and overall less self-management of health needs, 20 and smokers may in general show lesser belief in health care professionals, 21 which could also increase risk of glycemic variability and hypoglycemia.
The lack of information about alcohol consumption and other lifestyle factors is therefore a limitation. A limitation is the generalizability of our results as the population is not representative to all people with type 1 diabetes around the world. For example, data were obtained from a study conducted only in Europe and North America, and people from developing countries are thus not represented. Furthermore, people in this study are well controlled with respect to HbA1c due to the inclusion criteria, and addition of people with higher HbA1c levels could change the results dramatically. A limitation is that the relatively old Dexcom G4 sensor is inaccurate compared to today’s sensors. However, we anticipate that our conclusions would be the same even though a newer sensor was used. Another limitation is that only 49 smokers were present, which resulted in few severe hypoglycemic episodes for comparison and as a result, too little power for any statistical analysis. A strength of the study is the high validity of data.
Conclusions
In conclusion, smoking is associated with inadequate glycemic control and increased glycemic variability for people with type 1 diabetes manifested in an almost five-fold increased risk of not achieving glycemic target and increased time in hyperglycemia, decreased time in range, increased time in low hypoglycemia primarily driven by morning hypoglycemia, and increased fluctuation. Since randomization was not stratified according to smoking, it is important to emphasize that we cannot conclude on a causal relationship between smoking and the observations described above. It is important for clinicians to know that if the patient has type 1 diabetes, is smoking, and is suffering from hypoglycemia, a preemptive action to high HbA1c levels should not necessarily be treatment intensification due to the risk of hypoglycemia, especially during the morning. Focus could instead be on advices regarding basal insulin rate adjustment, carbohydrate intake, insulin-to-carb ratio adjustment, wearing a CGM with hypoglycemia alarms, and management of other lifestyle factors.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
