Abstract
Background:
The increasing offering of patch-based medical devices is accompanied by growing numbers of reported adverse skin reactions. Procedures for testing leachables according to ISO 10993 may not be optimal for lipophilic substances that can be mobilized on skin by sweat and sebum. We propose an improved extraction method for targeted analysis of leachables using low volumes of a sweat-sebum emulsion. The approach is illustrated by the analysis of isobornylacrylate (IBOA), a compound found in some devices and suspected for allergenic potential.
Method:
Three patch-based products were tested: an implantable device for continuous glucose monitoring (CGM), an intermittently scanned CGM (isCGM) device, and a micro-insulin pump. Quantification of IBOA was performed by gas chromatography and allergenic potential of IBOA levels was assessed by the KeratinoSens cell assay. Different combinations were used for extraction solvent (isopropanol, 5% ethanol-water solution, and sweat-sebum emulsion), extraction volumes (complete immersion vs partial immersion in 2 mm of solvent), and extraction time (3, 5, and 14 days).
Results:
Isobornylacrylate was only found in the isCGM device. About 20 mg/L IBOA were eluted after 3 days in isopropanol but only about 1 mg/L in ethanol-water. Sweat-sebum emulsion dissolves IBOA better and gives a more stable solution than ethanol-water. Decomposition of IBOA solutions requires adjusted extraction timing or correction of results. In the sweat-sebum extract, IBOA levels were about 20 mg/L after 3 days and about 30 mg/L after 5 days, clearly above the threshold found in the KerationSens assay for keratinocyte activation (10 mg/L).
Conclusion:
Extraction by low volumes of sweat-sebum emulsion can be a superior alternative for the targeted simulating-use assessment of leachables in patch-based medical devices.
Keywords
Introduction
In recent years, advances in technology have led to a steadily growing commercial offering of wearable or patch-based medical devices. 1 Especially in the area of diabetes therapy, there is an ever-increasing choice of such devices, eg, insulin pumps or continuously measuring glucose devices (continuous glucose monitoring [CGM], including intermittently scanned CGM [isCGM] devices). 2 In the area of self-monitoring of glucose, the use of CGM/isCGM is partially replacing classical spot monitoring by capillary blood glucose meters, even for patients with type-2 diabetes. 3 Also, for the treatment by subcutaneous insulin infusion, the use of (patch-based) insulin delivery systems is steadily replacing pens and is becoming more common. 4
These devices can greatly improve diabetes therapy (eg, the time-in-range for glucose level) and significantly improve the quality of life for people with diabetes.5,6 Some CGM systems are also combined with sensor-integrated and algorithm-supported pumps and represent a cornerstone of closed-loop systems on the way to an artificial pancreas. 7 However, another consequence of this trend is a higher frequency of use and a longer wear time of patches in contact with human skin. 8 Most patches are equipped with an adhesive layer that is applied directly on skin. Through sweat and humidity, skin can come in contact with compounds leaching out from the patches and medical devices themselves. This can be a threat to the patient because both patches and devices are complex mixtures of many material components. Thus, it is not surprising that together with the increasing use of patch-based devices, the number of reported skin-related issues and complications has increased.9-12 A number of patients have claimed to suffer from small effects like rashes but also more severe reactions were reported like itching, skin irritation, or even allergic reaction. Such effects are very annoying and often painful for the patients, frequently leading to discontinuation of the therapeutic use of the device. Additionally, these effects can be associated with an increased diabetes treatment burden and persistent health issues due to emotional distress. 13
The more frequent occurrence of adverse skin related issues has led to increasing research for the causes of these disorders. Several studies and numerous research experiments have been carried out to identify problematic substances in patch-based devices that may come in contact with skin. 8 As a possible root cause (among others), the substance class of acrylates was identified. 14 Acrylates are compounds very often used in glues and adhesive formulations but also frequently found as additives in plastic materials. In several cases, a specific representative of this class was named as being the culprit for allergic and sensitizing skin reactions of patch-based isCGM devices: the substance isobornylacrylate (IBOA, see Figure 1).11,12,15-18 Isobornylacrylate is frequently used as monomer in UV-light-curing resins, adhesives, and reactive diluents of inks and coatings.
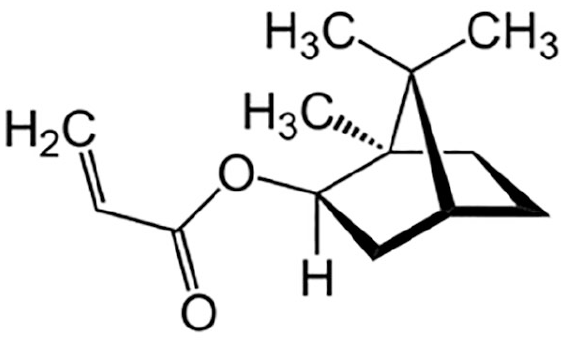
Structure of isobornylacrylate.
These circumstances and findings have encouraged us to investigate whether the methods currently used to test and qualify medical devices can be improved. For regulatory approval, medical devices must be in compliance with national and international laws and standards to avoid or minimize risks for patients. 19 European authorities base their assessment of biocompatibility on the norm ISO 10993 which sets standards for all aspects of testing materials that come in contact with the human body.20-23 In addition to biological testing, the ISO 10993 includes physico-chemical analysis of solutes and verifies compliance with limit values for leachables.24-26 Devices with noninvasive skin contact must be assessed for cytotoxicity, sensitization, and irritation. 27 The tests include both a worst-case extraction for general risk assessment and a simulating-use extraction under realistic conditions for the intended use case (detailed in the ISO 10993 Part 12 and Part 18). The simulating-use test is particularly important for risk evaluation, since it is assumed that the test conditions used here correspond to those during application. Much emphasis is therefore given to the appropriate choice of test conditions both for extraction (eg, amount of sample, sample preparation, choice of solvent, volume of solvent, duration, and temperature of extraction) as well as measurement procedure (eg, analytical method, instrument, and reference materials).
Unfortunately, due to the high variety of materials and applications, it is not possible to give a detailed test requirement and a standard test procedure for each device and intended use case. The ISO 10993 rather sets a general framework and describes guidelines for best test conditions. The choice of the optimal setting is left to the experts from industry and to the authorities involved in approval. In some cases, commercial laboratories specialized in this field assist manufacturers and recommend tests and procedures they can offer.
For the case we are discussing here—patch-based devices with skin contact—the choice of solvents should mimic the effect of skin secretions and of interstitial fluid (for invasive devices with a skin-penetrating component). The simulating-use test is often performed with saline or aqueous solutions with low content of an organic solvent (eg, ethanol in the range of 5%). The worst-case test is frequently carried out with a pure organic solvent like isopropanol or hexane, depending on device compatibility. This general approach is optimal for the overall risk assessment required for regulatory approval, but in the case of targeted analysis of a compound that needs closer investigation it might not be optimal. For example, IBOA is a lipophilic organic compound not soluble in water 28 and scarcely soluble in diluted aqueous alcoholic mixtures. As such, it would not be found in the eluate of an extraction in aqueous saline and is only partially dissolved in aqueous solutions of low alcohol content. Thus, it might be omitted or underestimated in the currently widespread methods for the assessment of leachables according to ISO 10993. However, in a several-day application of a patch on skin, the product is in prolonged contact to sweat. Sweat is a complex mixture wetting skin with a hydrolipidic film,29,30 and besides water, it contains the fat-dissolving component sebum.31,32 Sebum can thus promote continuous leaching of lipophilic compounds like IBOA from patches or devices and thus facilitate migration into skin and body.
To account for lipophilic leachables in patch-based devices, we assessed a new approach for the (targeted) simulating-use extraction. It is still based on the guidelines for sample preparation given by ISO 10993-18 and ISO 10993-12, but instead of using classical solvents it relies on an artificial sweat-sebum emulsion.33,34 Such emulsions are well known in material testing of products with intensive skin contact (eg, shoes, gloves, glasses, and jewelry). 35 They have a higher dissolution power for lipophilic substances and have been suggested for tests that need to mimic real skin conditions36,37 as they are closer to reality than, eg, freshly refined vegetable oil suggested by ISO 10993-12. The sweat-sebum emulsion used in the present study (trade name “Artificial eccrine perspiration/sebum emulsion,” Pickering Laboratories, CA, United States) is designed to mimic nonexercise induced skin-surface film liquids of human sweat. It contains (among other ingredients) a mixture of lipophile compounds like vegetable oils (olive oil and coconut oil), fatty acids (palmitic acid, linoleic acid, and stearic acid), paraffin wax, and squalene.
In addition to a different eluent, we also evaluated a lower extraction volume compared to ISO 10993, based on the actual skin contact surface rather than the total surface of the device. To demonstrate the possible benefit of the new approach, we compared the results of IBOA extraction based on the ISO 10993 (in ethanol-water mixture) and in sweat-sebum emulsion. For both solvents, we applied different extraction volumes and extraction times to include these optimized parameters in the final setting. As representative samples, we chose three patch-based products from the currently available medical devices for personal diabetes management: the transmitter of an implantable CGM sensor, a needle-type isCGM sensor, and a micropump for insulin delivery. The isCGM sensor and the insulin pump are applied with a transdermal inserter to be active in interstitial fluid for a few days. The wear time of the isCGM sensor is 14 days and that of the insulin pump 3 days. The CGM-sensor is subcutaneously implanted for 180 days and used with a noninvasive patch-based transmitter. The patch for the transmitter is available in two variants (white or transparent patch) and is renewed daily.
The results include IBOA analysis from the eluates by gas chromatography with flame-ionization detector (GC-FID) and an assessment of the allergenic potential of the found IBOA levels by a standardized cell assay (KeratinoSens assay).
We also performed stability tests of IBOA in solution, using the solvents and the same conditions as during extraction.
Skin exposure to a sensitizing allergenic substance may result in clinical manifestation of allergenic contact dermatitis. Adverse outcome pathways (AOPs) for skin sensitization describe the sequence of substance-induced pathophysiological events, starting with a molecular event that initiates a sequence of early cellular events ultimately resulting in an observable effect. 38 The four key events (KEs) of the AOP for skin sensitization are the covalent binding of electrophilic substances to skin proteins (also considered to be the molecular initiating event KE1), induction of cytoprotective gene pathways and inflammatory responses in keratinocytes (KE2), events in dendritic cells (eg, maturation of dendritic cells KE3), and events in lymph nodes (eg, T-cell priming and proliferation KE4). 39 Together, these four KEs reflect the sensitization phase of substance-induced allergic contact dermatitis. The activation of keratinocytes by KE1 results in transcriptomic changes. This altered gene expression pattern is induced by the activation of the nuclear factor erythroid 2-related factor 2 (Nrf2). In the presence of electrophiles, the repressor Kelch-like ECH-associated protein 1 (Keap1) normally binding to Nrf2 is modified at reactive cysteine residues on the surface 40 leading to the release of Nrf2. Free Nrf2 can then migrate to the nucleus and bind to its cognate DNA element, the antioxidant response element (ARE), thereby activating a battery of mainly cytoprotective genes. 41 The Nrf2 pathway is the molecular pathway most widely upregulated by sensitizers. 42 A cell-based method for the hazard identification and risk assessment of potential allergenic compounds uses human keratinocytes harboring the luciferase reporter gene under the control of an ARE. This commercially available KeratinoSens assay was adopted as OECD technical guideline 43 and was used in the present study to assess the allergenic potential of IBOA.
Methods
The tested products were purchased from pharmacy stores or from commercial suppliers for diabetes products. Samples were unpacked and subsequently assembled for the intended use according to the instructions given in the package leaflet. When ready, instead of application on skin, the products/samples (including patches) were introduced into the extraction vessels and the corresponding solvent was added (see subsequent section).
Extraction in a Mixture of Ethanol-Water (5:95 v/v) and Isopropanol
Airtight sealable vessels were chosen such that the vessel size was minimal but every sample could lie flat on the bottom of the vessel. Each sample was placed in an appropriate vessel and an individual volume of extraction solvent was added so that the sample was completely covered or only immersed in 2 mm of solvent. Solvent volumes are given in Table 1. Each product was extracted twice. Blanks were generated with the same vessels, solvents, and procedures but without samples. Samples were extracted for 72 hours/120 hours/236 hours (3 days/5 days/14 days) at 37°C (water-ethanol mixture), respectively, 50°C (isopropanol) in an incubator with very gentle shaking (60 rpm). An IBOA standard solution of 10 or 20 mg/L was included for stability testing to investigate any change in concentration over time, eg, due to temperature stress. Solutions were kept in the dark.
Extraction Volumes.
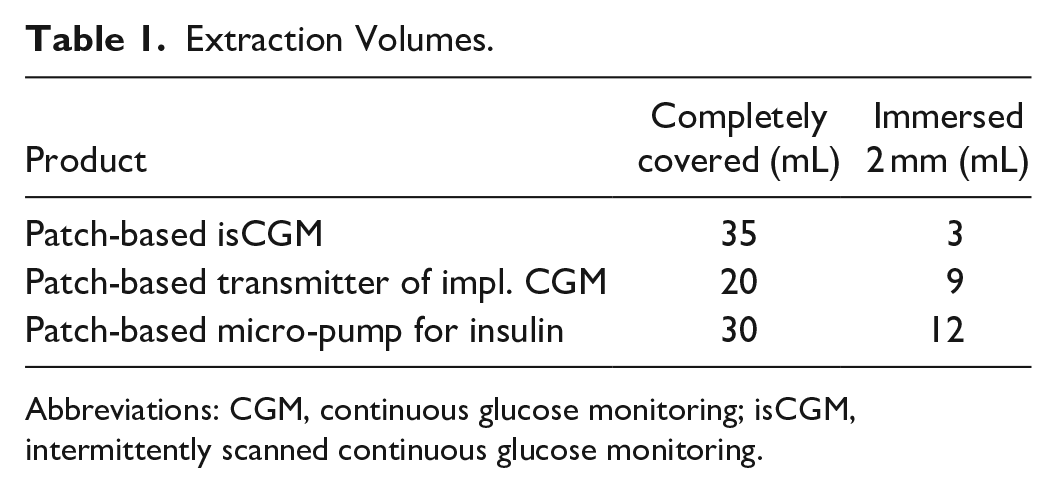
Abbreviations: CGM, continuous glucose monitoring; isCGM, intermittently scanned continuous glucose monitoring.
After extraction, the eluates of ethanol-water were diluted 2:1 (1+1) v/v with isopropanol to protect the column and the detector from overloading with water. The eluates of isopropanol were not changed. Each eluate was transferred to two GC-vials and two injections were performed from each vial. Solvent blanks were analyzed between samples to avoid carry-over. When carry-over was observed, additional ethyl-acetate blanks were injected. Settings of the GC-FID instrument are summarized in Table 2.
Experimental Settings for the Gas Chromatography With Flame-Ionization Detector Measurements.

Abbreviations: FID, flame-ionization detector; GC, gas chromatography.
For calibration, IBOA standard solutions were freshly prepared in the respective extraction solvent. A stock solution of 100 mg/L IBOA was diluted to 5 to 7 concentrations in the range of 0.5 to 100 mg/L.
Extraction in Sweat-Sebum Emulsion
The same vessels and procedures were used as described above. The extraction volume of sweat-sebum emulsion (Artificial eccrine perspiration/sebum emulsion, Pickering Laboratories, CA, United States) was adapted such that samples were only immersed in 2 mm of solvent. Extraction was performed at 37°C as described above. After extraction, an aliquot (500 µL) of the emulsion was diluted 2:1 (1+1) v/v with isopropanol and centrifuged for 15 minutes at 1000 rpm. The supernatant solution was transferred into two GC vials and analyzed as described above without further cleanup. A number of additional peaks from the ingredients of the sweat-sebum emulsion were visible in the chromatograms, but no interference occurred with the IBOA-peak (see example in Figure 2, retention time of the IBOA-peak is 18.05 minutes).
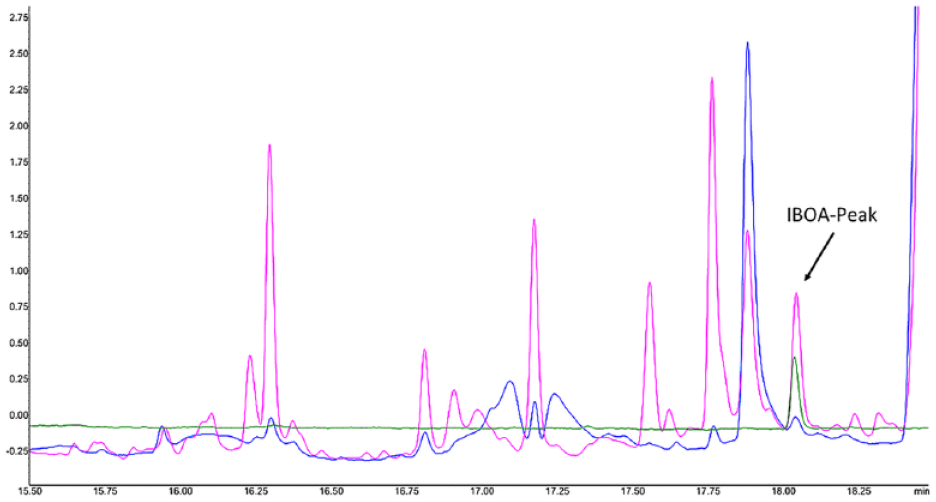
Example of gas chromatography with flame-ionization detector chromatograms of isobornylacrylate solutions (section from 15.5 to 18.5 minutes, isobornylacrylate-peak is at 18.05 minutes). Green line indicates freshly prepared IBOA solution of 10 mg/L in ethanol-water; blue line indicates freshly prepared solution of 0.5 mg/L isobornylacrylate in sweat-sebum emulsion; and purple line indicates solution of 20 mg/L isobornylacrylate in sweat-sebum emulsion after 7 days at 37°C. The peaks of sweat-sebum components do not interfere with the isobornylacrylate, neither in freshly prepared solution nor after a standing time of seven days.
For cell tests, IBOA was dissolved in dimethylsulfoxide (DMSO) and the solution was further diluted with culture medium resulting in a final concentration of 1% DMSO. The KeratinoSens assay was performed as described by OECD guideline. 44 This in vitro skin sensitization test makes use of a human keratinocyte cell line stably harboring a luciferase reporter gene under the control of the ARE of the human AKR1C2 gene. The luciferase signal reflects the activation by sensitizers of endogenous Nrf2-dependent genes. This allows quantitative measurement of luciferase gene induction by luminescence detection. The luciferase activity signal measured in the test reflects the activation of endogenous Nrf2-dependent genes by sensitizers after 48-hour test substance exposure. Test chemicals are considered positive in the KeratinoSens test method if they induce a statistically significant induction of the luciferase activity being equal or higher than 1.5-fold as compared to the solvent control and the cellular viability at the lowest concentration with a gene induction ≥1.5-fold is above 70%.
Results
Extraction in ethanol-water produced clear solutions and did not visually change the samples. This was also true for extraction with sweat-sebum emulsion, the eluates being similar milky solutions as the unused solvent. In the case of isopropanol, the extracts of the transmitter (including the white patch or transparent patch) were cloudy and the patches had separated into distinct layers on a part of the area.
The IBOA-content found in the extracts is summarized in Table 3. Isobornylacrylate was only found in extracts of the isCGM device. When no IBOA was detected, the table lists the limit of detection calculated from the calibration plot performed on the respective day (criteria according to ISO 32645). Isobornylacrylate was also not found in ethanol-water extracts of the isCGM after extraction times of 5 and 14 days (not explicitly shown in Table 3).
Results for Isobornylacrylate Levels in the Eluates After Extraction.

Abbreviations: CGM, continuous glucose monitoring; isCGM, intermittently scanned continuous glucose monitoring.
No isobornylacrylate was found in the worst-case extraction; wearing time is only one day.
No isobornylacrylate was found in the worst-case extraction; wearing time is only three days.
Stability tests showed that IBOA solutions are not stable over time. Concentrations decreased continuously in all solvents (see Figures 3 and 4). In ethanol-water, after 3 days, only 3% of the initial IBOA amount was measured. This was a faster decay than in sweat-sebum emulsion, where IBOA values only decreased to 23% of the initial value within 14 days. In isopropanol (being a nonaqueous, alcoholic solvent), the decay was also lower and stopped after 1 day, resulting in about 70% residual concentration after 3 days. This might be an equilibrium effect between the esters of isopropyl acrylate (formed by the solvent) and isobornyl acrylate. When stored at 5°C, a solution of IBOA in ethanol-water was stable for 24 hours. The relative stability of IBOA in the three solvents used here is compared in Figure 5.
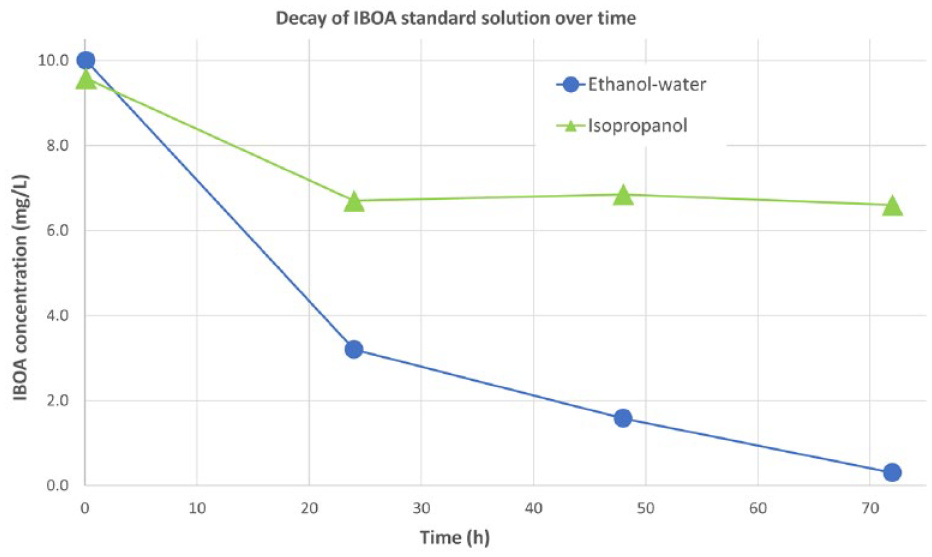
Comparison of the decay of isobornylacrylate concentration over three days’ time in a solution of ethanol-water and isopropanol. Error bars were omitted for clarity (error < 10%).
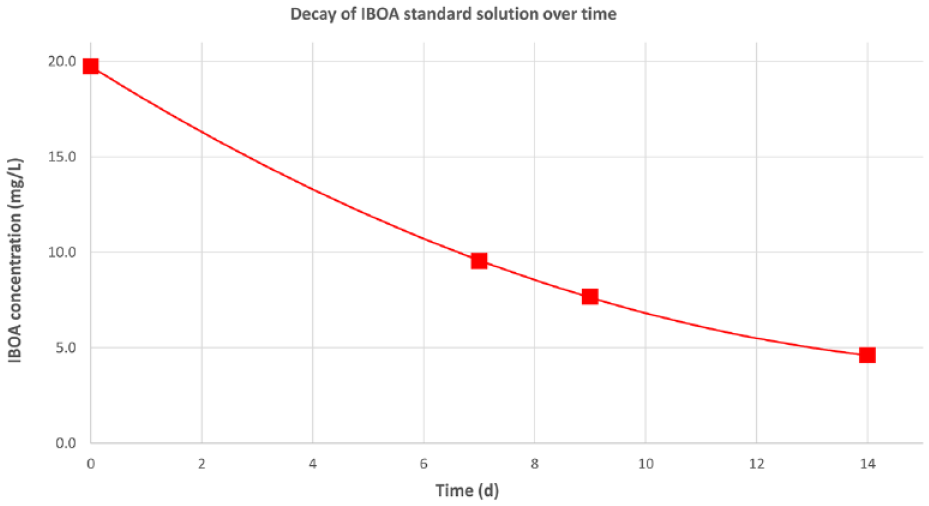
Decay of isobornylacrylate concentration over 14 days in sweat-sebum solution, starting at 20 mg/L. Error bars were omitted for clarity (error < 5%).
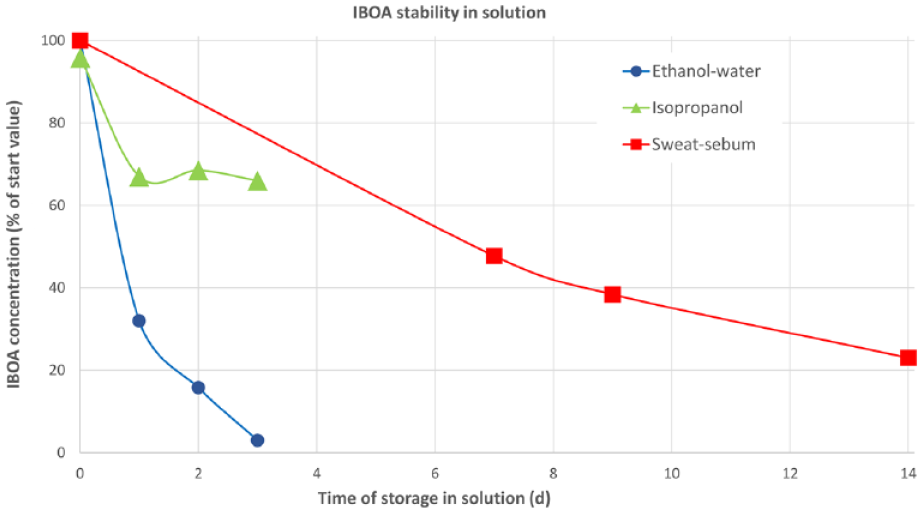
Comparison of isobornylacrylate stability when dissolved in the three solvents used. Error bars were omitted for clarity (error < 10%).
In the GC-data of standard solutions or eluates, no degradation products of IBOA were observed. Isoborneol was checked explicitly and was not detected, most likely due to lower FID detection sensitivity compared to IBOA. Acrylic acid is not sufficiently retained by the used column to be resolved in the elution chromatogram. In standard solutions of IBOA, no peaks were identified that increased during IBOA decay. As a result, calibration based on a sum-parameter of IBOA and degradation products was not possible.
Results of the keratinocyte activation potential of IBOA using the in vitro KeratinoSens assay are depicted in Figure 6. At a concentration of 5 mg/L, IBOA has no significant effect on luciferase expression and cell viability. Isobornylacrylate exposure at higher concentrations (10 and 15 mg/L) activates the keratinocytes causing an increase in luciferase expression greater than or equal to the 1.5-fold threshold value (blue line). In parallel, cell viability is considerably diminished. A concentration of 30 mg/L is toxic, the cells die and are therefore no longer able to induce the reporter gene luciferase. Thus, no luciferase activity is observed. Lactic acid was used as negative control and p-phenylenediamine as a positive control. During a patch test, a solution of 1% p-phenylenediamine (92 µM) showed a sensitization rate of 54% after 48 hours exposure. 45
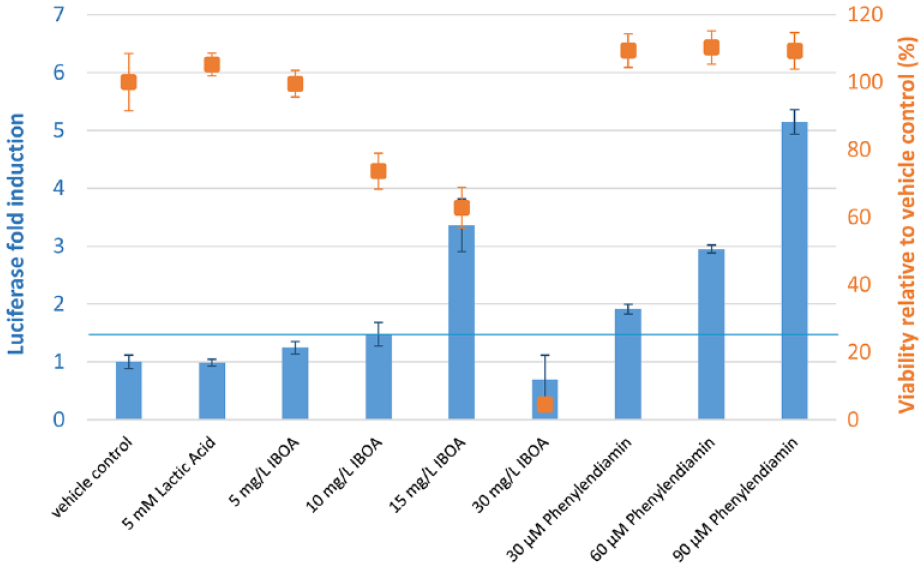
KeratinoSens in vitro cell assay performance toward isobornylacrylate, skin sensitizer p-phenylenediamine, and nonsensitizer lactic acid. Effect of increasing concentrations on the luciferase expression (blue bars) and viability (orange squares). Bars and dots represent mean values; the error bars the standard error of the mean (n = 6). The blue horizontal line marks the luciferase activity threshold being 1.5-fold compared to the solvent control (luciferase fold induction = 1). A total of 5 mg/L isobornylacrylate is considered positive in the KeratinoSens test method inducing a statistically significant 1.5-fold induction of the luciferase activity above the solvent control.
Discussion
With regard to the criteria of extraction solvent, extraction volume, and extraction time, the following aspects have to be considered:
Extraction solvent: Use of a “worst-case” solvent is a very helpful concept that was once again proved here. With this approach, the maximum quantities of extractables in the device can be estimated. Isopropanol is a good “worst-case” solvent for the described application, being nondestructive to the samples and a good solvent for IBOA.
Ethanol-water is not the solvent of choice for the simulated-use extraction of IBOA. The lipophilic sweat-sebum emulsion has the advantage of more readily dissolving IBOA into a more stable solution. Both aspects cause IBOA levels to be higher in sweat-sebum eluates than in ethanol-water. The levels from sweat-sebum eluates (17 mg/L after 3 days and 33 mg/L after 5 days) are above the threshold for keratinocyte activation measured by the KeratinoSens assay (10 mg/L). If IBOA concentrations were corrected for the decay found in the stability tests of the standard solutions, the levels of IBOA in the eluates would be in the range of 25 to 55 mg/L (see section "General discussion" and Table 4), very clearly indicating keratinocyte activation and a risk for skin sensitization. These levels of IBOA are already very toxic to the tested keratinocytes and therefore increase the risk for adverse skin effects.
Isobornylacrylate Concentrations in Solution After One Day of Extraction.

Abbreviation: IBOA, isobornylacrylate.
Extraction volume: Immersion in only 2 mm of solvent gives a better match to the use-case of a device patched on skin than complete immersion. Also, it is comprehensible to consider as relevant only the area of the patch and not the entire surface of the product. With the vessels used here, the immersion in 2 mm of solvent corresponds to an extraction ratio (ie, contact-surface to extraction-volume) of 3.2 cm2/mL which is very close to the recommendation of ISO-10993-12 based on sample surface (ie, sample surface to extraction volume of 3.0 cm2/mL). The amount of IBOA found in the ethanol-water extract of the isCGM device after 3 days complete immersion was about ten times higher than after 3 days immersion in only 2 mm of solvent. Although the result is hampered by the instability of IBOA in solution, this is a significant difference that can lead to a completely different risk assessment. The lower extraction volume is the more realistic approach. Thus, a lower extraction volume should generally be considered, also for the worst-case extraction (for which it was not applied here).
Extraction time: Instability of IBOA in solution and in biological environments has been established, eg, in animal clinical trials. 46 The compound is easily hydrolyzed to acrylic acid and isoborneol. However, in the analysis of leachables, this aspect is rarely discussed and only briefly mentioned in ISO 10993-12. Unstable analytes imply the need for even more closely matched extraction conditions and timing. One way to address this is repeated short-term extraction (eg, for periods of a few hours), closely simulating the effect of sweat on skin. Also, the hydrolysis products might be targeted by chemical analysis, eg, by derivatization of acrylic acid or isoborneol. Alternatively, results after longer elution times can be corrected using a decay-model derived from stability tests. We used the decay-model for IBOA derived from our stability test data to compare corrected mean eluate concentrations after one day for different extractions (see Table 4).
Again, the results clearly show that sweat-sebum emulsion is the preferable extraction solvent. To our knowledge, none of the research published about IBOA analysis in medical devices did discuss or account for a possible decomposition of IBOA during extraction.
General discussion: Although the dataset is limited (eg, only two extractions per test case), the results are sufficiently accurate for valid observations. The results confirm that a worst-case extraction of leachables from a medical device can strongly differ (by more than the tenfold) from a simulated-use extraction. Also, it is confirmed that experimental conditions are crucial for valid results and have to be optimized in detail.
It is all the more important to base risk assessments on a simulated-use test and not on a worst-case test. However, many of the investigations of IBOA in medical devices were performed by extractions using a worst-case approach, and the difference to a use-case was not discussed within the findings.11,12,47 Also in patch-tests for skin sensitization, the used IBOA concentrations are not adapted to levels expected during an application on skin.9,10,16 The usual IBOA concentration used in patch-tests is around 5% (eg, in acetone), with three orders of magnitude higher than concentrations found in the simulated-use extraction discussed here (about 50 mg/L). Such an approach can be misleading or even irrelevant. This is problematic as our results clearly confirm that IBOA has indeed a strong sensitizing potential and poses a serious risk for adverse skin effects.
Conclusion
Low volumes of sweat-sebum emulsion can be a superior alternative for targeted extraction of leachables in medical devices. This approach is a good simulation for testing a patch-based application, thus increasing relevance of the use-case assessment. However, stability of analytes in solution is an important issue that must be addressed in experimental planning.
This new approach should be considered as an industrial standard for the testing of patch-based medical devices in order to reduce patient risks for skin sensitization or other adverse skin effects.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Herbert Fink, Nuno M. de Barros Fernandes, and Jörg Weissmann are full-time employees of Roche Diabetes Care GmbH. Manfred Frey declares no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Roche Diabetes Care GmbH.
