Abstract
Introduction:
The objective of this study is to identify adverse events involving medical devices used in home environments with a focus on patients with diabetes and to categorize the probable causes.
Methods:
The chosen technologies that are fundamental for monitoring blood glucose and used in users’ decision-making were: blood glucose monitor (BGM), continuous glucose monitoring system (CGM) and Insulin Pump. In the search for evidence, two databases from the collection of technovigilance alerts between January 2019 and December 2024 were used: the Technovigilance Analytical Portal of the National Health Surveillance Agency (ANVISA) of Brazil and MAUDE-FDA—USA.
Results:
On the MAUDE-FDA platform, the total number of notifications were: 52,601 BGM, 1,624,664 of CGM, and 1,339,652 Insulin Pump. Strategies to mitigate the occurrence of adverse events were presented, related to human, technological, and environmental factors. Regarding the main problems reported with the patient, they were hypoglycemia and hyperglycemia.
Conclusion:
This paper highlights the need to encourage the practice of reporting to generate evidence and to present strategies to mitigate adverse events, such as developing user-centered technologies inserted in an interdisciplinary ecosystem in the form of a living laboratory; considering accessibility aspects in development and incorporation; developing guidance resources for users; considering metrological aspects to ensure technological reliability; and including sustainability, security, and data privacy actions. Interconnectivity opens up opportunities for the ubiquitous management of technological processes throughout the life cycle.
Keywords
Introduction
Diabetes mellitus is a long-term metabolic disease characterized by high blood glucose levels. 1 Controlling glucose levels is essential to safely manage diabetes and prevent the onset and progression of clinical complications. 2 Medical devices for diabetes, such as blood glucose monitors (BGM), continuous glucose monitoring systems (CGM) and insulin pumps, have been associated with improved glycemic control, quality of life, and user satisfaction.3,4
The conventional method for checking glucose is the finger prick method, performed by the medical device called a BGM system or also a glucometer. It is an electrochemical-based method in which small amounts of blood are extracted from the fingertip, placed on a test strip and the glucose value is generated by means of electrochemical sensors.2,5 The CGM determines blood glucose concentrations through a sensor inserted under the skin. The sensor measures electrical currents in the interstitial fluid, which are proportional to the blood glucose concentration. Additional features include alerts and alarms for rising or falling glucose levels and the ability to share data remotely with family, caregivers, and physicians. 4
Some technologies allow integration with the Insulin Pump, which consists of another medical device associated with diabetes management. This technology administers short-acting or rapid-acting insulin in basal and bolus forms that are programmed to adapt to the patient’s physiological insulin demands. Insulin is administered from a refillable reservoir in the subcutaneous tissue at preprogrammed rates.4,6
For people living with diabetes, access to affordable treatment is essential for their survival. 1 The increased use of medical devices raises concerns about the occurrence of unexpected and potentially fatal adverse events that pose risks to public health. 7 There are several regulations and documents related to the development of medical devices for diabetes for safety and performance, as some of them presented in Table 1. However, there are a variety of factors that interfere with the occurrence of adverse events that can trigger dangerous situations for the population. Adverse events are incidents that cause, or have the potential to produce, unexpected or undesirable results that affect the safety of the patient, operators and other people involved with the technologies.8,9
Regulations and Documents Related to the Development of Medical Devices for Diabetes for Safety and Performance.

The system for monitoring adverse events and technical complaints of health products available on the market is called technovigilance, which aims to protect and provide quality in the use of technology to the population. It consists of an important area of articulation between the actors involved in the life cycle of health technologies, from the development phase to their post-market use. 10 International studies have presented results of the analysis of adverse events in medical devices to alert patients to the potential risks when using different technologies and in different countries. Craig et al 11 present Australian data on adverse events in pulmonary ventilators and urogynecological mesh, Alsohime et al 12 discuss adverse events in an intensive care unit in Saudi Arabia, and Raghav et al 13 present a cross-sectional analysis of adverse events associated with medical devices and radiotherapy devices in India.
The use of real-world data has been used to compare technologies, ensure device quality, investigate safety issues in the user population, and analyze measurement accuracy. 14 Therefore, this article aims to analyze reports of adverse events related to medical devices used by people with diabetes. In addition, we will discuss the evidence that reports the main problems involving the technologies and present recommendations to improve accessibility, reliability, sustainability, and cybersecurity.
Methods
This is a descriptive and retrospective study with a quantitative approach. To perform the analysis of adverse events, the first stage consisted of planning, which involves identifying the medical devices that will be investigated, databases used, analysis period and information that will be extracted. The adverse event reports analyzed were related to the following medical devices used by people with diabetes: BGM, CGM, and Insulin Pump. The adverse event data analyzed were extracted from two databases. In the United States, adverse events are reported to the Food and Drug Administration (FDA) and cataloged in the Manufacturer and User Facility Device Experience (MAUDE) database, 15 which is an essential tool for post-market surveillance that requires high-quality and complete information, especially with regard to reporting sources. 7 In Brazil, adverse events must be reported to the ANVISA, an autarchy that aims to promote the protection of the population’s health, through the NOTIVISA notification system. 10
For each medical device, there are different codes and terms included for data extraction in the databases. The codes present at MAUDE and the definitions of each device are shown in Table 2.
Code of Each Medical Devices to Diabetes and Definition in MAUDE/FDA.
The data analyzed were analyzed between January 2019 and December 2024. The analysis was performed between February and March 2025. After data extraction, the data were exported to a spreadsheet containing the following information: date of notification, risk class, cause and effect of the occurrence, and technical name of the equipment. A spreadsheet was used to analyze the adverse event data to assist in the classification and categorization of data. Subsequently, graphical representations were made regarding the number of notifications, as well as the main problems reported with the device and effects on the patient. Strategies to mitigate the occurrence of adverse events in diabetes technologies have been established, based on human, technological, and environmental factors.
Results
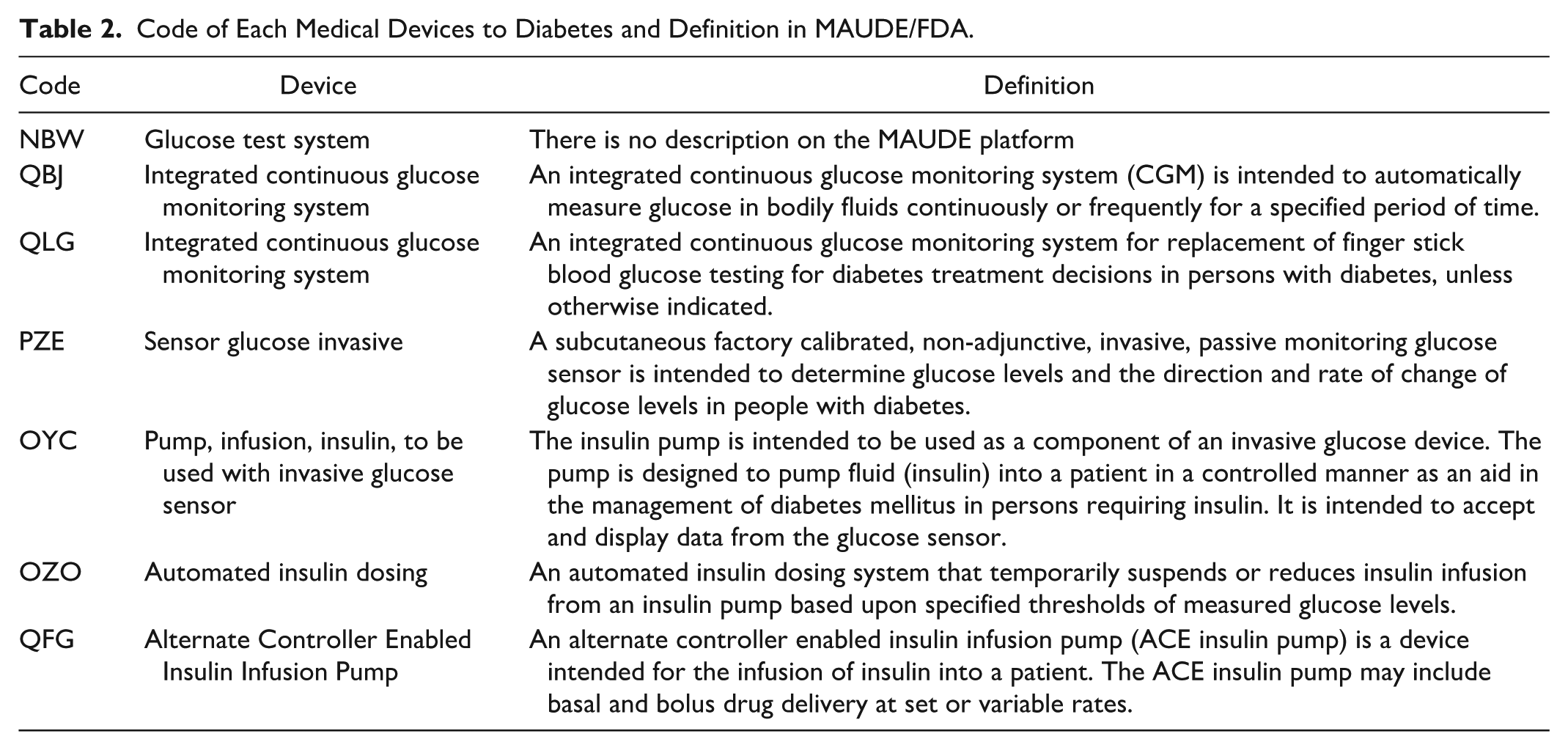
A total of 12,457,700 notifications were recorded between 2019 and 2024 in the MAUDE database. Regarding the medical devices with the highest number of associated notifications are presented by code in Figure 1. The technology with the highest total between the period 2019 and 2024 was Endosseous dental implant (code DZE) followed by infusion pump (code FRN). The other medical devices with the highest number of notifications are: Integrated continuous glucose monitoring system (code QBJ), alternate controller enabled insulin infusion pump (code QFG), automated insulin dosing device system (code OZP), insulin infusion pump (code OYC), automated insulin dosing (code OZO), Noncontinuous ventilator (code BZD), stimulator, spinal-cord (LGW), insulin infusion pump (LZG), prosthesis, breast (FTR), permanent pacemaker electrode (DTB), implantable cardioverter defibrillator (LWS), integrated continuous glucose monitoring system (QLG), pacemaker electrodes (NVN), invasive glucose sensor (PZE), Intravascular administration set (FPA), Continuous ventilator (CBK), automated external defibrillators (MKJ) and ventricular (assist) bypass (DSQ). When analyzing only in the year 2024, the DZE code continues to take first place, however, the QBJ code is in second place in number of notifications. The medical devices for diabetes are among the technologies with the highest number of notifications.
Twenty medical devices with the highest number of notifications in MAUDE/FDA.
The Integrated CGM System with code QBJ totaled more than 1,389,281 notifications during this period. In 2024 alone, there were 347,113 notifications, being the technology with the second highest number of incidents, behind only the bone dental implant this year, with 473,635 notifications. Alternate controller enabled insulin infusion pump (code QFG), automated insulin dosing device system (code OZP), insulin infusion pump (code OYC), automated insulin dosing (code OZO) remain on the list of medical devices with the highest number of notifications between 2019 and 2024.
The number of technical surveillance notifications for medical devices at ANVISA between 2019 and 2024, was 106,900. On the ANVISA platform, in this same period, the BGM and the CGM are among the medical equipment with the highest number of occurrences, being in fourth and seventh place, respectively. The number of notifications on the Brazilian platform is significantly lower compared to the US platform. Measures to encourage the reporting of problems involving medical devices need to be implemented, both at the organizational level and among citizens who can also report problems with the technology. A study conducted by Carlos de Souza et al 16 corroborates the discrepancy in the number of notifications between the ANVISA and MAUDE/FDA systems for ventilator and defibrillator technologies. The researchers are unaware of a comparative study between the databases for the different diabetes technologies, which will be presented in the following sections.
Below is the individual analysis for each of the medical devices, Glucose Monitor, CGM System, and Insulin Pump.
Analyses of Notifications in BGM
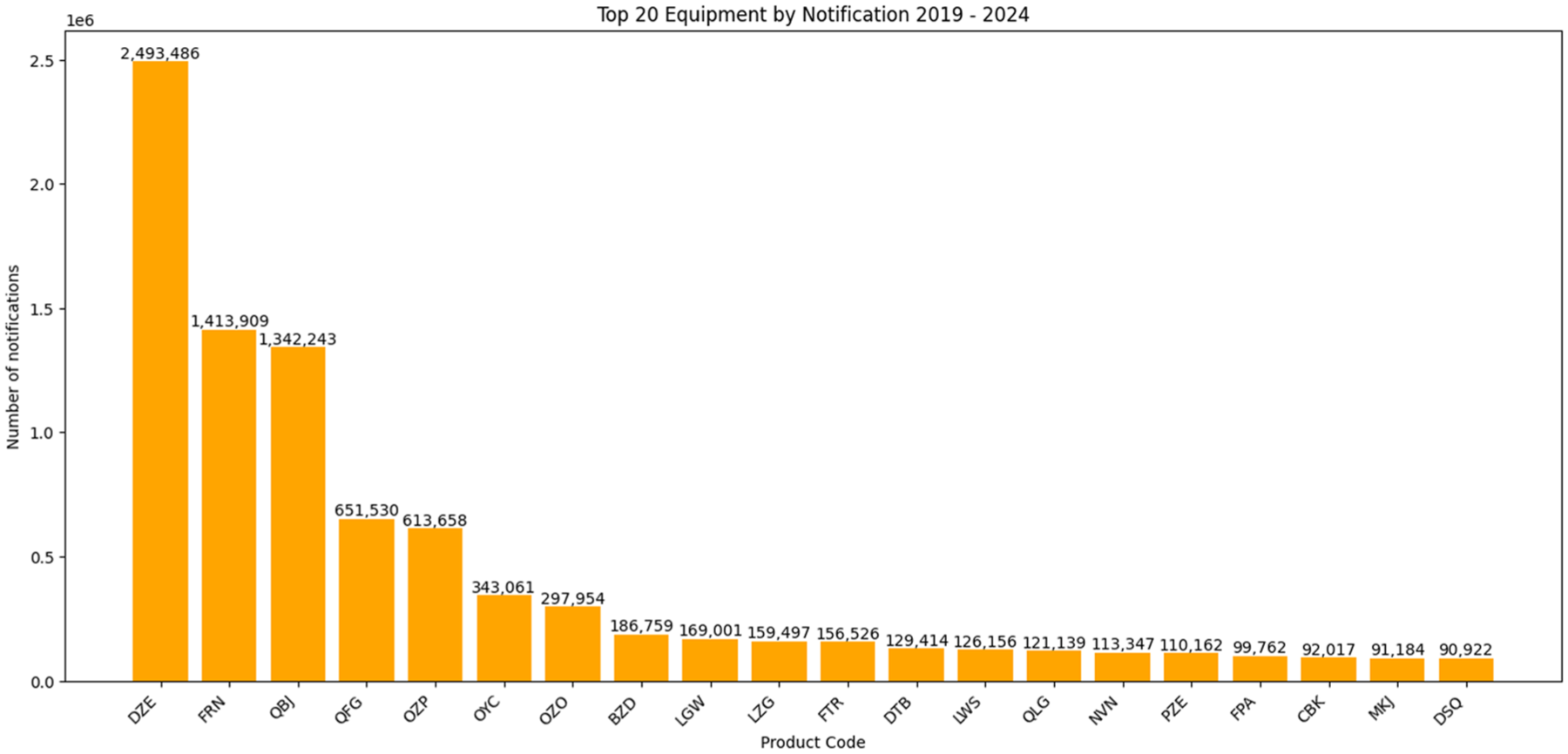
The total number of notifications per year regarding BGM on the MAUDE/FDA platform with the code NBW was 52,601, as shown in Figure 2.
Notifications on MAUDE/FDA in glucose monitor medical device with code NBW.
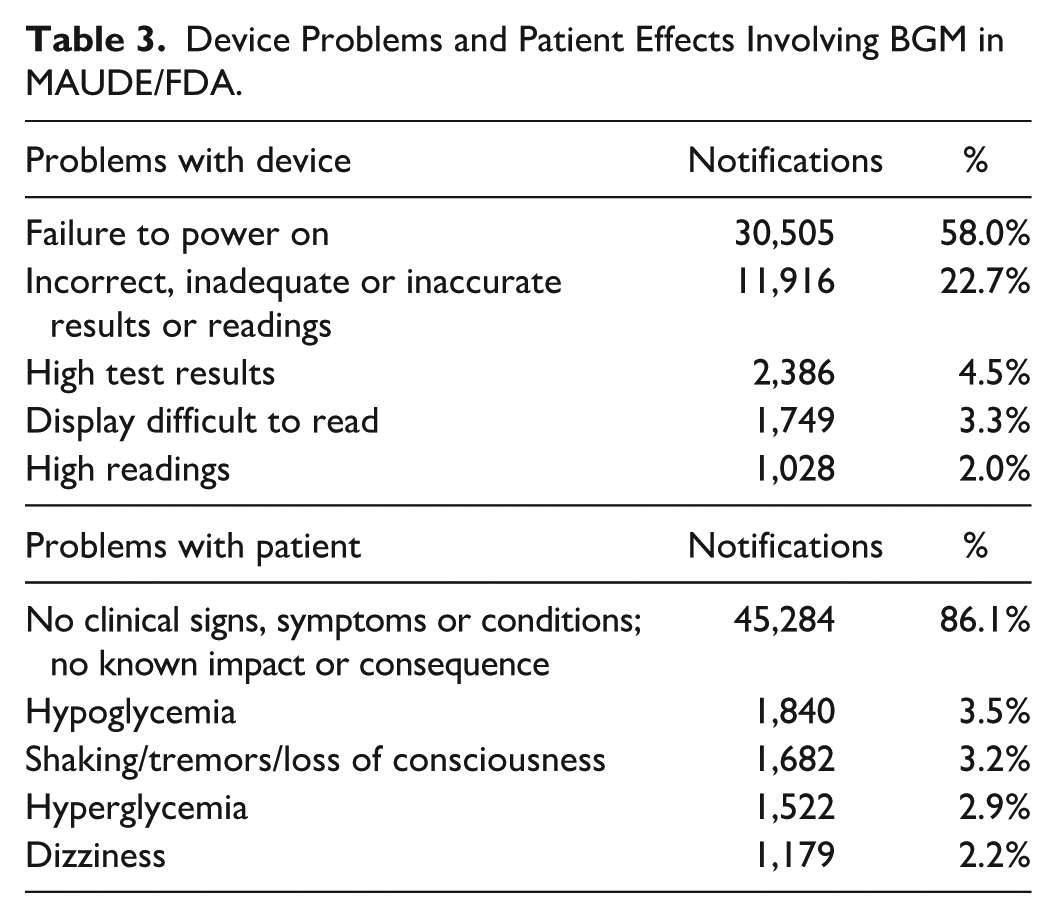
Regarding the reported problems related to the device and effects on the patient is shown in Table 3.
Device Problems and Patient Effects Involving BGM in MAUDE/FDA.
In the analysis of the technovigilance notifications in the public database at ANVISA, the data related to the glucose monitor with the respective terms involved are presented in Table 4.
Notification at ANVISA Regarding the Glucose Monitor.
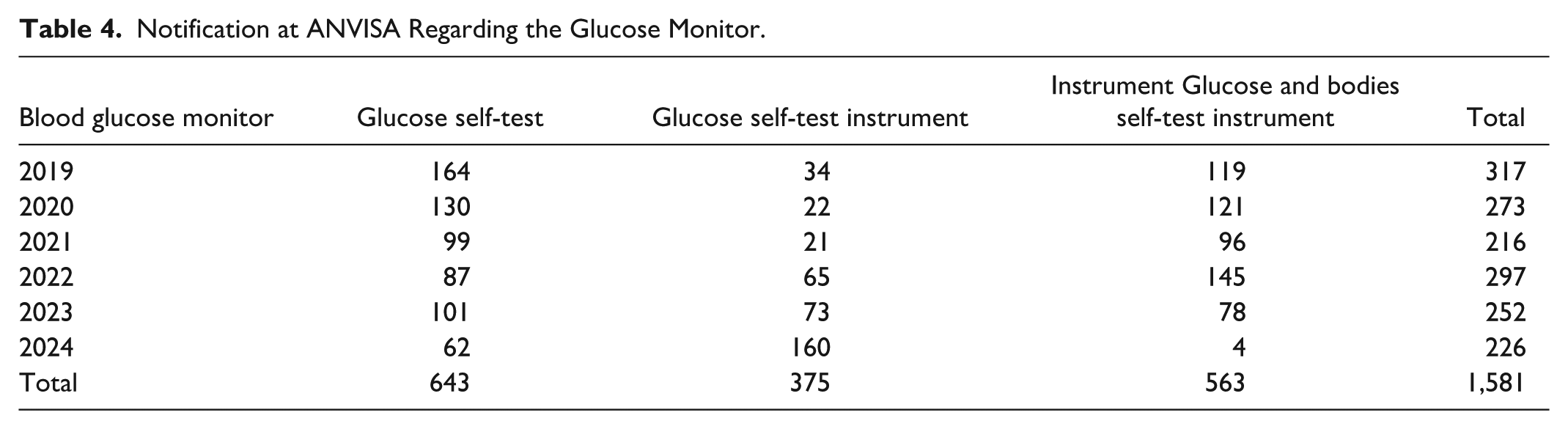
In both databases analyzed, a decrease in the number of technology notifications was observed between 2019 and 2024. One of the reasons for this decrease may be associated with the increase in other medical devices for diabetes on the market, such as CGMs.
Analysis of Notifications in CGM
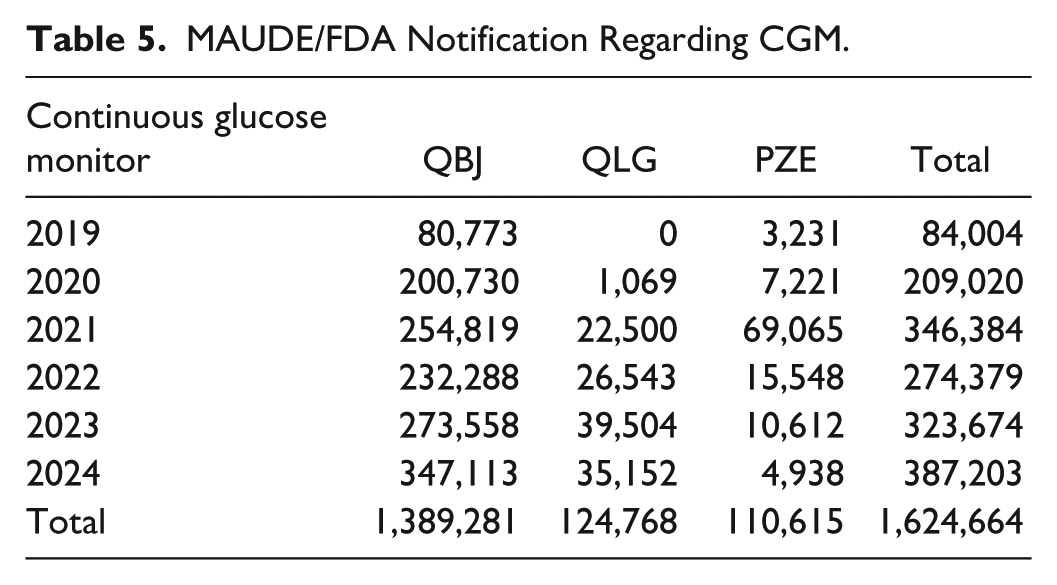
The total number of notifications per year regarding CGM on the MAUDE/FDA platform with codes QBJ, QLG and PZE was 1,624,664, as shown in Table 5. There is an increase per year in the total notification of this technology.
MAUDE/FDA Notification Regarding CGM.
When specifically analyzing the QBJ code, which had the highest number of notifications regarding this technology, an increase in the number of notifications can be observed in recent years, as shown in Figure 3.
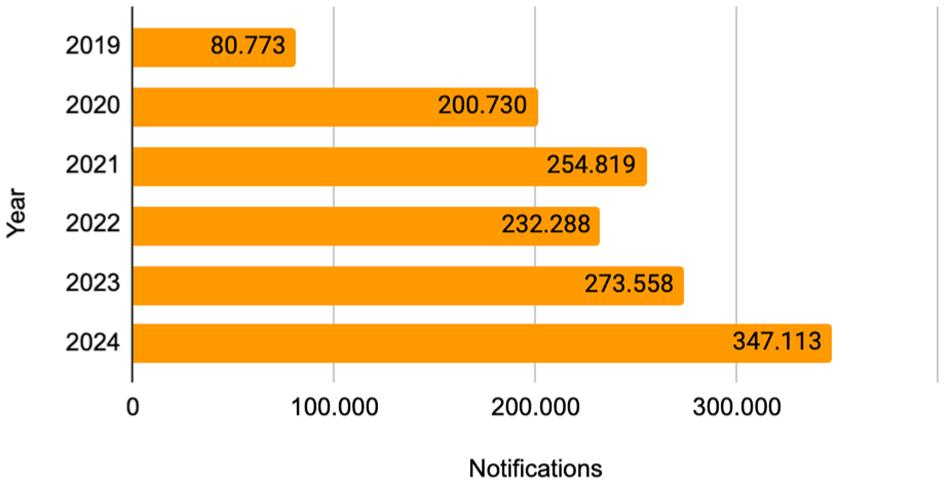
Notifications in MAUDE/FDA in glucose monitor with code QBJ.
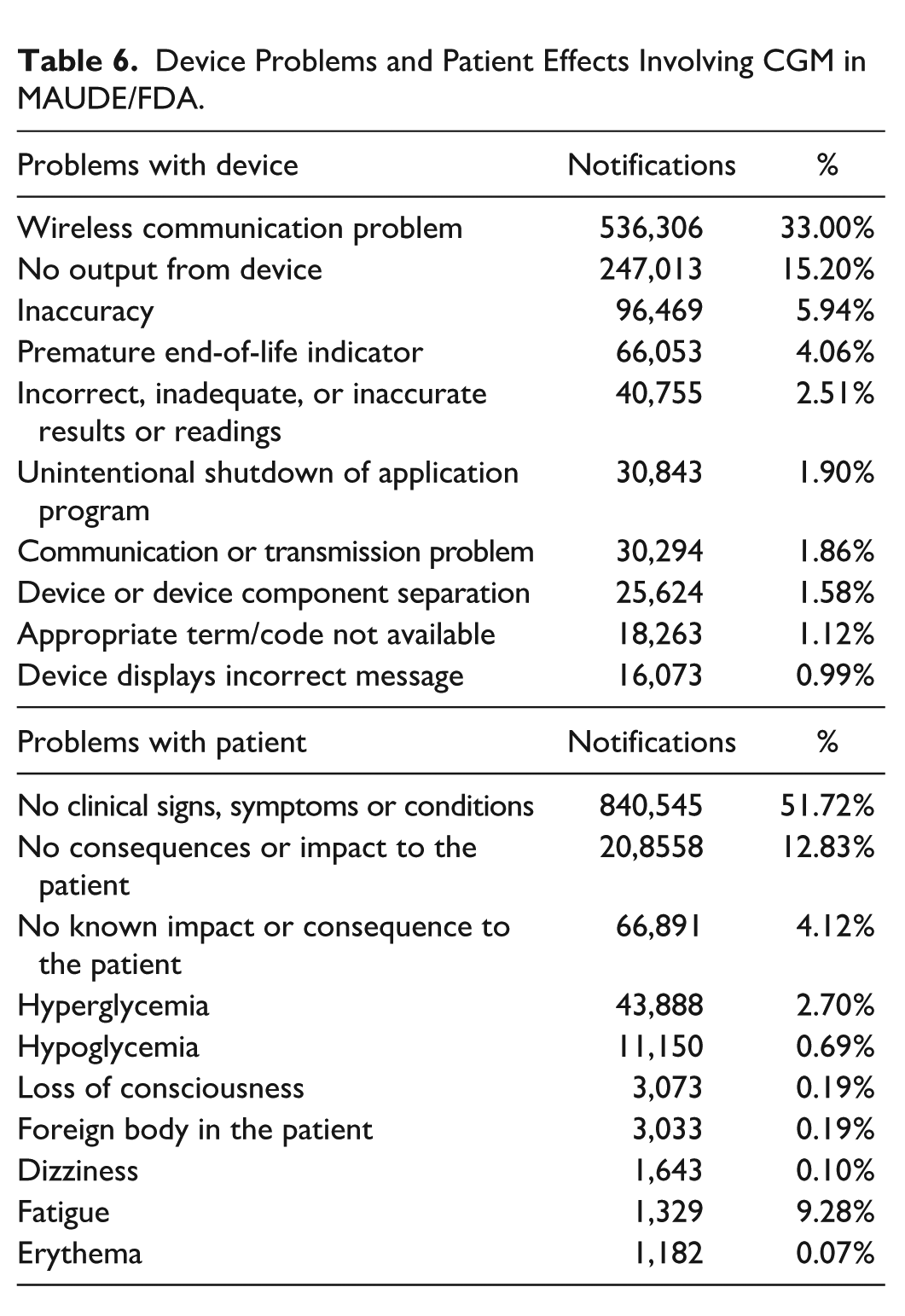
Regarding the reported problems related to the device and the effects on the patient is shown in Table 6.
Device Problems and Patient Effects Involving CGM in MAUDE/FDA.
In the analysis of technovigilance notifications in the public database at ANVISA, the data related to the CGM System are shown in Figure 4.
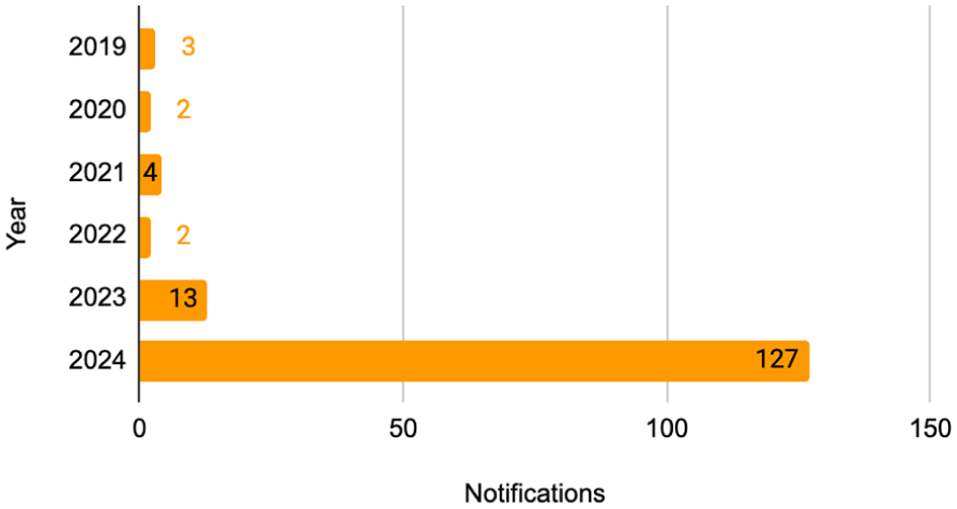
Notification to ANVISA regarding the CGM device with the name self-test for interstitial fluid glucose monitoring.
A total of 151 notifications were analyzed. A large increase in the number of notifications is in line with the increased availability of technology in society. A significantly smaller number when compared to the number of notifications in MAUDE/FDA, however, it shows significant growth in recent years. Output problems, incorrect results, unintended functions and incorrect messages are among the main problems reported, and in relation to the effect on the patient, convulsions and/or loss of consciousness, hypoglycemia and hyperglycemia.
Analysis of Notifications in Insulin Infusion Pump
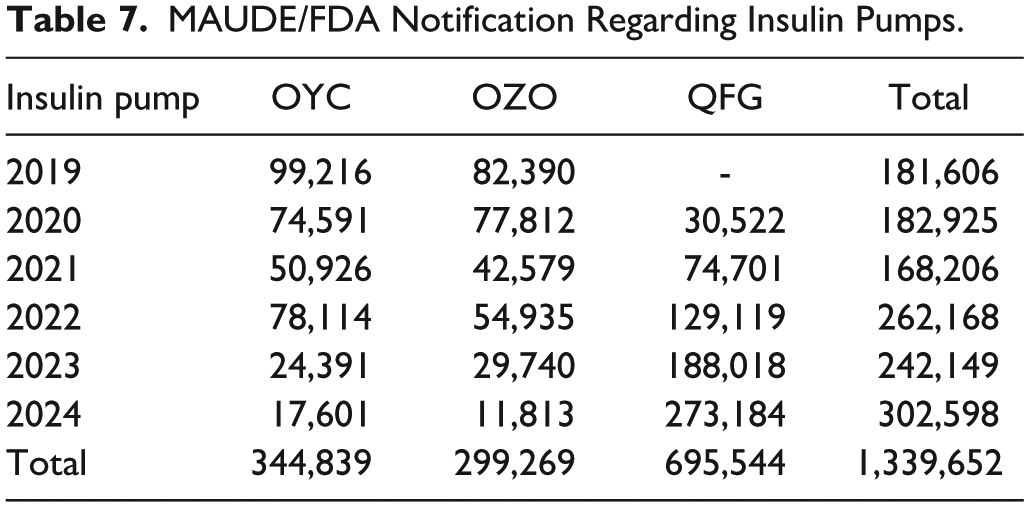
The total number of notifications per year regarding Insulin Pumps on the MAUDE/FDA platform with codes OYC, OZO and QFG was 1,339,652, as shown in Table 7.
MAUDE/FDA Notification Regarding Insulin Pumps.
Regarding the QFG code, as shown in Figure 5, a significant increase in the incidence of notifications was observed in insulin pumps.
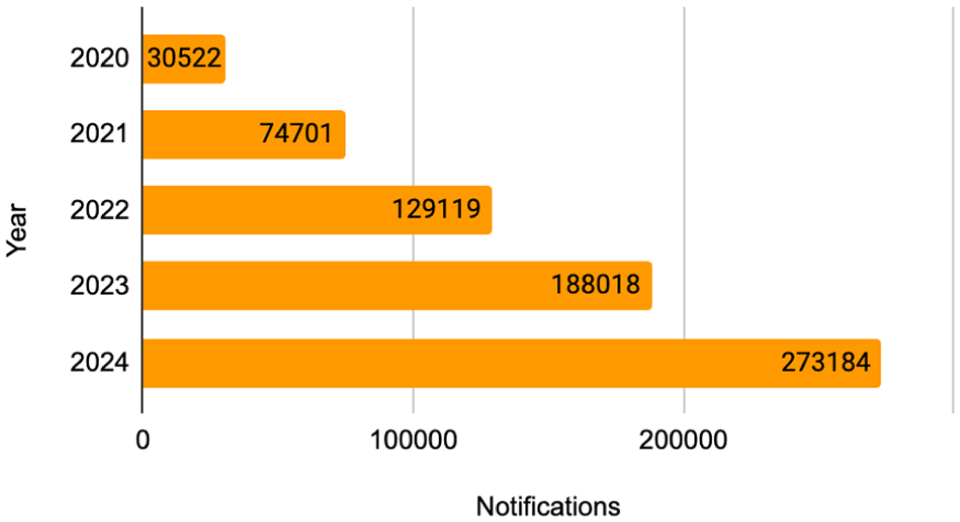
MAUDE/FDA notification regarding insulin pump with code QFG.
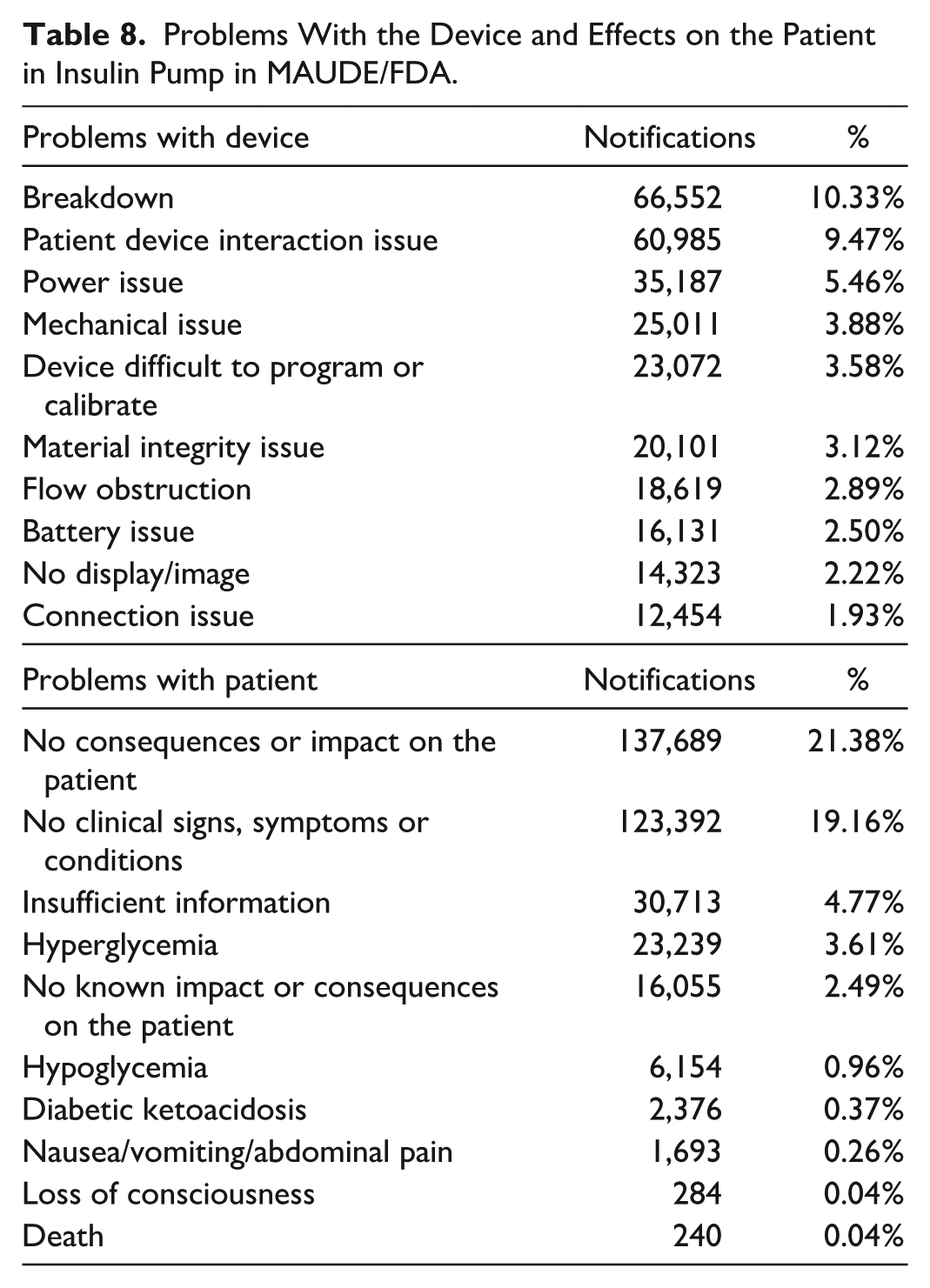
The main problems reported involving the device and the effects on the patient are shown in Table 8. Hyperglycemia, hypoglycemia and diabetic ketoacidosis appear next as the main damages to users, in convergence with what was reported with the insulin pump with code QFG.
Problems With the Device and Effects on the Patient in Insulin Pump in MAUDE/FDA.
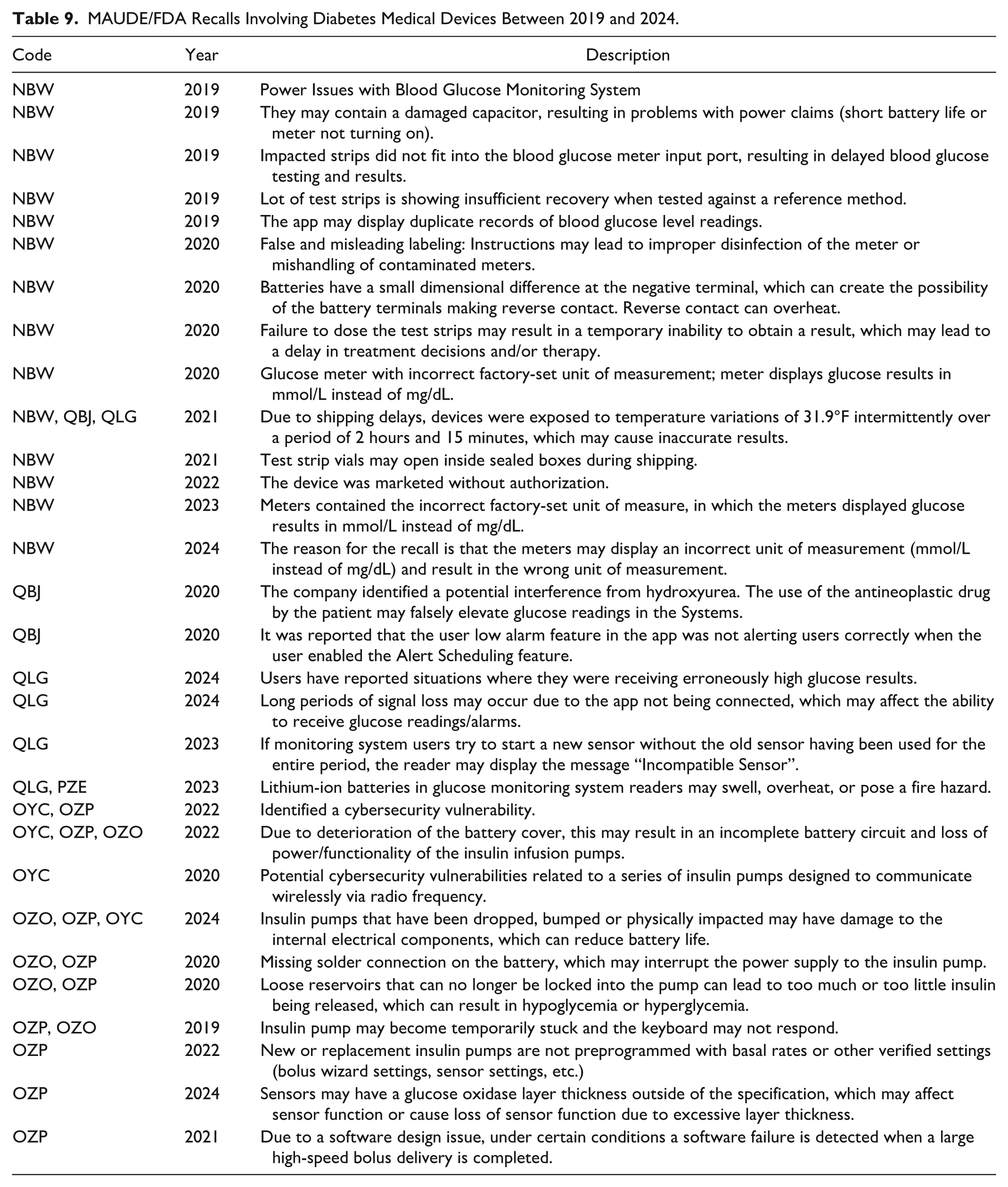
During this analysis period, it was analyzed 30 recalls (field actions carried out by the manufacturer) relating to the medical devices analyzed, as shown in Table 9. Some reports of safety implications were related to the battery, erroneous labeling, incorrect unit of measurement, damaged components that compromise the correct administration of insulin, use of the technology in environmental conditions that can cause inaccurate results, and cybersecurity vulnerability.
MAUDE/FDA Recalls Involving Diabetes Medical Devices Between 2019 and 2024.
An analysis by year was performed regarding recalls in BGM, CGM, and insulin pump, as shown in Figure 6.
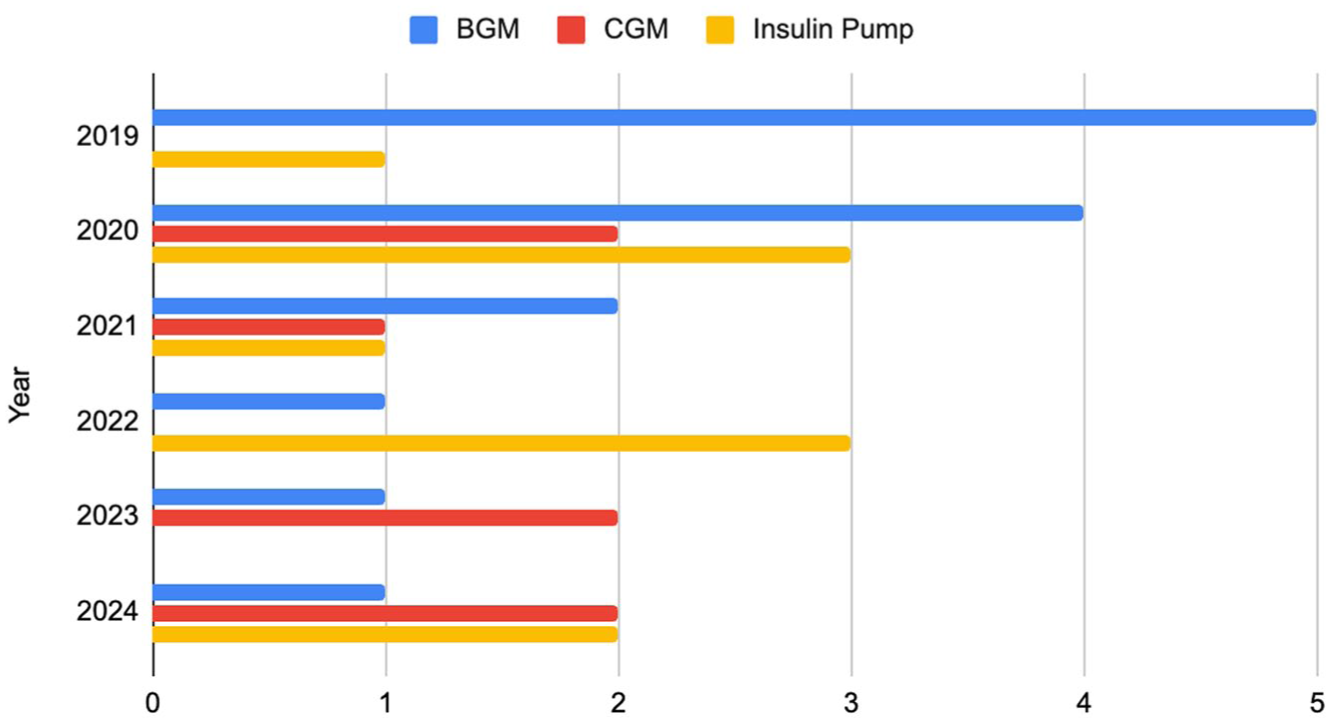
Recall in BGM, CGM, and insulin pump per year in MAUDE/FDA.
Strategies to Prevent Adverse Events in Medical Devices for Diabetes
After analyzing the adverse event, recommendations to preventive actions to mitigate adverse events in medical devices for diabetes were proposed based on human, technological, and environmental factors, reliability, and data interconnectivity, such as:
Provide user training on improper use: develop more interactive manuals with objective, accessible and easy-to-understand language for users;
Properly cleanse skin before use, as dirt can interfere with measurements;
Use with care to avoid dropping the equipment;
Supplies must be placed properly;
Develop accessible technologies with intuitive interfaces so that people with different profiles can use medical devices: consider population diversity in the development process
Periodically analyze device battery conditions;
Properly store devices and their supplies;
Properly dispose of supplies: Syringes, needles, lancets, disposable pens, insulin pump materials and other waste are involved with medical devices for diabetes and throwing these supplies in the regular trash, in addition to contaminating the environment, can also cause injuries and transmit diseases.
Analyze connection and data integration issues, as well as data security aspects such as cyber vulnerabilities to prevent data leaks and device programming changes.
Monitor the reliability of measurement results to avoid inaccurate diagnoses and inappropriate treatments.
Develop and implement protocols for safe use and reporting of adverse events, such as lack of procedures for proper use and adverse event reporting.
Discussion
The occurrence of adverse events in medical devices for diabetes has a direct impact on the quality of care, as these technologies are operated by the users themselves who use the technological resource to make decisions in the monitoring and/or treatment of diabetes. Problems involving medical devices have the potential to cause effects on the patient, ranging from discomfort and local pain to hypoglycemia and hyperglycemia that can lead to worsening of the clinical condition and even death of the patient. Therefore, strategies to mitigate the occurrence of adverse events in medical devices in homecare must be applied from the pre-marketing to the post-marketing stages.
Accessibility should be considered from pre-market development of new technologies to post-market. Developing accessible technologies is essential to ensure access to medical devices for all people. 17 People with visual impairments may not be able to operate properly without audible features and tactile indicators; people with hearing impairments may not understand alarms and auditory alerts, causing distress and impacting the appropriate use of the CGM system, as also discussed by Toschi and Munshi. 18
As discussed by Tsai et al, 3 it is important to assess and address potential cultural barriers. Recent studies demonstrate large racial, ethnic, gender, and socioeconomic disparities in the use of diabetes technology. 19 Daly and Hovorka 6 also discussed in her work that it is essential to ensure equity of access to diabetes technologies across nations and socioeconomic groups.
Regarding BGM, in convergence with Sly et al and the results obtained in the analysis of notifications in the databases, factors that interfere with the accuracy of glucose monitoring may be: inadequate storage of the monitor and test strips; improper preparation of the finger, with dirt; incorrect procedure; problems with the device battery, lack of maintenance and/or performance testing to assess measurement error; difficulty in reading the display; test strips with expired validity; operation of the device in environmental conditions (pressure, humidity and temperature) outside the range defined by the manufacturer; use of certain substances in the body that may influence the result as well as physiological aspects. 5
Regarding CGM, in convergence with the study conducted by Freckmann, transient connection failures between the transmitter and the receiver are among the most reported device problems in the study, in line with the analysis of the notifications in this work. Discrepancies in measurement results, system failures, problems when applying the sensor to the user’s skin, sensor dropped or dropped before the end of the wear period are other problems involving CGM. 20
Regarding the insulin pump, according to Heinemann, insulin infusion errors can occur due to pump failure, blockage of the insulin infusion set, problems at the infusion site, insulin stability problems, user error, or a combination of these. Users are exposed to significant and potentially fatal risks such as interruption of insulin infusion that can result in hyperglycemia and ketoacidosis, as well as excessive administration of insulin that can cause severe hypoglycemia. 21
Diabetes medical devices are increasingly connected to each other, on mobile devices, and on the Internet. This potentially exposes them to exploits such as unauthorized access, incorrect glucose readings, and incorrect insulin dose delivery. As discussed by Ho, several insulin pump manufacturers have faced cybersecurity issues in recent years involving the exposure of confidential patient information and vulnerabilities in the functionality of the devices, which have led to recalls and lawsuits. The unauthorized access could be used to administer too much or too little insulin, through the administration of an unintended insulin bolus or because insulin delivery is delayed or interrupted, which could lead to hypoglycemia or hyperglycemia. 22
Connectivity issues in diabetes technologies are among the most frequently reported, and this aligns with FDA guidance, which warns patients about a safety concern regarding diabetes devices such as CGMs and insulin pumps that rely on a smartphone to deliver critical safety alerts. An FDA statement stated that it has received reports from users that alerts are not being delivered or are not being heard, which is consistent with the data presented in this study. The absence of these alerts may have contributed to serious harms, including severe hypoglycemia, severe hyperglycemia, diabetic ketoacidosis, and death. 23 The Brazilian regulatory agency ANVISA also issued a safety report due to reports from users of continuous glucose monitors who did not receive or hear alarms from their smartphone or smartwatch apps. This may be related to the alarm setting on the smart devices, leading to interference in the emission of the alert signal from the apps associated with the CGM sensors/transmitters, whether in the detection of low sugar levels (hypoglycemia) or high sugar levels (hyperglycemia) in the blood. 24
Many incidents are not reported to regulatory agencies, resulting in significant underreporting. Studies estimate that only 0.5% of adverse events involving medical personnel are reported, and this number may be even lower in official reports. 25 There is a strong culture of underreporting among healthcare professionals and users, with the main barriers being fear of blame, lack of time, lack of perceived effectiveness in reporting, linguistic inequality, lack of accessibility, and lack of knowledge of the reporting system. Another limiting factor is the lack of standardized medical device nomenclature; sometimes, the same device has more than one name or code associated with it. Another limitation concerns the quality of reported data, which may not represent the reality of medical device problems. It is suggested that a culture of reporting adverse events in databases be disseminated and encouraged, raising awareness of its importance for preventing errors in healthcare settings.
To mitigate adverse events involving medical devices for diabetes, a holistic and interdisciplinary view is required. In convergence with Yadav et al, 26 collaborative efforts between regulatory agencies, manufacturers, healthcare professionals, and users are needed to overcome these challenges and establish a robust global framework for reporting adverse events in medical devices. To mitigate the occurrence of adverse events in diabetes technologies, we recommend developing medical devices within an interdisciplinary living lab ecosystem and involving diverse users in the development process to improve the technology’s accessibility and usability; developing accessible and intuitive best practice guides with universal design; periodically training users on medical devices by presenting potential failures they may encounter during use; considering the reliability of measurement results to avoid discrepancies in values that could lead to inaccurate diagnoses; properly disposing of waste to make the use of diabetes technologies more sustainable; and implementing measures to ensure data privacy and security.
Conclusion
This work highlights the need to analyze real-world data, such as adverse event reports, to explore device-related issues and mitigate the occurrence of harm to patients. The practice of reporting should be encouraged to generate evidence and develop strategies to improve the documentation and monitoring of adverse events, such as: developing user-centered technologies inserted in an interdisciplinary ecosystem in the form of a Living Laboratory; considering usability and accessibility aspects; developing guidance resources for users; considering metrological aspects to ensure technological reliability; and introducing sustainable actions and data security and privacy. Adverse events involving medical devices for diabetes can lead to serious harm to patients, ranging from hypoglycemia and hyperglycemia to even death.
The limitations of the work are essentially linked to underreporting related to the reporting of adverse events involving medical devices, incomplete reporting forms, diversity of data sources, codes and terms used to describe technologies and cause of the problems that can influence the quality of the data due to the lack of global standardization of nomenclature for medical devices. Reliable, high-quality data are essential for understanding problems and acting on continuous improvements. Interconnectivity opens up opportunities for the ubiquitous management of technological processes throughout the life cycle. Analyzing adverse events must be implemented to manage risks in medical devices used by people with diabetes to contribute to decision-making with safety and reliability.
Footnotes
Acknowledgements
None.
List of Abbreviations
ANVISA, Brazilian Health Regulatory Agency; BGM, blood glucose monitor; (BGM), CGM, continuous glucose monitoring (CGM),; FDA, Food and Drug Administration (FDA),; MAUDE, Manufacturer and User Facility Device Experience (MAUDE), Brazilian Health Regulatory Agency (ANVISA).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
