Abstract
Background:
Knowledge regarding the burden and predictors of hypoglycemia among older adults with type 1 diabetes (T1D) is limited.
Methods:
We analyzed baseline data from the Wireless Innovations for Seniors with Diabetes Mellitus (WISDM) study, which enrolled participants at 22 sites in the United States. Eligibility included clinical diagnosis of T1D, age ≥60 years, no real-time continuous glucose monitoring (CGM) use in prior three months, and HbA1c <10.0%. Blinded CGM data from 203 participants with at least 240 hours were included in the analyses.
Results:
Median age of the cohort was 68 years (52% female, 93% non-Hispanic white, and 53% used insulin pumps). Mean HbA1c was 7.5%. Median time spent in the glucose range <70 mg/dL was 5.0% (72 min/day) and <54 mg/dL was 1.6% (24 min/day). Among all factors analyzed, only reduced hypoglycemia awareness was associated with greater time spent <54 mg/dL (median time of 2.7% vs 1.3% [39 vs 19 minutes per day] for reduced awareness vs aware/uncertain, respectively,
Conclusions:
Over half of older T1D participants spent at least an hour a day with glucose levels <70 mg/dL. Those with reduced hypoglycemia awareness spent over twice as much time than those without in a serious hypoglycemia range (glucose levels <54 mg/dL). Interventions to reduce exposure to clinically significant hypoglycemia and increase time in range are urgently needed in this age group.
Introduction
Approximately 1.25 million people live with type 1 diabetes (T1D) in the United States. 1 The prevalence of T1D among older individuals is increasing worldwide, in part, because the incidence of T1D is increasing and, in part, because improvements in care have increased longevity.2,3 For example, in a follow-up analysis of the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) study, the all-cause mortality in the intensively treated group approached that of the general US population, likely attributed to overall improvements in diabetes management and cardiovascular risk reductions over the mean 27 years. 4 Other estimates, however, continue to demonstrate at least a two- to threefold increased risk of mortality in those with T1D compared to the broader population. 5 Despite the increasing prevalence and improved cardiovascular risk reduction strategies, risks associated with T1D in older adults remains understudied.
Hypoglycemia is one of the major factors limiting glycemic control and can lead to significant morbidity, mortality, reduced quality of life, and higher healthcare costs in patients with T1D across their lifespan.6-10 Older adults with T1D, particularly those with a long duration of disease, 11 are especially vulnerable to the sequelae of severe hypoglycemia, including arrhythmia, cognitive changes, falls leading to fractures, and even sudden death.12-18 Despite these dire consequences, hypoglycemia remains common in older individuals with T1D. Indeed, data from the T1D Exchange registry demonstrated high rates of severe hypoglycemia in older adults with longstanding T1D: 18% of registry participants 60 years or older reported hypoglycemia-related seizure or loss of consciousness within the last 12 months. 11 Thus, there is a pressing need to identify therapeutic approaches that mitigate hypoglycemia risks in this group.
Previous efforts to reduce hypoglycemia in older patients with T1D included strict avoidance of hypoglycemia and relaxed overall glucose targets.19,20 To achieve that, treatment guidelines/recommendations suggest aiming for a higher HbA1c level in patients with a long duration of diabetes and shorter life expectancy, on the assumption that this will reduce the frequency and severity of hypoglycemia.19,21 Recent guidelines from the Endocrine Society suggest that for patients 65 years and older, outpatient insulin therapy regimens should be designed specifically to minimize hypoglycemia, including setting an HbA1c goal of ≥7.0% for most patients, with an HbA1c goal 8.0%-8.4% or greater in those with comorbidities and shorter life expectancy. 22 However, data from the T1D exchange registry show that severe hypoglycemia is just as common among adult participants with an HbA1c >8.0% as in those with an HbA1c <7.0%. 23 Moreover, HbA1c does not indicate either the frequency, diurnal pattern, or severity of hypoglycemia in individual patients, making it an ineffective marker to assess their patterns of hypoglycemia, time in target range, or glucose variability. 24
Continuous glucose monitoring (CGM) technology has demonstrated clinical benefits, including reduced time in hypoglycemia for pediatric and adult patients with T1D25-27; however, data to support the use of CGM in older patients are limited. In the landmark JDRF CGM study, use of CGM was shown to improve HbA1c in adults ≥25 years old, especially those with a higher frequency of CGM use, although very few adults 60 years and older were enrolled in this study. 26 In a more recent study, a subgroup of subjects 60 years and older in a CGM intervention study had similar HbA1c improvements as younger adults, though in this study as well the number of older participants was relatively small. 27 Because Medicare did not cover CGM until 2017, access to this technology has been constrained. Furthermore, in an older adult population, clinicians may be reluctant to offer, and patients unaccustomed to trying, new health technology. CGM use has, nevertheless, expanded in the older T1D population, with over twice as much use in adults 50 years and older in 2016-2018 compared to 2010-2012. 23 Despite this, data remain limited on the extent to which adoption of CGM fosters reductions in hypoglycemia for this population.
The potential benefit of CGM to reduce hypoglycemia in older adults is currently being tested in the Wireless Innovation for Seniors with Diabetes Mellitus (WISDM) study. WISDM is a six-month parallel-group randomized controlled trial (RCT) in adults ≥60 years old with T1D (NCT03240432). The intervention group receives real-time CGM, while the control group follows usual care (self-monitoring of blood glucose). The primary objectives of WISDM are to determine if CGM can reduce hypoglycemia and improve quality of life in older adults with T1D. WISDM will also generate important data regarding other CGM measures such as time in target range, as well as hyperglycemia and glucose variability metrics.
The present analyses used blinded CGM data collected at baseline in the WISDM study to determine both the prevalence of hypoglycemia as well as the demographic, clinical, and psychosocial factors associated with CGM-measured hypoglycemia and overall glycemic control in older adults with T1D not currently using real-time CGM.
Methods
This report includes baseline data from 203 participants across 22 clinical sites who successfully completed their blinded CGM run-in as part of the WISDM study. The protocol and consent forms were approved by a centralized Institutional Review Board (The Jaeb Center for Health Research [JCHR], Tampa, FL, USA). Written informed consent was obtained from all participants prior to enrollment. Major eligibility criteria included clinical diagnosis of T1D, age ≥60 years, use of either an insulin pump or multiple daily insulin injections (MDIs), no use of real-time CGM in the three months prior to enrollment, and HbA1c <10.0% within 30 days prior to consent or at time of screening. A recruitment target was set to ensure that both insulin pump and MDI regimens were represented, requiring at least 40% of participants in each insulin delivery group.
Following enrollment, all participants wore a blinded (ie, glucose values not displayed to participants) Dexcom G4 Platinum Professional CGM (Dexcom G4 Platinum CGM System with an enhanced algorithm, software 505, Dexcom, Inc., San Diego, CA, USA) for 14-21 days as part of the baseline screening assessment. To continue in the WISDM trial, a criterion for inclusion in this baseline analysis, each participant needed to have at least 10 out of 14 days (240 hours) of CGM data and have calibrated the CGM using a blood glucose meter at least 1.8 times per day on average (manufacturer recommends calibration every 12 hours for the sensor used in the study).
An HbA1c and random C-peptide were collected following successful completion of the baseline CGM wear and analyzed at the University of Minnesota central lab. The detection limit of the C-peptide assay used was 0.003 nmol/L.
Statistical Analysis
Glucose metrics derived from international consensus reports were calculated for the 203 participants using the blinded CGM data from the screening period, including % time <70 mg/dL, % time <54 mg/dL, % time in range (70-180 mg/dL), % time >180 mg/dL, % time >250 mg/dL, and coefficient of variation (CV).28,29 The % time <70 mg/dL and % time <54 mg/dL were calculated both overall and separately for daytime (6
The following factors were tested for a potential association with the above glycemic metrics: age, sex, race/ethnicity, annual household income, education level, health insurance, living alone, employment status, body mass index, T1D duration, age at diagnosis, insulin delivery method, total daily insulin units per kilogram, prior CGM use, average blood glucose management (BGM) checks/day, detectable C-peptide, and reduced hypoglycemia awareness (defined as a score of 4 or more on the Clarke survey 30 ).
For each glycemic outcome and factor, a univariate regression model was fitted to assess their direct association, unadjusted for other potential confounders. Next, a multivariate linear regression model, with stepwise selection, was fitted for each glycemic outcome to determine which subset of factors was associated with the outcome, when considered together. Only factors with a
Data analyses were performed using SAS software version 9.4 (SAS Institute Inc., Cary, NC, USA). Metrics that were reasonably normally distributed were summarized using means ± SD. Skewed metrics were summarized using medians (interquartile range [IQR]) and were modeled using ranks.
Results
The analysis cohort included 203 participants, ranging in age from 60 to 86 years (median 68, IQR 65-71 years); 52% were female, 93% non-Hispanic white, and 53% pump users. Participant demographic and clinical characteristics are shown in Table 1. A median of 326 (IQR 308-390) hours of CGM data per participant were collected.
Participant Characteristics.
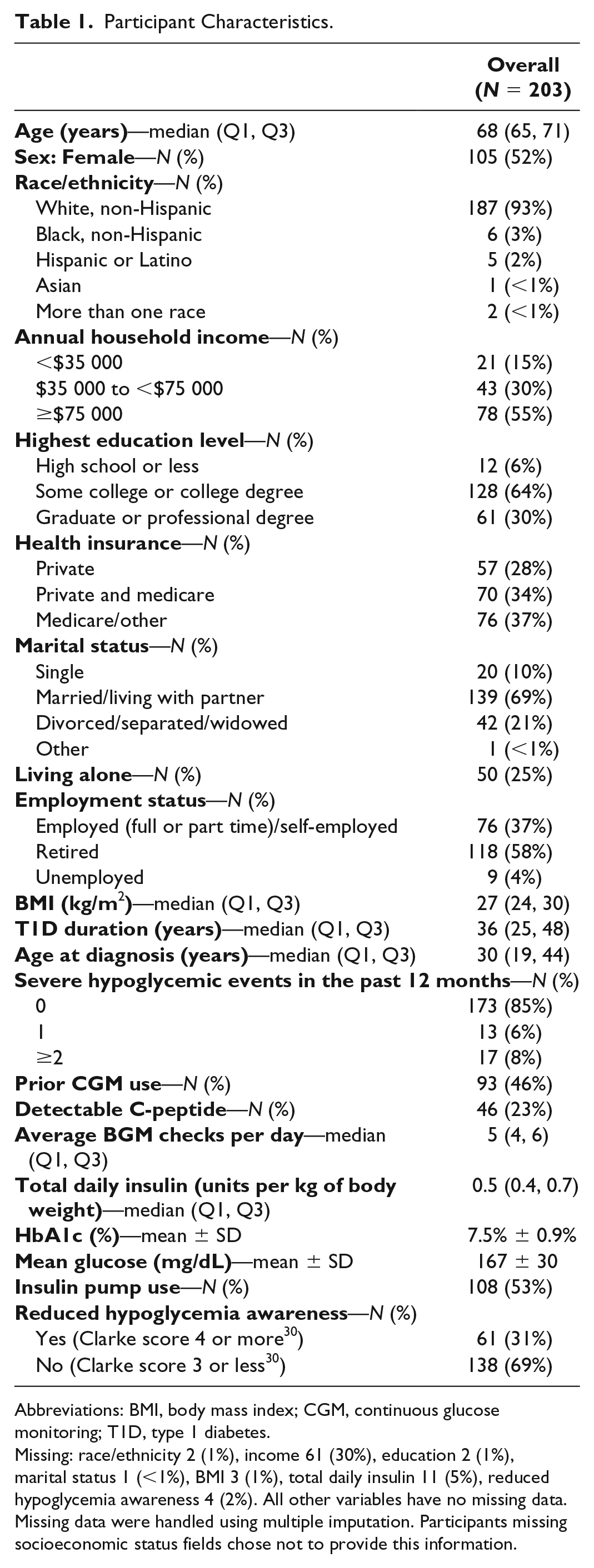
Abbreviations: BMI, body mass index; CGM, continuous glucose monitoring; T1D, type 1 diabetes.
Missing: race/ethnicity 2 (1%), income 61 (30%), education 2 (1%), marital status 1 (<1%), BMI 3 (1%), total daily insulin 11 (5%), reduced hypoglycemia awareness 4 (2%). All other variables have no missing data. Missing data were handled using multiple imputation. Participants missing socioeconomic status fields chose not to provide this information.
CGM-Measured Hypoglycemia
Overall, participants spent a median of 5.0% of time <70 mg/dL (72 minutes per day) and 1.6% of time <54 mg/dL (24 minutes per day) (Figure 1 and Table 2) and had a median of 2.4 hypoglycemic events per week (supplemental Table S1).
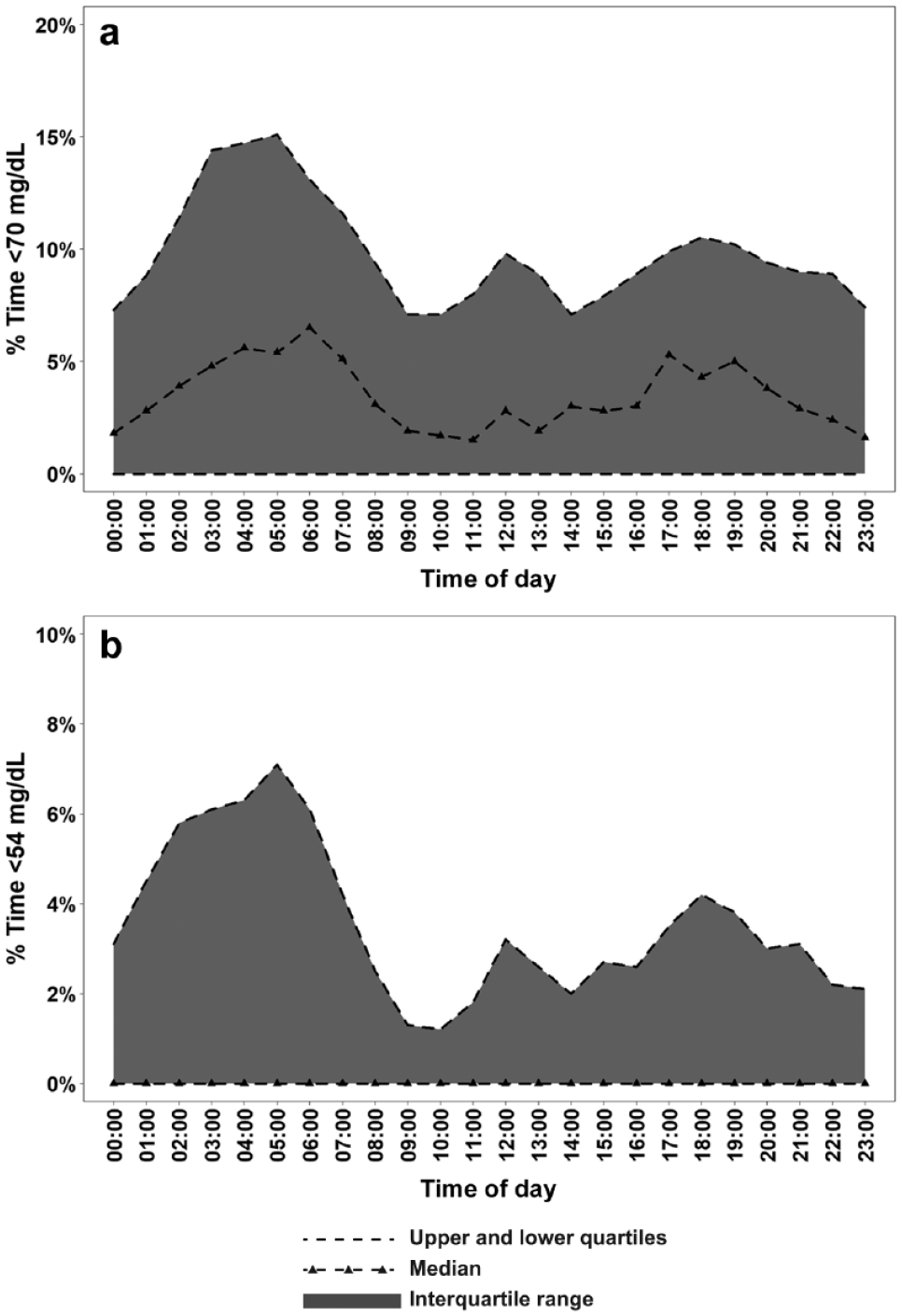
% Time <70 mg/dL (a) and % Time <54 mg/dL (b) by time of day.
CGM-Measured Hypoglycemia.
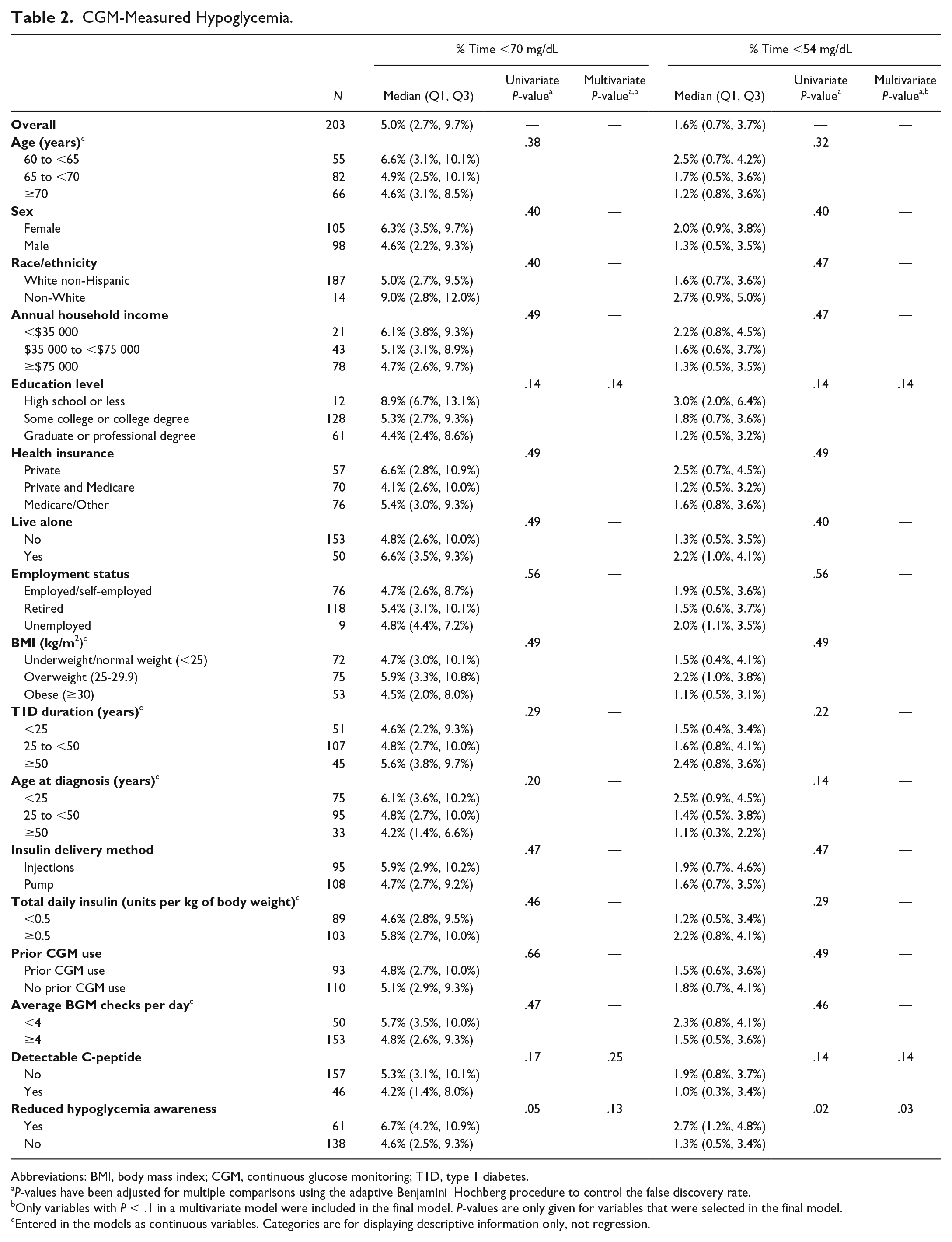
Abbreviations: BMI, body mass index; CGM, continuous glucose monitoring; T1D, type 1 diabetes.
Only variables with
Entered in the models as continuous variables. Categories are for displaying descriptive information only, not regression.
Reduced hypoglycemia awareness, defined as a score of 4 or more on the Clarke survey,
30
was associated with greater time spent with glucose levels <54 mg/dL (Figure 2 and Table 2). Median % time <54 mg/dL was 2.7% (39 minutes per day) vs 1.3% (19 minutes per day) (
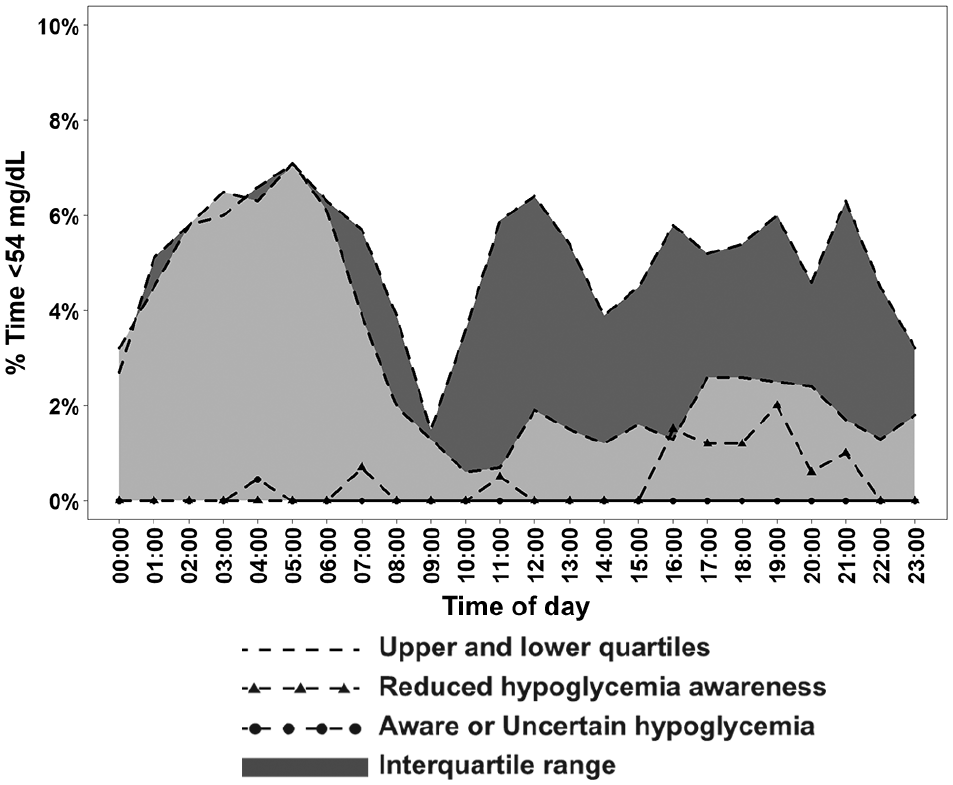
% Time <54 mg/dL by time of day and reduced hypoglycemia awareness.
The analysis was repeated separately for daytime and nighttime % time <70 mg/dL and % time <54 mg/dL (Table 3 and supplemental Table S2). The median % time <70 and <54 mg/dL were the same during both daytime and nighttime: 4.7% of time <70 mg/dL and 1.4% of time <54 mg/dL. During the day, those with reduced hypoglycemia awareness had a higher % time <70 mg/dL (
Daytime (6
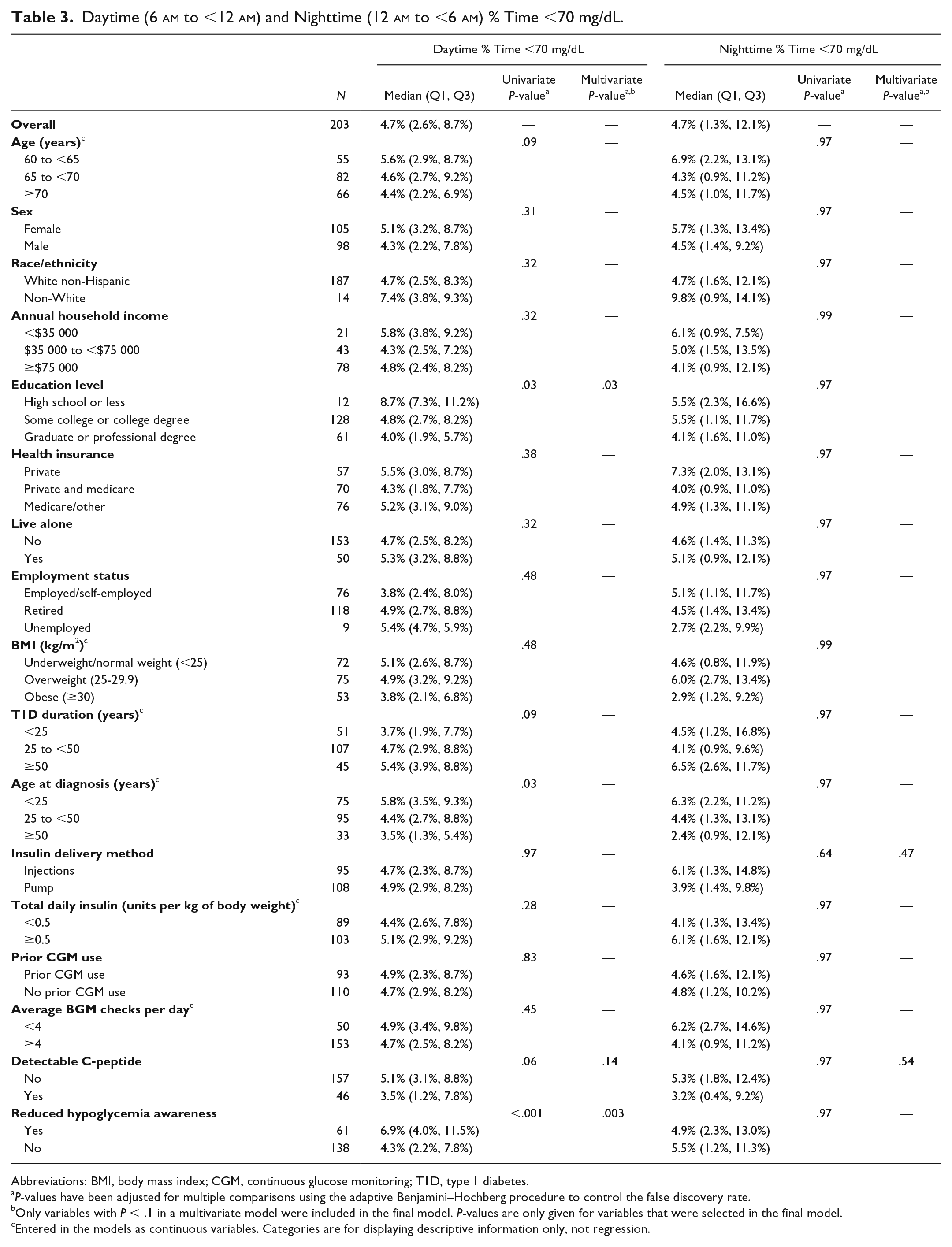
Abbreviations: BMI, body mass index; CGM, continuous glucose monitoring; T1D, type 1 diabetes.
Only variables with
Entered in the models as continuous variables. Categories are for displaying descriptive information only, not regression.
CGM-Measured Time in Range and Hyperglycemia
Overall participants spent a mean 56% of time in target glucose range of 70-180 mg/dL (13.4 hours per day), a mean 37% of time above 180 mg/dL (9.0 hours per day), and a median 12% of time above 250 mg/dL (2.8 hours per day).
Compared with participants with an employment status of “Employed” or “Unemployed,” participants reporting an employment status of “Retired” spent more time in target glucose range (
CGM-Measured Time in Range and Glucose Variability.
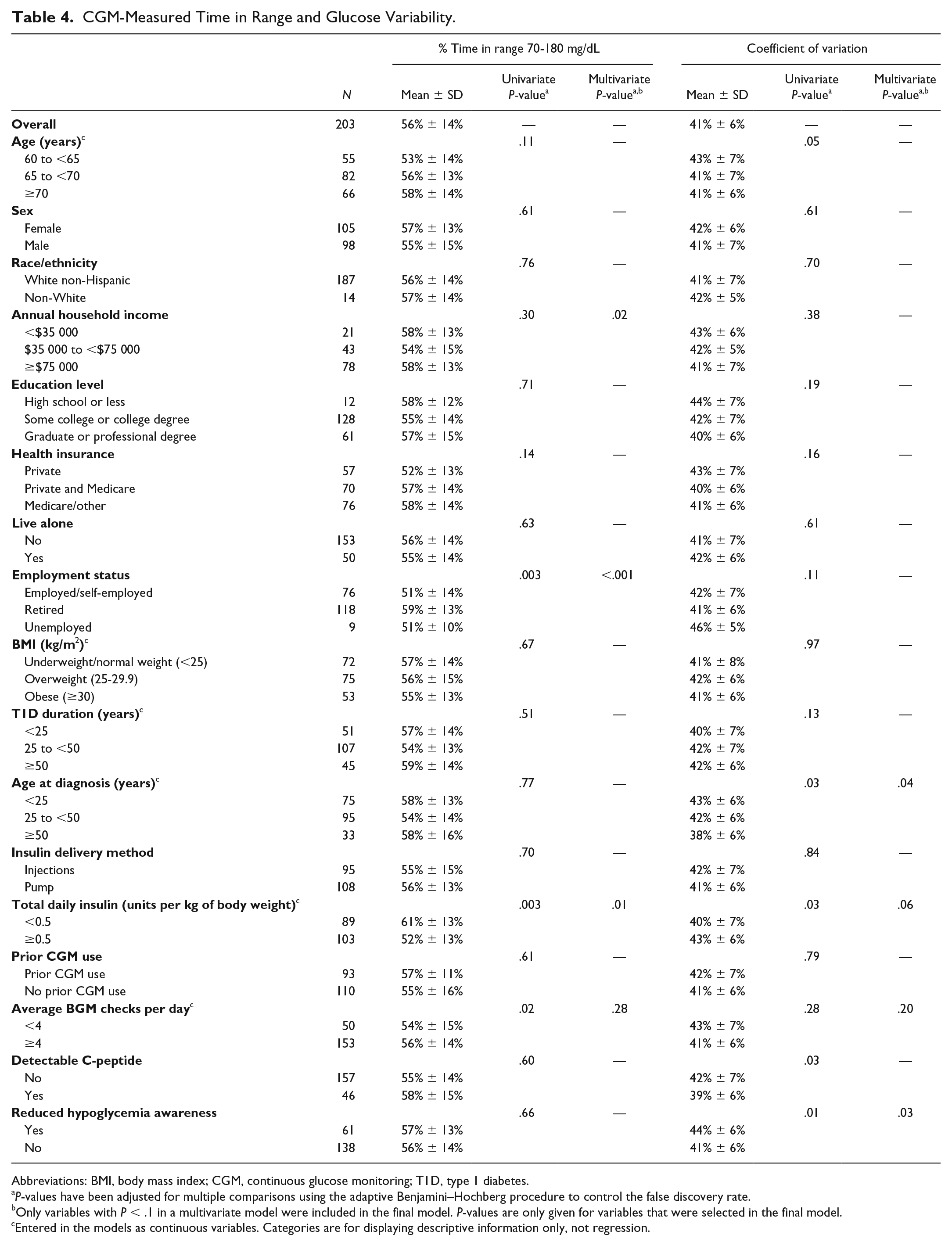
Abbreviations: BMI, body mass index; CGM, continuous glucose monitoring; T1D, type 1 diabetes.
Only variables with
Entered in the models as continuous variables. Categories are for displaying descriptive information only, not regression.
CGM-Measured Glucose Variability
Overall, participants had a mean CV of 41%. A higher CV was observed among participants who had reduced hypoglycemia awareness (
HbA1c
Overall, participants had a mean HbA1c of 7.5% ± 0.9%. The average HbA1c was lower among participants with an employment status of “Retired” (7.3%) compared with those who were employed (7.8%) or unemployed (8.1%) (
Discussion
The population of older adults living with T1D is growing, driven by improvements in diabetes and cardiovascular disease management over the last few decades. Little is known about this distinct population, however, since older adults with T1D have not been widely included in studies using modern CGM devices, and patients with hypoglycemia unawareness (which is associated with a longer duration of disease) are commonly excluded from clinical trials. Given the unique potential for devastating risks from hypoglycemia in an aging T1D population (including falls, fractures, arrhythmia, myocardial ischemia, cognitive impairment, among others), there exists an urgent unmet need to document the prevalence and severity of hypoglycemia in this population, identify those most at risk, and develop mitigation strategies that overcome these dangers. 16
In this analysis of baseline data from the WISDM trial, we observed that many adults ≥60 years old commonly spend 5% of time (over an hour per day) or more in a hypoglycemia range <70 mg/dL, with half of the participants in this cohort spending at least 24 minutes a day with very low glucose levels <54 mg/dL (median 1.6%). Especially noteworthy, a quarter of participants spent at least 53 minutes per day (3.7%) at this dangerously low glucose level. According to recent consensus statements and practice guidelines, a glucose <54 mg/dL is clinically significant and requires immediate action (level 2 hypoglycemia).29,32,33 When compared to the recently published International Consensus on Time in Range, these observed values exceed recommended amounts of hypoglycemia, where a recommended percent target time spent with a glucose <70 mg/dL is <4%, and time <54 mg/dL is <1%. 29
Also notable is that those reporting reduced hypoglycemia awareness spent more than twice as much time <54 mg/dL compared with those who reported intact awareness of hypoglycemia. This underscores the need for interventions specifically targeted to this group who are at high risk for severe hypoglycemia. Interestingly, there was no appreciable difference between median hypoglycemia duration at night and during the day; however, the IQR of hypoglycemia duration between patients was much higher at night. Also worth noting is that those with reduced hypoglycemia awareness did not demonstrate higher rates of nocturnal hypoglycemia at baseline, though they did have significantly higher amounts of time <70 mg/dL during the daytime. Nevertheless, nocturnal hypoglycemia was prevalent across the entire cohort, indicating substantial risk for experiencing clinically significant hypoglycemia even for those older adults with seemingly intact hypoglycemia awareness during daytime hours, which may be explained by the concept of nocturnal hypoglycemia associated autonomic failure. 34
Based on the above findings, strategies to reduce time spent in hypoglycemia, as being tested with real-time CGM in WISDM, may also be associated with improvements in glucose variability and hypoglycemia awareness that could lead to reductions in severe hypoglycemia events. Indeed, other studies have suggested a similar role for technology to aid in hypoglycemia avoidance.35,36 In addition to technology, this study highlights the need for better methods to identify those at highest risk for hypoglycemia and the necessary self-care education needed to assist them.
The extent that real-time CGM with low glucose alarms can reduce both day and nighttime hypoglycemia, and reduce the burden of hypoglycemia in those with reduced hypoglycemia awareness, will be revealed by the full RCT. Of note, we did not observe differences in levels of hypoglycemia or in other metrics of glucose control between insulin pump versus multiple daily injection users. There were several associations found between CGM metrics and demographic characteristics, including associations with annual income and employment status; however, the numbers of subjects are small in these groups. For instance, the association of retirement with higher time in range may be spurious or due to selection bias, or could reflect perhaps a group with more time to dedicate to their diabetes self-management. Also, there was no association with c-peptide status and hypoglycemia risk, suggesting that we may be detecting a very low c-peptide amount, and may suggest such low levels of c-peptide are not clinically meaningful. Worth noting is that the HbA1c levels in this analysis did not correspond to risk for time spent in hypoglycemia ranges. This further corroborates previous observations that HbA1c is not strongly associated with hypoglycemia, and perhaps setting higher HbA1c targets alone may not be the optimal strategy to reduce hypoglycemia burden. Future guidelines addressing hypoglycemia risk for older adults, therefore, should factor in CGM-derived time in range/time below range metrics in addition to, or replacing, HbA1c.
The strengths of this analysis include the large number of participants, enrolled from over 20 diabetes centers across the United States, all with comprehensive diabetes care programs. The inclusion of a considerable number of participants representing both insulin pump and MDI users allowed for a robust assessment of the impact of insulin delivery on hypoglycemia and other glucose measures in this age group. The inclusion of those with reduced hypoglycemia awareness is also valuable, since this population has historically been excluded from studies. The current report also has several limitations. First, the population enrolled in this study tended to have higher education (94% had some college or more) and had higher household income (55% reported ≥$75 000 per year) than a more general T1D population. The study population was also predominantly white (93%). The mean age was 68 years, which also limits assessment of the additional risk factors associated with aging into progressively older decades of life. Further, these data are from blinded CGM assessments and do not reflect glucose levels among participants already actively using real-time CGM.
Conclusion
These baseline findings from the WISDM trial highlight the high prevalence of hypoglycemia in older adults with T1D and also underscore the frequent and extreme hypoglycemia experienced by those with reduced hypoglycemia awareness. Specifically, this study demonstrates that, on average, older adults with T1D spend over an hour a day in the hypoglycemic range of <70 mg/dL. Individuals with reduced hypoglycemia awareness are particularly susceptible to additional time spent in serious hypoglycemia <54 mg/dL. Given the significant proportion of time spent in hypoglycemic ranges, even in this cohort with relatively higher levels of education and income, the need to further evaluate hypoglycemia risk prevention strategies, focusing especially on more socioeconomically disadvantaged and diverse populations, is clear. There remains an urgent need to reduce the risk of experiencing clinically significant hypoglycemia, and its complications, in older adults living with T1D; CGM use may be one such strategy for risk reduction. The results of the ongoing WISDM randomized clinical trial, comparing CGM use to a control group using BGM (with periodic blinded CGM), will further inform our understanding of hypoglycemia in older adults living with T1D.
Supplemental Material
Online_Supplemental_Material – Supplemental material for Hypoglycemia and Glycemic Control in Older Adults With Type 1 Diabetes: Baseline Results From the WISDM Study
Supplemental material, Online_Supplemental_Material for Hypoglycemia and Glycemic Control in Older Adults With Type 1 Diabetes: Baseline Results From the WISDM Study by Anders L. Carlson, Lauren G. Kanapka, Kellee M. Miller, Andrew J. Ahmann, Naomi S. Chaytor, Steven Fox, Lisa Kiblinger, Davida Kruger, Carol J. Levy, Anne L. Peters, Michael R. Rickels, Maamoun Salam, Viral N. Shah, Laura A. Young, Yogish C. Kudva and Richard Pratley in Journal of Diabetes Science and Technology
Footnotes
Author Contributions
ALC researched data and wrote/edited the manuscript. LGK performed statistical analyses and wrote/edited the manuscript. KMM, AJA, NSC, SF, LK, DK, CJL, ALP, MRR, MS, VNS, LAY, YCK, and RP researched data, contributed to discussion, and reviewed/edited the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ALC reports grants from T1DExchange, during the conduct of the study; grants and personal fees from Sanofi, grants and personal fees from NovoNordisk, grants and personal fees from Medtronic, grants and personal fees from Dexcom, grants and personal fees from Eli Lilly, personal fees from Insulet, grants from Abbott, outside the submitted work. In addition, ALC has a patent Treatment of hypoglycemia unawareness with intranasal insulin pending to HealthPartners Institute. LGK has no disclosures. KMM has no disclosures. AJA reports grants from Helmsley Charitable Trust and JDRF, during the conduct of the study; grants and personal fees from Dexcom, grants from Medtronic, grants and personal fees from Lilly, personal fees from Novo Nordisk, personal fees from MannKind, personal fees from Sanofi, outside the submitted work. NSC reports grants from Jaeb Center for Health Research, during the conduct of the study; personal fees from Eli Lilly, outside the submitted work. SF has no disclosures. LK does not currently act as a speaker for DexCom and has not worked with the company since March 2018. DK has no disclosures. CJL has no disclosures. ALP has no disclosures. MRR has no disclosures. MS reports other from JDRF, other from Helmsley Charitable Trust, other from DEXCOM, during the conduct of the study; other from Mylan, outside the submitted work. VNS reports grants from National Institute of Health, grants from Sanofi US, grants from Mylan/GmbH, grants from NovoNordisk, grants from VTV therapeutics, outside the submitted work. LAY has no disclosures. YCK reports grants from JDRF, other from Dexcom, during the conduct of the study; other from Dexcom, other from Medtronic, other from Tandem Diabetes, outside the submitted work; in addition, YCK has a patent Estimation of insulin sensitivity from CGM and subcutaneous insulin delivery in T1D issued. RP has no disclosures. Dr Kellee Miller is the guarantor of this work, as such, and had full access to all the data in the study and takes responsibility for the integrity of the data and accuracy of the data analysis. Material support was provided by Dexcom.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Juvenile Diabetes Research Foundation (grant 3-SRA-2017-425-M-R) and the Leona M. and Harry B. Helmsley Charitable Trust.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
