Abstract
Aims:
The aim was to investigate rebound hypoglycemic and hyperglycemic events, and describe their relation to other glycemic metrics.
Methods:
Data from intermittently scanned continuous glucose monitoring were downloaded for 90 days for 159 persons with type 1 diabetes. A hypoglycemic event was defined as glucose <3.9 mmol/l for at least two 15-minute periods. Rebound hypoglycemia (Rhypo) was a hypoglycemic event preceded by glucose >10.0 mmol/l within 120 minutes and rebound hyperglycemia (Rhyper) was hypoglycemia followed by glucose >10.0 mmol/l within 120 minutes.
Results:
A total of 10 977 hypoglycemic events were identified of which 3232 (29%) were Rhypo and 3653 (33%) were Rhyper, corresponding to a median frequency of 10.1, 2.5, and 3.0 events per person/14 days. For 1267 (12%) of the cases, Rhypo and Rhyper coexisted. The mean peak glucose was 13.0 ± 1.6 mmol/l before Rhypo; 12.8 ± 1.1 mmol/l in Rhyper. The frequency of Rhyper was significantly (P < .001) correlated with Rhypo (Spearman’s rho 0.84), glucose coefficient of variation (0.78), and time below range (0.69) but not with time above range (0.12, P = .13).
Conclusions:
The strong correlation between Rhyper and Rhypo suggests an individual behavioral characteristic toward intensive correction of glucose excursions.
Keywords
Introduction
Fear of hypoglycemia reduces quality of life in type 1 diabetes and induces behavioral changes that may occasionally be inappropriate.1,2 We define rebound hyperglycemia (Rhyper) as a hypoglycemic event followed by hyperglycemia, presumably due to excessive carbohydrate intake. Although the resulting highly variable glucose curve is commonly seen in clinical diabetes care, the phenomenon of Rhyper is infrequently described in the literature on continuous glucose monitoring (CGM). A recent publication reports that in observational studies, the frequency of Rhyper is reduced in patients with type 1 diabetes using real-time CGM (rtCGM) compared with blinded CGM and in those using rtCGM with a predictive low glucose alarm compared with rtCGM without alarms. 3 Two other studies have indicated that the need for carbohydrate to correct hypoglycemia is reduced in individuals with type 1 diabetes and insulin pumps with predictive low glucose suspension.4 -6 It is a well-known clinical observation that shortly after hyperglycemia sets in it may be followed by hypoglycemia mostly due to a too high correction dose of fast-acting insulin. In the following, this phenomenon is referred to as rebound hypoglycemia (Rhypo).
Intermittently scanned CGM (isCGM) was reimbursed in several countries in 2016 to 2017 with proven clinical benefits in epidemiological studies and probably remains the most widespread form of CGM.7 -9 The aim of the present study was to characterize Rhyper and Rhypo in adults with type 1 diabetes using CGM and to relate these events to other glycemic metrics.
Material and Methods
We had unrestricted access to isCGM (Freestyle Libre (version 1), Abbott, Whitney, UK) in the diabetes outpatient clinic for adults with type 1 diabetes in the Silkeborg Regional Hospital, Denmark.10,11 During routine clinical care, glucose data were downloaded from the software platform Diasend (Glooko + Diasend). We present results from a single download of glucose values covering 90 days for each of the patients with isCGM visiting the clinic from February through December 2019.
The patients had access to information of instant interstitial glucose values by swiping a receiver unit in close proximity to a sensor on the upper arm. In addition, glucose data recorded every 15 minutes for the preceding eight hours are presented graphically, and a trend arrow indicates if glucose is stable, rising, or falling. Mean glucose was calculated as the mean of glucose values (scanned and imported) for each 15-minute period. Active CGM time (%) was calculated for the 90-day period as the number of 15-minute periods with at least one glucose value divided by the total number of 15-minute periods (8640 = 90 × 24 × 4) multiplied by 100. Time in range (TIR)12 -14 was the number of 15-minute periods with a mean glucose value of 3.9 to 10 mmol/l divided by the number of periods with at least one glucose value multiplied by 100. Time below range (TBR) (glucose < 3.9 mmol/l) and time above range (TAR) (>10 mmol/l) were calculated similarly. Glycemic variability was given as the coefficient of variation (%) of 15-minute mean glucose values (standard deviation (SD)/mean glucose) multiplied by 100.
A hypoglycemic episode was defined as two or more consecutive 15-minute periods with glucose <3.9 mmol/l.15 -17 The duration was the number of periods <3.9 mmol/l × 15 minutes. The end of a hypoglycemic event was the last period with glucose <3.9 before at least two consecutive 15-minute periods with glucose ≥3.9 mmol/l. 16 No official definition of Rhypo and Rhyper exists. It seems logical to apply the upper limit of glucose TIR (10.0 mmol/l), but the time period (120 minutes) for the elevated glucose level to occur is less clear. We chose 120 minutes for the definition of Rhyper as previously suggested 3 and, for simplicity, applied the same definition for Rhypo. Rhypo was therefore defined as an episode with hypoglycemia preceded by glucose >10 mmol/l within 120 minutes before the first hypoglycemic value. The duration from the peak glucose value within 120 minutes before hypoglycemia to the hypoglycemic episode was calculated from the number of periods from (and including) the period with the maximal value and the following periods with glucose >3.9 mmol/l before a hypoglycemic episode. A Rhyper episode was defined as a hypoglycemic episode with the presence of at least one glucose value >10 mmol/l in the 120-minute period after the last hypoglycemic value. 3 The duration from the last hypoglycemic value to the peak value within 120 minutes after hypoglycemia was calculated as the number of periods from the first period with glucose >3.9 to the period including the peak glucose value. The frequency of hypoglycemic events per 14 days was calculated from the number of events in 90 days corrected for the number of days with available glucose values. As recommended, we also reported the hypoglycemic events with a trough value <3.0 mmol/l. 18 The rate of glucose decline in Rhypo per 15 minutes was calculated as the difference between the peak glucose 120 minutes before hypoglycemia and 3.9 mmol/l divided by the duration in minutes multiplied by 15. The rate of glucose increase in Rhyper was calculated similarly as the difference between 3.9 mmol/1 and the peak posthypoglycemic value within 120 minutes divided by the duration.
No ethical approval was needed for this observational study, which was approved by the local institution.
Statistical Analysis
Normally distributed data as assessed by visual inspection of QQ plots are presented as mean ± SD and compared with Student’s paired or unpaired t test as appropriate, and 95% confidence intervals (CIs) of the mean differences are presented. Parameters that could not adequately be described by a normal distribution (frequency of hypoglycemia, TBR, active CGM time, and diabetes duration) are presented as median (interquartile range [IQR]) and compared with a Mann-Whitney test. The median of paired differences was estimated as the Hodges-Lehman median difference (95% CI). Correlations are given as Spearman’s rho. Statistical analysis was performed with SPSS ver. 20. Data on hypoglycemic and hyperglycemic events and duration of specified time periods were extracted from the glucose download data based on our development of a program in the statistical package R ver. 4.2.1. If the glucose data were missing for ≤ 2 hours, the missing observations were imputed by means of interpolation. Missing observations for more than two hours were considered holes in the data that separate sections of uninterrupted data. A section starts with the first observation or the first observation after a hole in the data and ends before the next hole or at the end of glucose download. Therefore, a patient’s data may consist of only one or several sections. The hypoglycemic events start and end in same section. It follows that no rebound hypoglycemic measures were calculated if the episodes started at the beginning of a section. Similarly, no rebound hyperglycemic measures were calculated if the end of a section was within 120 minutes of the end of the episode.
Results
CGM data were downloaded for 169 persons with type 1 diabetes of whom 159 had an active CGM time >70% as recommended. 19 The mean age was 52 ±14 years, the median diabetes duration was 26 (IQR: 14-37) years, and the gender distribution was 88 (55%) men and 71 (45%) women. Insulin delivery was multiple daily injections (MDIs) for 134 (84%) and continuous subcutaneous insulin infusion (CSII) for 25 (16%) persons. The median active CGM time was 96% (IQR: 92-98), and the median maximal number of days with glucose values was 90 days (IQR: 90-90 days, range 71-90). The total number of days with available glucose values was 14 234. The median number of data sections was 7 (IQR: 4-16).
The total number of hypoglycemic events for 90 days was 10 977 including 3232 cases (29.4%) with Rhypo, 3653 (33.3%) with Rhyper, and 1267 (11.5%) with combined Rhypo-Rhyper. The frequencies of all hypoglycemic events, that is, those with Rhypo and Rhyper and related glycemic metrics, are shown in Table 1. The number of nadir hypoglycemic glucose measurements with the value of 2.2 mmol/l (the lowest to be registered with this and other monitors20,21) was 2004 corresponding to a fraction of 18.3%. The diurnal distribution of glycemic metrics is presented in Figure 1. The duration of hypoglycemia was highest in the two hours after midnight and decreased to a stable value in the daytime. In contrast, the frequency of all hypoglycemic events, Rhypo, and Rhyper was lower during the night and peaked in the afternoon.
Glycemic Characteristics for All Hypoglycemic Events and for Events With Rebound Hypoglycemia and Rebound Hyperglycemia in 159 Persons With Glucose Data Downloaded for 90 Days.
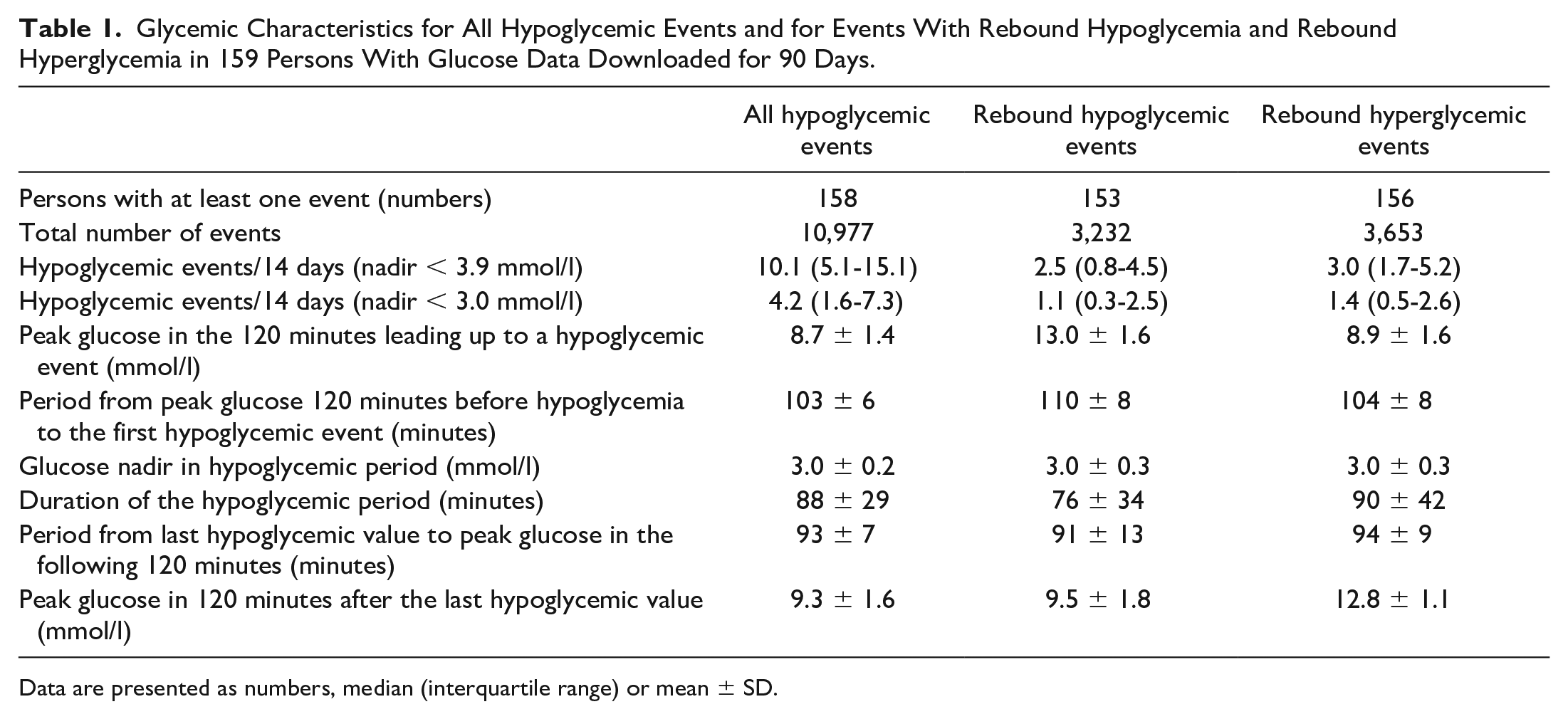
Data are presented as numbers, median (interquartile range) or mean ± SD.
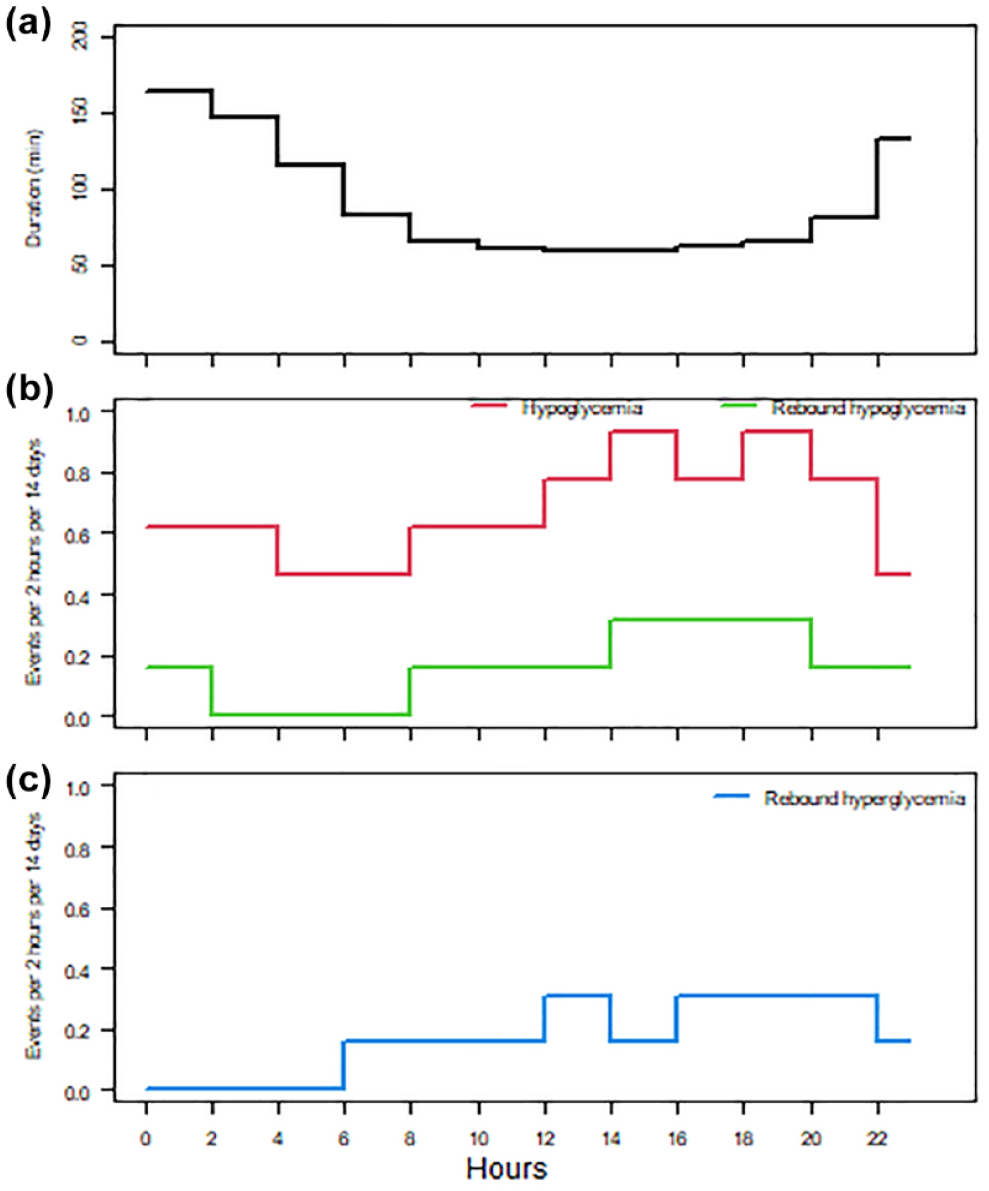
The diurnal distribution of glycemic metrics for two-hour periods. (a) The mean duration of hypoglycemic events (black). (b) The median frequency of hypoglycemic events (red) and Rhypo (green) per person allocated according to the time stamp indicating the initiation of the relevant hypoglycemic event. (c) The median frequency of rebound hyperglycemia (blue) per person allocated according to the time stamp for the peak glucose values. Note that due to overlap, the frequencies of Rhypo and rebound hyperglycemia cannot be summed up.
The duration of hypoglycemia for events with Rhypo (76 ± 34 minutes) was compared with the duration of events with hypoglycemia that were not preceded by glucose >10 mmol/l within 120 minutes (91 ± 28 minutes). The 15-minute difference found was statistically significant (CI: 11-20, P < .001).
The rate of glucose increase in Rhyper of 1.29 ± 0.18 mmol/l per 15 min, which was significantly higher than the rate of glucose decline in Rhypo was 1.24 ± 0.23 mmol/l per 15 minutes (CI: 0.02-0.009, P = .006).
The individual frequency of Rhyper for the whole study period was significantly (P < .001) correlated with both the rate of Rhypo (Spearman’s rho: 0.84, Figure 2), glucose coefficient of variation (0.71), and TBR (0.69) but not with TAR (–0.12, P = .13). We divided the population into group A with 0-4 hyperglycemic rebound episodes per 14 days (n = 103) and group B with >4 episodes (n = 56). Patients in group B were younger, had a significantly higher rate of Rhypo, a higher peak glucose before hypoglycemia, a slightly lower glucose nadir, and a higher posthypoglycemic peak value (Table 2). In group B, TBR was significantly (4.1 percentage point [CI: 3.0-5.1]) higher and CV was 7.1 percentage point (CI: 5.4-8.7) higher than in group A (P < .001 for both). TAR was 40% ± 13% in group B and 42% ± 18% in group A (difference: –2%, CI: –7% to 4%, P = .51).
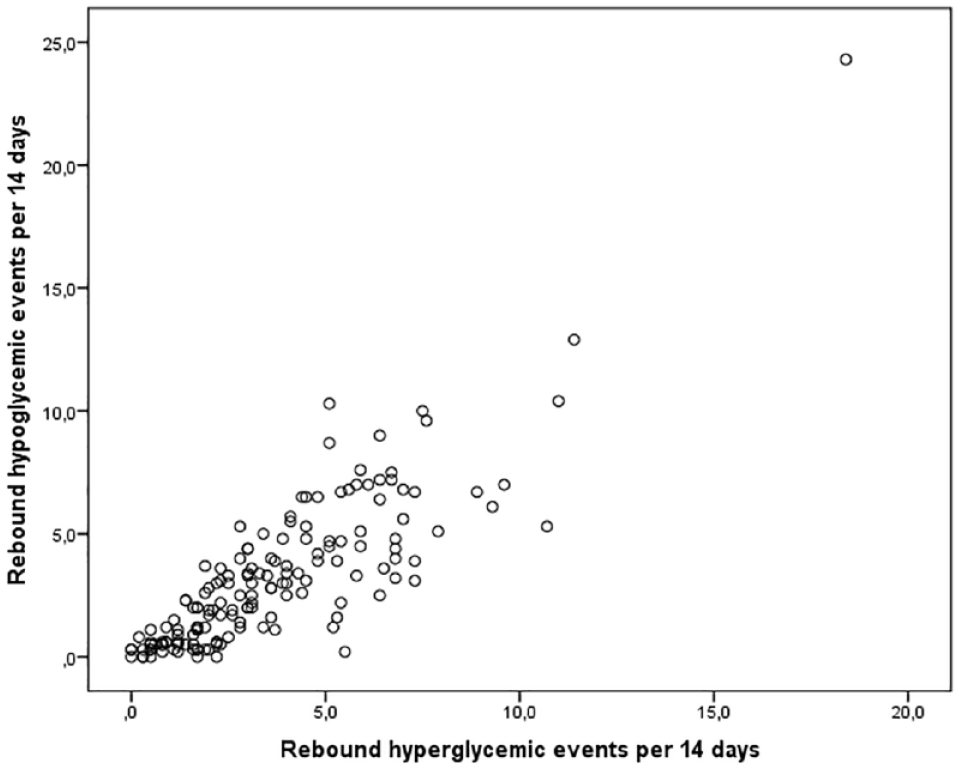
A scatterplot of frequency of rebound hypoglycemic events per 14 days versus the frequency of rebound hyperglycemic events per 14 days.
Glycemic and Clinical Characteristics of Persons With A Low or High Frequency of Rebound Hyperglycemia.
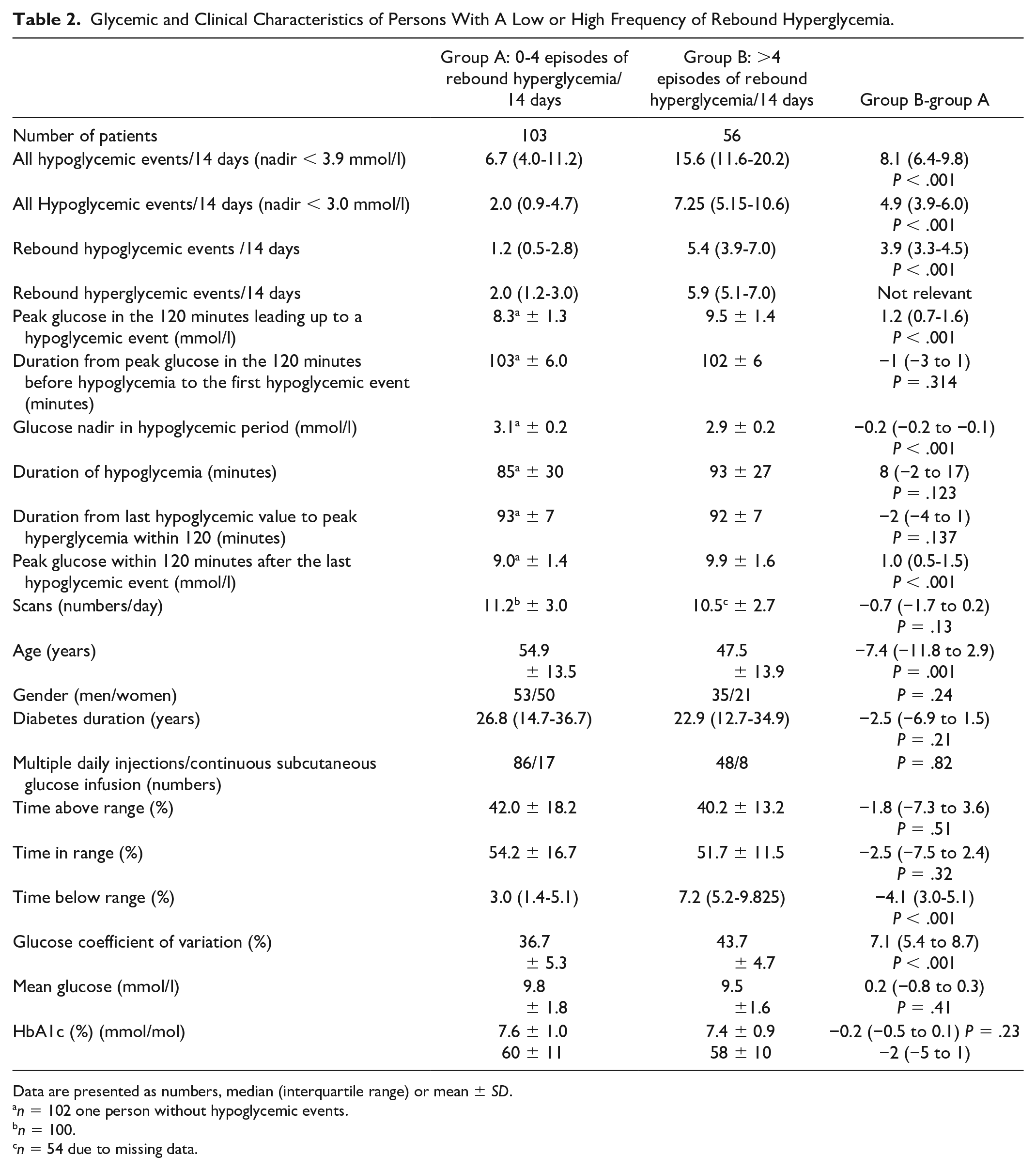
Data are presented as numbers, median (interquartile range) or mean ± SD.
n = 102 one person without hypoglycemic events.
n = 100.
n = 54 due to missing data.
Discussion
The main finding was that 1/3 of the hypoglycemic events were preceded by a glucose value >10.0 mmol/l within two hours and that 1/3 of the events were followed by hyperglycemia. Importantly, the two phenomena coexisted in 1/9 of the hypoglycemic cases and the individual frequency of Rhypo and Rhypo were correlated.
Our finding of a strong correlation between Rhypo and Rhyper supports the hypothesis that persons who overcompensate in terms of carbohydrate intake in the hypoglycemic state also treat hyperglycemia more aggressively, indicating a behavioral connection between the two phenomena. The incidence of Rhypo followed by hyperglycemia underlines the clinical importance of the so-called glycemic “roller coaster” phenomenon, which may be associated with increased oxidative stress and cardiac strain.22,23
We and others 3 found that the frequency of Rhyper was positively correlated with glycemic variability. Thus, the frequency of Rhyper belongs to the group of glycemic metrics that is associated with treatment safety. 24 Patients with more than four rebound hyperglycemic episodes per two weeks are characterized by an unfavorable combination of a slightly lower hypoglycemic nadir followed by a higher glycemic peak. The consequence is a high TBR and CV in comparison with the significantly older group of patients with less frequent Rhyper. Our observation is in line with an observational study reporting younger age in the group with excessive carbohydrate intake but no difference in diabetes duration. 2
The presence of high TBR, a high CV, and consequently a wide 25th, 75th percentile glucose band on the ambulatory glucose profile (AGP) is the hallmark of Rhypo and Rhyper. These findings should prompt the clinician to search, for examples, in the individual’s daily glucose profiles, which can be a time-consuming effort. We suggest extending the AGP report25,26 to include information about the frequency of Rhypo and Rhyper. Such information may facilitate rapid identification of an important problem that may be corrected by reinstruction in how to calculate the correction bolus and the amount of carbohydrate needed for hypoglycemia treatment.
The duration of hypoglycemia was shorter for events with Rhypo than for events with Rhyper without preceding Rhypo. This may be so because of a difference in patients’ expectations for hypoglycemia. Rhypo is a result of a recent action (insulin dose and exercise), and patients who are well aware of the risk of hypoglycemia will likely react earlier.
The downloaded data consist of glucose information only without information about insulin dose, carbohydrate intake, or exercise. Our definition of Rhyper is based on the most likely assumption that the steep rise in glucose is caused by carbohydrate intake in excess of what is needed. In cases of hypoglycemia before a planned meal, omission or reduction of bolus insulin may partly explain the rise in glucose. Without reporting of exercise, we can only assume that Rhypo is mostly the consequence of too much fast-acting insulin.
The frequency of total hypoglycemic events decreases during the night, but the duration of each event was longer (Figure 1a and b). The rise in Rhypo at 08:00 to 10:00 (Figure 1b) may reflect that some persons overdose fast-acting insulin to correct a high glucose level when they wake up. Although disputed, the term Rhyper has also been used to describe an early morning rise in glucose after nocturnal hypoglycemia. 27 The increase in Rhyper in the period from 06:00 to 08:00 (Figure 1c) may potentially also be due to carbohydrate overload.
We defined a 120-minute time span before and after hypoglycemia for characterization of Rhypo or Rhyper. Since the mean duration from the peak glucose before Rhypo to hypoglycemia is 110 minutes and the mean duration after hypoglycemia to Rhyper is 89 minutes, the number of events will obviously increase considerably if the time period is extended to, for example, 150 minutes. In our opinion, it is relevant to adhere to a definition that implies a rather high rate of decrease or increase in glucose. The mean rate of declining glucose in Rhypo from the peak value and the rate of increasing glucose in Rhyper are both well above the rate (±0.9 mmol/l per 15 minutes) needed for the display of a declining or rising glucose trend arrow in Freestyle Libre. 20
A drawback of the study is the 15-minute glucose sampling frequency by isCGM, which introduces imprecision in estimation of the true nadir and peak glucose and the duration of the time periods compared with rtCGM with sampling every 5 minutes. We believe, however, that this does not fundamentally affect the interpretation of our results. The fraction of all hypoglycemic events with glucose nadir ≤ 2.2 mmol/l was 18%, which seems an unrealistically high proportion and may indicate that the monitor underestimates very low glucose values 28 and the frequency of hypoglycemia may be overestimated. Even so, the frequency of hypoglycemia <3.0 mmol/l in our study (median: 4.2 [mean 5.0] per 14 days) was comparable with frequencies reported in other studies with an unblinded sensor (mean 2.0-7.8 per 14 days).3,15
Glucose data from isCGM can be missing for several reasons, including delayed application of a new sensor, warm-up time for sensors (60 minutes), loss of sensor connection, or a time span between two scans exceeding eight hours. This imposes a limitation on the calculation of the number of total hypoglycemic events, which must be minimum estimate due to problems related to the edges of the sections of complete data. With a median number of seven sections per person, this seems to be of minor importance.
Inter-study comparison of the frequency of hypoglycemia is difficult because it critically depends on the performance of the monitor in the low glucose range, the definition of a hypoglycemic event, and the population characteristics. We defined a hypoglycemic event as at least two 15-minute periods with glucose <3.9 mmol/l, which is in line with the study by Bolinder et al 15 but opposite a recent study that used a definition of one 5-minute period <3.9 mmol/l. 3 This explains why the duration of hypoglycemia (88 minutes) in our study and in the study by Bolinder et al (which can be estimated to approximately 92 minutes) is longer than the durations reported by other unblinded CGM monitors (40-48 minutes).
Despite differences in monitors, patient population, and definitions of hypoglycemia, we noticed a uniformity across studies in the frequency of Rhyper (3-4 events per 14 days) and in the ratio of Rhyper to all hypoglycemic events (24%-40%). 3 We have not been able to find previously reported frequencies of Rhypo.
It is important to realize that the Freestyle Libre monitor version used in the present study had no any low or high glucose alarm. Furthermore, the majority of the patients were treated with MDI and a minority with CSII, which did not have low glucose suspend or automated insulin delivery. Our results, therefore, cannot be generalized to a population using CGM with alarms or to patients using insulin pumps with a closed loop system. The frequency of Rhyper was reduced moderately, by 7%, when patients changed from a system with no such alarm to an rtCGM system with predictive low glucose alarm. 3
The strength of this study is the large number of hypoglycemic (≈11 000), Rhyper (≈3600) and Rhypo (≈3200) events available for analysis. Furthermore, we were able to combine glucose data in a “real-world” setting with other clinical data, and the active CGM time was high.
Conclusions
Rhypo and Rhyper are present in approximately one third of all hypoglycemia cases and a strong correlation exists between these phenomena. This may indicate an individual behavioral attitude of rapid correction of glucose excursions; an attitude that may be modified in dialogue with the healthcare provider. The presentation of the frequency of Rhypo and Rhyper in the AGP report may facilitate the interpretation of glucose data from CGM.
Footnotes
Acknowledgements
The authors express their gratitude to statistician Aparna Udupi, Biostatistical Advisory Service, Faculty of Health, Aarhus University, Denmark, for data management and to MD Esben Grove Laugesen, Diagnostic Center Silkeborg Regional Hospital and Steno Diabetes Center Aarhus, Denmark, for comments on the manuscript.
Abbreviations
CSII, continuous subcutaneous insulin infusion; isCGM, intermittently scanned continuous glucose monitoring; MDI, multiple daily injections; Rhyper, rebound hyperglycemia; Rhypo, rebound hypoglycemia; rtCGM, real-time continuous glucose monitoring; TAR, time above range; TBR, time below range; TIR, time in range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KWH has received honorarium as an advisory board member for Abbott Laboratories A/S, Denmark. BMB has no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported financially by the Rosa and Asta Jensen Foundation.
