Abstract
Background:
The pain associated with pricking the fingertip for blood glucose self-testing is considered to be a major burden in diabetes treatment. This study was performed to evaluate the system accuracy of the invasive TensorTip Combo Glucometer (CoG) device component in accordance with ISO15197:2015 requirements and to explore the accuracy of the noninvasive tissue glucose prediction component.
Methods:
One hundred samples were obtained from people with type 1 and type 2 diabetes and healthy volunteers (43 females, 57 males; age: 53 ± 16 years), with glucose distribution as requested by the ISO standard. Three strip lots were tested twice by healthcare professionals in comparison to YSI 2300 Stat Plus reference method followed by a noninvasive tissue glucose reading (NI-CoG). Mean Absolute (Relative) Difference (MARD) was calculated and a consensus error grid (CEG) analysis was performed.
Results:
The ISO system accuracy criteria were met with the invasive strip technology by 586/600 of the data points (97.1%) and for each strip lot separately. All invasive results (100%) were within CEG-zone A and total MARD was calculated to be 7.1%. With the noninvasive reading, 99% of raw data points were in A + B (91.1% and 7.8%), and the total MARD was calculated to be 18.1%.
Discussion:
The invasive component of the CoG device was shown to be in full compliance with the current ISO15197 criteria. Good results were also obtained with the NI-CoG tissue glucose prediction. This noninvasive technology would potentially be suitable for frequent pain-free glucose monitoring in many people with diabetes.
Keywords
Introduction
Measurement of blood glucose by means of test strips requiring a capillary blood sample is one of the most frequent procedures in the daily life of insulin-treated patients with type 1 or type 2 diabetes.1,2 The sample is obtained by pricking the fingertip with a lancet. During the life of a patient with type 1 diabetes, this painful procedure is performed four to five times a day, which amounts to ~1800 finger-pricks per year, and 90 000 finger-pricks during a period of 50 years. Over the years, the continuously induced skin lesions result in a scarred and thickened skin at the fingertips, even when the patients regularly change the location of the sampling process. The development of alternative ways to obtain the glucose information that is necessary to guide and monitor the treatment interventions is without doubt one of the most desired current research targets and represents a well-established unmet practical medical need for people with diabetes.
A huge variety of direct and indirect physical methods have been investigated in the past decades to reach this target.3-5 The challenge is immense, and the hurdles are high. Not only that the employed direct or indirect technology has to be specific for glucose. In contrast to blood glucose, the noninvasively measured—or better predicted—glucose concentration is derived from different physiological compartments and the signal may be composed of a mixture of the current arterial, capillary, venous, interstitial, and intracellular glucose concentrations. It is well established that the glucose concentrations in these different compartments are subject to physiological differences, in particular in times of frequent dynamic blood glucose changes like in the postprandial phase.6,7 The contribution of different sources to the glucose information may vary based on the employed technology. This situation raises, eg, the question, whether a blood glucose reference method is really the optimal reference method to evaluate the accuracy of a noninvasive device: The system accuracy standards for the current invasive devices for patient self-testing of blood glucose by means of invasive technologies have been developed over the past decades and are nowadays well established and clearly defined by the regulatory agencies.8,9 No such standards or guidelines exist as of today that consider the specific circumstances of noninvasive tissue glucose prediction. In the lack thereof, the guidelines for invasive devices are usually taken for accuracy assessment of noninvasive technologies.
A new glucose meter offering the option to obtain blood glucose information via the standard invasive way or to predict tissue glucose by means of an optical noninvasive technology has recently been developed (TensorTip Combo Glucometer, Cnoga Medical Ltd., Cesarea, Israel) and tested in clinical trials.10,11 This present study was performed to determine the system accuracy of the invasive device component by means of a study protocol following the ISO15197:2015 standard. In addition, the performance of the noninvasive device component was also analyzed in accordance to the analytical procedures suggested by the ISO standard and using a point-of-care standard capillary glucose reference method.
Patients and Methods
This study was performed following the suggested system accuracy protocol of the ISO15197:2015 standard 9 and in compliance with the guidelines for Good Clinical Practice, the Declaration of Helsinki, and all applicable legal and ethical requirements. The responsible ethical review board approved the study protocol and participants signed written informed consent prior to study enrollment.
Study Device
The TensorTip Combo Glucometer (Cnoga Medical Ltd., Cesarea, Israel) consists of a noninvasive tissue glucose prediction module and an add-on invasive module that is identical to the approved Okmeter match device K090609 (OK Biotech Co., Ltd., Hsinchu City, Taiwan, data on file). The noninvasive module uses four monochromatic light source and color image sensors absorbing continuous wavelength light usually in the range from blue to infrared after transmission through or reflection by the tissue. The color image raw data are acquired by the color image sensor and stored in a memory buffer to be used for the computation of a dedicated algorithm executed by the device software. A more detailed presentation regarding the underlying technology has been published recently. 10
Study Conduct
The primary objective of the study was to investigate the system accuracy for the invasive Combo Glucometer (CoG) component in comparison to the YSI 2300 STAT PLUS (YSI Ltd., Yellow Springs, UT, United States). Another aim of the study was to explore the performance of the noninvasive device component when analyzed by the same statistical methods. Only adult patients with type 1 or type 2 diabetes or healthy subjects could be enrolled if they had an HbA1c-value <10%, a hematocrit between 20% and 60% and if their actual glucose values fell within the glucose concentrations between 50 and 399 mg/dL with a distribution as required by the ISO protocol. They were excluded if they were regularly taking anticoagulation drugs, thrombin inhibitors, or larger quantities of ascorbic acid. Other exclusion criteria were pregnancy or breast feeding, HIV or Hepatitis C infections. In addition, patients undergoing glucose adjustment procedures (glucose uptake or insulin administration), to reach different required glucose ranges, were not allowed to have insulin resistance (daily insulin dose >80 U), history of frequent hypoglycemic events or an actual blood pressure >165/95 mmHg. Any anatomic condition at the fingers preventing the measurement with the noninvasive device component also led to patient exclusion.
During the screening visit, the participants were informed about the study and signed informed consent. Thereafter, they were trained about the device operations and how to perform the calibration procedure for the noninvasive device component at home (56 parallel invasive double measurements/noninvasive readings within 7-10 days). In addition, the participant history was taken, and blood was drawn for assessment of the biochemical inclusion and exclusion criteria.
After completion of the calibration, the patient returned to the study site. An optional screening measurement with a strip-based point-of-care device (StatStrip, NovaBiomedical, Waltham, MA, United States) determined in which distribution bin of the ISO protocol the patient would fell in and the decision regarding a potential glucose adjustment intervention was made. Thereafter, the hematocrit was determined, and at the desired glucose range, a first capillary sample was acquired for the YSI reference method. Then, a healthcare professional used three devices, which were randomly selected out of a total of six devices, to invasively measure glucose with three strip lots (double determinations). The patient performed another invasive reading himself with his own device (randomly assigned strip lot) and an additional noninvasive reading. There was no interaction between the invasive and the noninvasive readings during these experiments. Finally, a second sample was obtained for another YSI reference measurement. The far extreme blood glucose samples (<50 mg/dL/2.7 mmol/L and ≥400 mg/dL/22.2 mmol/L) were obtained by laboratory manipulations as described in the ISO15197 standard. 8 No noninvasive results were available for these extreme samples.
Statistical Methods
Data analysis of system accuracy was performed according to EN ISO 15197:2015 8 using YSI 2300 STAT PLUS as the reference method, including bias analysis according to Bland and Altman 12 and regression analysis according to Passing and Bablok. 13 In addition, a consensus error grid (CEG) analysis according to Parkes et al and Pfützner et al14,15 as well as a surveillance error-grid according to Klonoff et al 16 was performed. The analyses with the noninvasive results were performed with the raw data, which were analyzed without the exclusion of outliers.
Different results were compared by means of appropriate parametric and nonparametric statistical methods and a P-value <.05 was considered to be statistically significant.
Results
The ISO system accuracy criteria were met by 586/600 of the data points (97.1%) with the invasive device component and also individually by each strip lot as shown in Table 1. Bland Altman analysis results are shown in Figure 1 and the Consensus Error Grid Results are depicted in Figure 2(a). All invasively collected values (100%) were within CEG-zone A, and the total Mean Absolute (Relative) Difference (MARD) was calculated to be 7.1%. The regression curve for this data set was y = 1.02 * x + 2.08 with R² = .991.
TensorTip Combo Glucometer System Accuracy in Accordance with ISO15197:2015.
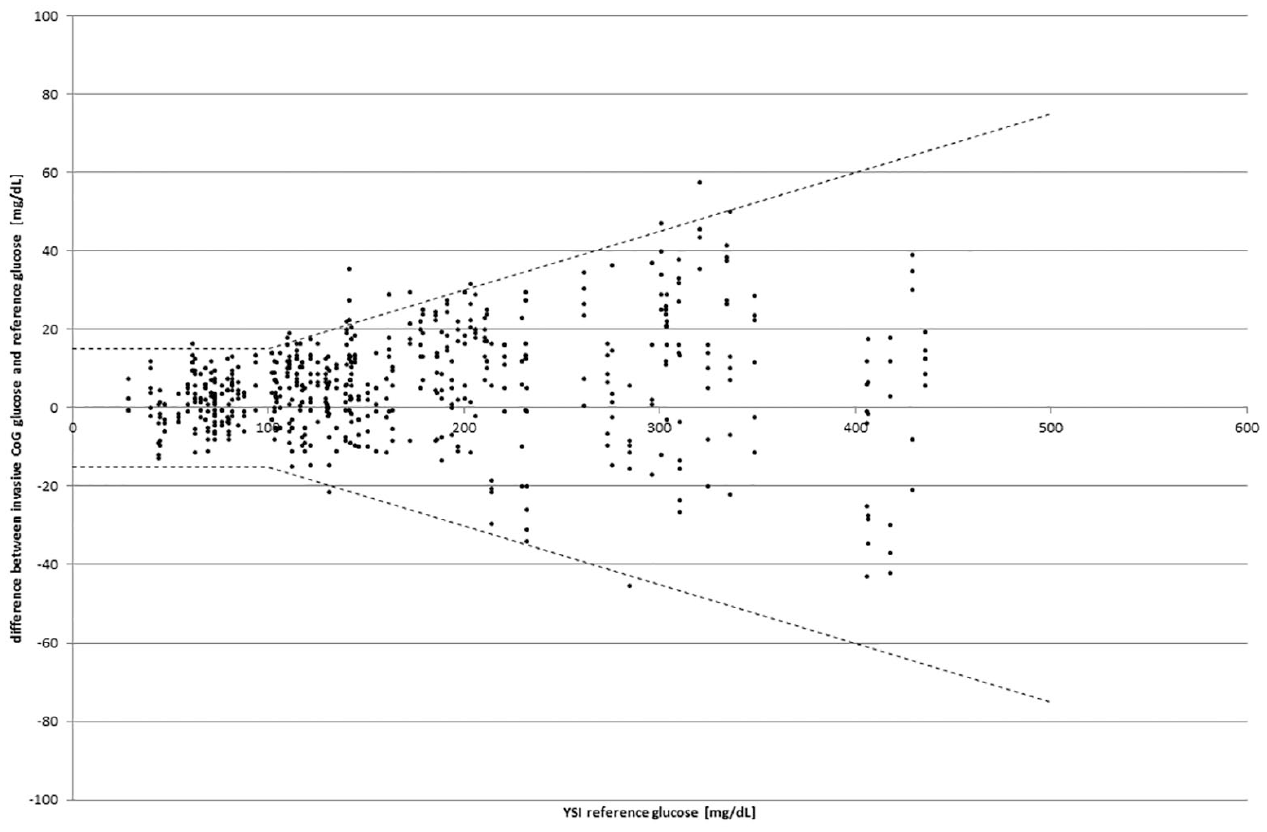
Difference plot (modified Bland-Altman analysis 12 ) for the invasive device component.
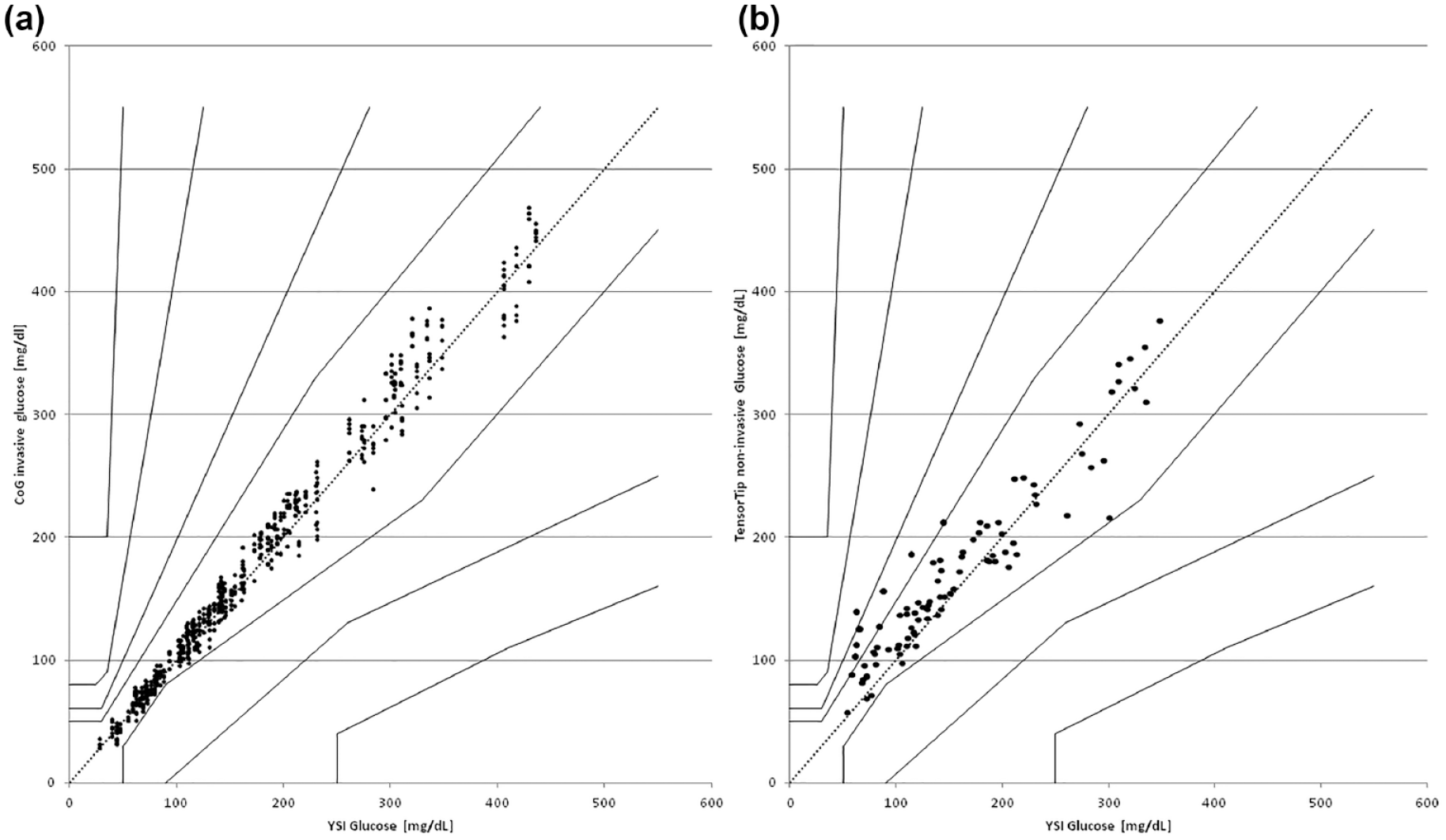
Results of the consensus error grid analysis according to Parkes et al. for the invasive (a) and the noninvasive (b) device component (raw data).
One requirement for accurate noninvasive prediction of tissue glucose by the TensorTip Combo Glucometer is the performance of a proper calibration procedure with the collection of 56 paired data sets (one noninvasive reading and two parallel invasive readings) over a period of seven to ten days and with varying glucose concentrations. It is a result of this study that the noninvasive technology worked best when operated within the range of glucose values occurring during the calibration period. As an example, an appropriate prediction of tissue glucose during a period of hypoglycemia was only achievable, if hypoglycemic values have been seen used during the calibration period as well. Such cases where the tested range was not covered in the calibration manifold and that were showing a difference between the YSI reference blood glucose value and the noninvasively predicted tissue glucose ≥40% for values ≥100 mg/dL/5.55 mol/L and ≥40 mg/dL for values <100 mg/dL/5.55 mol/L were detected, but left in the analysis. Such deviations were seen in 8/90 data sets (8.9%). The raw noninvasive data analysis revealed that 70% of all noninvasive data sets (63/90) were within ISO acceptance criteria. The regression curve for the noninvasive results was 0.881 * x + 32.09, with R² = 0950. The Consensus Error Grid of the noninvasive results is displayed in Figure 2(b). In the raw data analysis, there were 91.1% in Zone A, 7.8% in Zone B, and 1.1% in Zone C. Mean Absolute (Relative) Difference of the raw data set was 18%. The associated surveillance error-grid with the noninvasive data set is provided in Figure 3.
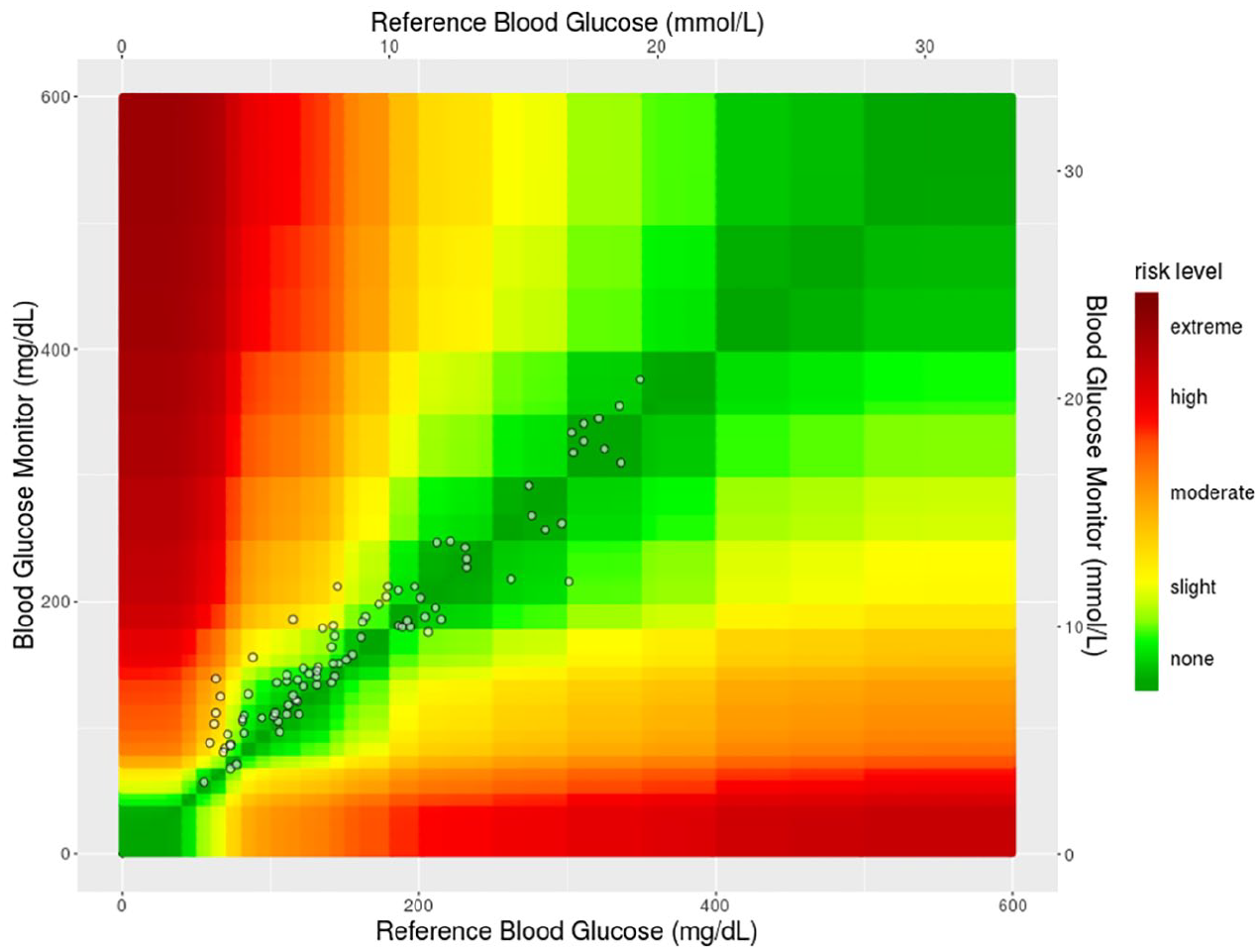
Surveillance error grid for the noninvasive data set.
The study procedures were well tolerated. No general adverse events or device-related adverse events were reported from the study visits or the previous calibration periods.
Discussion
This system accuracy study for the invasive component was conducted in accordance with the suggested ISO15197 protocol. 9 The trial was performed with three strip lots and six devices and with 100 samples that were distributed over the entire blood glucose measurement range, and with a distribution of glucose values as requested by the ISO standard. It is of note that this invasive glucose-oxidase based blood glucose meter system is an adapted version of the approved OK Meter (OK Biotech Co., Ltd., Hsinchu City, Taiwan) and technically identical to that system. It was therefore expected that the results would be similar to those obtained with the originating meter technology. This was indeed the case with 97.1% of the data pairs meeting overall ISO criteria. A total MARD of 7.1% when operated by healthcare professionals and the same MARD of 7.1% when operated by users indicate the robustness of the invasive CoG component performance, which can therefore be considered to present state of the art technology for patient self-testing of blood glucose. After performing several different statistical analyses as requested by ISO15197:2015, it can be confirmed that the invasive TensorTip CoG component meets all acceptance criteria for system accuracy as defined by the ISO15197:2015 standard.
A major advantage of the noninvasive tissue glucose reading approach is the access to an unlimited number of pain-free glucose readings enabling easy glucose trend monitoring. It should be emphasized that the ISO 15197 requirements have not been designed to evaluate the performance of noninvasive glucose reading devices. Applying the same analysis conducted on the invasive component according to ISO 15197, however, revealed that 70% to 80% of the noninvasive tissue glucose predictions when paired with capillary blood glucose reference readings were meeting the ISO accuracy criteria. In addition, it was seen in the analysis that the results were substantially better, if the calibration procedure was correctly performed prior to the experiment. An experimental error that was detected was that patients were tested in this study with an actual blood glucose concentration outside of the glucose concentration range that had been covered during the previous calibration period. This mismatch of test vs calibration concentrations may be the reason for the observed gaps of more than 40 mg/dL/40% in the comparison between YSI lab reference results and invasive results on one side and the noninvasive CoG readings on the other side.
The results of this study were used by the manufacturer to improve the calibration procedure. In the next generation, an initial calibration procedure with 25 comparator measurements is performed over three to four days. The device software is now programmed with a self-learning algorithm and requests additional invasive confirmatory readings whenever an unusual pattern of signals is determined by the noninvasive technology. The accuracy of this renewed calibration procedure is currently subject of ongoing prospective clinical trials. Another potential solution can also be an intentional glucose titration into the hypoglycemic range including performance of additional calibration measurements under medical supervision.
Our study has several limitations which need to be addressed, when discussing the results. As accepted by the ISO protocol, extreme glucose levels were tested by means of manipulated samples prepared in the laboratory. We have shown that in particular glucose-oxidase based test strip technologies are subject to oxygen interference, which requires a very careful in vitro handling of such artificially prepared samples before and during the testing procedures. 17 In addition, this study was a cross-sectional single spot comparison with a blood glucose reference method ignoring the well-established lag time between blood glucose and tissue glucose in the case of dynamic changes.18,19 On the other side, the trend information that becomes available by more frequent readings was also not included in this evaluation. It is important to note that the CoG was calibrated at home by the participant by using the invasive glucometer. Despite this condition, the postcalibration accuracy to YSI lab device still resulted in >70% match of the noninvasive results with the restrictive ISO15197:2015 criteria for invasive devices, and the detection of most of the readings in zone A of the consensus error grid analysis in the vast majority of the participants with prior correctly performed calibration. Even in the raw data set, 98.9% were found to be in A + B of the Consensus Error Grid, which to our knowledge represents the best performance shown in such an analysis for a noninvasive glucose prediction so far.
In conclusion, the invasive component of the CoG device fulfilled ISO criteria and the tissue glucose prediction by the noninvasive device component showed a good accuracy even when tested against a capillary blood glucose reference method. These results are providing an encouraging perspective for patients with diabetes to gain frequent and pain-free glucose information suitable to help improving glycemic control.
Footnotes
Author Contributions
Andreas Pfützner planned and conducted the study, researched the data, and prepared the manuscript. Filiz Demircik and Alexander Lier contributed to study conduct, data analysis, and manuscript discussion. Johannes Pfützner, Kim Kessler, and Jan Spatz contributed to study conduct and data analysis. Stephanie Strobl contributed to study conduct, data analysis and discussion, and manuscript preparation. Anke H. Pfützner contributed to study planning and regulatory submission, and to result interpretation and discussion.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The TensorTip CoG device is manufactured by CNOGA Medical Ltd., Ceasarea, Israel (CNOGA). Andreas Pfützner received a research grant for study conduct and travel support from CNOGA. Anke H. Pfützner is the co-worker and spouse of Andreas Pfützner. The other co-authors have no conflict of interest to report.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: APF received a research grant from CNOGA Medical for the performance of the study. AHPF is the spouse of APF. The other authors received no financial support for the research, authorship, and/or publication of this article.
