Abstract

New guidelines have recently been introduced (ISO 15197:2013(E)) by the International Organization for Standardization to establish acceptable performance for blood glucose monitoring systems (BGMS). 1 In this study the accuracy of the blood glucose readings provided by the AgaMatrix WaveSense JAZZ BGMS, which is a handheld BGMS that requires 0.5µl capillary whole blood, was studied when compared to a laboratory reference method. The BGMS test strip contains glucose oxidase with a redox chemical mediator that produces an electrochemical signal in proportion to the concentration of glucose in the blood. The meter measures the signal using multiple measurements and reanalyzing the input stimulation signal in response to how the chemistry is progressing to correct for common interferents.
The study was performed within the Diabetes Research Network Wales, Swansea University in compliance with good clinical practice (GCP) guidelines and approved by the relevant ethics committee.
System accuracy was evaluated for 100 subjects spanning a wide concentration range (1.50-24.75 mmol/L). Fresh capillary blood samples were collected from the fingertip of the subject’s hand. A predetermined number of samples were assigned to each of 7 concentration subgroups to ensure even distribution across the range. In accordance with the ISO 15197 guidelines an agreed number of samples were modified to meet the extreme glucose concentrations and adjusted to have pO2 concentrations equivalent to capillary blood prior to assay.
Plasma glucose was analyzed using a YSI 2300 Stat Plus (Yellow Springs Instruments, Fleet, UK) as the reference method and blood glucose was determined on 6 different BGMS meters with 3 lots of test strips, which were all provided by the manufacturer. For YSI measurement, blood samples were collected into capillary collection tubes (Lithium Heparin Microvette, Sarstedt, Leicester, UK) and centrifuged prior to plasma glucose measurement. In addition, hematocrit was measured using a Hemo Control analyzer (EKF, Cardiff, UK). Daily control measures were carried out on all meters and instruments. Other than in the modified samples, all blood sampling and analysis were performed within a 5-minute time frame.
The blood glucose results from 3 different lots of test strips (200 samples/lot) showed excellent agreement and linearity with results from the YSI (
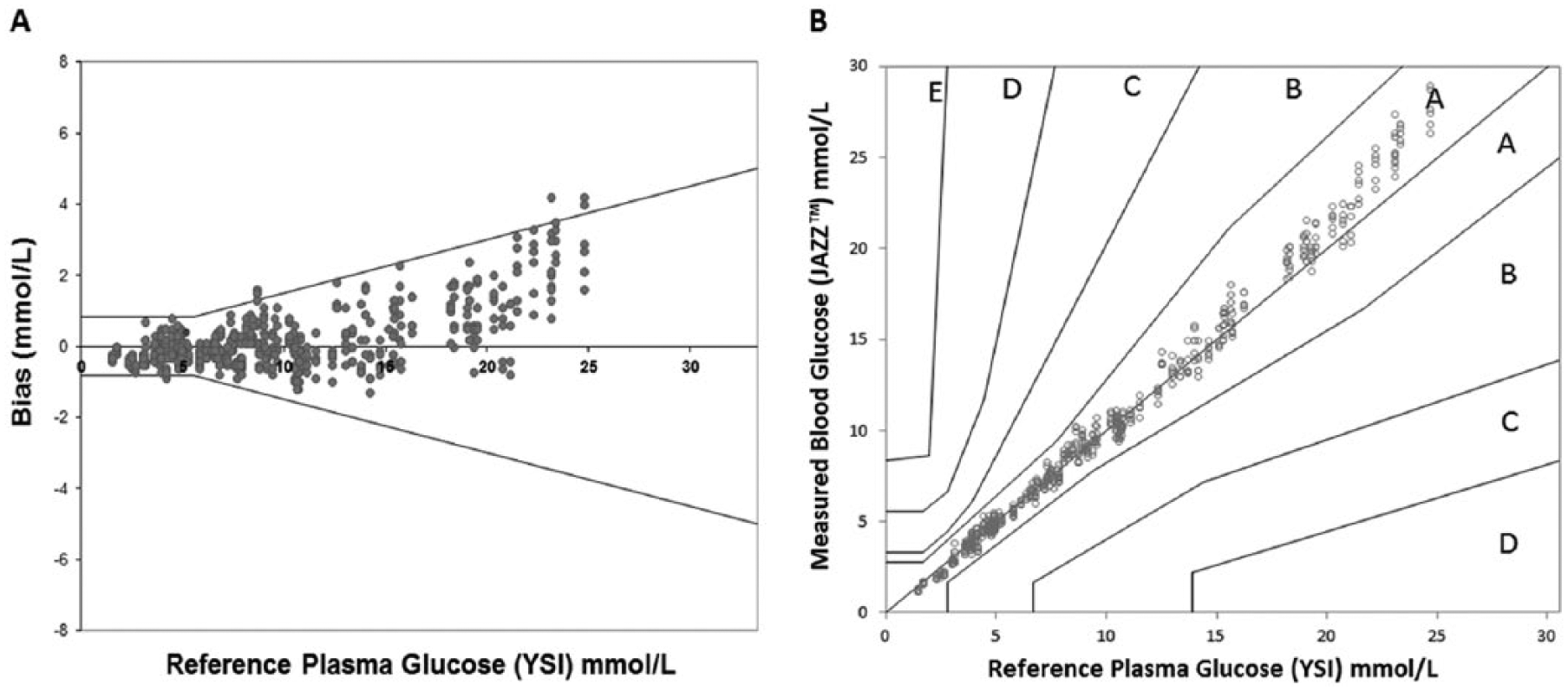
(A) System accuracy plot for BGMS glucose vs YSI 2300 plasma glucose concentration. Data shown are from all 3 test strip lots. Area inside the solid lines represents minimum acceptable accuracy from ISO 15197:2013. (B) Consensus error grid plot of BGMS glucose concentrations vs YSI 2300 plasma glucose. In all, 600 data points shown (100 samples measured in duplicate on each meter).
This study demonstrates that the system has excellent agreement with a reference laboratory method and exceeds the minimum acceptable system accuracy criteria specified by ISO 15197:2013.
Footnotes
Abbreviations
BGMS, blood glucose monitoring system; GCP, good clinical practice; ISO, International Organization for Standardization; YSI, Yellow Springs Instruments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
