Abstract
New European medical device regulations require the performance of postmarketing surveillance evaluations for blood glucose meters (BGMs). We conducted an ISO15197:2015-conform system performance evaluation with the approved glucose dehydrogenase (GDH)-based Wellion NEWTON BGM. One hundred subjects were enrolled into the study (44 female, 56 male, 43 healthy subjects, 23 type 1 diabetes, 34 type 2 diabetes, age: 53.7 ± 15.8 years). In addition, manipulated heparinized whole blood was used for a laboratory interference test with ten selected substances (interference definition: substance-induced bias > 10%). The mean absolute relative difference (MARD) was 4.7%, and 100% of the values were in zones A (99.7%) and B (0.3%), respectively, of the consensus error grid. Interference was observed with xylose only, which is a known interfering substance for GDH-based BGMs.
Introduction
Routine blood glucose monitoring is a key component in diabetes management to avoid the development of health complications, such as blindness, kidney disease, heart disease, and nerve damage.1-3 For approval of meters for self-monitoring of blood glucose (SMBG), a clinical system performance evaluation in accordance with ISO15197:2015 4 needs to be conducted. In Europe, a general requirement for a more thorough postmarketing surveillance (PMS) for marketed medical products has been introduced in May 2022. 5 At this stage, there are no recommendations or guidelines existing, how such a PMS program should be conducted for blood glucose meters (BGMs) and strips. The Austrian Diabetes Supply Company MedTrust has recently introduced a new SMBG (Wellion NEWTON), which is based on a glucose dehydrogenase (GDH) sensing technology. This clinical and laboratory study was performed by an external institute as part of the company’s PMS program to re-confirm the system accuracy in compliance with ISO 15197:2015 guidelines and to identify the possible impact of key substances that have been described previously as interferents with other SMBG technologies.
Patients and Methods
The clinical study was performed in compliance with all applicable regulatory and ethical requirements. The responsible institutional review board (IRB) (Ethikkommission der Landesärztekammer Rheinhessen) approved the study (Application No. 2022-16712) and all participants signed informed consent prior to any study procedure. The study was registered in the German Clinical Trial Registry (DRKS No. DRKS00031124).
Clinical System Performance Evaluation
Blood was taken from the fingertip for the reference method (YSI 2300Statplus Glucose Analyzer, YSI Inc., Yellow Springs, OH), and health care professionals conducted three readings with the study of SBGM with three different strip lots and three different devices (with randomized combinations). Thereafter, another reference measurement was performed. The reference readings before and after the test were not allowed to differentiate by more than 5% to be eligible for the analysis. To meet ISO15197:2015 requirements, the blood glucose concentrations tested in this study had to be distributed as follows: 5%: < 50 mg/dL (< 2.77 mmol/L); 15%: > 50 to 80 mg/dL (> 2.77-4.44 mmol/L); 20%: > 80 to 120 mg/dL (> 4.44-6.66 mmol/L); 30%: > 120 to 200 mg/dL (> 6.66-11.10 mmol/L); 15%: > 200 to 300 mg/dL (> 11.10-16.65 mmol/L); 10%: > 300 to 400 mg/dL (> 16.65-22.20 mmol/L); 5%: > 400 mg/dL (> 22.20 mmol/L). If needed, glucose adjustments were allowed by of means insulin or glucose administration. The samples with extreme values were obtained by manipulating heparinized whole blood from five healthy donors each, by incubating at 37°C (< 50 mg/dL) or by careful spiking with a 20% physiological glucose solution (> 400 mg/dL), respectively.
Interference Testing
Heparinized whole blood samples provided by ten of the healthy volunteers were adjusted to have three different blood glucose concentrations (50-80, 130-160, and 250-300 mg/dL). Two concentrations (50% and 100% of the maximally tested concentration) of the selected ten substance candidates for interference were separately prepared with each glucose concentration. The final glucose concentration was determined with the reference method, and the degree of oxygenation (85%-100%) and the hematocrit (35%-45%) of the prepared samples were confirmed to be in the target ranges. Three meters and 3 strip lots (in randomized combinations) were employed to perform 10 parallel measurements (reference: mean YSI reading obtained before and after the interference measurements).
Statistical Analysis
Data analysis of system accuracy was performed according to EN ISO 15197:2015 4 using YSI 2300 STAT PLUS as the reference method, including: bias analysis according to Bland and Altman, 5 regression analysis according to Passing and Bablok, 6 MARD calculation, and consensus error grid analysis.7,8 For the interference analysis, a mean absolute bias > 10% (substance interference factor [SIV]) was defined as indicative for interference.
Results
One hundred participants were enrolled into the study (44 female, 56 male, 43 healthy subjects, 23 type 1 diabetes, 34 type 2 diabetes, age: 53.7 ± 15.8 years). Blood glucose levels were distributed over the entire measurement range and in accordance with the ISO requirements. The glucose concentration of the samples ranged between 37 mg/dL (2.06 mmol/L) and 477 mg/dL (26.50 mmol/L). Thirty-four samples had glucose concentrations ≤ 100 mg/dL (5.55 mmol/L). There were no adverse events or serious adverse events observed during the study conduct.
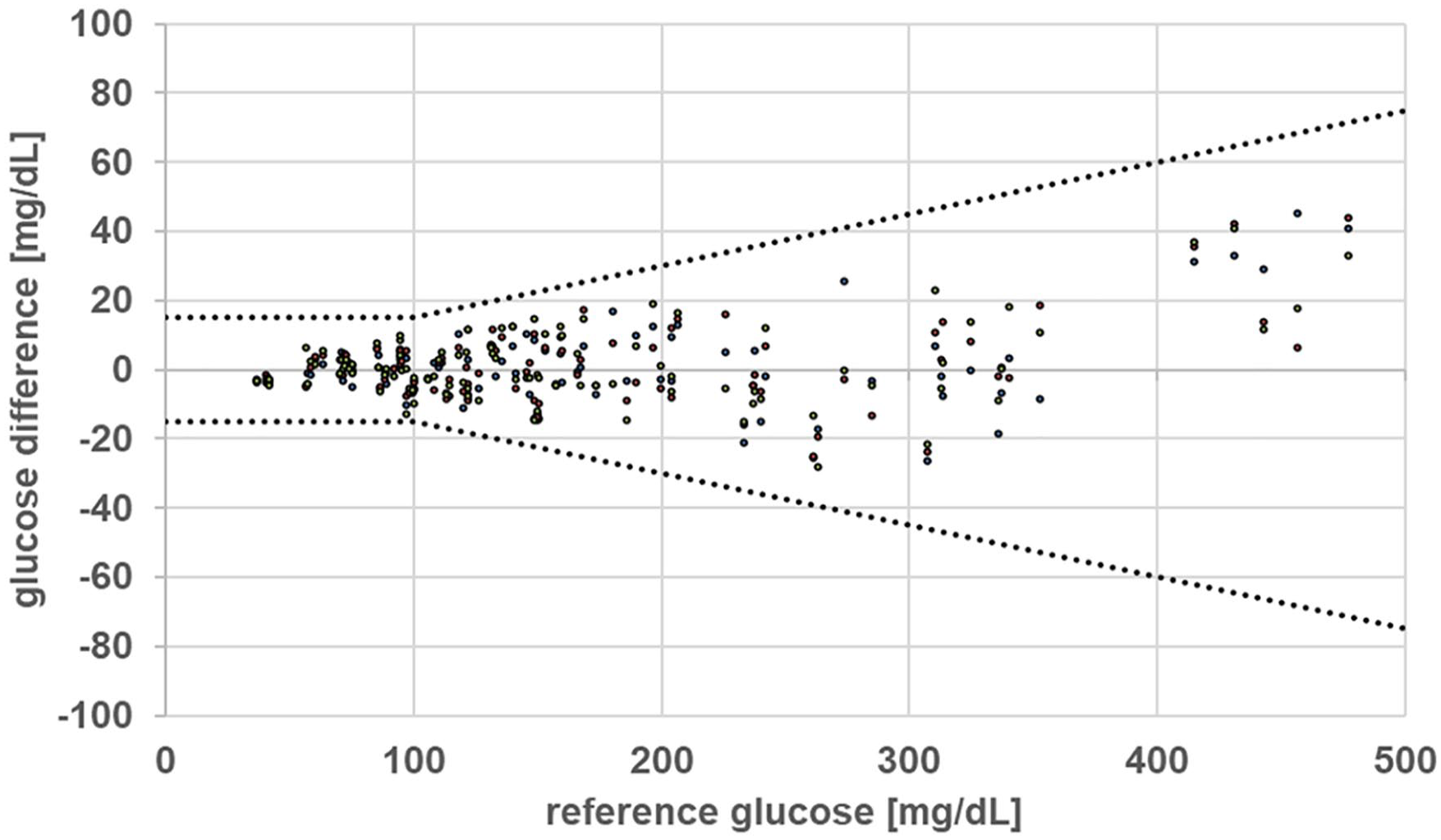
The study showed a high accuracy of the study device with an MARD: 4.7%. The different percentages of values with varying differences of < 5% to < 30% are provided in Table 1.
Accuracy Evaluation for Different Blood Glucose Ranges and Different Acceptance Criteria.
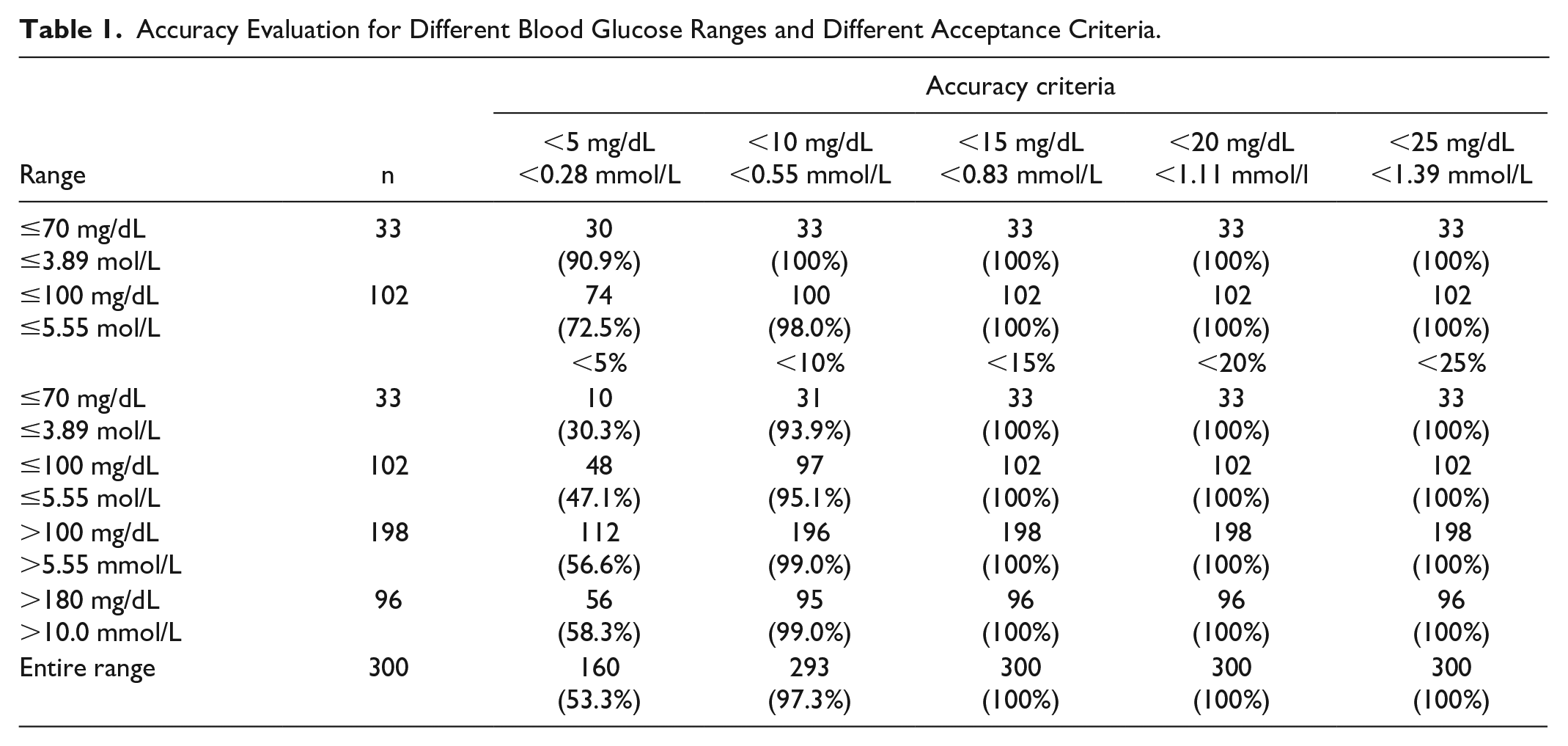
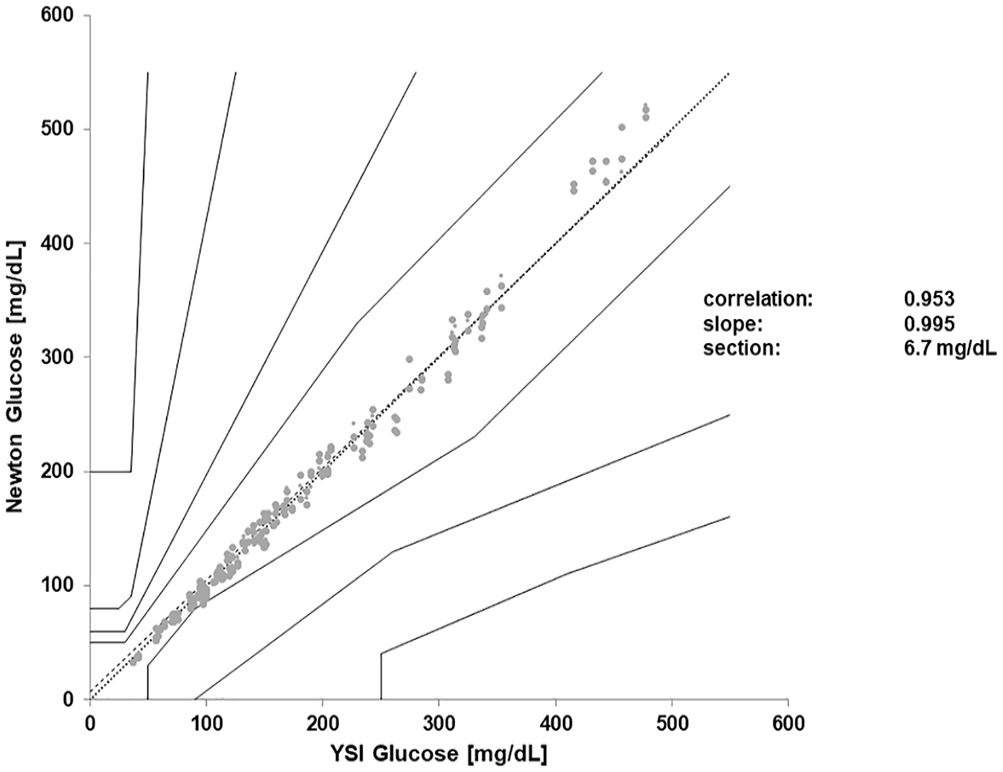
Regression analysis revealed a slope of 0.953 and a correlation coefficient of 0.995 (see Figure 1). In the consensus error grid analysis for type 1 diabetes,7,8 299/300 (99.7%) of the data points were found in zone A and 1/300 (0.3%) was found to be in zone B (see Figure 1). In the Bland-Altman analysis, 100% of the data pairs were within the acceptance boundaries, difference of ±15 mg/dL (0.7 mmol/L) for values ≤ 100 mg/dL (5.55 mmol/L) and ±15% for values > 100 mg/dL (5.55 mmol/L,), and there was no observable trend for general over-reading or under-reading (see Figure 2). The results of the in vitro interference tests are provided in Table 2 and Figure 3. Interference was observed with xylose but not with any of the other nine substances tested.
Regression analysis and consensus error grid analysis with 100 samples and three strip lots (n = 300). The trend line is indicated by the thicker dotted line.
Bland-Altman analysis. All results (n = 300) were within the acceptance boundaries, which are indicated by the dotted lines.
Results of the In Vitro Interference Tests.
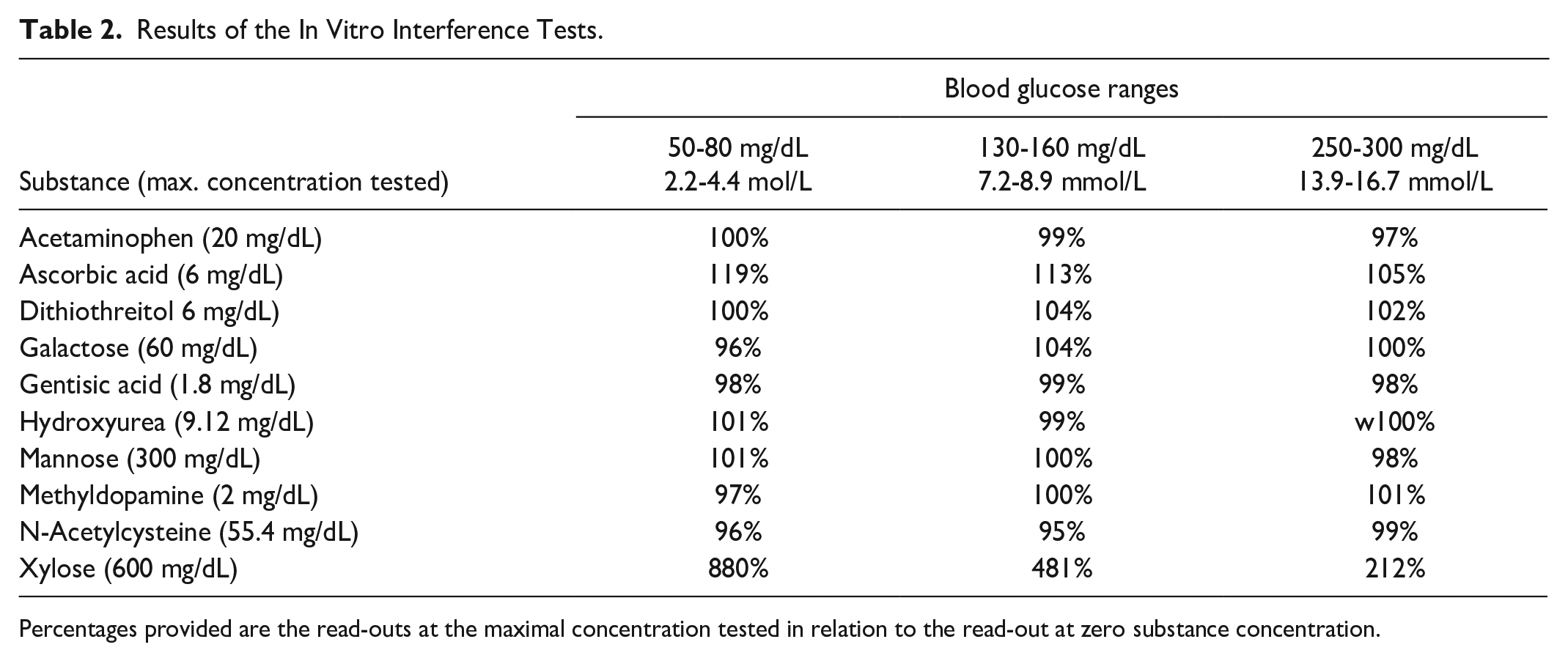
Percentages provided are the read-outs at the maximal concentration tested in relation to the read-out at zero substance concentration.
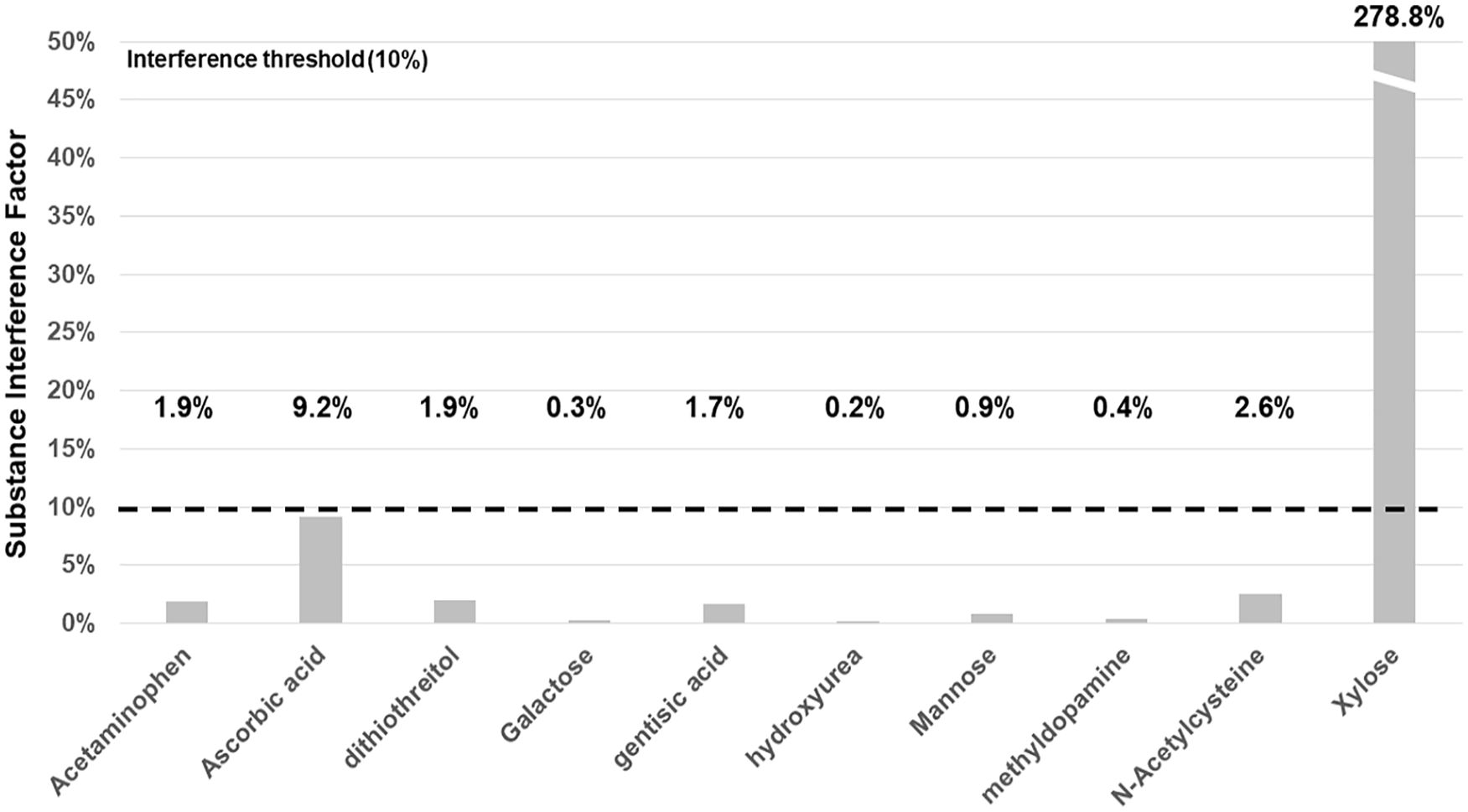
Substance interference factors of all tested substances (SIV: mean absolute relative difference for all tested blood glucose concentrations at the highest interferent concentration).
Discussion
Success of diabetes care depends on accurate and robust SMBGs, 1 which are needed for correct insulin dosing, detection and prevention of hypoglycemic and hyperglycemic episodes,1,9,10 and prevention of secondary complications.2,3,11 Over the past decades, the analytical accuracy of SMBG systems has constantly improved. In parallel, the regulatory bodies have increased the approval hurdles and while meters in the beginning of the millennium were still accepted with MARD levels of 10% to 15%, they nowadays have to show a MARD < 10%. The acceptance range of the previous ISO15197:2003 4 was narrowed down with ISO15197:2015 to be ±15 mg/dL (0.83 mmol/L) for values ≤ 100 mg/dL (5.55 mmol/L) and ±15% for values > 100 mg/dL (5.55 mmol/L). 4
In this study, the GDH-based SMBG Wellion NEWTON was subjected to a clinical and laboratory accuracy study in accordance with the ISO15197:2015 protocol and successfully passed this evaluation with a MARD of 4.7% and with 100% of the data pairs in the acceptable zones A and B of the consensus error grid analysis. In addition, we also conducted an abbreviated interference test with ten substances, known to potentially interfere with GDH-based SMBGs or needle sensors for continuous glucose monitoring.12-16 The investigated SBGM was only affected by xylose.
Xylose is a known substrate of GDH, and it is, for example, not recommended to use GDH-based devices when performing a standard malabsorption test procedure with application of 15 g of xylose, which can result in 1-hour blood xylose concentrations of up to 50 to 60 mg/dL.16-18 The observed xylose interference is in line with previous investigations with GDH-based meter and strip technologies,12,19,20 and is appropriately addressed by a warning advice in the instructions for use (IFU) of the SMBG. Xylose interference can also affect glucose oxidase-based needle sensors for continuous glucose measurement. 21 Before drawing ultimate conclusions regarding the clinical relevance of our findings, it is required to perform confirmatory clinical studies with oral xylose uptake and SMBG use in accordance with the IFU.
Conclusions
The investigated GDH-SMBG was observed to be very accurate and to meet all applicable regulatory US and EU requirements. Only xylose had an influence on the meter results, which is a known phenomenon for GDH-based glucose measurement technologies.
Footnotes
Declaration of Conflicting Interests
APF has received a research grant from Medtrust GmbH. SW is an employee of Medtrust GmbH. The other author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by an unrestricted grant from Medtrust GmbH, Austria.
